Research Article - Biomedical Research (2016) Volume 27, Issue 4
Efficacy of minimally invasive transforaminal lumbar interbody fusion for single-segment lumbar degenerative disease
Aiguo Gao1,2*, Peng Zhao3, Yingchuan Zhou1, Qing Zhang1, Li Cheng11Department of Orthopedics, Wuxi People’s Hospital Affiliated to Nanjing Medical University, 299 Qingyang Road, Wuxi City, Jiangsu Province, 214023, PR China
2Department of Orthopedics, The First Affiliated Hospital of Soochow University, 188 Shizi Jie, Suzhou, Jiangsu Province, 215006, PR China
3Department of Orthopedics, The Affiliated Jiangyin Hospital of Southeast University Medical College, 163 Shoushan Road, Jiangyin City, Jiangsu Province, 214400, PR China
- *Corresponding Author:
- Aiguo Gao
Department of Orthopedics
Wuxi People’s Hospital Affiliated to Nanjing Medical University
China
E-mail: gaoaiguo899@163.com
Accepted date: April 17, 2016
Abstract
Lumbar degenerative disease is the major cause of low back pain. Currently, surgical treatment remains the standard care for lumbar degenerative disease. This study aimed to evaluate the efficacy and complications of minimally invasive transforaminal lumbar interbody fusion (MIS-TLIF) for the treatment of single-segment lumbar degenerative disease. A total of 75 patients with single-segment lumbar degenerative disease that underwent MIS-TLIF from 2011 to 2014 in Wuxi People’s Hospital were enrolled, while 120 cases undergoing open transforaminal lumbar interbody fusion (O-TLIF) during the study period were randomly sampled as controls. The duration of surgery, intraoperative blood loss, duration of intraoperative X-ray scan, creatine kinase level, time to first post-surgical ambulation, incidence of post-surgical complications, rate of interbody fusion, Visual Analogue Scale (VAS) score and Oswestry disability index (ODI) were compared between MIS-TLIF and O-TLIF groups. The mean intraoperative blood loss, time to first post-surgical ambulation and serum creatine kinase level were significantly lower in MIS-TLIF group than in O-TLIF group (P<0.05), while longer duration of surgery and duration of intraoperative X-ray scan were observed in MIS-TLIF group than in O-TLIF group (P<0.001). However, no significant differences were detected in the incidence of postsurgical complications and the rate of interbody fusion one year post-surgery between these two groups (P>0.05). VAS score and ODI significantly reduced one year post-surgery compared to those before surgery in both groups (P<0.05). In conclusion, MIS-TLIF is an effective treatment for lumbar degenerative disease, which alleviates surgical invasiveness, reduces intraoperative blood loss, and accelerates recovery.
Keywords
Lumbar degenerative disease, Minimally invasive transforaminal lumbar interbody fusion, Open transforaminal lumbar interbody fusion, Efficacy
Introduction
Lumbar degenerative disease refers to the symptoms of lower back pain and/or radiating pain, numbness or weakness in the legs stemming from a degenerated spinal disc [1]. Currently, transforaminal lumbar interbody fusion (TLIF) is a major approach for the treatment of lumbar degenerative disease, and its adequate compression and bone grafting results in satisfactory clinical efficacy [2-4]. However, such a surgery has problems of high invasiveness, large blood loss and long duration to recovery, and these shortcomings limit the clinical efficacy of TLIF.
Recently, minimally invasive transforaminal lumbar interbody fusion (MIS-TLIF) has been employed in clinical therapy of degenerative diseases. This approach is reported to have advantages of minimal invasiveness, minor intraoperative blood loss and rapid recovery. However, there is little knowledge on the long-term efficacy and complications of MIS-TLIF for lumbar degenerative disorders. This study was designed to evaluate the clinical effectiveness and complications of MIS-TLIF for the treatment of singlesegment lumbar degenerative disease.
Subjects and Methods
Subjects
This study was approved by the ethics review committee of Wuxi People’s Hospital Affiliated to Nanjing Medical University (XRY-2010028). Signed informed consent was obtained from all participants following a detailed description of the purpose and potential benefits from the study.
The patients with lumbar degenerative disease admitted to Wuxi People’s Hospital Affiliated to Nanjing Medical University (Wuxi, China) during the period from January, 2011 through December, 2014 were enrolled. Inclusion criteria were: (1) grade I or II single-segment lumbar spondylolisthesis without neural symptoms or with symptoms in unilateral nerve; (2) discogenic lumbar pain or low back pain in which conservative treatment for over 6 months were ineffective; (3) lumbar pain caused by post-surgical recurrent lumbar intervertebral disc prolapse; (4) intervertebral disc prolapse complicated by lumbar segmental instability; (5) formation of intervertebral pseudoarthrosis; and (6) patients with written informed consent. Those who met the following criteria were excluded from the study: (1) grade III or multiple-segment lumbar spondylolisthesis together with lumbar intervertebral disc prolapse; (2) a history of posterior lumbar decompression or combined with lumbar tumors; (3) severe hyperosteogeny and extensive dpidural fibrosis; (4) severe osteoporosis; and (5) those who rejected surgery. Finally, a total of 195 eligible patients with single-segment lumbar degenerative disease were enrolled in this study, including 75 cases undergoing MIFTLIF as study subjects and 120 cases undergoing open transforaminal lumbar interbody fusion (O-TLIF) that were served as controls. All surgeries were performed by the same groups of surgeons.
MIS-TLIF
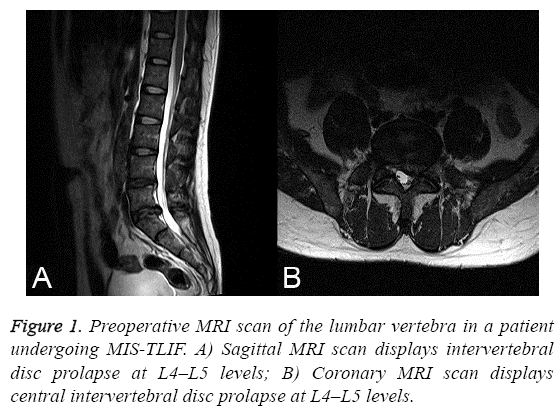
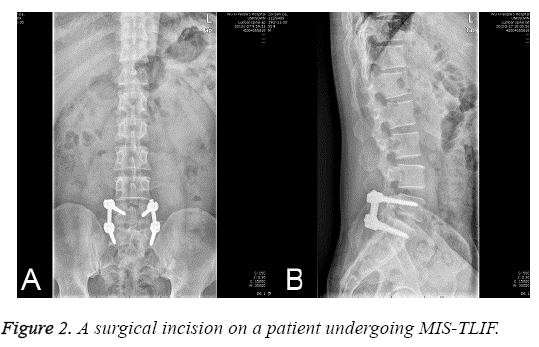
Following epidural anesthesia, patients were placed in a supine position. The chest and bilateral anterior superior iliac spine were placed onto a soft pillow, and the abdomen was suspended. C-arm posteroanterior radiography and MRI scanning was performed to position the target segment and identify the projection on the surface of the upper and lower vertebral pedicles (Figure 1). An incision with approximately 2.5 cm in length was cut on the exterior margin of the projection on the surface of vertebral pedicle of the affected vertebral body (Figure 2). The anadesma was incised, and the spatium intermusculare between multifidus and longissimus was identified. The articular process of the target segment was identified with fingers. The spatium intermusculare and tissues surrounding the articular process were bluntly dissociated using a periosteum detacher. The articular process was fully exposed, and soft tissue expanders were inserted. The Pipeline spreader and light source were implanted, and X-ray scan was performed again to confirm the location of the spreader in the target segment. The soft tissues embedded in the spreader were removed, and inferior articular process and part of superior articular process were removed. Part of ligamentum flavum was removed, and the dural sac and nerve root were exposed and retracted. The compressive intervertebral disc and hyperplastic osteophyte were removed. Then, the dural sac and nerve root were retracted, and intervertebral spreaders were implanted to spread intervertebral space. The width of intervertebral space was measured, and the upper and lower endplates were removed. Bones were grafted, and lumbar intervertebral fusion device filled with autogenous cancellous bone (Johnson & Johnson, New Brunswick, NJ, USA) was implanted. X-ray scan was done to confirm the location of the intervertebral fusion device. X-ray scan was done again to confirm the position of internal fixation and intervertebral fusion device.
The percutaneous pedicle screw was inserted. Briefly, the vertebral pedicle puncture needle was implanted through the exterior margin of the upper and lower vertebral pedicles under the guide of C-arm scan. The insertion site was identified using X-ray, and the puncture needle was screwed to the vertebral body. The inner core was withdrawn, and the guiding needle was inserted and pushed to the bony fixation to validate nonperforation of vertebral pedicle and anterior margin of the vertebral body. The puncture needle was removed, and the soft tissues were expanded and protected with an expander. A hollow pedicle screw was inserted according to the thread length. Following insertion of ipsilateral pedicle screw, an appropriate fixation rod was placed. After X-ray revealed satisfactory position of the rod, the rod was compressed and the screw cap was fastened. The pedicle screw and internal fixation rod were inserted and compressed using the same method described above in the contralateral side, and the screw cap was fastened and fixed. The location of screw rod and interbody infusion device was identified using X-ray scanning. The wound was rinsed and sutured.
O-TLIF
Following epidural anesthesia, the patients were placed in supine position. Then, the abdomen was suspended. C-arm Xray scan was performed to identify the segment of lesion, and a median vertical incision was cut. The zygapophyseal joint in the affected segment was exposed, and the lateral inferior articular process and part of superior articular process were removed. A portion of ligamenta flava was cleared, and dural sac and nerve root were exposed and retracted. Compressive intervertebral disc and hyperplastic osteophyte were incised. Dural sac and nerve root were retracted, and intervertebral expanders were implanted to expand intervertebral space. The width of intervertebral space was estimated, and the upper and lower endplates were removed. Bones were grafted, and lumbar intervertebral fusion device filled with autogenous cancellous bone was implanted. The location of the intervertebral fusion device was confirmed by X-ray scan. Bilaeral paravertebral muscle was detached, and crista lambdoidalis was positioned. An appropriate vertebral pedicle screw was implanted. X-ray scan was performed to confirm the position of the screw. The pre-bending titanium rod was implanted. X-ray scan was performed to identify the position of the rod, and the nut of the screw was tightened. X-ray scan was done again to confirm the position of internal fixation and intervertebral fusion device. The wound was rinsed and sutured.
Perioperative management
All subjects were given the same pre-surgical preparation and post-surgical treatment. Antibiotics were intravenously administered for 12 hours after surgery for prevention of infections, and wound dressing change was given one day postsurgery. In addition, low-dose hormone and mannitol was given to patients with residual nerve root to alleviate nerve root edema.
Assessment of clinical efficacy
The duration of surgery, duration of intraoperative X-ray scan, intraoperative blood loss, time to first post-surgical ambulation, and incidence of post-surgical complications were observed, and serum creatine kinase level was determined before surgery and 3 and 7 days post-surgery. Visual Analogue Scale (VAS) score and Oswestry disability index (ODI) were employed to quantify pre- and post-surgical pain and dysfunction. Anterioposterior and lateral lumbar X-ray scan was performed at the day of surgery, and 1, 3, 6 months and 1 year post-surgery, and three-dimensional computed tomography (CT) reconstruction of the lumbar spine was done one year post-surgery to evaluate interbody fusion.
Statistical analysis
All data were expressed as mean ± standard deviation (SD). Differences of proportions were tested for statistical significance with the chi-square test, and Student’s t test was used to compare between groups. All statistical analyses were performed using statistical software SPSS version 19.0 (SPSS Inc.; Chicago, IL, USA), with a P value <0.05 indicative of statistical significance.
Results
Baseline characteristics of the patients
Of the 75 patients undergoing MIS-TLIF, there were 32 men and 43 women, with a mean age of 53 ± 8 years (range, 36 to 72 years). There were 45 cases with discogenic low back pain or lumbar pain, 22 cases with lumbar spondylolisthesis and 8 cases with lumbar intervertebral disc prolapsed combined with lumbar spondylolisthesis. MIS-TLIF was performed at L3–L4 in 4 cases, at L4–L5 in 31 cases, and at L5–S1 in 30 cases. Among the 120 cases receiving O-TLIF, there were 48 men and 72 women, with a mean age of 55 ± 10 years (range, 42 to 78 years). There were 53 cases with discogenic low back pain or lumbar pain, 40 cases with lumbar spondylolisthesis and 27 cases with lumbar intervertebral disc prolapsed combined with lumbar spondylolisthesis. O-TLIF was performed at L3–L4 in 4 cases, at L4–L5 in 60 cases, and at L5–S1 in 56 cases (Table 1). There were no significant differences in the gender, age, type of disease and surgical site between the two groups (all P values>0.05).
| Characteristics | MIS-TLIF group (n=75) |
O-TLIF group (n=120) |
P value | |
|---|---|---|---|---|
| Male/female | 30/45 | 48/72 | 0.964 | |
| Mean age (years) | 53 ± 8 | 55 ± 10 | 0.472 | |
| Type of disease | Lumbar spondylolisthesis | 22 (29.3%) | 40 (33.3%) | 0.492 |
| Discogenic low back pain | 45 (60%) |
53 (44.2%) | ||
| Intervertebral disc disorder combined with lumbar spondylolisthesis | 8 (10.7%) |
27 (22.5%) | ||
| Surgical site | L3–L4 | 4 (5.3%) | 11 (9.2%) | 0.832 |
| L4–L5 | 41 (54.7%) | 60 (50%) | ||
| L5–S1 | 30 (40%) | 49 (40.8%) | ||
Table 1: Demographic and clinical characteristics.
Clinical efficacy
All subjects were followed up for 12 months. The patients undergoing MIS-TLIF had a mean intraoperative blood loss of 195 ± 203 ml and a mean time to post-surgical ambulation of 2.8 ± 1.3 days, which were significantly lower than those of OTLIF (P=0.017 and <0.01). MIS-TLIF group had a significantly longer duration of surgery and duration of exposure to intraoperative X-ray scan than O-TLIF group (P<0.01). However, no significant difference was detected in the rate of interbody fusion one day post-surgery between these two groups (P>0.05). In addition, there were no significant differences in VAS score and ODI before surgery and one day post-surgery between MIS-TLIF group and O-TLIF group (P>0.05). However, VAS score and ODI significantly reduced one year post-surgery compared to those before surgery in both groups (P<0.05) (Table 2). There were no significant differences in serum creatine kinase level pre-surgery and 7 days post-surgery between MIS-TLIF group and O-TLIF group (P>0.05), and a significantly lower serum creatine kinase level was detected in MIS-TLIF group than in O-TLIF group 3 days post-surgery (P < 0.05) (Table 3).
| Duration of surgery (min) | Intraoperative blood loss (ml) | Duration of exposure to intraoperative radiation (s) | VAS score | ODI (%) | Time to first post-surgical ambulation (d) | Rate of interbody fusion at final follow-up (%) | ||
|---|---|---|---|---|---|---|---|---|
| 191 ±3 2 | 195 ± 203 | 102 ± 25 | Pre-surgery | Final follow-up | Pre-surgery | Final follow-up | ||
| 7.3 ± 1.2 | 1.7 ± 1.1 | 56.3±14.7 | 16.8±8.0 | 2.8±1.3 | 100 | |||
| 7.0 ± 1.3 | 1.7 ± 1.0 | 59.8±12.1 | 17.8±5.9 | 6.7±1.2 | 100 | |||
| 150 ± 18 | 298 ± 100 | 65 ± 11 | ||||||
| 4.479 | 2.461 | 6.321 | 0.612 | 0.064 | 0.922 | 0.493 | 11.428 | - |
| <0.01 | 0.017 | <0.01 | 0.544 | 0.949 | 0.361 | 0.624 | <0.01 | >0.05 |
Table 2: Comparison of final follow-up between MIS-TLIF and O-TLIF groups.
| Time | MIS-TLIF group (n=75) |
O-TLIF group (n=120) |
P value |
|---|---|---|---|
| Pre-surgery | 89.3 ± 36.3 | 92.0 ± 43.0 | 0.815 |
| 3 days post-surgery | 247.9 ± 91.1 | 329.9 ± 106.4 | 0.006 |
| 7 days post-surgery | 101.3 ± 44.6 | 126.9 ± 51.8 | 0.073 |
Table 3: Comparison of serum creatine kinase level between MISTLIF and O-TLIF groups.
Post-surgical complications
Five cases receiving O-TLIF developed nerve root irritation post-surgery, which disappeared following administration of mannitol, dexamethasone, and nerve growth factors. Fives case undergoing MIS-TLIF developed would infection, which was healed following treatment with antibiotics. There was no significant difference in the incidence of post-surgical complications between MIS-TLIF group and O-TLIF group (χ2=1, P>0.05).
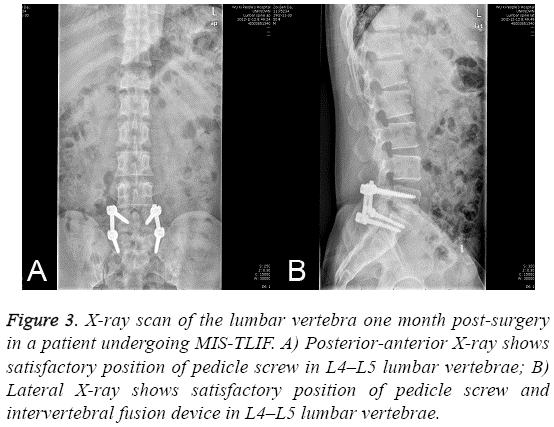
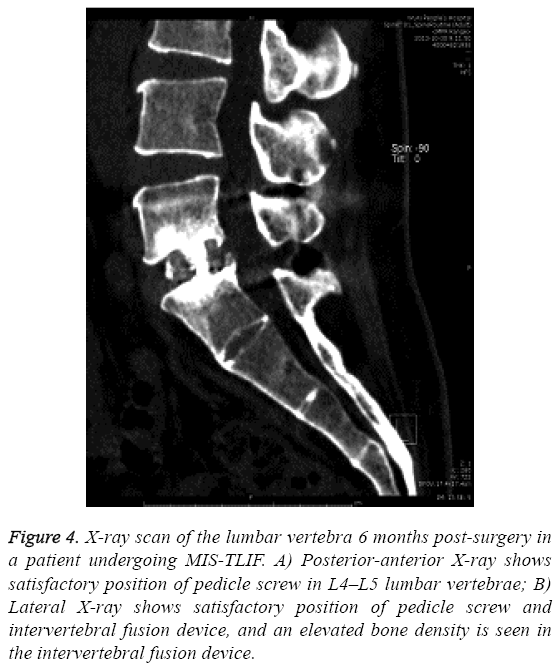
X-ray scan of the lumbar vertebra 1 month (Figure 3) and 6 months (Figure 4) post-surgery showed satisfactory position of pedicle screw in lumbar vertebrae of patients undergoing MISTLIF, and 3-dimensional CT reconstruction image of the lumbar vertebra one year post-surgery revealed bone bridge formation between vertebral body, and fusion of intervertebral space in patients undergoing MIS-TLIF (Figure 5). The rate of interbody fusion was 100% one day post-surgery in both groups (Table 2).
Figure 3: X-ray scan of the lumbar vertebra one month post-surgery in a patient undergoing MIS-TLIF. A) Posterior-anterior X-ray shows satisfactory position of pedicle screw in L4–L5 lumbar vertebrae; B) Lateral X-ray shows satisfactory position of pedicle screw and intervertebral fusion device in L4–L5 lumbar vertebrae.
Figure 4: X-ray scan of the lumbar vertebra 6 months post-surgery in a patient undergoing MIS-TLIF. A) Posterior-anterior X-ray shows satisfactory position of pedicle screw in L4–L5 lumbar vertebrae; B) Lateral X-ray shows satisfactory position of pedicle screw and intervertebral fusion device, and an elevated bone density is seen in the intervertebral fusion device.
Discussion
Lumbar degenerative disease is the major cause of low back pain [5]. Currently, TLIF is a major approach for the treatment of lumbar degenerative disease [2-4]. Since TLIF only requires the removal of unilateral zygapophyseal joints through intervertebral foramen and has little retraction to dural sac and nerve root, the incidence of complications such as dural sac rupture and nerve root injury is significantly lower compared to posterior lumbar interbody fusion (PLIF) [6,7]. Although OTLIF significantly reduces damages to posterior bone tissues and ligament, paravertebral muscle is required to be detached from spinous process and vertebral plate to the exterior side of facet articular process [8]. To reduce intraoperative injury to paravertebral muscle, MIS-TLIF has been recently introduced for lumbar interbody fusion via intervertebral foramen through the natural space between multifidus and erector spinae. This surgery has achieved satisfactory clinical efficacy [9,10]. However, there is little knowledge on the comparative efficacy of MIS-TLIF versus O-TLIF for lumbar degenerative disease.
MIS-TLIF has the following advantages to O-TLIF. First, this surgery causes minor damages to paravertebral muscle [11]. MIS-TLIF is performed through the spatium intermusculare approach by means of Pipeline spreader, and has no requirement of detaching paravertebral muscle from spinous process and vertebral plate, resulting in minor injury to paravertebral muscle [11]. Creatine kinase is a sensitive marker of muscle damage, and a higher level indicates more severe damage [12-14]. In this study, a significantly higher serum creatine kinase level was observed in O-TLIF group than in MIS-TLIF group 3 days post-surgery, indicating that O-TLIF caused a greater damage to paravertebral muscle than MISTLIF. It has been shown that excessive damage to paravertebral muscle insertion site and damage to spinal never may cause denervation atrophy of deep back muscles, postsurgical muscle fiber scarring, and reduced muscular function, which affects post-surgical recovery of lumbar and back muscles and induces lumbar and back pain [15]. Second, this approach causes little intraoperative blood loss. MIS-TLIF directly exposes facet articular process and transverse process via multifidus muscle space, and has little detachment of multifidus muscle. Thus the damage to lumbar muscle soft tissues by conventional large incision approach could be avoided, resulting in better protection of blood circulation and reduced intraoperative blood loss [16]. Our findings showed a significantly lower inraoperative blood loss (195 ± 203 ml) in MIS-TLIF group than in O-TLIF group (298 ± 100 ml). Third, MIS-TLIF leads to mild post-surgical pain and rapid recovery [17]. It has been shown that the patients undergoing MIS-TLIF have remarkably alleviated post-surgical pain, which may be associated with the specific design of the adjustable Pipeline spreader [18-20]. The inverted “V”-shape structure exposes and decompresses zygapophyseal joints through expanding the natural muscle space between multifidus and erector spinae while it does not affect surgical visual field and reduces surgical incision [21]. In addition, the surgery has significantly reduced detachment of the multifidus insertion site compared to conventional TLIF. After surgery, multifidus naturally adheres to erector spinae. Furthermore, MIS-TLIF reduces the detachment of paravertebral muscle, which helps reserves the function of paravertebral muscle. Alleviated post-surgical pain and recovery of paravertebral muscle function promote earlier ambulation in patients undergoing MIS-TLIF [21]. In this study, the time to first post-surgical ambulation was significantly lower in MIS-TLIF group (2.8 ± 1.3 days) than in O-TLIF group (6.7 ± 1.2 days), suggesting that MIS-TLIF leads to milder post-surgical pain and quicker recovery from surgery compared to O-TLIF.
Several limitations of this study should be pointed out. First, our case number is limited and further studies involving larger cases are needed to confirm our conclusion. Second, MIS-TLIF has two problems: long duration of surgery and long duration of intraoperative X-ray scan [22]. In this study, a longer duration of surgery was observed in MIS-TLIF group than in O-TLIF group. Logarithmic curve fitting analysis revealed that the duration of MIS-TLIF reduced with the accumulation of cases undergoing surgery, and the duration tended to be 140 min after the surgeon performed MIS-TLIF in over 30 cases [23]. In this study, the mean duration of MIS-TLIF was 191 ± 32 min, which may be associated with lack of surgical experiences and inadequate proficiency with surgical instruments [22]. The duration of intraoperative X-ray scan was found to be significantly longer in MIS-TLIF group than in TLIF group, which was strongly associated with surgeon’s experiences [20]. In addition, MIS-TLIF is reported to excessively depend on the guiding of intraoperative X-ray scan, resulting in exposure to high-dose radiation [24]. Consistent with previous studies, our findings showed that MIS-TLIF caused longer duration of intraoperative X-ray scan, which may be associated with surgeon’s lack of experiences, and excessive dependence on guiding of intraoperative X-ray scan. More intensive protective interventions should be implemented to reduce the damage caused by radiation.
In summary, MIS-TLIF and O-TLIF are effective for the treatment of lumbar degenerative disease, and they have no significant difference with regard to the incidence of postsurgical complications. Although MIS-TLIF requires a longer duration and causes a longer duration of intraoperative X-ray scan compared to O-TLIF, this surgical approach leads to less intraoperative blood loss, milder damage to paravertebral muscle, quicker post-surgical recovery from surgery, and shorter time to first post-surgical ambulation.
Acknowledgements
This study was supported by the grants from the Wuxi Bureau of Science & Technology. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- Modic MT, Ross JS. Lumbar degenerative disk disease. Radiology 2007; 245: 43-61.
- Taher F, Essig D, Lebl DR, Hughes AP, Sama AA, Cammisa FP, Girardi FP. Lumbar degenerative disc disease: current and future concepts of diagnosis and management. AdvOrthop 2012; 970752, 2012.
- Silber JS, Anderson DG, Hayes VM, Vaccaro AR. Advances in surgical management of lumbar degenerative disease. Orthopedics2002; 25: 767-771.
- An H, Boden SD, Kang J, Sandhu HS, Abdu W, Weinstein J. Summary statement: emerging techniques for treatment of degenerative lumbar disc disease. Spine 2003; 28: S24-S25.
- Modic MT, Ross JS. Lumbar degenerative disc disease. Radiology 2007; 245: 43-61.
- Cole CD, McCall TD, Schmidt MH, Dailey AT. Comparison of low back fusion techniques: transforaminal lumbar interbody fusion (TLIF) or posterior lumbar interbody fusion (PLIF) approaches. Curr Rev Musculoskelet Med 2009; 2: 118-126.
- Barbagallo GM, Albanese V,Raich AL, Dettori JR, Sherry N, Balsano M. Lumbar lateral interbody fusion (LLIF): comparative effectiveness and safety versus PLIF/TLIF and predictive factors affecting LLIF outcome. Evid Based Spine Care J 2014; 5: 28-37.
- Dhall SS, Wang MY, Mummaneni PV. Clinical and radiographic comparison of mini-open transforaminal lumbar interbody fusion with open transforaminal lumbar interbody fusion in 42 patients with long-term follow-up. J Neurosurg Spine 2008; 9: 560-565.
- Kim JS, Jung B, Lee SH. Instrumented minimally invasive spinal-transforaminal lumbar interbody fusion (MIS-TLIF); minimum 5-years follow-up with clinical and radiologic outcomes. J Spinal Disord Tech 2012.
- Rouben D, Casnellie M, Ferguson M. Long-term durability of minimal invasive posterior transforaminal lumbar interbody fusion: a clinical and radiographic follow-up. J Spinal Disord Tech 2011; 24: 288-96.
- Goldstein CL, Macwan K, Sundararajan K, Rampersaud YR. Comparative outcomes of minimally invasive surgery for posterior lumbar fusion: a systematic review. ClinOrthopRelat Res 2014; 472: 1727-1737.
- Kim KT, Lee SH, Suk KS and Bae SC. The quantitative analysis of tissue injury markers after mini-open lumbar fusion. Spine 2006; 31: 712-716.
- Kumbhare D, Parkinson W, Dunlop B, Ryan E, Denkers M, Shah AA. Biochemical measurement of muscle injury created by lumbar surgery. Clin Invest Med 2007; 30: 12-20.
- Kumbhare D, Parkinson W, Dunlop B. Validity of serum creatine kinase as a measure of muscle injury produced by lumbar surgery. J Spinal Disord Tech 2008; 21: 49-54.
- Stevens KJ, Spenciner DB, Griffiths KL, Kim KD, Zwienenberg-Lee M, Alamin T. Comparison of minimally invasive and conventional open posterolateral lumbar fusion using magnetic resonance imaging and retraction pressure studies. J Spinal Disord Tech 2006; 19: 77-86.
- Parker SL, Mendenhall SK, Shau DN, Zuckerman SL, Godil SS, Cheng JS. Minimally invasive versus open transforaminal lumbar interbody fusion for degenerative spondylolisthesis: comparative effectiveness and cost-utility analysis. World Neurosurg2014; 82: 230-238.
- Adogwa O, Parker SL, Bydon A, Cheng J, McGirt MJ. Comparative effectiveness of minimally invasive versus open transforaminal lumbar interbody fusion: 2-year assessment of narcotic use, return to work, disability, and quality of life. J Spinal Disord Tech 2011; 24: 479-484.
- Wang J, Zhou Y, Zhang ZF, Li CQ, Zheng WJ, Liu J. Minimally invasive or open transforaminal lumbar interbody fusion as revision surgery for patients previously treated by open discectomy and decompression of the lumbar spine. Eur Spine J 2011; 20: 623-628.
- Wang J, Zhou Y, Feng Zhang Z, Qing Li C, Jie Zheng W, Liu J. Comparison of the clinical outcome in overweight or obese patients after minimally invasive versus open transforaminal lumbar interbody fusion. J Spinal Disord Tech 2014; 27: 202-206.
- Wang J, Zhou Y, Zhang ZF, Li CQ, Zheng WJ, Liu J. Comparison of one-level minimally invasive and open transforaminal lumbar interbody fusion in degenerative and isthmic spondylolisthesis grades 1 and 2. Eur Spine J 2010; 19: 1780-1784.
- Shunwu F, Xing Z, Fengdong Z, Xiangqian F. Minimally invasive transforaminal lumbar interbody fusion for the treatment of degenerative lumbar diseases. Spine 2010; 35: 1615-1620.
- Karikari IO, Isaacs RE. Minimally invasive transforaminal lumbar interbody fusion: a review of techniques and outcomes. Spine 2010; 35: S294-S301.
- Silva PS, Pereira P, Monteiro P, Silva PA,Vaz R. Learning curve and complications of minimally invasive transforaminal lumbar interbody fusion. Neurosurg Focus 2013; 35: E7.
- Peng CW, Yue WM, Poh SY, Yeo W, Tan SB. Clinical and radiological outcomes of minimally invasive versus open transforaminal lumbar interbody fusion. Spine (Phila Pa 1976) 2009; 34: 1385-1389.