Research Article - Biomedical Research (2017) Volume 28, Issue 17
Evaluation of positional error during radiotherapy for breast cancer after modified radical mastectomy
Hui-Juan Zhang1#, Chun Zhang2#, Rui-Gang Ge1, Xiao-Liang Liu1, Xiao-Hu Cong1, Bao-Lin Qu1 and Lin Ma1*
1Department of Radiation Oncology, the General Hospital of the People's Liberation Army, Beijing, PR China
2Department of Medicine, Xinjiang Uigur Autonomous Region Corps Hospital, Chinese People’s Armed Police Force, Urumqi, PR China
#These authors contributed equally to this work
- *Corresponding Author:
- Lin Ma
Department of Radiation Oncology
The General Hospital of the People's Liberation Army, PR China
Accepted on August 11, 2017
Abstract
Accurate positioning of Radiation Therapy (RT) is critical to its safety and efficacy for postmastectomy patients. We measured the Planning Target Volume (PTV) margins using the daily Cone-Beam Computed Tomography (CBCT) for patients who received Modified Radical Mastectomies (MRMs). We analysed setup error data using CBCT for 58 postmastectomy patients, including 28 with left-sided breast cancers (15 patients who received single chest wall (CW) RT and 13 who received CW plus supraclavicular region (CW/SR) RT) and 30 with right-sided cancers (15 who received single CW RT and 15 who received CW/SR RT). The positional error was corrected and compared in x, y, and z translational planes for different breast cancer locations (left or right) and different target volumes (single CW or CW/SR). We analysed a total of 925 digital images using the On Board Imaging system (OBI). The Z translational errors in treating CW/SR were significantly greater than in treating CW alone for both left-sided (P=0.035) and right-sided (P=0.048) cancers. The Y translational errors were significantly greater for the left-sided cancers than for the right-sided cancers (P=0.001) for both CW alone (P=0.026) and CW/SR (P=0.001). The Z translational errors of CW/SR were significantly greater than of single CW, and the Y translational errors were significantly greater on the left side than on the right side. In treating postmastectomy patients with daily image-guided radiation therapy, positioning errors should be checked and corrected.
Keywords
Breast neoplasms, Radiotherapy, Mastectomy, Radiotherapy setup errors, Cone-beam computed tomography.
Introduction
For breast cancer patients who have undergone MRM, postoperative adjuvant Radiation Therapy (RT) has been shown to reduce the risk of local recurrence [1-4]. However, precise targeting for RT after mastectomy can be problematic because of the irregularly shaped target volumes that include thin and curved Chest Walls (CWs); surrounding heart, lung, brachial plexus, contralateral breast, and other organs at risk (OAR); and non-coplanar supraclavicular and axillary regions. These require the use of sophisticated techniques that generate optimal dosimetry to meet clinical requirements. Three- Dimensional Conformal RT (3D-CRT), Intensity-Modulated RT (IMRT), and Volumetric Modulated Arc Therapy (VMAT) were shown to provide superior target volume coverage and dose conformity while reducing the dose delivered to the surrounding normal tissues [5,6]. However, these advanced techniques require accurate treatment delivery, as even slight errors in patient position caused by treatment setup errors and inter-fractional motions can significantly affect dose delivery to target volume and OARs. Therefore, Image-Guided Radiation Therapy (IGRT) is a fundamental prerequisite to the safe practice of the post-MRM 3D-CRT, IMRT, and VMAT. Cone-Beam Computed Tomography (CBCT) image sets are acquired by two-dimensional projection images at different positions that rotate around the patient and reconstructed through software algorithms into a three-dimensional volume. The co-registration by CBCT enhances the patient position accuracy and reduces the random deviations better than by traditional two-dimensional portal imaging, and it can offer smaller Planning Target Volumes (PTV), improved target coverage, and lower doses to OARs [7-9]. Adequate target dose coverage is needed for the local control of breast cancer.
An International Commission on Radiation Units and Measurements (ICRU) report [10] recommends that PTV shape absorbed-dose distributions to ensure that the prescribed absorbed dose will actually be delivered to all parts of the CTV with a clinically acceptable probability, despite geometrical uncertainties such as organ motion and setup variations. However, large PTV margins may also increase the dose delivered to the surrounding OARs. For example, a larger PTV volume was associated with greater increases in risk fibrosis and cardiac disease [11,12]. The risk in expanding PTV margins could be limited by reducing the systematic setup error [13].
Postmastectomy RT has different considerations that the RT in use to treat other tumors, including the considerable possibility of positional error due to the rare use of thermoplastic mask immobilization and the presence of skin folds between the breast boards and skin. Furthermore, the overwhelming majority of studies of setup accuracy for breast cancer patients concentrated on patients who had undergone breast-conserving procedures (lumpectomy with whole breast RT). Few studies have investigated the setup accuracy in MRM patients who required CW and Supraclavicular Region (SR) irradiation and faced potential setup errors such as patient positional movement.
This study evaluated these positional errors using quantitative analysis of the images using CBCT and online correction protocols.
Materials and Method
Patients
Patients who underwent MRMs from March 2013 to October 2015 at our institution, including 28 patients with left-sided cancers (15 patients receiving CW alone RT and 13 receiving CW+SR RT) and 30 with right-sided cancers (15 CW alone RT and 15 CW+SR RT) were included in this study. The characteristics of these patients and their treatments are summarized in Table 1. This study was conducted in accordance with the declaration of Helsinki, and was approved by the Ethics Committee of the Chinese PLA General Hospital (No. S2014-001-02). Written informed consent was obtained from all participants. The characteristics of these patients and their treatments are summarized in Table 1.
| Characteristics | Left side | Right side |
|---|---|---|
| N (%) | 28 (48.3) | 30 (51.7) |
| Age (years) | ||
| Mean (standard deviation) | 51.5 (12.2) | 53.0 (8.7) |
| Dose and fractionation (%) | ||
| 50 Gy/25 F | 10 (17.2) | 13 (29.3) |
| 36.5 Gy/10 F | 18 (31.1) | 17 (22.4) |
| PTV volume (cc) | ||
| Mean (standard deviation) | 644 (11.2) | 623 (13.4) |
| Target volume | ||
| Chest wall | 15 (25.9) | 15 (25.9) |
| Chest wall+supraclavicular | 13 (22.3) | 15 (25.9) |
| TNM stage (%) | ||
| II stage | 10 (17.2) | 11 (19.0) |
| III stage | 18 (31.0) | 19 (32.8) |
| Body mass index | ||
| Median (standard deviation) | 25.8 (9.7) | 25.6 (7.9) |
Table 1. Patients and treatment characteristics.
We included patients (a) with confirmed pathological invasive breast carcinoma; (b) who received MRM with/without axillary dissection or sentinel node biopsy alone; (c) tolerated RT well, without bone marrow suppression after chemotherapy; (d) could be immobilized with a breast board due to good activity function of ipsilateral upper limb; and (e) had no poor wound healing or axillary effusion. We excluded patients (a) whose Karnofsky performance score was <70; (b) for whom RT was not indicated; (c) who did not have complete CBCT imaging data available; or (d) were unwilling to participate in the study.
Treatment technique
All patients were treated in a supine position and immobilized with a breast board, and both arms elevated above the head holding hands grips, and individually positioned using an armsupport device. Three skin marks defined the position of patients, breast board, and treatment couch on patient’s left, right, and thorax by a laser beam, representing X, Y, and Z axes, respectively. A 5.0 mm bolus was added to the CW, and the surgical scar was marked with a radiopaque clip. Patients underwent simulation Computed Tomography (CT) scanning (Brilliance TM CT BigBore, Philips) with 3.0 mm slice thickness. CT images were transferred to the Pinnacle system (Pinnacle3 version 9.6, Phillips Medical Systems, Andover, MA, USA), on which we performed target delineation and treatment planning. The prescription doses were 50 Gy in 25 fractions (23 cases) and 36.5 Gy in 10 fractions (35 cases), respectively, which were the fractionation regimens of a phase II clinical trial that was being carried out at our institution.
Online IGRT protocol
Treatment was delivered using a Varian RapidArc™ linear accelerator (Varian Medical Systems, Inc., Palo Alto, CA) equipped with a kilo voltage CBCT image-guidance system. The image matching function was computed by the system software. X, Y, and Z represent the right-left, superior-inferior, and anterior-posterior directions, respectively. The online images of the target volumes and adjacent anatomic structures were acquired daily with OBI before each treatment. The rigid matching anatomic landmarks for the OBI were the vertebrae, sternum, and ribs. The acquired CBCT images were automatically compared with the planning images and then manually repositioned to determine the inter-fractional motions and setup errors, which were corrected online by performing a couch shift. The positional errors were recorded in the X, Y, and Z translational directions (Figure 1).
Statistics
Statistical analysis was performed using the IBM SPSS (Version 20.0, SPSS Inc., Chicago, IL, USA) statistical data package. Systematic error was computed for each patient’s positional error by averaging all the fractions; Standard Deviation (SD) as random error was also calculated. Systematic error of patient groups (Σ), (right or left breast patient groups and those undergoing CW or CW+SR radiation), was derived to describe random errors (σ) of motion parameters for all fractions of each group. Data are presented as the mean ± standard deviation. Paired t-tests were used to compare mean displacement between left and right groups. A Wilcoxon signed-rank test was used to determine significant differences for all the parameters of systematic and random errors. A P-value <0.05 was considered significant.
Results
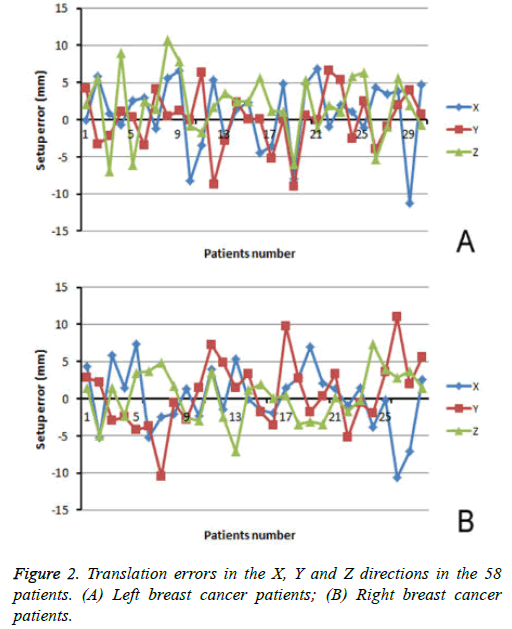
Altogether, we analysed 925 OBIs for the 58 breast cancer patients. Table 2 summarizes the positional error parameters in three directions (X, Y, and Z), based on the different target volumes (CW and CW/SR) in patients with left- and rightsided breast cancers (Figure 2) and the translational errors in the X, Y, and Z directions in the 58 patients (A, left-side breast cancer patients; B, right-side breast cancer patients).
| Left CW | Left CW + SC | Right CW | Right CW + SC | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| X | Y | Z | X | Y | Z | X | Y | Z | X | Y | Z |
| 4.2 ± 1.12 | 2.1 ± 1.09 | -0.1 ± 1.12 | 0.1 ± 0.51 | 5.6 ± 1.36 | -4.4 ± 0.90 | 2.8 ± 0.85 | 1.4 ± 0.69 | 4.4 ± 1.48 | -3.6 ± 1.25 | 0.1 ± 1.68 | -1.9 ± 1.72 |
| -3.3 ± 0.78 | 5.6 ± 0.69 | 5.8 ± 1.29 | -5.2 ± 0.76 | 1.2 ± 0.68 | -3.6 ± 0.65 | 2.2 ± 1.08 | -4.9 ± 1.08 | -5.2 ± 1.47 | 9.8 ± 0.46 | 0.4 ± 0.78 | 1.4 ± 1.41 |
| -2.2 ± 1.46 | -7.0 ± 1.70 | 0.8 ± 0.92 | 0.1 ± 1.05 | 1.1 ± 1.09 | 4.8 ± 2.40 | -2.9 ± 0.99 | 1.4 ± 0.86 | 5.8 ± 2.13 | 2.7 ± 0.92 | -3.4 ± 2.3 | 2.8 ± 2.53 |
| 1.1 ± 1.05 | 9.0 ± 0.92 | -0.7 ± 1.02 | -9.0 ± 2.05 | -6.0 ± 1.62 | -8.0 ± 1.77 | -2.4 ± 2.55 | -2.3 ± 1.06 | 1.4 ± 0.85 | -1.8 ± 1.19 | -3.1 ± 1.2 | 7.0 ± 0.95 |
| 0.3 ± 0.80 | -6.1 ± 1.22 | 2.6 ± 0.99 | 0.6 ± 0.67 | 5.4 ± 1.31 | 4.9 ± 0.99 | -4.2 ± 0.90 | 3.5 ± 0.86 | 7.4 ± 1.00 | 0.3 ± 1.20 | -3.5 ± 0.89 | 2.1 ± 1.25 |
| -3.4 ± 0.52 | 2.4 ± 0.67 | 2.9 ± 0.81 | 0 ± 0.86 | -1.2 ± 1.62 | 6.8 ± 0.85 | -3.7 ± 0.83 | 3.7 ± 0.73 | -5.2 ± 0.83 | 3.3 ± 0.5 | 0.2 ± 0.52 | 1.3 ± 0.82 |
| 4.1 ± 1.58 | 1.4 ± 1.19 | -1.2 ± 1.01 | 6.6 ± 1.15 | 1.9 ± 1.02 | -1.0 ± 0.68 | -10.5 ± 1.26 | 4.8 ± 1.52 | -2.4 ± 1.58 | -5.2 ± 0.65 | -1.7 ± 0.99 | -0.9 ± 0.64 |
| 0.5 ± 1.04 | 10.8 ± 0.99 | 5.6 ± 1.45 | 5.4 ± 1.78 | 1.1 ± 1.99 | 1.9 ± 1.06 | -0.6 ± 0.99 | 1.7 ± 0.80 | -2.1 ± 1.14 | -0.5 ± 1.29 | -0.2 ± 1.30 | 1.5 ± 1.36 |
| 1.2 ± 1.12 | 7.8 ± 1.37 | 6.6 ± 0.86 | -2.6 ± 1.11 | 5.8 ± 2.66 | 1.1 ± 1.39 | -2.8 ± 0.44 | -2.6 ± 0.75 | 1.3 ± 0.50 | -2 ± 1.05 | 7.3 ± 2.23 | -3.8 ± 1.41 |
| -0.1 ± 0.53 | -0.8 ± 1.23 | -8.2 ± 0.99 | 2.5 ± 0.79 | 6.3 ± 0.47 | -1.1 ± 0.60 | 1.5 ± 0.75 | -3.0 ± 1.39 | -2.3 ± 1.45 | 3.6 ± 0.67 | 4.0 ± 0.94 | -0.2 ± 1.24 |
| 6.4 ± 1.11 | -1.7 ± 0.84 | -3.5 ± 0.54 | -3.9 ± 1.19 | -5.3 ± 0.75 | 4.3 ± 1.11 | 7.2 ± 0.59 | 3.6 ± 0.91 | 3.9 ± 0.67 | 11.0 ± 0.99 | 2.8 ± 1.05 | -10.6 ± 2.33 |
| -8.7 ± 1.12 | 1.7 ± 0.97 | 5.3 ± 1.36 | -1.0 ± 1.12 | -0.9 ± 1.63 | 3.4 ± 1.20 | 4.9 ± 0.92 | -2.4 ± 0.92 | -1.4 ± 0.90 | 1.9 ± 0.75 | 3.7 ± 0.92 | -7.1 ± 0.62 |
| -2.8 ± 1.08 | 3.6 ± 0.88 | -2.7 ± 1.13 | 1.9 ± 1.90 | 5.6 ± 1.45 | 3.8 ± 1.09 | 1.5 ± 1.13 | -7.1 ± 1.27 | 5.3 ± 1.08 | 5.6 ± 0.73 | 1.5 ± 1.87 | 2.6 ± 1.89 |
| 2.3 ± 0.87 | 2.5 ± 0.75 | 1.5 ± 0.52 | 4.0 ± 1.14 | 2.0 ± 1.03 | -11.3 ± 0.60 | 3.3 ± 0.92 | 1.2 ± 0.63 | -0.1 ± 0.51 | NA | NA | NA |
| 0.1 ± 1.30 | 2.4 ± 1.20 | 2.3 ± 1.27 | 0.7 ± 1.17 | -0.7 ± 1.38 | 4.7 ± 0.80 | -1. 8 ± 0.88 | 2.0 ± 1.34 | -1.6 ± 1.23 | NA | NA | NA |
Table 2. Translation errors in X, Y and Z directions for different target volumes and primary tumor location.
The translational errors for the different target volumes (CW vs. CW/SR) are compared in Table 3. The translational errors in the Z direction for CW/SR were significantly larger than those for CW alone, depending on whether the primary tumor was located on the left or right side (left: P=0.035 and right: P=0.048). The translational errors in the X and Y directions for the different target volumes were not statistically significant.
| Displacement direction | Left | Right | ||||
|---|---|---|---|---|---|---|
| CW | CW+SC | P | CW | CW+SC | P | |
| X | 3.8 ± 0.25 | 4.2 ± 0.28 | 0.285 | 4.1 ± 0.29 | 4.6 ± 0.31 | 0.22 |
| Y | 5.0 ± 0.31 | 4.6 ± 0.29 | 0.599 | 3.6 ± 0.24 | 3.7 ± 0.29 | 0.708 |
| Z | 4.1 ± 0.26 | 4.9 ± 0.31 | 0.035 | 4.1 ± 0.27 | 5.0 ± 0.35 | 0.048 |
Note: Data presented as mean ± standard deviation. CW: Chest Wall; SC: Supraclavicular.
Table 3. Comparison of translational errors of different target volumes (chest wall vs. chest wall+supraclavicular region) for left and right breast cancer.
The left-sided translational errors in the Y direction were significantly larger than those on the right side (P=0.001), both for CW alone (P=0.026) and CW/SR (P=0.001) as target volume. However, the translational errors in the X and Z directions for the different target volumes were not statistically significant (Tables 4 and 5).
| Displacement direction | CW | CW+SC | ||||
|---|---|---|---|---|---|---|
| Left | Right | P | Left | Right | P | |
| X | 3.8 ± 0.25 | 4.1 ± 0.29 | 0.385 | 4.2 ± 0.28 | 4.6 ± 0.31 | 0.277 |
| Y | 5.0 ± 0.31 | 3.6 ± 0.24 | 0.001 | 4.6 ± 0.29 | 3.7 ± 0.29 | 0.026 |
| Z | 4.1 ± 0.26 | 4.1 ± 0.27 | 0.805 | 4.9 ± 0.31 | 5.0 ± 0.35 | 0.829 |
Note: Data presented as mean ± standard deviation. CW: Chest Wall; SC: Supraclavicular.
Table 4. Comparison of translational errors of different primary tumor location (left breast vs. right breast) for target volumes of chest wall and chest wall+supraclavicular region.
| Displacement direction | Primary tumor location | P | |
|---|---|---|---|
| Left | Right | ||
| X | 4.0 ± 0.19 | 4.3 ± 0.20 | 0.19 |
| Y | 4.8 ± 0.22 | 3.6 ± 0.18 | 0.001 |
| Z | 4.5 ± 0.20 | 4.5 ± 0.22 | 0.844 |
Table 5. Comparison of translational errors of different primary tumor location (left breast vs. right breast).
Discussion
Adjuvant radiotherapy is a critical therapy for breast cancer patients who have undergone MRM. As primary RT technologies, 3D-CRT and IMRT offer superior target volume coverage and dose conformity, with a limited dose to the normal structures [14-17]. In the process of radiotherapy for breast cancer patients, the position repeatability of the target volume is affected by many factors, such as breathing, weight change, treatment setup error, inter-fractional motion, which lead to an incomplete irradiation of the target volume or a higher irradiation dose of the normal organs, resulting in not only a lower local control rate, but also serious complications [18-20]. Guckenberger et al. reported that a mean reduction of PTV minimal dose errors were rotational errors alone in 4.4%, translational errors alone in 6%, and combined translational and rotational errors in 7% [7]. Studies have shown that increased positional errors result in a loss of target volume coverage and increased OAR maximal dose [7,21,22].
CBCT [23], a major non-crystal X-ray, plate-based digital imaging equipment, is advantageous due to its small volume, light weight, and open architecture features; it can also be directly integrated into the linear accelerator. A Gantry rotation cycle will be able to acquire and reconstruct CT images of a volume within the scope of the reconstructed threedimensional image, which can be matched with a threedimensional image of the treatment plan to assess the positional error. CBCT has a high resolution and can recognize the soft tissue structure. CBCT can accurately calculate the three-dimensional translational directions of each center on the setup errors and reflect the location and changes to the irradiation area (its structure and organization) in the image processing system through the software data processing and control the mobile treatment bed position error correction, which can effectively improve the accuracy of radiotherapy. Therefore, breathing, chest motion, and other physiological activities that are captured on the image result in the tissue image being larger than the CT image, thus causing errors when matching the image. In addition, when the image matching is successful, the rotation error cannot be corrected by rotating the therapeutic bed in the Y-axis direction, only by analyzing the rotation of the image. IGRT [24], the medical accelerator CT combined with imaging acquisition equipment, obtains the target information, verifies whether the treatment plan with the image information for measuring position errors and correct guidance, guides the follow-up treatment, and reduces the setup error. A linear accelerator equipped with CBCT is the most widely used image guidance technology and often used in precise clinical radiotherapy on tumor positioning error correction where it is very effective. IGRT is integral to ensuring the safe delivery of 3D-CRT and IMRT by decreasing systematic error using online verification.
The margins expanded by CTV-to-PTV and planning risk volume (PRV) were guided by measuring the position errors using CBCT online positional verification. Breast patient setup errors affected not only irradiated regions and neighboring critical organs but also PTV and PRV dose distribution. Breast patient setup errors mainly reflected the accuracy of the mechanical isocenter, treatment isocenter, resolution of couch positioning, laser position, and immobilization devices [22]. Other setup errors were related to patient factors, including age, performance status, body shape and movement, and compliance.
Patient setup errors were composed of the systematic and random deviations during inter-fractional variations in patient posture and position. A CTV-to-PTV margin size that ensures at least 95% dose to 99% of the CTV (on average) was equal to about 2 Σ+0.7σ based on the study of Stroom et al. [22]. According to the formula, the mean motion displacements for 58 patients in our study were 6 mm (X), 5 mm (Y), and 6 mm (Z). Our results for the setup errors were very similar to others published studies. Lozano et al. quantifiably analysed the systematic and random patient setup errors in conventional RT for breast and head-and-neck cancers with a no-action level protocol and found translational margin deviations of 6.3 mm (X), 7.2 mm (Y), and 4.8 mm (Z) for breast cancers [25]. Semaniak and Kukolowicz investigated postmastectomy position errors for two patient groups and found PTV margins to be 5.1 mm (X) and 4.9 mm (Y) for the first group and 5.4 mm (X) and 6.4 mm (Y) for the second group [26]. Increasingly, in clinical treatment settings, additional CBCT images are taken to verify the patients’ setups and to assess the daily dose delivery.
In the present study, we considered the motion and setup errors of breast cancer irradiation for two different target volumes: CW alone and the CW/SR. The Z translational errors of CW/SR were greater than those for the CW alone, for both the left side (P=0.035) and the right side (P=0.048). These differences in the Z direction had three possible causes. First, a greater target volume increased the motion errors due to generating uncertainty in the image registration. Chung et al. showed that a large target volume (breast size) was significantly associated with an extensive setup error (P=0.022) and recommended that meticulous caution is paid to patients with large breasts on every fraction [27]. Second, the non-coplanar target volumes of CW and SR lead to motion errors in the Z direction during image registration. Third, the position alignment of CW/SR was more difficult because variations in the arm position make it difficult to establish an exact correlation between the bony anatomy and the target volume position [28,29]. Feng et al. reported PTV margins were consistently larger by 0.4-1.2 mm in all axes for large target volumes including the supraclavicular and axillary nodal regions, with calculated PTV margins of 7.1 mm (Z), 4.3 mm (X), and 8.9 mm (Y) [29].
This distinction between the CW and SR positioning in the Z direction is important after MRM, as it affects the total dose to the CW target and adjacent OARs (lung and heart). The lung and heart volumes irradiated in the auxiliary RT after MRM could be significantly affected by positioning errors in the Zaxis direction due to the adjacent ipsilateral lungs and/or heart, and because the CW is obviously thinned by the resection of the breast and some pectoralis muscle. Larger PTV margins may assure a sufficient coverage of the target volumes, but it could also increase the radiation dose to OARs. Thus, reducing the position errors in the Z-axis direction is necessary to improve setup technique and to carry out daily IGRT safely [28].
To our knowledge, no study has explicitly reported on positional errors in maintaining required margins for PTV for different breast tumor locations for post-MRM RT by various IGRT image registrations. We first reported different positional errors for patients who had undergone left or right mastectomies. Translational Z errors were not significant (left: 4.5 ± 0.20 mm; right: 4.5 ± 0.22 mm; P=0.844), nor were X errors (left: 4.0 ± 0.10 mm; right: 4.3 ± 0.21 mm; P=0.190). However, Y translational differed significantly (left: 4.8 ± 0.22 mm; right: 3.6 ± 0.18 mm; P=0.001). Similarly, Y translational errors differed significantly left and right breasts for target volumes of the CW alone versus the CW/SR.
Reportedly, the setup margins required for the breast cancer patients are largest in the Y (superior-inferior) direction [29,30], possibly because cardiac impulses produce Y translational errors in the left CW (with or without the SR) as the thinner CW after mastectomy is more sensitive to impulses of the heart and main blood vessels. Alderliesten et al. [31] found a moderate correlation between the breast surface and heart displacement and considerable geometric variations of the heart relative to the breast surface (particularly in the Y direction). Variable heart positions in the Y direction at least had the same effect on setup errors in the Y direction, which may have been exacerbated by the tilted body position of patients being treated on an angled breast board. We also found that CTV-PTV margins significantly varied in the Y direction for CW RT (with or without the SR) in patients treated for leftside versus right-side breast cancers. The motion and setup error differences between RT for the right- and left-sided breast cancers have not been widely studied. Yue et al. [32] also reported differences in the motion and setup errors between the right- and left-sided breast cancers treated with accelerated partial breast irradiation. The use of the bony anatomy-based reference points indicated that the mean three-dimensional vector motion magnitudes differed significantly (left: 7.5 mm; right: 6.4 mm, P=0.005). The mean systematic errors of the bony anatomy based on the motion for the left-versus rightside breast patients were as follows: X: 2.6 vs. 3.1 mm (P=0.075); Y: 4.2 vs. 3.1 mm (P=0.010); and Z: 3.9 vs. 2.8 mm (P=0.000). This study confirmed obvious differences in setup errors between the left- and right-side breast RT. However, due to the anatomic structures being essentially changed, the resultant setup errors in the X and Z directions, with a significant difference for both sides, could not be applied to the patients after mastectomy irradiation. Based on our findings, the CTV-PTV margins used in clinical practice should be adapted to consider the differences in setup errors for the leftand right-sided breast cancers.
The main limitation of this present research was that the respiratory control facilities were not applied in our study. Thus, we lacked the information on the intra-fraction movements (such as respiration), which have been associated with significant setup errors [33-35]. However, Richter et al. reported that breathing-induced chest motions were small in planning four-dimensional CT studies and were found to be reproducible for the total treatment course [36]. Similarly, Brouwers et al. reported that data from both setup and twodimensional electronic portal imaging device dosimetry indicate that RT for patients treated during free breathing and voluntary moderately deep inspiration breath-holding was reproducible without significant differences [34].
Conclusion
The Z translational errors of irradiating the CWs plus the SR of breast cancer patients were significantly greater than that of the CWs alone, and the left-sided Y-translational errors in these patients were significantly greater than for the right-sided errors. In treating breast cancer patients after MRM, positioning errors in IGRT should be anticipated and corrected.
Acknowledgements
This study was supported by Clinical Research Support Funds of Chinese PLA General Hospital (No.2016FC-TSYS-1008), Wu Jieping Medical Foundation (No.320675012636) .The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Conflict of Interests
All authors declare that they have no conflicts of interests.
References
- Clarke M, Collins R, Darby S, Davies C, Elphinstone P, Evans V, Godwin J, Gray R, Hicks C, James S, MacKinnon E, McGale P, McHugh T, Peto R, Taylor C, Wang Y, Early Breast Cancer Trialists Collaborative Group (EBCTCG). Effects of radiotherapy and of differences in the extent of surgery for early breast cancer on local recurrence and 15-year survival: an overview of the randomised trials. Lancet 2005; 366: 2087-2106.
- Overgaard M, Hansen PS, Overgaard J, Rose C, Andersson M, Bach F, Kjaer M, Gadeberg CC, Mouridsen HT, Jensen MB, Zedeler K. Postoperative radiotherapy in high-risk premenopausal women with breast cancer who receive adjuvant chemotherapy. Danish Breast Cancer Cooperative Group 82b Trial. N Engl J Med 1997; 337: 949-955.
- Recht A, Edge SB, Solin LJ, Robinson DS, Estabrook A, Fine RE, Fleming GF, Formenti S, Hudis C, Kirshner JJ, Krause DA, Kuske RR, Langer AS, Sledge GW Jr, Whelan TJ, Pfister DG, American Society of Clinical Oncology. Postmastectomy radiotherapy: clinical practice guidelines of the American Society of Clinical Oncology. J Clin Oncol 2001; 19: 1539-1569.
- McGuire SE, Gonzalez-Angulo AM, Huang EH, Tucker SL, Kau SW, Yu TK, Strom EA, Oh JL, Woodward WA, Tereffe W, Hunt KK, Kuerer HM, Sahin AA, Hortobagyi GN, Buchholz TA. Postmastectomy radiation improves the outcome of patients with locally advanced breast cancer who achieve a pathologic complete response to neoadjuvant chemotherapy. Int J Radiat Oncol Biol Phys 2007; 68: 1004-1009.
- Wang J, Li X, Deng Q, Xia B, Wu S, Liu J, Ma S. Postoperative radiotherapy following mastectomy for patients with left-sided breast cancer: A comparative dosimetric study. Med Dosim 2015; 40: 190-194.
- Ma C, Zhang W, Lu J, Wu L, Wu F, Huang B, Lin Y, Li D. Dosimetric comparison and evaluation of three radiotherapy techniques for use after modified radical mastectomy for locally advanced left-sided breast cancer. Sci Rep 2015; 5: 12274-12283.
- Guckenberger M, Meyer J, Vordermark D, Baier K, Wilbert J, Flentje M. Magnitude and clinical relevance of translational and rotational patient setup errors: a cone-beam CT study. Int J Radiat Oncol Biol Phys 2006; 65: 934-942.
- Topolnjak R, Sonke JJ, Nijkamp J, Rasch C, Minkema D, Remeijer P, van Vliet-Vroegindeweij C. Breast patient setup error assessment: comparison of electronic portal image devices and cone-beam computed tomography matching results. Int J Radiat Oncol Biol Phys 2010; 78: 1235-1243.
- White EA, Cho J, Vallis KA, Sharpe MB, Lee G, Blackburn H, Nageeti T, McGibney C, Jaffray DA. Cone beam computed tomography guidance for setup of patients receiving accelerated partial breast irradiation. Int J Radiat Oncol Biol Phys 2007; 68: 547-554.
- Gregoire V, Mackie TR. State of the art on dose prescription, reporting and recording in Intensity-Modulate Radiation Therapy (ICRU report No. 83). Cancer Rdaiother 2011; 15: 555-559.
- Darby SC, Ewertz M, McGale P, Bennet AM, Blom-Goldman U, Brønnum D, Correa C, Cutter D, Gagliardi G, Gigante B, Jensen MB, Nisbet A, Peto R, Rahimi K, Taylor C, Hall P. Risk of ischemic heart disease in women after radiotherapy for breast cancer. N Engl J Med 2013; 368: 987-998.
- Collette S, Collette L, Budiharto T, Horiot JC, Poortmans PM, Struikmans H, Van den Bogaert W, Fourquet A, Jager JJ, Hoogenraad W, Mueller RP, Kurtz J, Morgan DA, Dubois JB, Salamon E, Mirimanoff R, Bolla M, Van der Hulst M, Wárlám-Rodenhuis CC, Bartelink H, EORTC Radiation Oncology Group. Predictors of the risk of fibrosis at 10 years after breast conserving therapy for early breast cancer: a study based on the EORTC Trial 22881-10882 boost versus no boost. Eur J Cancer 2008; 44: 2587-2599.
- Mukesh M, Harris E, Jena R, Evans P, Coles C. Relationship between irradiated breast volume and late normal tissue complications: a systematic review. Radiother Oncol 2012; 104: 1-10.
- Zhang Q, Yu XL, Hu WG, Chen JY, Wang JZ, Ye JS, Guo XM. Dosimetric comparison for volumetric modulated arc therapy and intensity-modulated radiotherapy on the left-sided chest wall and internal mammary nodes irradiation in treating post-mastectomy breast cancer. Radiol Oncol 2015; 49: 91-98.
- Haciislamoglu E, Colak F, Canyilmaz E, Dirican B, Gurdalli S, Yilmaz AH, Yoney A, Bahat Z. Dosimetric comparison of left-sided whole-breast irradiation with 3DCRT, forward-planned IMRT, inverse-planned IMRT, helical tomotherapy, and volumetric arc therapy. Phys Med 2015; 31: 360-367.
- Canney PA, Deehan C, Glegg M, Dickson J. Reducing cardiac dose in post-operative irradiation of breast cancer patients: the relative importance of patient positioning and CT scan planning. Br J Radiol 1999; 72: 986-993.
- Yang B, Wei XD, Zhao YT, Ma CM. Dosimetric evaluation of integrated IMRT treatment of the chest wall and supraclavicular region for breast cancer after modified radical mastectomy. Med Dosim 2014; 39: 185-189.
- Ahmad R, Hoogeman MS, Quint S, Mens JW, de Pree I, Heijmen BJ. Interfraciton bladder filling variations and time trends for cervical cancer patients assessed with a portable 3-dimensional ultrasound baldder scanner. Radiother Oncol 2008; 89: 172-179.
- Siddiqui F, Shi C, Papanikolaou N, Fuss M. Image-gudidance protocol comparison: supine and prones setup accuracy for pelvic radiation therapy. Acta Oncol 2008; 47: 1344-1350.
- Laursen LV, Muren LP, Etstren UV. Residual rotational setup errors after daily cone-beam CT image guiduance in lovally advanced cervical cancer (poster) Physics and technology: adaptive and image/dose guided RT. Tadiother Oncol 2010; 96: 419.
- Wang H, Shiu A, Wang C, Odaniel J, Mahajan A, Woo S, Liengsawangwong P, Mohan R, Chang EL. Dosimetric effect of translational and rotational errors for patients undergoing image-guided stereotactic body radiotherapy for spinal metastases. Int J Radiat Oncol Biol Phys 2008; 71: 1261-1271.
- Stroom JC, de Boer HC, Huizenga H, Visser AG. Inclusion of geometrical uncertainties in radiotherapy treatment planning by means of coverage probability. Int J Radiat Oncol Biol Phys 1999; 43: 905-919.
- Ahmad R, Hoogeman MS, Bondar M, Dhawtal V, Quint S, De Pree I, Mens JW, Heijmen BJ. Increasesing treament accuracy for cervical cancer patients using correlations between bladder-filling patients change and cervical-uterus displacements: proof of principle. Radiother Oncol 2011; 98: 340-346.
- Yu Y, Michaud AL, Sreeraman R, Liu T, Purdy JA, Chen AM. Comparison of daily versus nondaily image-guided radiotherapy protocols for patients treated with intensity-modulated radiotherapy for head and neck cancer. Head Neck 2014; 36: 992-997.
- Lozano EM, Pérez LA, Torres J, Carrascosa C, Sanz M, Mendicote F, Gil A. Correction of systematic set-up error in breast and head and neck irradiation through a no-action level (NAL) protocol. Clin Transl Oncol 2011; 13: 34-42.
- Semaniak A, Kukolowicz P. Set-up uncertainty during postmastectomy radiotherapy with Segmented Photon Beams Technique. Rep Pract Oncol Radiother 2015; 20: 181-187.
- Chung MJ, Lee GJ, Suh YJ, Lee HC, Lee SW, Jeong S, Lee JW, Kim SH, Kang DG, Lee JH. Setup error and effectiveness of weekly image-guided radiation therapy of tomo direct for early breast cancer. Cancer Res Treat 2015; 47: 774-780.
- Gierga DP, Riboldi M, Turcotte JC, Sharp GC, Jiang SB, Taghian AG, Chen GT. Comparison of target registration errors for multiple image-guided techniques in accelerated partial breast irradiation. Int J Radiat Oncol Biol Phys 2008; 70: 1239-1246.
- Feng CH, Gerry E, Chmura SJ, Hasan Y, Al-Hallaq HA. An image-guided study of setup reproducibility of postmastectomy breast cancer patients treated with inverse-planned intensity modulated radiation therapy. Int J Radiat Oncol Bio Phys 2015; 91: 58-64.
- Laaksomaa M, Kapanen M, Skytta T, Peltola S, Hyödynmaa S, Kellokumpu-Lehtinen PL. Estimation of optimal matching position for orthogonal kV setup images and minimal setup margins in radiotherapy of whole breast and lymph node areas. Rep Pract Oncol Radiother 2014; 19: 369-375.
- Alderliesten T, Betgen A, Elkhuizen PH, van Vliet-Vroegindeweij C, Remeijer P. Estimation of heart-position variability in 3D-surface-image-guided deep-inspiration breath-hold radiation therapy for left-sided breast cancer. Radiother Oncol 2013; 109: 442-447.
- Yue NJ, Goyal S, Kim LH, Khan A, Haffty BG. Patterns of intrafractional motion and uncertainties of treatment setup reference systems in accelerated partial breast irradiation for right- and left-sided breast cancer. Pract Radiat Oncol 2014; 4: 6-12.
- Kinoshita R, Shimizu S, Taguchi H, Katoh N, Fujino M, Onimaru R, Aoyama H, Katoh F, Omatsu T, Ishikawa M, Shirato H. Three-dimensional intrafractional motion of breast during tangential breast irradiation monitored with high-sampling frequency using a real-time tumor-tracking radiotherapy system. Int J Radiat Oncol Biol Phys 2008; 70: 931-934.
- Brouwers PJ, Lustberg T, Borger JH, van Baardwijk AA, Jager JJ, Murrer LH, Nijsten SM, Reymen BH, van Loon JG, Boersma LJ. Set-up verification and 2-dimensional electronic portal imaging device dosimetry during breath hold compared with free breathing in breast cancer radiation therapy. Pract Radiat Oncol 2015; 5: 135-141.
- Betgen A, Alderliesten T, Sonke JJ, van Vliet-Vroegindeweij C, Bartelink H, Remeijer P. Assessment of set-up variability during deep inspiration breath hold radiotherapy for breast cancer patients by 3D-surface imaging. Radiother Oncol 2013; 106: 225-230.
- Richter A, Sweeney R, Baier K, Flentje M, Guckenberger M. Effect of breathing motion in radiotherapy of breast cancer: 4D dose calculation and motion tracking via EPID. Strahlenther Onkol 2009; 185: 425-430.