Research Article - Biomedical Research (2017) Volume 28, Issue 10
Is elevated red blood cell distribution associated with mortality in superelderly patients with community-acquired pneumonia?
Mevlüt Karataş1* and Songül Özyurt2
1Atatürk Chest Diseases and Thoracic Surgery Training and Research Hospital, Ankara, Turkey
2Department of Pulmonology, Faculty of Medicine, Recep Tayyip Erdoğan University, Rize, Turkey
- *Corresponding Author:
- Mevlüt Karataş
Atatürk Chest Diseases and Thoracic Surgery Training and Research Hospital
Ankara, Turkey
Accepted date: February 13, 2017
Abstract
Introduction: The purpose of this study was to determine the association between red blood cell distribution width (RDW) and mortality in super-elderly subjects (≥ 90 years) diagnosed with community-acquired pneumonia (CAP).
Methods: One hundred twenty subjects in the super-elderly age group diagnosed with CAP were followed up for at least one year after first diagnosis time. It was investigated whether high RDW level and advanced age were associated with mortality.
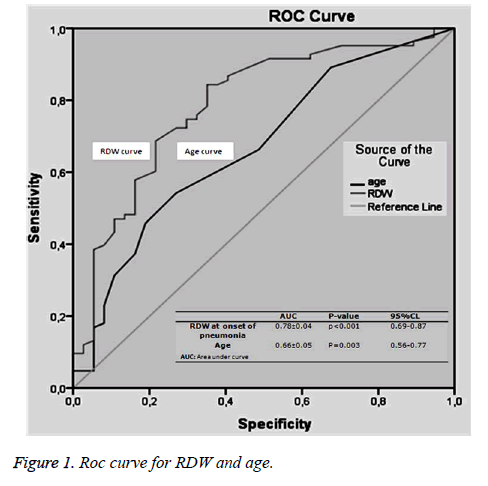
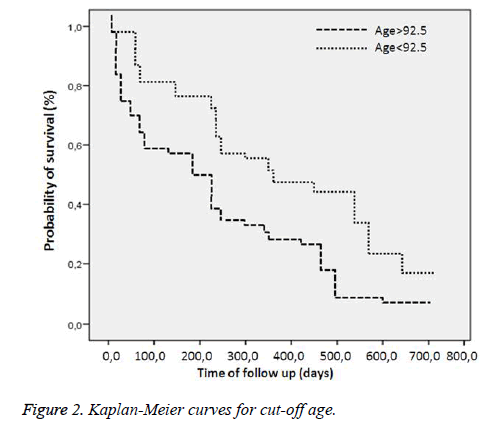
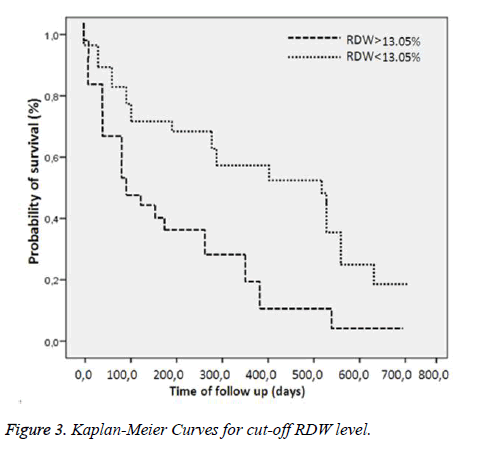
Results: Eighty-three subjects (69%) died, and 37 (31%) survived. Basal RDW level was significantly correlated with mortality (p<0.001). A cut-off value>13.05% for RDW predicted mortality with specificity of 64.86% and sensitivity of 84.33%, with a negative predictive value (NPV) of 64.86% and a positive predictive value (PPV) of 84.33%. A cut-off value>92.5 for age predicted mortality with specificity of 72.97% and sensitivity of 54.21%, with NPV of 41.53% and PPV of 81.8%. Cox regression analysis revealed that a RDW cut-off>13.05%, age cut-off>92.5 and presence of Alzheimer’s disease increased mortality independently 2.6-fold (p=0.002), 1.5-fold (p=0.040) and 2.4-fold (p=0.003), respectively.
Conclusions: This is the first study to examine the relationship between RDW, age and mortality specifically in a super-elderly subject aged over 90 with a diagnosis of CAP. RDW level, advanced age and presence of Alzheimer’s disease are correlated with mortality in super-elderly subjects (≥ 90 years) with CAP.
Keywords
Super-elderly, Red cell distribution width, Mortality, Risk factors, Community-acquired pneumonia, Survive
Introduction
Community-acquired pneumonia (CAP) is a significant cause of mortality and morbidity, particularly in the elderly. Pneumonia in elderly subjects prolongs hospitalization and increases complications [1]. Annual mortality levels as high as 40% are seen in subjects with CAP [1]. Morbidity and mortality levels decrease with vaccination and appropriate antibiotic use in several diseases secondary to infection, but the desired decrease is not observed in pneumonia. Pneumonia is the sixth most common cause of death worldwide, the most common cause of infection-related deaths and the second main cause of hospitalization [2-4].
Red blood cell distribution width (RDW) is a routinely used hematological marker analyzed with complete blood count and showing the volume of erythrocytes in the circulation [5]. RDW is routinely employed in the diagnosis of thalassemia and iron deficiency [6]. Numerous studies have reported that RDW levels are correlated with poor prognosis in congestive heart failure (CHF), coronary heart disease (CHD) [7,8], rheumatoid arthritis [9], acute stroke [10], chronic renal failure (CRF) [11], pulmonary embolism [12] and septic shock [13]. While the cause of RDW elevation is uncertain, recent studies have shown that inflammatory processes raise RDW by affecting erythropoiesis and that a high RDW level is associated with poor prognosis in CAP [14,15]. RDW is also known to rise together with advanced age and disease burden [16].
The association between RDW and mortality in the super-elderly age group (≥ 90 years) with a diagnosis of CAP is still unclear. We hypothesized that high RDW and advanced age are associated with mortality in super-elderly subjects with CAP.
Materials and Methods
Following receipt of local ethical committee approval, this study was performed as a retrospective investigation of 120 subjects hospitalized with a diagnosis of CAP between 1 January, 2014, and 1 January, 2016 and was followed up for at least one year after first diagnosis time, at the Recep Tayyip Erdoğan University Faculty of Medicine Chest Diseases Department, Turkey. Subjects’ demographic characteristics, basal complete blood count and RDW values at time of diagnosis, accompanying comorbid diseases, laboratory findings, and pneumonia symptoms and findings were collected retrospectively from medical records and recorded. Survival data for subjects dying in hospital were obtained from the hospital archive, and survival data for other subjects were obtained by telephone from the subjects themselves or their families. Diagnosis of CAP was based on Infectious Diseases Society of America and American Thoracic Society guidelines [17]. Following subject archive records examination, at least one clinical finding (dark, yellow phlegm, cough, body temperature >37.8°C) or at least two minor criteria in addition to pulmonary infiltrations at x-ray (tachypnea, dyspnea, impaired orientation, pleural pain, pulmonary consolidation or a leukocyte number >12,000 cells/microL) were adopted as diagnostic for CAP.
Exclusion criteria: Subjects with a history of pneumonia in the previous 30 days, with active pulmonary tuberculosis and known HIV positivity and chronic immunosuppressive subjects (subjects immunosuppressive due to solid organ transplantation, post-splenectomy subjects, subjects receiving 10 mg prednisolone or equivalent medications daily for longer than 30 days, subjects being treated with other immunosuppressive drugs and subjects with severe neutropenia) were excluded from the study.
Laboratory analysis
Complete blood count (CBC) was performed using an automatic haematology analyzer (Cell-Dyn Ruby 100 test/h, 2012, Abbott). RDW analysis was assessed as a component of CBC. The normal reference interval for RDW in our hospital laboratory is 11.6-14.8%.
Statistical analysis
Subjects were divided into two groups depending on RDW (cut-off:13.05) and age (cut-off:92.5) values at time of diagnosis. Constant and category basic characteristics were compared between the groups using the independent-samples ttest and x2 test. Survival analyses between the groups were performed using Kaplan-Meier survival analysis. Prognostic variables for mortality were analyzed using the Cox proportional hazards model. A P-value of <0.05 was considered significant. The results of the univariate and multivariate Cox regression analyses are presented as hazard ratios (HRs) with 95% confidence intervals (CIs). ROC curve analysis was performed for RDW and age.
Results
One hundred twenty subjects diagnosed with CAP were included in the study, 85 (70.8%) women and 35 (29.2%) men, with a mean age of 93.6 ± 3.9. At 24-month retrospective investigation, 83 (69%) subjects had died and 37 (31%) were still living. Mean duration of hospitalization was 8.6 ± 6.2 days. Subjects’ demographic and basal laboratory data based on their RDW and age cut-off values are shown in (Tables 1 and 2).
| RDW<13.05 n=37 |
RDW>13.05 n=83 |
P† value | |
|---|---|---|---|
| Female sex,% | n=28 (75) | n=57 (68) | |
| Age, years | 92.6 ± 3.2 | 94.1 ± 4.0 | 0.041 |
| Survival/days | 437.9 ± 264.4 | 279.7 ± 285.7 | 0.004 |
| Hospitalization time/days | 7.7 ± 5.2 | 9.0 ± 6.4 | 0.222 |
| Luekocyte number, ×103 | 11.5 ± 4.9 | 13.7 ± 10.6 | 0.116 |
| Lymphocyte, ×103 | 2.6 ± 6.7 | 7.7 ± 8.0 | 0.092 |
| Neutrophil, ×103 | 9.3 ± 4.9 | 9.7 ± 5.7 | 0.708 |
| Monocyte, ×103 | 0.7 ± 0.3 | 0.8 ± 0.3 | 0.434 |
| Eosinophil, ×103 | 0.2 ± 0.8 | 0.2 ± 0.1 | 0.858 |
| Basophil, ×103 | 2.3 ± 0.9 | 0.1 ± 0.9 | 0.047 |
| MCV, fL | 86.3 ± 4.7 | 87.0 ± 8.7 | 0.576 |
| MCH, pg | 28.7 ± 1.7 | 28.6 ± 2.5 | 0.74 |
| Platelet, K/uL | 258.00 ± 119.7 | 241.34 ± 112.7 | 0.473 |
| MPV, fL | 7.6 ± 2.4 | 14.2 ± 4.5 | 0.194 |
| Hemoglobin, g/dL | 11.9 ± 1.7 | 11.6 ± 2.0 | 0.488 |
| Hematocrit, % | 35.8 ± 5.1 | 35.8 ± 6.1 | 0.977 |
| Values are given with mean and standard deviation P†: P value for RDW subgroups RDW: Red Cell Distribution Width; MCV: Mean Corpuscular Volume; MPV: Mean Platelet Volume; MCH: Mean Corpuscular Hemoglobin; fL: Femtoliter; pg: pikogram; dL:desiliter |
|||
Table 1: Basal and laboratory characteristics according to cut-off level of RDW.
| age<92.5 n=65 |
age>92.5 n=55 |
P‡ value | |
|---|---|---|---|
| Female sex,% | n=44 (67) | n=41 (74) | |
| Age,years | 90.9 ± 0.7 | 96.8 ± 3.7 | <0.001 |
| Survival/days | 373.8 ± 294.6 | 275.09 ± 272.4 | 0.059 |
| Hospitalization time/days | 8.4 ± 6.4 | 8.8 ± 5.8 | 0.725 |
| Luekocyte number, ×103 | 13.3 ± 9.4 | 12.8 ± 9.1 | 0.76 |
| Lymphocyte, ×103 | 4.9 ± 6.6 | 5.2 ± 6.8 | 0.853 |
| Neutrophil, ×103 | 9.9 ± 5.8 | 9.2 ± 5.1 | 0.5 |
| Monocyte, ×103 | 0.7 ± 0.4 | 0.7 ± 0.5 | 0.927 |
| Eosinophil, ×103 | 0.4 ± 0.1 | 0.8 ± 1.5 | 0.119 |
| Basophil, ×103 | 1.3 ± 7.2 | 2.2 ± 1.1 | 0.927 |
| MCV, fL | 86.0 ± 9.1 | 87.8 ± 5.4 | 0.175 |
| MCH, pg | 28.6 ± 2.5 | 28.6 ± 2.0 | 0.675 |
| Platelet, K/uL | 252.25 ± 122.5 | 239.65 ± 102.4 | 0.543 |
| MPV, fL | 12.6 ± 4.1 | 11.7 ± 3.4 | 0.903 |
| Hemoglobin,g/dL | 11.6 ± 1.9 | 11.9 ± 1.9 | 0.478 |
| Hematocrit, % | 35.6 ± 5.7 | 36.1 ± 5.8 | 0.634 |
| Values are given with mean and standard deviation P‡: P value for age subgroups RDW: Red Cell Distribution Width; MCV: Mean Corpuscular Volume; MPV: Mean Platelet Volume; MCH: Mean Corpuscular Hemoglobin; fL: Femtoliter; pg: pikogram; dL:desilite |
|||
Table 2: Basal and laboratory characteristics according to cut-off level of age.
Comorbid diseases accompanying pneumonia were hypertension in 43 cases (35.5%), CHF in 27 (22.5%), cerebrovascular disease in 22 (18.3%), Alzheimer’s disease in 21 (17.5%), type II diabetes mellitus in 14 (11.6%), CHD in 14 (11.6%), and CRF in 31 (25.8%) (Table 3). Mean total RDW level was 15.8 ± 3.4%. RDW level in the non-surviving subjects was 15.96 ± 3.6%, compared to 13.12 ± 2.0% in the surviving subjects (p<0.001).
| N | % | |
| Arterial hypertension | 43 | 35.5 |
| Chronic renal failure | 31 | 25.8 |
| Congestive heart failure | 27 | 22.5 |
| Cerebrovascular disease | 22 | 18.3 |
| Alzheimer's Disease | 21 | 17.5 |
| Malignity | 15 | 12.5 |
| Coronary heart disease | 14 | 11.6 |
| Tip II diabetes mellitus | 14 | 11.6 |
| Chronic liver disease | 12 | 10 |
| Prostate diseases | 7 | 5.8 |
| Anemia | 5 | 4.1 |
Table 3: Additional diseases accompanying pneumonia.
Basal RDW levels at time of hospitalization were significantly correlated with some blood parameters such as; platelet number, MCV, MCH, eosinophil numbers, neutrophil-eosinophil ratio and eosinophil-basophil ratio (p=0028, p<0.001, p=0.007, p=0.001, p=0.001 and p<0.001, respectively).
Comparison of the surviving and non-surviving subjects in terms of accompanying comorbid factors revealed that Alzheimer’s disease affected mortality (p=0.02). No correlation was observed between other comorbid factors and mortality. At ROC analysis, basal RDW levels and the age factor were observed to affect mortality (Figure 1). Survival curves of both cut-off points of RDW and age were statistically different (p<0.001) (Figures 2 and 3). Area under the curve (AUC) for RDW was calculated at 0.78 (95%CI: 0.698-0.878) (p<0.001), and AUC for age at 0.66 (95%CI: 0.565-0.773) (p=0.003). A cut-off value for RDW of 13.05% exhibited 64.86% specificity for mortality and 84.33% sensitivity, with a negative predictive value (NPV) of 64.86% and a positive predictive value (PPV) of 84.33%. A cut-off value for age of 92.5 exhibited 72.97% specificity and 54.21% sensitivity for mortality, with NPV of 41.53% and PPV of 81.81%. Cox regression analysis was performed to determine the extent to which RDW cut-off>13.05%, age cut-off>92.5 and presence of Alzheimer’s disease independently affected mortality. RDW>13.05% increased mortality 2.6-fold (p=0.002) (95%CI: 1.435-4.814), advanced age >92.5increased mortality 1.5-fold (p=0.040) (95% CI: 1.022-2.452) and presence of Alzheimer’s disease increased mortality 2.4-fold (p=0.003) (95%CI: 1.336-4.326) (Table 4).
| P value | OR | 95% CI | ||
| Lower | Upper | |||
| Age>92.5 cut-off | 0,040 | 1.58 | 1,022 | 2,452 |
| RDW>13.05 cut-off | 0,002 | 2.62 | 1,435 | 4,814 |
| Alzheimer’s disease | 0,003 | 2.4 | 1,336 | 4,326 |
| OR: Odds ratio | ||||
Table 4: Cox regression results modeling mortality by Alzheimer’s disease, RDW and age cut-off values.
Discussion
The risk of pneumonia is greater in subjects of advanced age compared to other age groups. CAP can lead to severe mortality and morbidity in the geriatric age group. This study involved super-elderly (age ≥ 90-108) subjects hospitalized in the chest diseases clinic with a diagnosis of pneumonia. To the best of our knowledge, this is the first study to investigate basal RDW, age and mortality in subjects aged over 90, with a mean age of 93.6 ± 3.9 and diagnosed with CAP. The most significant study finding is that RDW>13.05% and age>92.5 were significantly correlated with poor prognosis in super-elderly subjects with CAP.
Various mechanisms have been proposed to explain the association between RDW and mortality, although the biological mechanisms between high RDW levels and mortality are still unclear. The relation between RDW levels and CAP is also unclear. Lippi et al. suggested that chronic subclinical inflammation might be involved in the relation between RDW and mortality [18], while Weiss et al. reported that inflammation may increase RDW by compromising erythrocyte half-life, erythropoiesis and the red cell membrane [19]. Some studies have also interpreted blood RDW elevation as a result of exposure to oxidative stress. Inflammation and oxidative stress affect erythrocyte haemostasis. Although erythrocytes possess an antioxidant capacity, oxidative stress causes erythrocyte destruction and reduced life span. Eshler et al. reported that high RDW may be associated with increased oxidative stress in subjects with Down syndrome, respiratory insufficiency or receiving dialysis [20]. Patel et al. showed that decreased serum antioxidant levels were associated with RDW elevation [16]. Katsoulis et al. showed significantly lower total antioxidant status in subjects with CAP compared to a control group [21].
Braun et al. reported that high RDW>14.5% was associated with higher mortality and severe morbidity, independently of blood WBC and hemoglobin levels at time of presentation to hospital, in 637 subjects with CAP and a mean age of 46 [22]. Braun et al. reported that RDW>15% was correlated with mortality in subject with CAP and a mean age of 69.9 [23]. Lee et al. showed that high RDW>15.2% was associated with high mortality in 744 subjects with CAP and a mean age of 70.1 [14]. Patel at al. reported that high RDW>14.05% was associated with high mortality in 8175 subjects with an age ranging between 52 and 66 and dying from various causes [16]. In a meta-analysis of almost 4000 subjects dying of various causes and with a mean age ranging between 73.6 and 79.1, Patel et al. reported that highest mortality was observed in the group with RDW>16.0% [24]. Velilla et al. reported that high mortality was associated with advanced age in 125 geriatric subjects with a mean age of 85, and that the risk of mortality increased as RDW levels rose [25].
Other studies have reported correlations between RDW levels considered independently of other risk factors and anaemia [26], CHF [27,28], CRF [29], CHD [30], and metabolic syndrome [31]. Analysis of the entire subject group in our study revealed that RDW levels were correlated with anaemia (p=0.094) but not correlated with the other co-morbid diseases.
We determined a significantly higher mortality in subjects with Alzheimer’s disease and higher RDW level. This finding is compatible with Öztürk et al.’s study [32]. Cox regression analysis was performed in order to determine how RDW level, age, accompanying comorbid diseases and other subject-related factors affected mortality independently of one another.
High RDW>13.05% was observed to increase mortality 2.6- fold (p=0.002), advanced age>92.5 increased mortality 1.6- fold (p=0.04) and Alzheimer’s disease increased mortality 2.5- fold (p=0.003). A direct correlation between high RDW levels and advanced age and mortality was compatible with the results of other studies [14,23,24]. The difference between our research and previous studies is that all our subjects were of advanced age (≥ 90 years), with a mean age of 93.6 ± 3.9. Mean ages in previous studies were lower. To the best of our knowledge, this is the first study performed in a chest diseases clinic and specific to the super-elderly age group. Another difference is that the RDW cut-off value we calculated was also lower than those of other studies, and mortality increases significantly above that value. We therefore think that advanced age (>92.5) and high RDW (>13.05%) may be of clinical and subclinical importance independently of other factors in subjects with CAP, that they can be a predictor of mortality in the super-elderly age group and that these findings can make a significant contribution to the literature.
There are a number of limitations to this study. One is that it was performed in a single center. The fact that the study group was selected from the chest diseases department alone resulted in a relatively low subject number. The results are not therefore binding on subjects hospitalized in other departments with CAP. Another limitation of this study is that nutritional status, such as iron, Vitamin B12 and folate deficiencies, which might lead to RDW elevation, could not be documented. Nonetheless, the presence of anemia did not emerge as a factor affecting mortality at multivariate analysis.
In conclusion, this study shows that basal RDW levels are correlated with mortality in super-elderly subjects with CAP, and that RDW>13.05% and age>92 significantly increase mortality. In addition, this is the first study to examine the relationship between RDW, age and mortality specifically in a super-elderly subject group aged over 90 hospitalized in the chest diseases department with a diagnosis of CAP.
Acknowledgements
The authors wish to thank to Recep Tayyip Erdoğan University Medical Faculty staff in Rize, for their assistance.
References
- Kaplan V, Clermont G, Griffin MF. Pneumonia: still the old man's friend? Arch Intern Med 2003; 163: 317-323.
- Niederman MS, Mandell LA, Anzueto A, Bass JB, Broughton WA, Campbell GD. Guidelines for the management of adults with community-acquired pneumonia. Diagnosis, assessment of severity, antimicrobial therapy, and prevention. Am J Respir Crit Care Med 2001; 163: 1730-1754.
- Mandell LA. Epidemiology and etiology of community-acquired pneumonia. Infect Dis Clin North Am. 2004; 18: 761-776.
- Waterer GW, Rello J, Wundernk RG. Management of community-acquired pneumonia in adults: concise clinical review. Am J Respir Crit Care Med 2011; 183: 157-164.
- Evans TC, Jehle D. The red blood cell distribution width. J Emerg Med 1991; 9: 71-74.
- Lin CK, Lin JS, Chen SY, Jiang ML, Chiu CF. Comparison of hemoglobin and red blood cell distribution width in the differential diagnosis of microcytic anemia. Arch Pathol Lab Med 1992; 116: 1030-1032.
- Förhécz Z, Gombos T, Borgulya G, Pozsonyi Z, Prohászka Z, Jánoskuti L. Red cell distribution width: a powerful prognostic marker in heart failure. Eur J Heart Fail 2010; 128: 415.
- Van Kimmenade RR, Mohammed AA, Uthamalingam S, van der Meer P, Felker GM, Januzzi JL Jr. Red blood cell distribution width and 1-year mortality in acute heart failure. Eur J Heart Fail 2010; 12: 129-136.
- Lee WS, Kim TY. Relation between red blood cell distribution width and inflammatory biomarkers in rheumatoid arthritis. Arch Pathol Lab Med 2010; 134: 505-506.
- Ani C, Ovbiagele B. Elevated red blood cell distribution width predicts mortality in persons with known stroke. J Neurol Sci 2009; 277: 103-108.
- Oh HJ, Park JT, Kim JK, Yoo DE, Kim SJ, Han SH. Red blood cell distribution width is an independent predictor of mortality in acute kidney injury patients treated with continuous renal replacement therapy. Nephrol Dial Transplant 2012; 27: 589-594.
- Zorlu A, Bektasoglu G, Guven FM, Dogan OT, Gucuk E, Ege MR. Usefulness of admission red cell distribution width as a predictor of early mortality in patients with acute pulmonary embolism. Am J Cardiol 2012; 109: 128-134.
- Pierce CN, Larson DF. Inflammatory cytokine inhibition of erythropoiesis in patients implanted with a mechanical circulatory assist device. Perfusion 2005; 20: 83-90. Erratum in: Perfusion. 2005; 20: 183.
- Lee JH, Chung HJ, Kim K, Jo YH, Rhee JE, Kim YJ, Kang KW. Red cell distribution width as a prognostic marker in patients with community-acquired pneumonia. Am J Emerg Med 2013; 31: 72-79.
- Braun E, Kheir J, Mashiach T, Naffaa M, Azzam ZS. Is elevated Red cell distribution width a prognostic predictor in adult patients with community acquired Pneumonia? BMC Infectious Diseases 2014; 14: 129.
- Patel KV, Ferrucci L, Ershler WB, Longo DL, Guralnik JM. Red blood cell distribution width and the risk of death in middle-aged and older adults. Arch Intern Med 2009; 169: 515-523.
- Mandell LA, Wunderink RG, Anzueto A, Bartlett JG, Campbell GD, Dean NC, Dowell SF, File TM Jr, Musher DM, Niederman MS, Torres A, Whitney CG; Infectious Diseases Society of America; American Thoracic Society. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community acquired pneumonia in adults. Clin Infect Dis 2007; 44: 27-72.
- Lippi G, Targher G, Montagnana M, Salvagno GL, Zoppini G, Guidi GC. Relation between red blood cell distribution width and inflammatory biomarkers in a large cohort of unselected outpatients. Arch Pathol Lab Med 2009; 133: 628-632.
- Weiss G, Goodnough LT. Anemia of chronic disease. N Engl J Med 2005; 352: 1011-1023.
- Ershler WB, Sheng S, McKelvey J, Artz AS, Denduluri N, Tecson J, Taub DD, Brant LJ, Ferrucci L, Longo DL. Serum erythropoietin and aging: a longitudinal analysis. J Am Geriatr Soc. 2005; 53: 1360-1365.
- Katsoulis K, Kontakiotis T, Baltopoulos G, Kotsovili A, Legakis IN. Total antioxidant status and severity of community-acquired pneumonia: are they correlated? Respiration 2005; 72: 381-387.
- Braun E, Domany E, Kenig Y, Mazor Y, Makhoul BF, Azzam ZS. Elevated red cell distribution width predicts poor outcome in young patients with community acquired pneumonia. Crit Care 2011; 15: R194.
- Braun E, Kheir J, Mashiach T, Naffaa M, Azzam ZS. Is elevated red cell distribution width a prognostic predictor in adult patients with community acquired pneumonia? BMC Infect Dis 2014; 14: 129.
- Patel KV, Semba RD, Ferrucci L, Newman AB, Fried LP, Wallace RB, Bandinelli S, Phillips CS, Yu B, Connelly S, Shlipak MG, Chaves PH, Launer LJ, Ershler WB, Harris TB, Longo DL, Guralnik JM. Red cell distribution width and mortality in older adults: a meta-analysis. J Gerontol A Biol Sci Med Sci 2010; 65: 258-265.
- Martínez-Velilla N, Ibáñez B, Cambra K, Alonso-Renedo J. Age (Dordr). 2012; 34: 717-723.
- Urrutia A, Sacanella E, Mascaro J, Formiga F. Anemia in the elderly. Rev Esp Geriatr Gerontol 2010; 45: 291-297.
- Felker GM1, Allen LA, Pocock SJ, Shaw LK, McMurray JJ, Pfeffer MA, Swedberg K, Wang D, Yusuf S, Michelson EL, Granger CB; CHARM Investigators. Red cell distribution width as a novel prognostic marker in heart failure: data from the CHARM Program and the Duke Databank. J Am Coll Cardiol 2007; 50: 40-47.
- Allen LA, Felker GM, Mehra MR, Chiong JR, Dunlap SH, Ghali JK, Lenihan DJ, Oren RM, Wagoner LE, Schwartz TA, Adams KF. Validation and potential mechanisms of red cell distribution width as a prognostic marker in heart failure. J Card Fail 2010; 16: 230-238.
- Lippi G, Targher G, Montagnana M, Salvagno GL, Zoppini G, Guidi GC. Relationship between red blood cell distribution width and kidney function tests in a large cohort of unselected outpatients. Scand J Clin Lab Invest 2008; 68: 745-748.
- Dabbah S, Hammerman H, Markiewicz W, Aronson D. Relation between red cell distribution width and clinical outcomes after acute myocardial infarction. Am J Cardiol 2010; 105: 312-317.
- Sánchez-Chaparro MA, Calvo-Bonacho E, González-Quintela A, Cabrera M, Sáinz JC, Fernández-Labandera C, Aguado LQ, Meseguer AF, Valdivielso P, Román-García J; Ibermutuamur Cardiovascular Risk Assessment Study Group. Higher red blood cell distribution width is associated with the metabolic syndrome: results of the Ibermutuamur Cardiovascular risk assessment study. Diabetes Care 2010; 33: e40. 32.
- Öztürk ZA, Ünal A, Yiğiter R, Yesil Y, Kuyumcu ME, Neyal M, Kepekçi Y. Is increased red cell distribution width (RDW) indicating the inflammation in Alzheimer's disease (AD)? Arch Gerontol Geriatr 2013; 56: 50-54.