- Biomedical Research (2011) Volume 22, Issue 2
Expression of vascular endothelial growth factor on neovascularization during experimental tooth movement by magnets
Toshitsugu Kawata*, Shinya Kohno, Masato Kaku, Tadashi Fujita, Junji Ohtani, Msahide Motokawa and Kazuo Tanne
Department of Orthodontics and Craniofacial Developmental Biology, Graduate School of Biomedical Sciences, Hiroshima University, 1-2-3 Kasumi, Minami-ku, Hiroshima 734-8553, Japan
- *Corresponding Author
- Toshitsugu Kawata
Department of Orthodontics and
Craniofacial Developmental Biology
Graduate School of Biomedical Sciences
Hiroshima University, 1-2-3 Kasumi
Minami-ku, Hiroshima 734-8553
Japan
Tel: +81-82-257-5686
Fax: +81-82-257-5687
Email: tenzan@hiroshima-u.ac.jp
Accepted date: January 07 2011
Abstract
Vascular endothelial growth factor (VEGF) can induce neovascularization. We recent found that the number of osteoclasts was enhanced of VEGF with the application of mechanical force for experimental tooth movement. In this study, the VEGF was detected in osteoblasts and periodontal tissues on the tension and pressure side of the alveolar bone. Moreover, the neovascularization was significantly increased in the tooth movement by magnet groups compared with the control. These results suggest that VEGF, highly expressed by mechanical stimuli, enhances the number of neovascularizations as the paracrine factor is accelerated by magnetic force.
Keywords
Vascular endothelial growth factor (VEGF), Osteoclasts, Magnetic force, Tooth movement
Introduction
Tooth movement in orthodontic treatment is achieved by a combination of mechanical and physiologic forces acting on the teeth and their supporting structures to effect the biologic changes that cause teeth to move through bone. Othodontic tooth movement is achieved by a repeated process of alveolar bone resorption on the pressure side and new bone formation on the tension side [1,2]. Originally, the orthodontic forces to teeth were the orthodontic wires. Orthodontics with the magnet was tried by various researchers in the 1980's [3,4]. Generally, orthodontic treatment with the magnet is few pains in teeth. As the reason, it was also reported that osteoblasts produce VEGF, and it is well-understood from these findings that experimental tooth movement is a useful in vivo model for investigating the expression and role of VEGF in the periodontal tissues with active remodeling [5,6,7].
Vascular endothelial growth factor (VEGF) is the most important and essential mediator for angiogenesis [8]. It was reported that VEGF expression was detected in the vascularization process of developing embryos [9], and the pathophysiologic process of inflammation and wound healing [10]. The specific receptors for VEGF, two receptor tyosine kinases, fms-like tyrosine kinase (Flt-1) and fetal liver kinase (Flk-1), have been identified [11,12,13,14]. Our recent study [15] demonstrated that osteoclasts markedly cxpressed Flt-1, and that recombinant human (rh)VEGF (AQ) induced many osteoclasts in op/op mice.
We conducted this study to investigate the expression of VEGF and neovascularization within periodontal tissues during experimental tooth movement using an immunohistochemical approach.
Material and Methods
C57BL/6J mice, each weighing about 15 g, were obtained from Jackson Laboratory (Bar Harbor, ME, USA) for use in this experiment. All animals wrere treated under ethical regulations for animal experiments, defined by the Ethics Committee, Hiroshima University Faculty of Dentistry.
The animals were fed a granular diet (CA1; Japan Clea, Tokyo, Japan) to prevent the influence of masticatory forces on the incisors. They were divided into three groups and an experimental appliance for tooth movement exerted an initial force of 1.0g applied in the distal direction (Figure 1A and B).
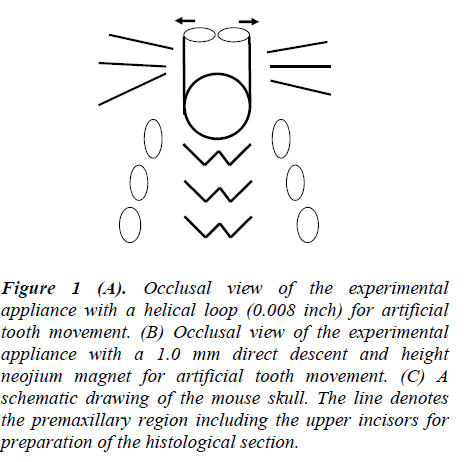
Figure 1: (A). Occlusal view of the experimental appliance with a helical loop (0.008 inch) for artificial tooth movement. (B) Occlusal view of the experimental appliance with a 1.0 mm direct descent and height neojium magnet for artificial tooth movement. (C) A schematic drawing of the mouse skull. The line denotes the premaxillary region including the upper incisors for preparation of the histological section.
Expression of VEGF of Experimental Tooth Movement
First, 30-day-old mice were divided into three groups. The first group served as the controls (n=5). The second group, upper Incisors were the initial experimental tooth movement using a wire (n=5). In the third group, upper Incisor were beginning of experimental tooth movement using magnets (n=5). An experimental wire appliance with a helical loop was made of 0.008-inch round stainless steel wire (3M Unitek, Monrovia, CA, USA). The wire appliance was bonded onto the lateral surfaces of the upper incisors (Figure 1A) In the present study, new magnetized orthodontic brackets were designed. The magnet works to move teeth distally and is not merely a mechanical force. It is well adapted to the biophysical system of tooth and bone interrelationships. The experimental magnet appliance was a 1.0 mm direct descent and height neojium magnet (Hitachi metals, Tokyo, Japan). The magnetic flux density of this magnet was a 150 m tesla. The magnet appliance was bonded onto the frontal surfaces of the upper incisors (Figure 1B). The force magnitudes were calibrated by a tension gauge (Shinpo Cprp., Tokyo, Japan) to exert an initial force of 1.0 g applied in the distal direction. Experimental tooth movement was continued for 14 days in the three groups. The distances between the left and right appliances’ tips, bonded onto the upper incisors, were measured every three days on dorsovetral cephalograms. A rat-and-mouse cephalometric X-ray apparatus (Asashi Roentgen Ind. Co., Kyoto, Japan) was used at 20-25 kV and 6mA with an exposure time of 30. Sec on Kodak Dental Ultra-speed film (Eastman Kodak Co., Rochester, NY, USA). We measured the width of the midpalatal suture every three days on the frontal sections (n=5) and subtracted it from the distance of tooth movement according to a previous study [16].
On days 3, 7, 14 and 21 after initiating the experiment, we investigated the changes in neovascularization number in the three groups (n=5). The neovascularization number that appeared in the PDL space on the pressure side of the incisors was counted on 5 sections at 35 μm intervals for each specimen.
Histological Examination
The mice were killed under general anesthesia with sodium pentobarbital, fixed in 4% paraformaldehyde, and rinsed in distilled water. The specimens were then decalcified in 14% EDTA (pH 7.4) for 14 days and embedded in paraffin. The premaxillary bone, including the upper incisors, were cut into frontal sections of 7μm thickness (Figure 1C). The expression of VEGF on the tension and pressure were examined on sections immunohistochemically stained with a rabbit anti-mouse VEGF polyclonal antibody (LAB VISION, Vector Laboratories, Burlingame. CA, USA), and counterstained with methyl green. Normal rabbit IgG (Santa Cruz Biotechnology, Santa Cruz, CA, USA) was used as the control for the polyclonal antibody. The neovascularization number in a 100 X 3600μm2 area of the PDL space on the tension and pressure side of the incisor was counted in 5 sections at 35μm intervals for each specimen.
Statistical Analysis
To examine differences in the neovascularization number and the amount of tooth movement among multiple groups, we performed analysis of variance (ANOVA) and multiplecomparison tests (Fisher) using Statview (Abacus Concepts, Inc., Berkeley, CA, USA)
Results
Expression of VEGF
Figure 1 and 2 shows the expression of VEGF within the periodontal tissues 10 days after the experimental tooth movement was initiated.
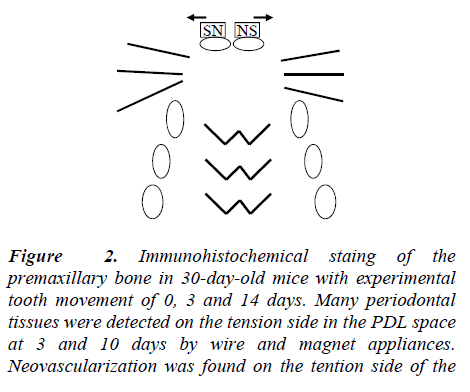
Many osteoblasts were detected on the tension and pressure side in the PDL space. In the osteoblasts, VEGF was highly expressed, as noted by the intense immunostaining with the VEGF antibody (Figure 2 and 3).
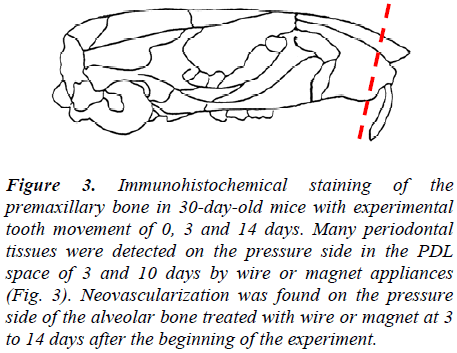
Meanwhile, no immunoreactivity was detected in the negative control (Figure 2 and 3)
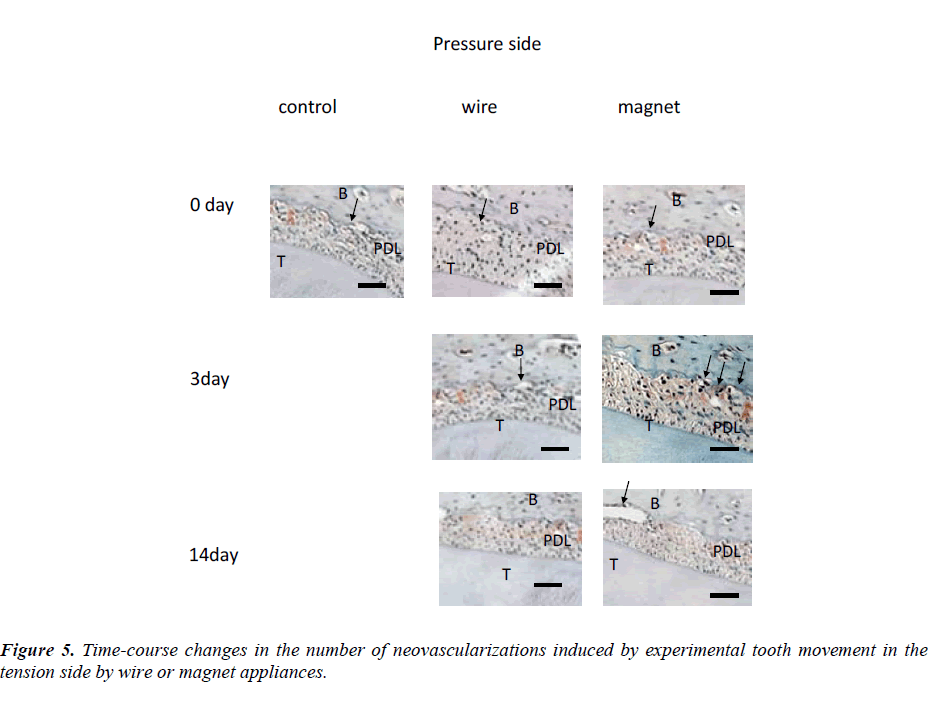
Figure 2:Immunohistochemical staing of the premaxillary bone in 30-day-old mice with experimental tooth movement of 0, 3 and 14 days. Many periodontal tissues were detected on the tension side in the PDL space at 3 and 10 days by wire and magnet appliances. Neovascularization was found on the tention side of the Orthodontic treatment and VEGF.
Figure 3: Immunohistochemical staining of the premaxillary bone in 30-day-old mice with experimental tooth movement of 0, 3 and 14 days. Many periodontal tissues were detected on the pressure side in the PDL space of 3 and 10 days by wire or magnet appliances (Fig. 3). Neovascularization was found on the pressure side of the alveolar bone treated with wire or magnet at 3 to 14 days after the beginning of the experiment.
Changes in the Neovascularization Number and the Amount of Tooth Movement
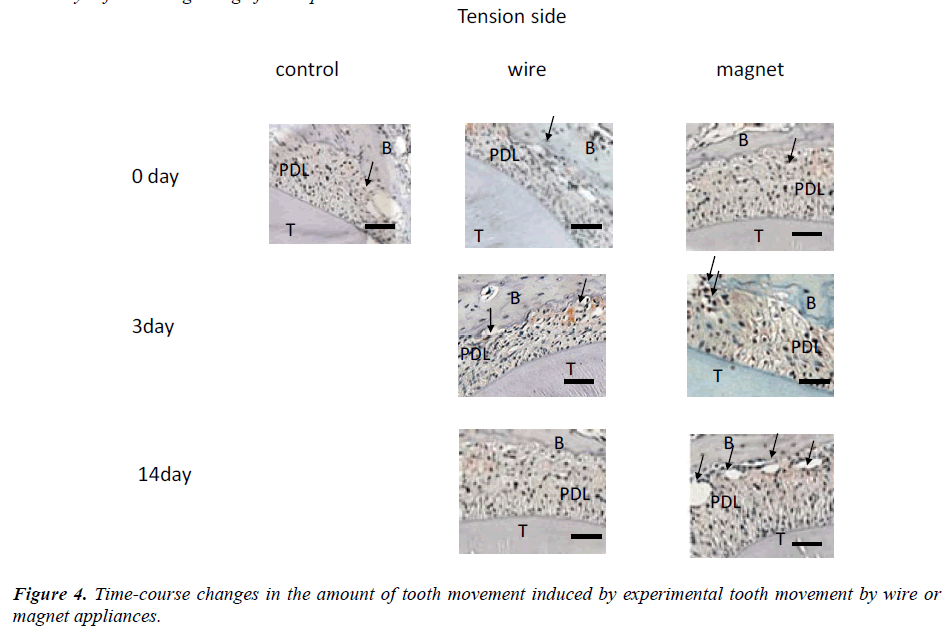
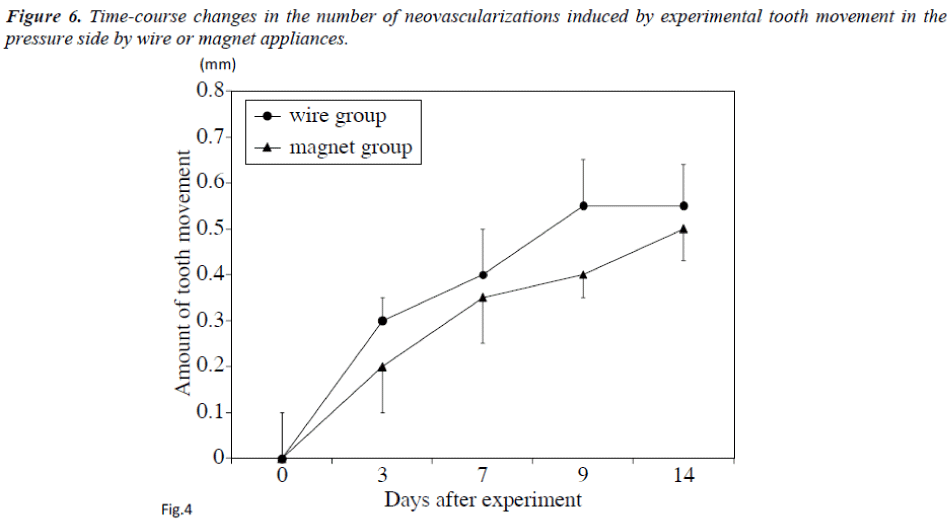
Changes in the amount of tooth movement every three days (Figure 4) and changes in the numbers of neovascularization on days 3, 7, 9 and 14 are shown in Figure 5.
An average of 6 neovascularizations were found on the pressure side of the alveolar bone treated with wire at 3 to 14 days after the beginning of the experiment. On day 14, about 4 neovascularizations were detected. Many resorption lacunae were also detected on the pressure side in the PDL space in the wire group (Figure 5). In contrast, about 3 neovascularizations were found in controls on the same days. Significant differences in the numbers of neovascularizations were found between the control or wire groups on days 3 and 7 (Figure 5). On day 14, the amount of tooth movement was about 0.44 mm in the magnet group (Figure 4). On days 0, 7, 9 and 14, the difference was not statistically significant un the control, wire or magnet groups. On the other hand, on day 3, the number of neovascularizations was significantly higher in the magnet group than in other groups (p<0.01).
The same result was also obtained on the pressure side (Figure 6).
Discussion
VEGF, which isalso known as vascular permeability factor [17], induces angiogenesis in several physiological and pathological es. VEGF is a crucial regulator of angiogenesis, and its mRNA is expressed in various tissues, such as the lung and heart [18,19]. Previous studies showed that EGF mRNA was expressed in osteoblasts in in vitro experiments [20], suggesting that osteoblastic activity may be closely related to angiogenesis, which is the main function of VEGF. VEGF has various biological functions, such as vascular permeability and migration of human monocytes [17,21,22]. In this study, many periodontal tissues were detected on the tension and pressure side in the PDL space 3 days after the beginning of the experiment, and VEGF was highly expressed in these periodontal tissues. Thus, it is assumed that VEGF expression in periodontal tissues was promoted by mechanical stimuli to the periodontium, suggesting that VEGF may participate in the regulation of periodontal tissue metabolism and wound healing during orthodontic tooth movement.
Very recently, we demonstrated that local administration of rhVEGF enhanced the number of osteoclasts during experimental tooth movement, and the number of nuclei per osteoclast in rhVEGF-injected mice was significantly higher than in control mice [23]. Several experimental approaches have been attempted to induce more efficient tooth movement by increasing the number of osteoclasts and resultant bone resorption [24,25]. We speculate that several elements play a role in orthodontic tooth movement. In this study, it was emphasized that a combination of orthodontic stimuli by magnets accelerated differentiation of periodontal tissues, and it is apparent that these cells function to increase neovascularization. From the past research, the blood vessel rebirth of the dentistry orthodontic therapy with a magnet was not observed. In this study, pains in teeth by the orthodontics treatment might relate to VEGF. Therefore, natural induction of VEGF by magnetism may be effective for clinical orthodontic treatment by enhancing the rate of orthodontic tooth movement.
Conclusion
The authors showed that the natural induction of VEGF by magnetism may be effective for clinical orthodontic treatment by enhancing the rate of orthodontic tooth movement. Current study results suggest that VEGF, highly expressed by mechanical stimuli, enhances the number of neovascularizations as the paracrine factor is accelerated by magnetic force.
Acknowledgments
The authors are grateful to the staff of the Research Facility of Laboratory Animal Science, Hiroshima University School of Medicine, for their assistance.
This study was conducted in part with the support of a Grant-in-aid (15791213) from the Ministry of Education, Science, Sports and Culture in Japan.
References
- Choi J, Baek SH, Lee JI, Chang YI. Effects of clodronate on early alveolar bone remodeling and root resorption related to orthodontic forces: a histomorphometric analysis. Am J Orthod Dentofacial Orthop. 2010; 138: 548-549.
- Milne TJ, Ichim I, Patel B, McNaughton A, Meikle MC. Induction of osteopenia during experimental tooth movement in the rat: alveolar bone remodelling and the mechanostat theory. Eur J Orthod. 2009; 31:221-231.
- Kawata T, Hirota K, Sumitani K, Umehara K, Yano K, Tzeng HJ, Tabuchi T A new orthodontic force system of magnetic brackets. Am J Orthod Dentofacial Orthop.1987;92:241-248.
- Sandler PJ, Meghji S, Murray AM, Springate SD, Sandy JR, Crow V, Reed RT. Magnets and orthodontics. Br J Orthod. 1989; 16:243-249.
- Yamasaki K, Shibata Y, Imai S, Tani Y, Shibasaki Y, Fukuhara T. Clinical application of prostaglandin E1 (PGE1) upon orthodontic tooth movement. Am J Orthod 1984; 85: 508-518.
- Collins MK, Sinclair PM. The local use of vitamin D to increase the rate of orthodontic tooth movement. Am J Orthod Dentofacial Orthop 1988; 94: 278-284.
- Takano-Yamamoto T, Kawakami M, Kobayashi Y, Yamashiro T, Sakuda M. The effect of local application of 1,25-dihydroxycholecalciferol on osteoclast numbers in orthodontically treated rats. J Dent Res 1992; 71: 53-59.
- Leung DW, Cachianes G, Kuang WJ, Goeddel DV, Ferrara N. Vascular endothelial growth factor is a secreted angiogenic mitogen. Science 1989; 246: 1306-1309.
- Dumont DJ, Fong GH, Puri MC, Gradwohl G, Alitalo K, Breitman ML. Vascularization of the mouse embryo: a study of flk-1, tek, tie, and vascular endothelial growth factor expression during development. Dev Dyn 1995; 203: 80-92.
- Dvorak HF, Detmar M, Claffey KP, Nagy JA, van de Water L, Senger DR. Vascular permeability factor/vascular endothelial growth factor: an important mediator of angiogenesis in malignancy and inflammation. Int Arch Allergy Immunol 1995; 107: 233-235.
- Shibuya M, Yamaguchi S, Yamane A, Ikeda T, Tojo A, Matsushime H, Sato M. Nucleotide sequence and expression of a novel human receptor-type tyrosine kinase gene (flt) closely related to the fms family. Oncogene 1990; 5: 519-524.
- de Vries C, Escobedo JA, Ueno H, Houck K, Ferrara N, Williams LT. The fms-like tyrosine kinase, a receptor for vascular endothelial growth factor. Science 1992; 255: 989-991.
- Terman BI, Dougher-Vermazen M, Carrion ME, Dimitrov D, Armellino DC, Gospodarowicz D, Bohlen P. Identification of the KDR tyrosine kinase as a receptor for vascular endothelial cell growth factor. Biochem Biophys Res Commun 1992; 187: 1579-1586.
- Millauer b, Wizigmann-Voos S, Schnurch H, Martinez R, Moller NP, Risau W. High affinity VEGF binding and developmental expression suggest Flk-1 as a major regulator of vasculogenesis and angiogenesis. Cell 1993; 72: 835-846.
- Kaku M, Niida S, Kawata T, Maeda N, Tanne K. Dose- and time-dependent changes in osteoclast induction after a single injection of vascular endothelial growth factor in osteopetrotic mice. Bioomed Res 2000; 21:67-72.
- Stark TM, Sinclair PM. Effect of pulsed electromagnetic fields on orthodontic tooth movement. Am J Orthod Dentofac Orthop. 1987; 91:91-104.
- Senger DR, Galli SJ, Dvorak AM, Perruzzi CA, Harvey VS, Dvorak HF. Tumor cells secrete a vascular permeability factor that promotes accumulation of ascites fluid. Science 1983; 219:983-985.
- Berse B, Brown LF, Van de Water L, Dvorak HF, Senger DR. Vascular permeability factor (vascular endothelial growth factor) gene is expressed differentially in normal tissues, macrophages, and tumors. Mol Biol Cell 1992; 3: 211-20.
- Ferrara N, Houck K, Jakeman L, Leung DW (1992). Molecular and biological properties of the vascular endothelial growth factor family of proteins. Endocr Rev 1992; 13: 18-32.
- Harada S, Nagy JA, Sullivan KA, Thomas KA, Endo N, Rodan GA, Rodan SB. Induction of vascular endothelial growth factor expression by prostaglandin E2 and E1 in osteoblasts. J Clin Invest 1994; 93: 2490-2496.
- Barleon B, Sozzani S, Zhou D, Weich HA, Mantovani A, Marme D. Migration of human monocytes in response to vascular endothelial growth factor (VEGF) is mediated via the VEGF receptor flt-1. Blood 1996; 87:3336-3343.
- Clauss M, Weich H, Breier G, Knies U, Rockl W, Waltenberger J, Risau W. The vascular endothelial growth factor receptor Flt-1 mediates biological activities. Implications for a functional role of placenta growth factor in monocyte activation and chemotaxis. J Biol Chem 1996; 271:17629-1734.
- Kaku M, Kohno S, Kawata T, Fujita I, Tokimasa C, Tsutsui K, Tanne K. Effects of vascular endothelial growth factor on osteoclast induction during tooth movement in mice. J Dent Res. 2001;80: 1880-1883.
- Collins MK, Sinclair PM. The local use of vitamin D to increase the rate of orthodontic tooth movement. Am J Orthod Dentofacial Orthop. 1988 ; 94: 278-284.
- Takano-Yamamoto T, Kawakami M, Yamashiro T. Effect of age on the rate of tooth movement in combination with local use of 1,25(OH)2D3 and mechanical force in the rat. J Dent Res 1992; 71: 1989-1992.