- Biomedical Research (2007) Volume 18, Issue 1
Influence of Experimental Diabetes Mellitus on Secretary Granules in ß-Cells in the Dog Pancreatic Islet
Yuji Asai1, Hiroyuki Morimoto1, Yoshio Mabuchi1, Eisuke Sakuma1, Nobuyuki Shirasawa3, Ikuo Wada2, Osamu Horiuchi2, Atsushi Sakamoto4, Chiharu Tanaka1, Damon C. Herbert5 and Tsuyoshi Soji1
1Functional Morphology, Nagoya City University Medical Science, 1 Kawasumi, Mizuho-cho, Mizuho-ku, Nagoya City, Aichi, 467-8601, Japan
2Musculoskeletal Medicine, Nagoya City University Medical Science, 1 Kawasumi, Mizuho-cho, Mizuho-ku, Nagoya City, Aichi, 467-8601, Japan
3Department of Anatomy and Structural Science, Yamagata University School of Medicine, 2-2-2 Iida-nishi, Yamagata 990-9585, Japan
4Division of Forensic Medicine, Center for the Community Medicine, Jichi Medical School, Tochigi 329-0498, Japan
5Department of Cellular and Structural Biology, Mail Code 7762, The University of Texas Health Science Center at San Antonio, San Antonio, Texas 78229-3900 USA
- Corresponding Author:
- Yuji Asai
Functional Morphology Nagoya City University Medical Science 1 Kawasumi
Mizuho-cho, Mizuho-ku Nagoya City, Aichi, 467-8607, Japan
Tel: +81-52-853-8121
Fax: 81-52-842-3210
E-mail: soji_t@med.nagoya-cu.ac.jp
Accepted Date: November 07, 2006
Abstract
The secretory granules of the β-cells (β-granules) in the pancreatic islets differ in shape in different animals. Whether β-granules are made entirely of insulin or not is unknown. Ten mixed-breed dogs were used and separated into two groups. One was a control group and the other served as those with experimental diabetes mellitus (DM) and received injections of Streptozotocin. After the injections, blood glucose was checked on day 14. The pancreas was removed after fixation by perfusion and post-fixed with Caulfield’s fixative. The specimens were then processed and observed under an transmission electron microscope (TEM). Immunocytochemistry using antibodies to insulin was also performed. The β-cells in the DM animals were reduced in size and displayed fewer electron dense granules. Additionally, almost all of the cell organelles had decreased in number Immunocytochemical observations of the control β-cells showed a strong positive reaction on the rodshaped and spherical granules. The number of rodshaped granules in control dogs ranged from 3299 to 3559, while in DM animals they varied from 374 to 602. The spherical granules in the controls numbered between 168 and 295 as compared to the DM dogs which ranged from 394 to 706 in each animal. The ratio of rodshaped and spherical granules in the controls was 5:1 and in the DM dogs 1:1. It was understood that the diminution in the total number of granules in the DM dogs was mainly due to a decrease of rodshaped granules. It is assumed that the rodshaped granule corresponded to proinsulin, the storage form of the insulin, and the spherical granules to the active form of insulin. However, there have been no reports discussing this relationship. The present study demonstrated that the βgranules, especially rodshaped granules decreased in DM animals, whereas the percentage of spherical granules remained the same or were even elevated compared to the normal, controls. Thus, it is believed that Streptozotocin mainly attacked the pathway where preproinsulin is converted to proinsulin.
Keywords
Dog pancreas, diabetes mellitus, β-cell, β-granule, ultrastructure
Introduction
The secretory granules of β-cells (β-granules) in the pancreatic islets are of different shape in different animals. Those of rabbits, rats, and mice are mostly round, while those of humans, dogs, bats, and snakes are slender rod, hexagonal, or round in the same animals/individuals [1]. Whether β-granules are composed entirely of insulin or not is not known. Kvistberg et al [2] reported that rod-shaped β-granules were closely related to insulin. The relationship between rod-shaped granules and other shapes of β-granules is also unknown. A morphological characteristic of β-granules in the dog pancreatic islet is their conspicuous rod shape. Although there are numerous studies using an ultra-high-voltage electron microscope in which the three-dimensional structure of rod-shaped or polygonal β-granules of the yellowtail, mouse, and snake have been noted [3, 4], we cannot find a report describing the functional difference between rod-shaped and spherical granules in the dog pancreatic β-cell. We also failed to find a report to analyze rod-shaped β-granules statistically. It has been suggested that the morphology of the granules in the dog may signal certain physiological characteristics implying that the rod-shaped granules reflect a different function from those that are round or spherical. In the present report, we present results from the statistical analyses of the rod-shaped β-granules using electron-microscopic serial sections and show a possible function of these granules using of experimental diabetes mellitus (DM).
Materials and Methods
Ten mixed breed dogs weighing 1.5 5 to 2.0 kg were employed in this study and separated into two groups. The all animals were housed in 12h light and 12h dark. The animals were fed and given water at ad libitum. One was the control group which were injected physiological saline, and the other was for the one shot injection of experimental DM using Streptozotocin [2-deoxy-2-(methyl-3-nitosoureido)-D-glucopyranose]; 35mg/Kg of dog] at sub cutaneous. After the injection of Streptozotocin, which is known to damage and/or destroy pancreatic beta cells [5], blood glucose was checked on day 14. The ex-erimental animals were selected if their blood glucose concentrations were over 300mg/dl. Since four animals of the initial five animals the received Streptozotocin did not have blood glucose concentrations above 300mg/dl, an additional 24 were treated to obtain the five experimental animals. All animals were treated according to “The Guidelines for Animal Experimentation” of the Experimental Animal Science Center of the Nagoya City University and were sacrificed under deep anesthetized with Nembutal (pentobarbital, Abbot Lab., North Chicago, IL, U.S.A.).
The pancreases of both control and experiment dogs were fixed for electron microscopic analysis. The thorax of the animals was opened under Nembutal anesthesia, a catheter inserted into the left ventricle of the heart and the fixative (0.05 M cacodylate-buffered 2.5% glutaraldehyde, pH 7.2) infused via the aorta for 30 min [6]. The pancreas from each animal was removed, cut into cubes of about 1 mm3, and post fixed with Caulfield’s fixative [7]. The tissues were then dehydrated in increasing concentrations of ethanol, embedded in epoxy resin [8], cut into ultra-thin serial sections (about 80 nm thick) and observed under an H-500 and H-7000 transmission electron microscope (TEM).
Immunocytochemistry for the identification of intracellular insulin was also performed. The antibody of insulin and colloidal gold were commercially obtainned (insulin: 10ug/ml, mouse anti-human insulin. The solution was adjusted for the pathological use, Biomeda Co., USA and colloidal gold: EY Laboratories, Inc., USA). Anti-insulin was used at 1:1, and reacted 8 h. at room temperature and then after 3 min of rinse of PBS, the specimens were reacted the colloidal gold (15nm in diameter) diluted at 1:50 for 30 min. in room temperature. Then the specimens were stained by uranyl acetate and lead citrate. The specificity of the immunocytochemical reaction was checked by substitution of the antibody by PBS. No specific reactions were observed in the control specimens for insulin.
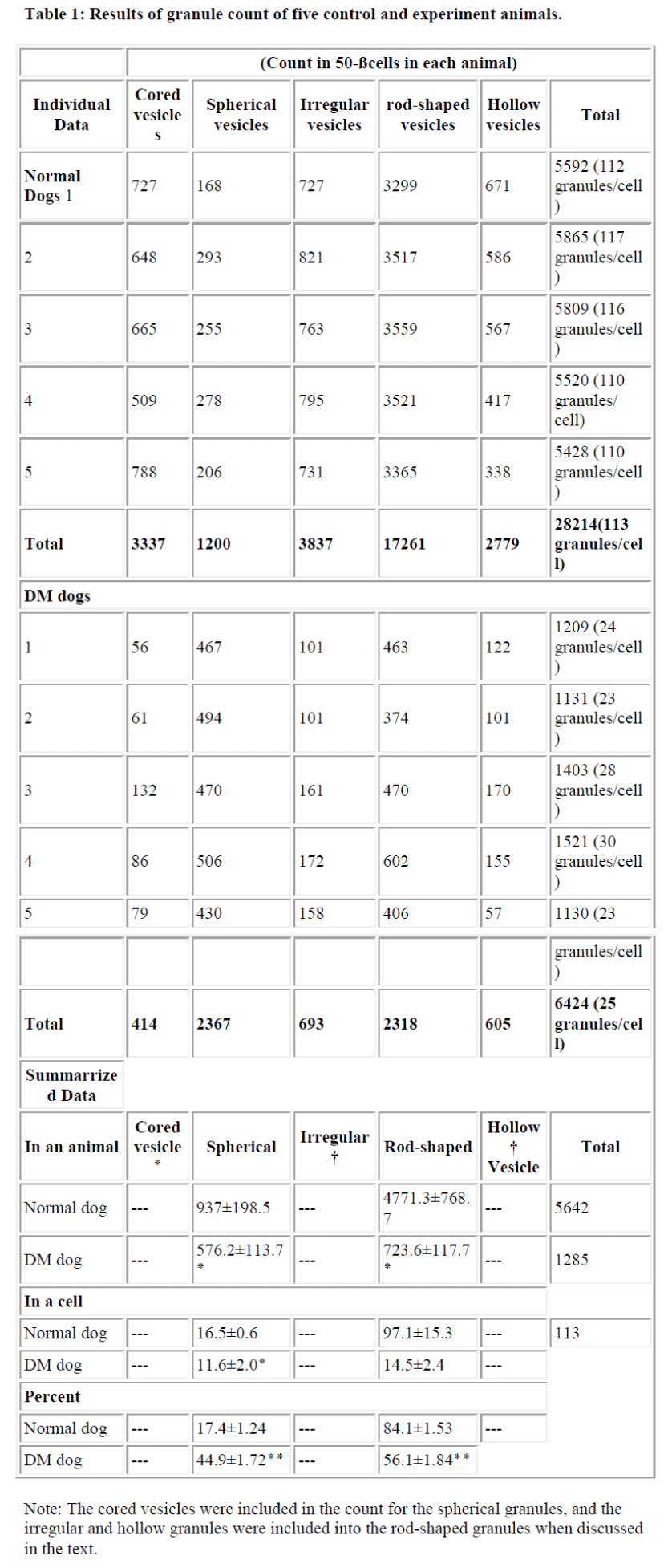
Each set of data consisted of six electron microscopic specimens. To confirm the granule type, each was identified using serial sections and measured as follows: From each of the 30 sets of six serial specimens, six electron micrographs (x3,000 magnification) were obtained. A total of 50 β-cells which displayed nuclei were selected and the granules counted. The total granule number in the control islets varied about from 4630 to 5862 compared with the DM islet which varied from1011 to 1721. The data obtained were then analyzed statistically by the Student t-test.
Results
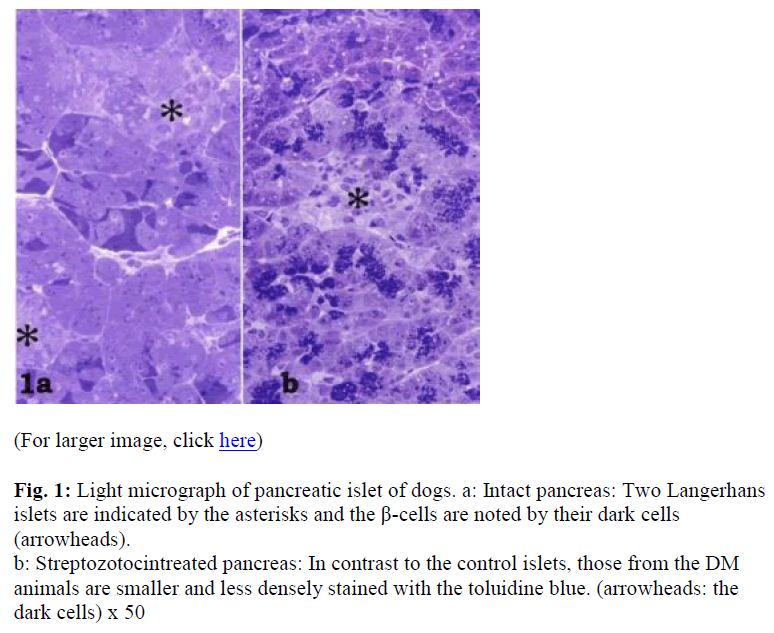
Though the pancreatic islets were easily identified in the controls (Fig. 1a), they were somewhat more difficult to locate in the experimental animals (Fig. 1b). The islets in the DM dogs were smaller and showed a lower distribution density (Figs. 1a, b). In the control islets, toluidine blue-dense cells were observed especially in the central area (Fig. 1a). The profile of the DM dog islets indicated the presence of fewer secretory granules in the endocrine β-cells, since few toluidin blue-dense cells were noted (Fig. 1b).
Figure 1: Light micrograph of pancreatic islet of dogs. a: Intact pancreas: Two Langerhans islets are indicated by the asterisks and the β-cells are noted by their dark cells (arrowheads).
b: Streptozotocintreated pancreas: In contrast to the control islets, those from the DM animals are smaller and less densely stained with the toluidine blue. (arrowheads: the dark cells) x 50
The secretary granules of dog pancreatic β-cells were categorized as being of five types; cored vesicles, spherical granules, irregular granules, hollow and rod-shaped granules. In this study, the irregular-shaped and hollow granules were included in the category of rod-shaped and the cored in the category termed spherical.
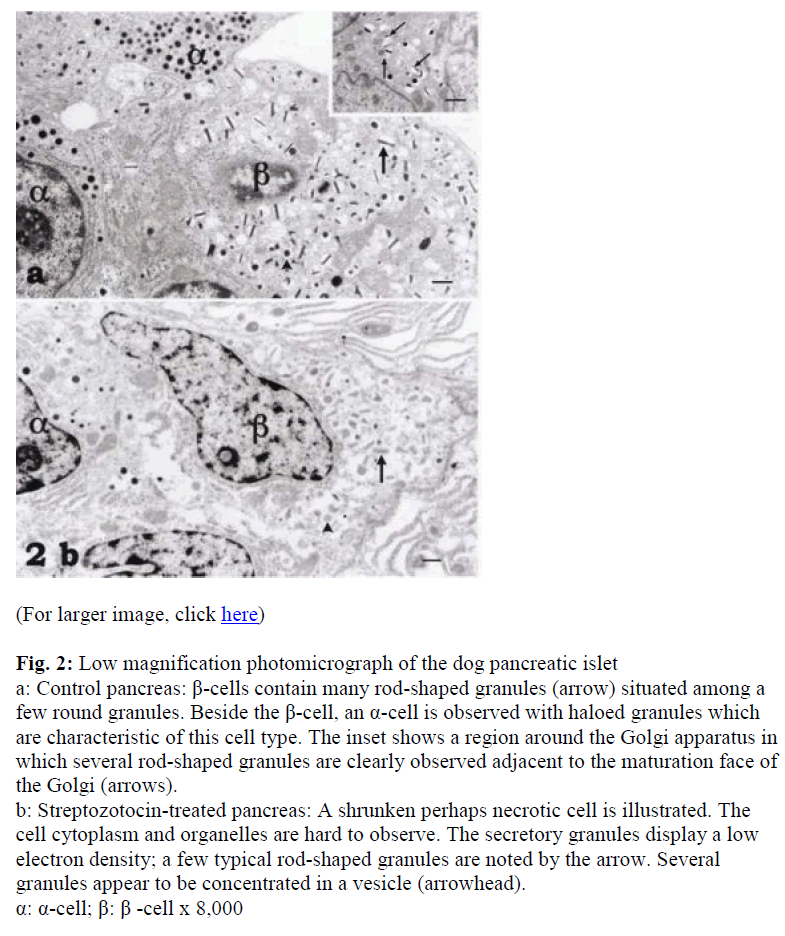
The β-cells of the dog pancreatic islets displayed many rod-shaped secretory granules with a few that were spherical, so identification of the cell was not difficult (Fig. 2a). These granules were mainly located around a well-developed Golgi apparatus (Fig. 2a). Although the β-granules in the pancreas of the control dogs showed high electron density, they were less dense and fewer in the DM animals (Fig. 2b). A few oval mitochondria were also scattered in the cytoplasm (Figs. 2a, b).
Figure 2: Low magnification photomicrograph of the dog pancreatic islet a: Control pancreas: β-cells contain many rod-shaped granules (arrow) situated among a few round granules. Beside the β-cell, an α-cell is observed with haloed granules which are characteristic of this cell type. The inset shows a region around the Golgi apparatus in which several rod-shaped granules are clearly observed adjacent to the maturation face of the Golgi (arrows).
b: Streptozotocin-treated pancreas: A shrunken perhaps necrotic cell is illustrated. The cell cytoplasm and organelles are hard to observe. The secretory granules display a low electron density; a few typical rod-shaped granules are noted by the arrow. Several granules appear to be concentrated in a vesicle (arrowhead). α: α-cell; β: β -cell x 8,000
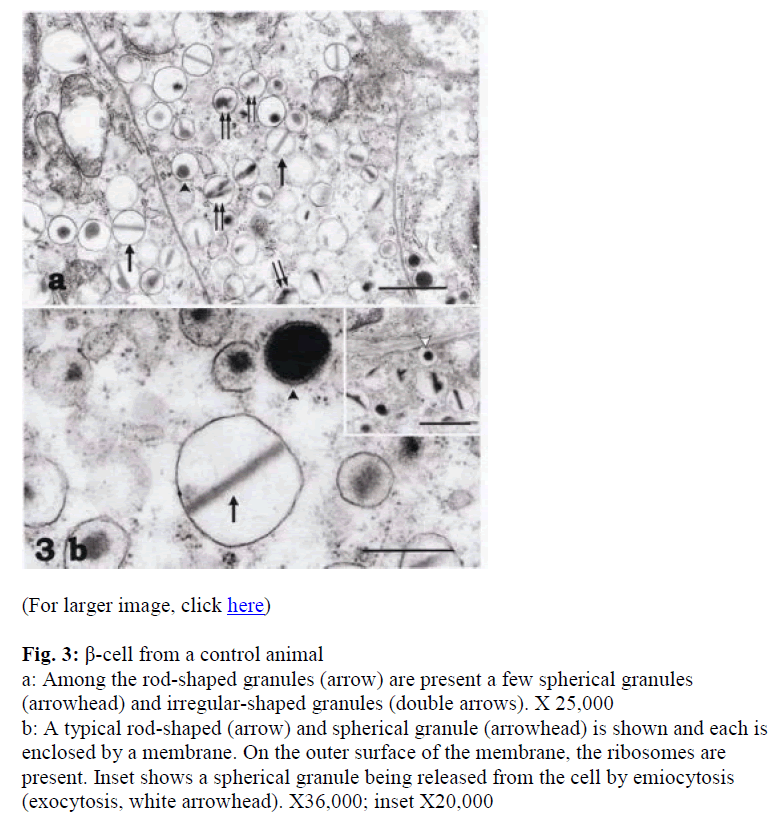
In the β-cells of the control dogs, the spherical, rod-shaped, and irregular-shaped granules were compactly arranged in the cells. The rod-shaped granules were the most numerous of the granule type (Fig. 3a). Scattered among these were a few spherical (arrowheads in Figs. 3a, b) and irregular-shaped granules (double arrows in Fig. 3a). A typical rod-shaped granule (arrow) and the spheri-cal granules with or without halos (arrowhead) are shown in Figure 3b enclosed by a membrane whose outer surface was often studded with ribosomes (Fig. 3b). Besides β-cells, α-cells were also observed with haloed granules (Fig. 3a).
Figure 3: β-cell from a control animal a: Among the rod-shaped granules (arrow) are present a few spherical granules (arrowhead) and irregular-shaped granules (double arrows). X 25,000
b: A typical rod-shaped (arrow) and spherical granule (arrowhead) is shown and each is enclosed by a membrane. On the outer surface of the membrane, the ribosomes are present. Inset shows a spherical granule being released from the cell by emiocytosis (exocytosis, white arrowhead). X36,000; inset X20,000
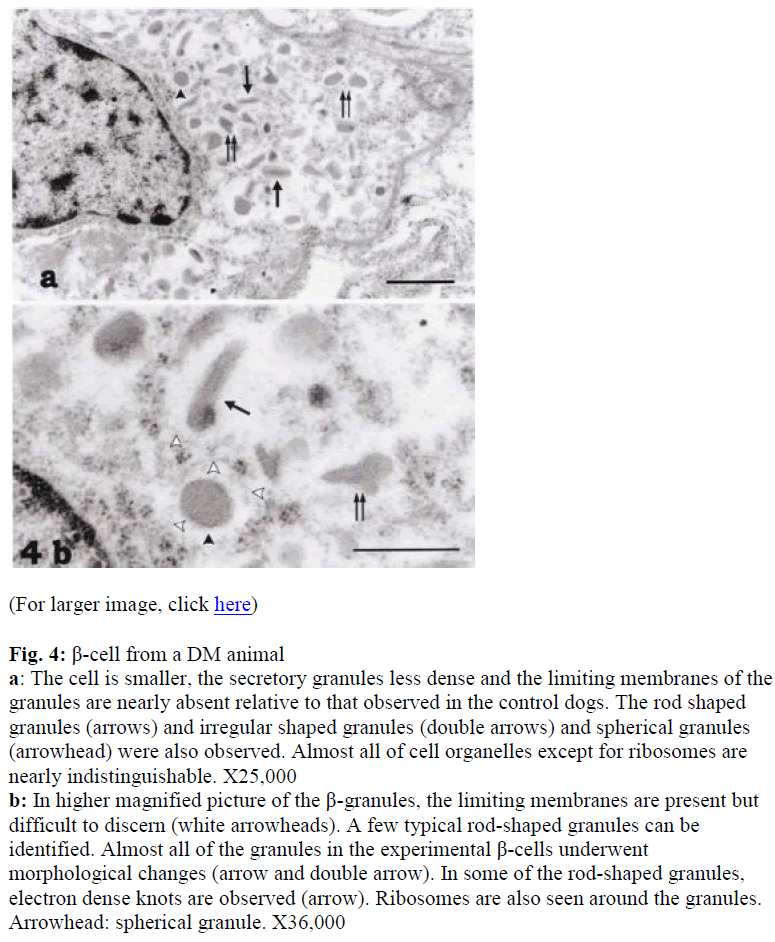
Additionally, almost all of the cell organelles had decreased in number. Few typical rod-shaped granules were observed. Almost all of the granules in the experimental β-cells suffered from the drug treatment (Figs. 4a, b). Although the granular membranes were not clear, studded ribosomes were obseved around the granules in the DM β-cells (Fig. 4). Compared with the control β-cells, the ratio of irregular type granules to the total granules in the immunoreactivity of the DM β-cells to insulin was weaker than the control β-cells. The positive sites DM animals increased (Fig. 4a).
Figure 4: β-cell from a DM animal a: The cell is smaller, the secretory granules less dense and the limiting membranes of the granules are nearly absent relative to that observed in the control dogs. The rod shaped granules (arrows) and irregular shaped granules (double arrows) and spherical granules (arrowhead) were also observed. Almost all of cell organelles except for ribosomes are nearly indistinguishable. X25,000
b: In higher magnified picture of the β-granules, the limiting membranes are present but difficult to discern (white arrowheads). A few typical rod-shaped granules can be identified. Almost all of the granules in the experimental β-cells underwent morphological changes (arrow and double arrow). In some of the rod-shaped granules, electron dense knots are observed (arrow). Ribosomes are also seen around the granules. Arrowhead: spherical granule. X36,000
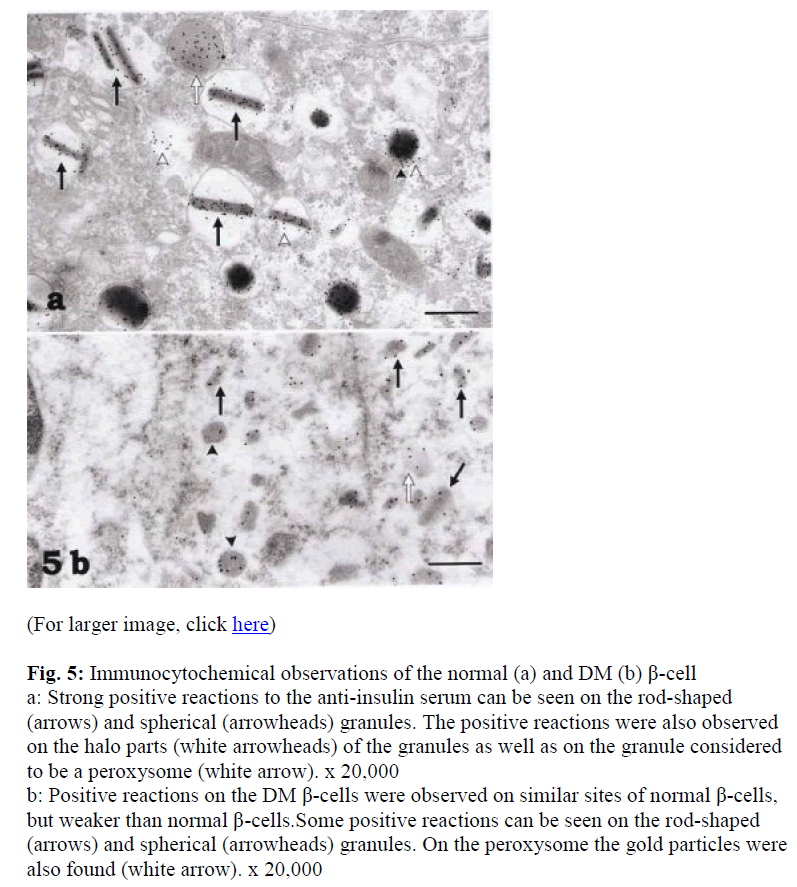
Immunocytochemical observations of the control cells using antibodies to insulin showed strong positive reactions on the rod-shaped (arrows) and spherical (arrow-heads) granules. Positive reactions were also present on the halo parts (white arrowheads) of the granules (Fig. 5a) as well as on structures considered to be peroxisomes (white arrow in Fig. 5a). Immunoreactivity of the DM β-cells to insulin was weaker than the control β-cells. The positive sites were, however, observed on similar parts of control β-cells such as on rod-shaped (arrows) and spherical (arrowheads) granules (Fig 5b).
Figure 5: Immunocytochemical observations of the normal (a) and DM (b) β-cell a: Strong positive reactions to the anti-insulin serum can be seen on the rod-shaped (arrows) and spherical (arrowheads) granules. The positive reactions were also observed on the halo parts (white arrowheads) of the granules as well as on the granule considered to be a peroxysome (white arrow). x 20,000
b: Positive reactions on the DM β-cells were observed on similar sites of normal β-cells, but weaker than normal β-cells.Some positive reactions can be seen on the rod-shaped (arrows) and spherical (arrowheads) granules. On the peroxysome the gold particles were also found (white arrow). x 20,000
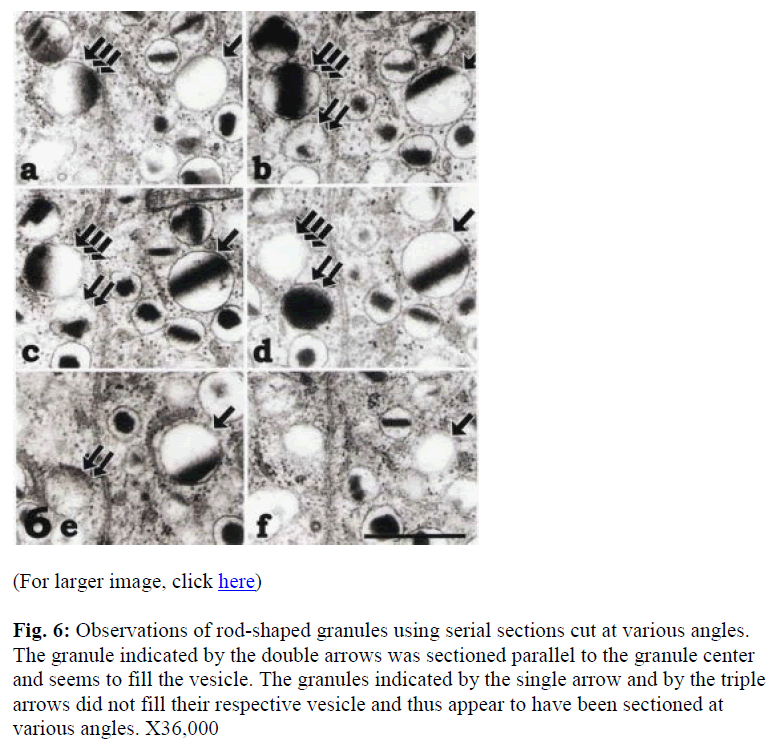
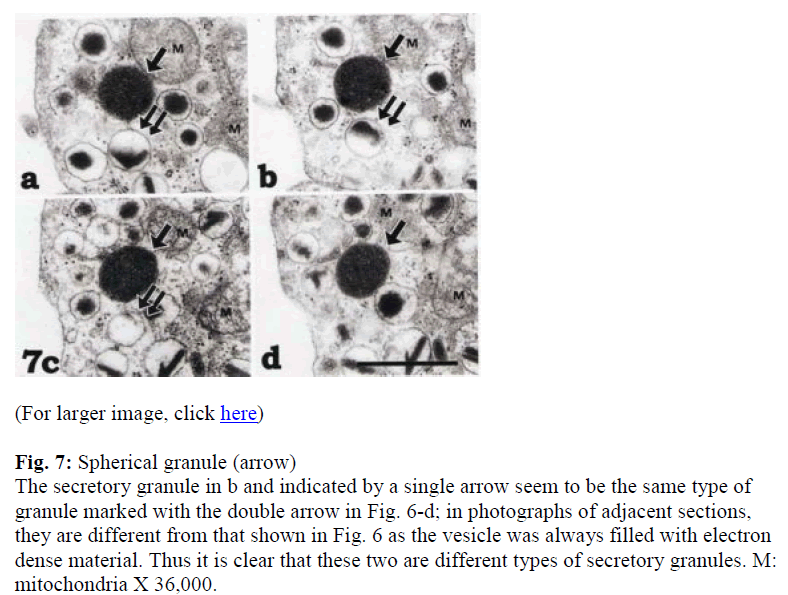
To distinguish between the peroxisome-like granule, spherical granules or tangential sections of the rod-shaped granules, serial sections were employed (Figs. 6 and 7). Although the granules indicated by single and triple arrows were easily distinguished as rod-shaped, the granule indicated by the double arrow in Figure 6d was once identified as spherical. However, serial sections indicated that it was rod-shaped (Fig. 6b-e). On the other hand, Figure 7 illustrates a spherical granule, which is indicated by a single arrow. The four micrographs in Figure 7 show serial sections through a secretory granule as indicated by single arrows which was categorized as being spherical while the granule indicated by double arrow in figure 7a-c was rod-shaped.
Figure 6: Observations of rod-shaped granules using serial sections cut at various angles. The granule indicated by the double arrows was sectioned parallel to the granule center and seems to fill the vesicle. The granules indicated by the single arrow and by the triple arrows did not fill their respective vesicle and thus appear to have been sectioned at various angles. X36,000
Figure 7: Spherical granule (arrow)
The secretory granule in b and indicated by a single arrow seem to be the same type of granule marked with the double arrow in Fig. 6-d; in photographs of adjacent sections, they are different from that shown in Fig. 6 as the vesicle was always filled with electron dense material. Thus it is clear that these two are different types of secretory granules. M: mitochondria X 36,000.
As mentioned above, the β-cells in the experiment animals were reduced in size and in electron density of their granules (Fig. 4a); the limiting membranes of the granules were very difficult to identify (Figs. 4a, b).
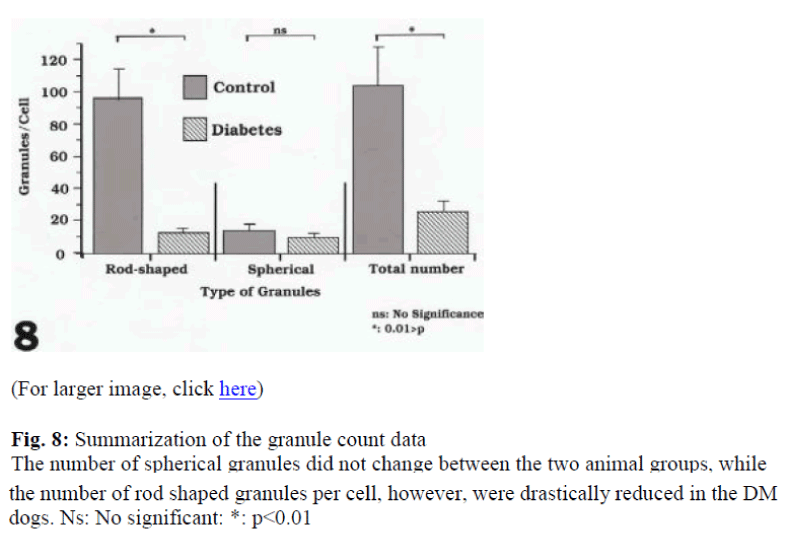
The granules classified as rod-shaped, irregular-type, spherical, cored and hollow were counted. The number of granules in the β-cells were counted in 50 cells in each animal as summarized in Table 1. The total granule number per cell in the control dogs ranged from 109 to 117 granules (5520 to 5862 granules were recorded in the 50 cells counted) and in the DM dogs they ranged 23 to 30 granules per cell, with a total of 1130 to 1521 in the 50 cells counted. Clearly, the granule number in the experimental animals was drastically decreased. Besides the rod- shaped granules in the control dogs ranging from 3299 to 3559 and in DM dogs from 374 to 602, the spherical granules in the control and experimental animals varied from 168 to 295 and 394 to 706, respectively. In the control dogs, the ratio of rod-shaped to spherical granules was 5:1, while in the drug treated animals, the ratio was 1:1. It was felt that the diminution of in the total granule numbers in the DM dogs was mainly due to a decrease in the frequency of rod-shaped granules (Fig. 8).
Number of the Granules in the β-cells of the Normal and DM Dog
Discussion
β-cells of the pancreatic islets of various animals have been extensively studied [3,4,9,10,11,12]. Watari [4], in particular, observed these granules in detail using an ultra-high-voltage electron microscope, and classified them into 10 types. He reported that the three-dimensional structure of the granules in the snake contains hexagonal crystalline cores shaped like a rhombic dodecahedron. Electron microscopic study of a hexagon should give a variety of shapes if the rhombic dodecahedron is sectioned at various angles. However, in our observation of the normal dog pancreatic islets, such a variety was not seen. Most of the granules were not polygonal but were slender rods, such as pentagons or hexagons. The rod-shaped granules of the dog had more circular electron microscopic profiles depending on the angle of the section.
It is generally known that the snake pancreatic β-cell contains conspicuous rod-shaped granules [3, 12] which often show a crystalline structure. In general, crystalline structures have a certain hexagonal [13] or rectangular profile [14] or are rod shaped. In our study the rod-shaped granules contained in the dog β-cell showed a crystalloid structure which was interpreted as representing stability and purity of the molecule. It was believed that the crystals were a storage form of a certain protein [13] such as proinsulin such as in the dog, snake or human.
To address a relationship between the rod-shaped and spherical granules in the dog, the current investigators studied the ultrastructure of the DM dog pancreas. The present investigation clearly showed that the pancreatic β-granules especially rod-shaped granule decreased drastically in DM dog. It is commonly accepted that the direct perfusion of the pancreas of an experimental animal with glucose results in degranulation of β-cells [15]. Lacy and his co-workers [9,16,17] and Orci et al. [18] reported that the emiocytosis (exocytosis) is the major and probably the only mechanism of insulin secretion. In dog, there were roughly categorized two types of granule (spherical and rod-shaped). Exocytosis in the β-cell of rodents was demonstrated [19] where all of the granules were spherical. We did not observe the exocytosis of rod-shaped granule in this study. We therefore suggest that these granules might be a storage form of insulin, not destined for exocytosis.
In DM dog β-cells, the granules were drastically decreased, especially those that were rod-shaped. It is commonly accepted that insulin is synthesized first as preproinsulin, then proinsulin and finally insulin. It was not clear that this insulin family of molecules correspond to any of the various shaped granules in the dog or human such as those that are spherical or rod-shaped. Preproinsulin is synthesized in the rough endoplasmic reticulum and then converted to proinsulin in the Golgi apparatus. Subsequently, proinsulin is converted to the insulin again in Influence of Diabetes Mellitus on β-Cells the Golgi apparatus and discharged from the cell [19]. Knowing that the rod-shaped granules were crystalline, it is rather reasonable to presume these granules corresponded to proinsulin, the storage form of the hormone, and the spherical granule correspond to active insulin. However, there are no reports discussing or confirming this relationship.
While the number of secretory granules decreased in the DM cells, treatment with Streptozotocin also appeared to induce destruction of the pancreatic cell itself. Electron microscopic observation of the experimental cells clearly showed abnormalities such as reduced cell size, and decreased electron density and numbers of the granules. It was clear that the β-cell activity or function was extremely compromised in the experimental animals. The present study revealed that in the DM animals the total number of granules was approximately one fifth of that found in the normal animals, and that the number of rod-shaped granules decreased to one tenth of that in the controls. However, an increased percentage of spherical granules in DM animals in relation to the controls has been shown. On average 5642 granules were counted in a control animal and 1285 granules in a DM dog.
As mentioned above, the rod-shaped granule appeared to correspond to proinsulin the storage form of the insulin, and the spherical to the active form of insulin. The pathway of synthesis of preproinsulin was working in the DM dogs because studded ribosomes were preserved. It is still unclear where Streptozotocin acts in the insulin pathway. Possible points range from nuclear DNA to the release of insulin [20]. After the treatment of Streptozotocin, some β-cells become necrotic, including those in certain tumors such as inslinoma [20]. In conclusion, from morphological data it is postulated that Streptozotocin mainly acts on the pathway from preproinsulin to proinsulin.
References
- Herman L, Satao T, Fitzgerald PJ. The pancreas. In: Electron Microscopic Anatomy. Kurtz SM (ed.) Chapter 3 Academic press, New York, pp 59-95, 1964.
- Kvistberg D, Lester A, Lazarow A: Staining of insulin with aldehyde fuchsin. J Histochem Cytochem 1966; 14: 609-611.
- Watari N, Hotta Y, Mabuchi Y. Morphological studies on vitamin A-strong cell and its complex with macrophage observed in mouse pancreatic tissue following excess vitamin A administration. Okajima Folia Anat Jpn, 1982, 58: 837-858.
- Watari N. Crystalline structures observed in both the exocrine and endocrine pancreatic cells. In: Recent Progress in Electron Microscope of Cell and Tissue. Yamada E, Mizuhira V, Kurosumi K, Nagano T (ed), Igaku Shoin Ltd. Tokyo, pp 109、1976.
- Elliott WC, Houghton DC, Gilbert DN, Baines-Hunter J, Bennet WM. Experimental gentamicin nephrotoxicity: effect of of Streptozotocin-induced diabetes. J Phaaol Exp Ther 1985; 233: 264-270
- Soji T, Herbert DC. Intercellular communication between rat anterior pituitary cells. Anat Rec 1989; 224: 523-533.
- Caulfield J.B. Effect of varying the vehicle for OsO4 in tissue fixation. J. Biophys. Biochem. Cytol 1957; 3: 827-833.
- Luft JH. Improvement in epoxy resin embedding methods. J Biophys Biochem Cytol 1961; 9: 409-414.
- Lacy PF. Electron microscopic identification of different cell type in the islets of Langerhans of the ginea pig, rat rabbit and dog. Anat Rec 1957; 128: 255-267.
- Howell SL, Kostianovsky M, Lacy PF. A study by electron microscopic radioaurtography. J Cell Biol 1969; 42: 695.
- Misugi K, Howell SL, Greider MH, Lacy PF, Sorenson GD. The pancreatic beta cell. Demonstration with peroxidase labeled antibody technique. Arch Pthol 1970; 89: 97-102.
- Fujita T, Takamatsu K, Tokunaga J. Pancreatic Islets. In: SEM Atlas of Cells and Tissues. Igaku Shoin, Tokyo, pp 306-310, 1981.
- Fawcett, DW. Cytoplasmic inclusions: In: The Cell, second edition. W.B. Saunders Company, Philadelphia, pp 668-685, 1981.
- Soji T, Fujita T, Yoshimura F. Crystals in Hyperfunctioning rat parathyroid cells. Endocrinol Japon 1974; 21: 551-553.
- Brown EM Jr, Dohan FC, Freedman LR, De Moor P, Jukens FD. The effect of prolonged infusion of the dog’s pancreas with glucose. Endocrinology 1952; 50:644-656.
- Lacy PF. Beta cell secretion from the standpoint of pathobiologist. Diabetes 1970; 19: 895-905.
- Lacy PF: Endocrine secretory mechanisms. A review. Am J Pathol 1975; 79: 170-187.
- Orci L, Amherdt M, Malaisse-Lagae F, Rouiller C. Renold AE. Insulin release by emiocytosis: demonstration with freeze-etching technique. Science 1973; 179: 82-84.
- Bonquis t L. Fine structure of the endocrine pancreas in newborn rodents. Diabetes 1972; 21:1051-1059.
- Chang AY, Diani AR. Chemically and hormonally induced Diabetes Mellitus. In: The Diabetic Pancreas Ed: Volk BW and Arquilla ER, chapter 19, pp 415-422, Prenum Medical Book Company, NY and London, 1985.