Research Article - Biomedical Research (2017) Volume 28, Issue 4
Efficacy of autologous adipose tissue-derived stem cells with extracellular matrix and hyaluronic acid on human hip osteoarthritis
Jaewoo Pak1,2,3, Jung Hun Lee1,4, Kwang Seung Park4 and Sang Hee Lee4*1Stems Medical Clinic, Chungdam-dong, Gangnam-gu, Seoul, Republic of Korea
2TPH (TEDA-TICH affiliated hospital), Third Ave, TEDA, Tianjin, China
3Life Science Institute, Komplek Permata Senayan Jl, Tentara Pelajar, Patal Senayan-Kebayoran Lama Jakarta, Indonesia
4Department of Biological Sciences, National Leading Research Laboratory, Myongji University, Myongjiro, Yongin, Gyeonggido, Republic of Korea
- *Corresponding Author:
- Sang Hee Lee
Department of Biological Sciences
Myongji University, Republic of Korea
Accepted date: September 14, 2016
Abstract
The aim of this study is to demonstrate that percutaneous injections of autologous Adipose Tissue- Derived Stem Cells (ASCs) and Extracellular Matrix (ECM) in the form of adipose Stromal Vascular Fraction (SVF), along with Hyaluronic Acid (HA) and Platelet-Rich Plasma (PRP) activated by calcium chloride could regenerate cartilage-like tissue in human hip Osteoarthritis (OA) patient. Autologous lipoaspirates were obtained from adipose tissue of the abdominal origin. Afterwards, the lipoaspirates were homogenized to obtain ECM. This homogenized lipoaspirates were then mixed with collagenase and incubated. The ASCs/ECM-containing SVF was mixed with calcium chloride-activated PRP plasma and HA, and this ASCs mixture was injected into a hip of a patient with OA. Pre-treatment and posttreatment MRI T2 views, functional rating index, range of motion, and pain score data were analysed. The patient’s Magnetic Resonance Imaging (MRI) data showed significant positive changes. Cartilagelike tissue regeneration was evident in MRI studies of the patient. Along with MRI evidence, the measured physical therapy outcomes in terms of range of motion, subjective pain, and functional status, all improved. These results indicate that percutaneous injection of the ASCs mixture is a promising minimally invasive therapy for OA of human hip.
Keywords
Adipose tissue-derived stem cells, Extracellular matrix, Human cartilage regeneration, Osteoarthritis.
Introduction
Adipose Tissue-Derived Stem Cells (ASCs) in the form of Stromal Vascular Fraction (SVF) have been widely used in Korea over decades by plastic surgeons as a semi-permanent volume expander. In June 2009, Korean Food and Drug Administration (KFDA) had allowed adipose SVF to be used as a medical procedure when obtained and processed within a same medical facility with minimal processing [1]. Adipose SVFs contain various components, including Mesenchymal Stem Cells (MSCs) and Extracellular Matrix (ECM) [2]. Such MSCs are termed Adipose Tissue-Derived Stem Cells (ASCs) and have shown to regenerate cartilage in vitro [3,4]. In 2011, Pak has showed that ASCs contained in the adipose SVF can regenerate cartilage-like tissue in human Osteoarthritis (OA) patients [5]. Since then, many studies have confirmed that autologous ASCs in human adipose SVF are potential agents capable of regenerating cartilage in OA patients [6]. Due to lack of standardization in autologous adipose tissue processing procedures and large variations in the numbers of ASCs within the adipose tissue, the resultant cartilage regeneration capacity may vary widely [7]. Among the many reasons, some of the factors that may affect the result of cartilage regeneration by ASCs can be due to ASC viability, adherence, and growth.
It is well recognized that ECM may work as a scaffold and enhance stem cell adherence at the site of the cartilage lesion [8]. Also, ECM has been shown to excrete various growth factors which may also enhance the survival and growth of stem cells following direct local injections [8].
In this report, Platelet-Rich Plasma (PRP) was used as a source of growth factors and as a differentiating agent for the ASCs that were administered locally. PRP contains various growth factors including transforming growth factor-beta (TGF-beta). These growth factors in PRP have been shown to have positive effects on growth and differentiation of various cells including chondrocyte formation [9-11]. Hyaluronic Acid (HA) and ECM were used as scaffolding materials [8,12]. Furthermore, we present a Magnetic Resonance Imaging (MRI) evidence of cartilage-like tissue regeneration in human hip OA patient using the ASCs mixture (ASCs and ECM in the form of adipose SVF along with HA and PRP activated by calcium chloride).
Case Report
The new rules and regulation by the KFDA has made it possible to use adipose tissue as a source of ASCs in Korea [1]. Informed patient consent was obtained prior to the surgical procedures. The approval and consent to report single cases were waived by the Myongji University Institutional Review Board committee (HSC) for case report. Further, this clinical study was in compliance with the Declaration of Helsinki and regulation guidelines of the KFDA.
Inclusion and exclusion criteria, and outcome endpoints
The inclusion criteria, exclusion criteria, and outcome endpoints are listed as follows. Inclusion criteria: MRI evidence of the hip OA; orthopaedic evaluation that determined that patient was a candidate for a Total Hip Replacement (THR) surgery; males or females; 50 years of age or over; unwillingness to proceed with THR; failure of conservative management; and on-going disabling pain. Exclusion criteria: active inflammatory or connective tissue disease thought to impact pain condition (i.e., lupus, rheumatoid arthritis, fibromyalgia); active endocrine disorder that might impact pain condition (i.e., hypothyroidism, diabetes); active neurologic disorder that might impact pain condition (i.e., peripheral neuropathy, multiple sclerosis); active cardiac disease; and active pulmonary disease requiring medications. Outcome endpoints: will be studied using pain score, physical therapy and MRI both pre- and post- treatment. The pain score and physical therapy will involve Visual Analog Scale (VAS), Functional Rating Index (FRI), and Range of Motion (ROM) that were determined as previously described [13,14].
Restriction of medications
Patient was restricted from taking steroids, aspirin, Non- Steroidal Anti-Inflammatory Drugs (NSAIDs), and Asian herbal medications for one week prior to the procedure.
Liposuction
In the operating room, approximately 50 ml of packed adipose tissue were obtained by liposuction of the subcutaneous layer of the lower abdominal area using manual techniques [5]. The adipose tissue was treated with collagenase (0.07% type 1, at 37°C for 40 min; Adilase, Worthington, Lakewood, NJ, USA) and the Stromal Vascular Fraction (SVF) containing ASCs was separated from the lipoaspirates by a fat stem cell isolator (CPLTM, Medicamatch, Ansan, Korea).
Extracellular matrix (ECM) preparation
In order to homogenize the lipoaspirates for even penetration of collagenase and also to obtain additional ECM, the packed adipose tissue was transferred back to two 60 ml Luer-Lock syringes connected to a manual homogenizer/ECM extractor (CPLTM, Medicamatch, Ansan, Korea), and were cut 20 times to mince the lipoaspirates.
Preparation of autologous adipose-derived stem cells (ASCs) mixture
ASCs were extracted through the use of digestive enzymes (0.07% type 1 collagenase; Adilase, Worthington, Lakewood, NJ, USA) and centrifugation (300 g for 5 min) to separate and remove collagenase [3-5]. The total volume of the solution containing ASCs and ECM was 8.5 ml from 50 ml of packed adipose tissue. While preparing the ASCs and ECM, 30 ml autologous blood were drawn along with 2.5 ml anticoagulant citrate dextrose solution (0.8% citric acid, 0.22% sodium citrate, and 0.223% dextrose; Baxter Healthcare Corp., Marion, NC, USA). After centrifugation (300 g at room temperature for 15 min, then 1200 g at room temperature for 5 min using supernatant), 4.4 ml of Platelet-Rich Plasma (PRP) along with the Buffy coat were obtained and 3% (w/v) calcium chloride (0.1 ml; Choongwae Pharmaceutical Co., Gyeonggido, Korea) was added to the mixture to activate PRP. 0.5% (w/v) hyaluronic acid (2 ml; Huons, Chungbuk, Korea) was added as a scaffold to this mixture. The ASCs mixture comprised of separated ASCs and ECM, together with activated PRP and Hyaluronic Acid (HA).
ASCs mixture-based treatment
After the hip was cleaned with 5% povidone-iodine (Choongwae Pharmaceutical Co., Seoul, Korea) and draped in a sterile fashion, the injection site was anesthetized with 0.25% ropivacaine (Huons, Chungbuk, Korea) superficially outside of joint capsule and diluted 0.125% lidocaine (Daehan Pharmaceutical Co., Gyeonggido, Korea) was used to anesthetize inside the joint capsule. On the same day of liposuction, the ASCs mixture (14 ml) was injected into the hip joint via anterior approach using a 90 mm, 18 gauge spinal needle under an ultrasound guidance. The patient was, then, instructed to remain still for 60 minutes to allow settlement of the ASCs mixture at the site of lesion [5,6]. Upon discharge from the clinic, the patient was instructed to exercise minimal activities for one week. The patient returned for three additional injections of PRP activated by calcium chloride at weekly intervals for three weeks.
Patient and treatment outcome
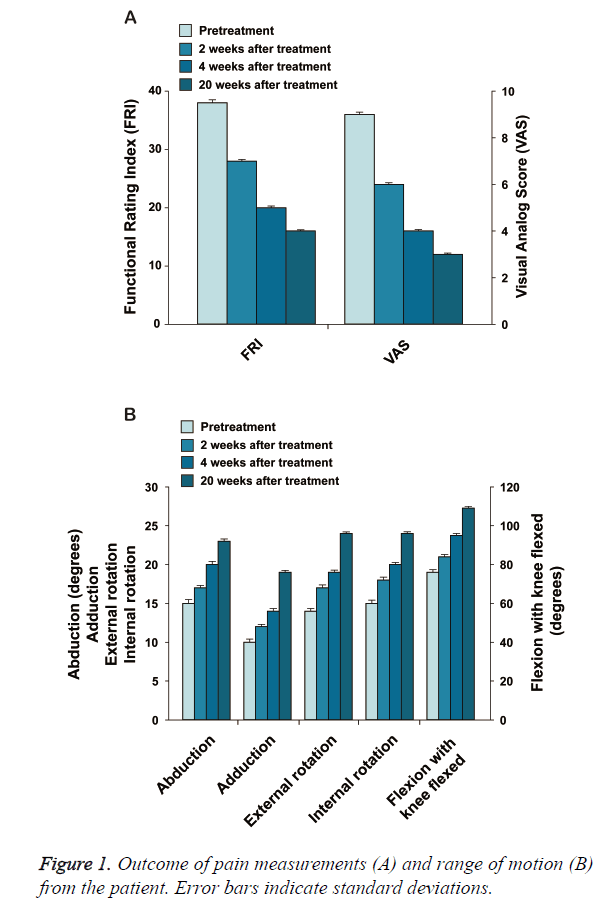
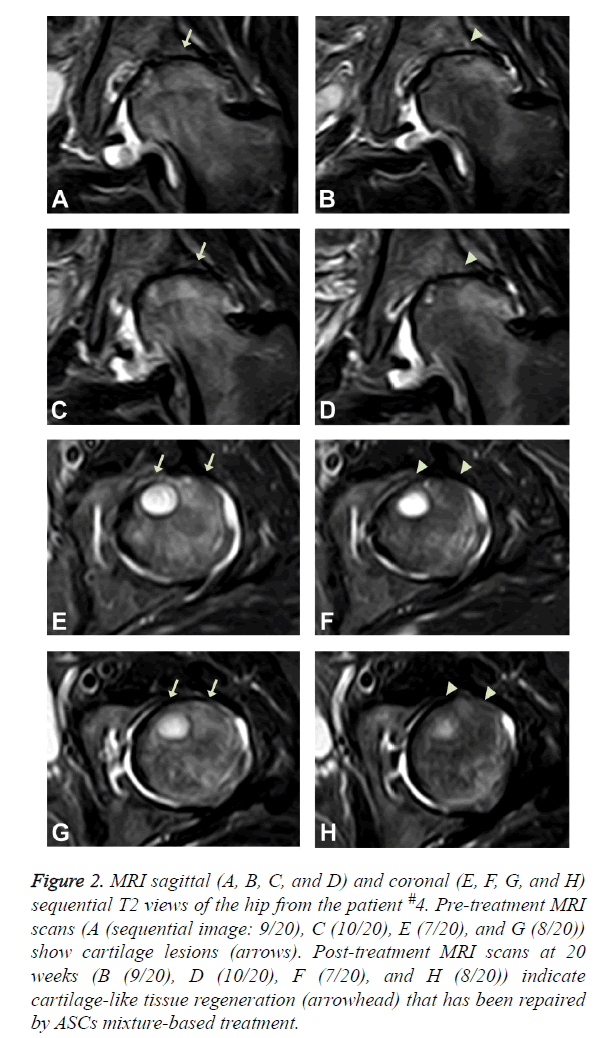
The patient is a 50-year-old Korean female with bilateral hip osteoarthritis. The patient had the chronic bilateral hip pain over 7 years that gradually increased. The patient was apparently better with use of chronic NSAID and occasional injections of steroids and HA. However, approximately one year prior to the office visit, the patient’s symptoms deteriorated. The patient underwent arthroscopic debridement/ lavage but was not completely relieved of pain. The patient noticed worsening of the pain after the procedure and was offered THR surgery by the orthopaedic surgeon. However, the patient was reluctant to go through THR surgery due to potential side effects. At the time of initial evaluation, the patient reported severe pain (VAS score: 9; Figure 1A and could not walk well without an assistant device. ROM (Figure 1B) was decreased. A pre-treatment MRI demonstrated joint space narrowing, irregular cortical margin with diffuse bone marrow oedema (Figures 2A, 2C, 2E, and 2G).
Figure 2: MRI sagittal (A, B, C, and D) and coronal (E, F, G, and H) sequential T2 views of the hip from the patient #4. Pre-treatment MRI scans (A (sequential image: 9/20), C (10/20), E (7/20), and G (8/20)) show cartilage lesions (arrows). Post-treatment MRI scans at 20 weeks (B (9/20), D (10/20), F (7/20), and H (8/20)) indicate cartilage-like tissue regeneration (arrowhead) that has been repaired by ASCs mixture-based treatment.
This patient went through the identical procedure of receiving autologous ASCs mixture as described in the Experimental Methods section. After the 2nd week of ASCs mixture injection, the patient’s pain improved over 30% and the ROM improved more than 20% (Figures 1A and 1B). By the 4th week, pain was relieved by more than 50% and ROM improved over 30% (Figures 1A and 1B). When the patient returned at 20 weeks after the injection, pain was relieved by more than 70% and ROM was improved by 50% (Figures 1A and 1B), and cartilage-like tissue regeneration was evident on the MRI (Figures 2B, 2D, 2F, and 2H).
Discussion
This clinical case report provides clear MRI evidence of cartilage-like tissue regeneration in OA of human hip by using the ASCs mixture. Based on MRI features, the newly formed tissue resembled that of cartilage; however, the true nature of the newly formed tissue cannot be confirmed without a biopsy. While cartilage regeneration in human knees using ASCs in the form of SVF have been shown in numerous human patients, this case report is the very first study suggesting possibly a better improvement in current strategy of cartilage regeneration by employing the ASCs mixture in human hip. In addition to the MRI evidence, the patient’s symptoms and signs improved as early as two weeks than previously reported by Pak [5]. The patient reported symptom improvement by the second week of the treatment. The patient's reported improvement in symptoms may probably be due to the presence of ECM and its factors in addition to ASCs.
ECM has been shown to work as a scaffold and shown to excrete various growth factors [8]. Thus, ECM can potentially improve the symptoms earlier by enhancing the cell adherence to the lesion. After the adherence, it is possible that the various growth factors released by the ECM can stimulate the adhered cells and surrounding tissue to grow. Therefore, the patient noticed the early clinical improvements. As shown previously, the patient did not report 100% resolution of their symptoms. This may be due to the fact that OA is a disease of the whole joint and not just cartilage. Thus, with improved cartilage regeneration, the patient may need to strengthen the related tendons, ligaments, and muscles to further improve already improved joint conditions.
Cartilage regeneration with adipose SVF depends on numerous factors: number of stem cells and their viability, adherence, growth and differentiation of the injected ASCs. The number of stem cells obtained from each individual patient is very much influenced by collagenase enzyme used. Stem cells exist within the ECM of the adipose tissue and by breaking down the ECM with collagenase; stem cells are released [3,4]. However, use of collagenase at high concentrations is detrimental to the stem cell viability, and lesser concentrations are not effective in releasing stem cells from the matrix [15]. Therefore, standardizing with correct mixture and dosage of the collagenase is very important. Standardization of the collagenase is further complicated by individual patient variations. The number of stem cells that can be obtained from each individual patient varies greatly [7]. Aging and the degree of obesity, for example, may affect the texture of subcutaneous tissue of each individual patient. Thus, collagenase may have different effects on each individual patient’s adipose tissue [16], and standardizing the correct amount of collagenase to be applied to general patient population would be very difficult, if not impossible. Since high concentration of collagenase may be detrimental to viability of stem cells, it would be a better strategy to use both collagenase at lower concentrations and homogenizer. Homogenizer helps to cut the fat tissue into small pieces which in turn facilitate even penetration of collagenase. With better and even penetration of collagenase, it is possible to obtain more stem cells and more ECM, although further verification is needed.
PRP was used as a source of growth factor and a differentiating factor for ASCs. PRP contains various growth factors including TGF-beta. These growth factors have shown to stimulate growth of the stem cells as well as differentiation of stem cells to chondrocytes [11]. Although HA was used as an additional scaffold for stem cells to attach to the lesion, symptom improvement can be attributed to the HA injected. It is very certain that HA probably played a role in improving the symptoms.
Although further studies are needed to verify this procedure presented in this report, this is the very first case report attempting to improve the current strategy of cartilage regeneration in human hip with percutaneous injection of the ASCs mixture. Currently, no non-surgical curative therapy is available for the treatment of osteoarthritis. Percutaneous injection of stem cell therapy with the ASCs mixture may therefore provide an alternative to current treatment strategy for the treatment of hip osteoarthritis.
Conclusion
In the present case, percutaneous injections of autologous ASCs mixture to hip OA provided significant relief from earlier symptoms. All clinical criteria of FRI, VAS score, and ROM improved on the patient, along with significant MRI changes. Although no biopsy of the regenerated tissue was performed, the tissue can be estimated to be cartilage-like in nature by comparing with the surrounding cartilage and other tissues. Although further studies are necessary to verify this new procedure, percutaneous injections of autologous ASCs mixture present a promising, minimally invasive option of treating OA of human hip by regenerating cartilage-like tissue.
Conflict of Interests
The authors declare that they have no competing interests.
Acknowledgements
The author acknowledges the support from the staff of Stems Medical Clinic. This work was supported by research grants from the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (No. 2011-0027928 and No. 2016R1C1B2010308) and Marine Biotechnology Program (20150581, Development of Technology for Biohydrogen Production using Hyperthermophilic Archaea) Funded by Ministry of Oceans and Fisheries in Republic of Korea.
References
- Korean Food and Drug Administration (KFDA). Cell therapy: Rules and regulations. KFDA 2009.
- Alexander RW. Understanding Adipose-Derived Stromal Vascular Fraction (AD-SVF) cell biology and use on the basis of cellular, chemical, structural and paracrine components: A concise review. J Prolother 2012; 4: e855-e869.
- Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, Benhaim P, Lorenz HP, Hedrick MH. Multilineage cells from human adipose tissue-implications for cell-based therapies. Tissue Eng 2001; 7: 211-228.
- Zuk PA, Zhu M, Ashjian P, De Ugarte DA, Huang JI, Mizuno H, Alfonso ZC, Fraser JK, Benhaim P, Hedrick MH. Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell 2002; 13: 4279-4295.
- Pak J. Regeneration of human bones in hip osteonecrosis and human cartilage in knee osteoarthritis with adipose tissue derived stem cells. J Med Case Rep 2011; 5: 296.
- Pak J, Lee JH, Kartolo WA, Lee SH. Cartilage regeneration in human with adipose tissue-derived stem cells-current status in clinical implications. Biomed Res Int 2016; 2016: 4702674.
- Baer PC, Geiger H. Adipose derived mesenchymal stromal/stem cells-tissue localization, characterization, and heterogeneity. Stem Cells Int 2012; 2012: 812693.
- Benders E, van Weeren PR, Badylak SF, Saris DB, Dhert WJ, Malda J. Extracellular matrix scaffolds for cartilage and bone regeneration. Trends Biotechnol 2013; 31: 169-176.
- Parsons P, Hesselden K, Butcher A, Maughan J, Milner R, Horner A. The biological effect of platelet rich-plasma on the fracture healing process. J Bone Joint Surg Br 2009; 91: 293.
- Li NY, Yuan RT, Chen T, Chen LQ, Jin XM. Effect of platelet-rich plasma and latissimus dorsi muscle flap on osteogenesis and vascularization of tissue-engineered bone in dog. J Oral Maxillofac Surg 2009; 67: 1850-1858.
- Wu W, Chen F, Liu Y, Ma Q, Mao T. Autologous injectable tissue-engineered cartilage by using platelet-rich plasma: experimental study in a rabbit model. J Oral Maxillofac Surg 2007; 65: 1951-1957.
- Uzuki M, Sawai T. A comparison of the affinity of sodium hyaluronate of various molecular weights for degenerated cartilage: A histochemical study using hyaluronic acid binding protein. Int Congr Ser 2001; 1223: 279-284.
- Childs JD, Piva SR. Psychometric properties of the functional rating index in patients with low back pain. Eur Spine J 2005; 14: 1008-1012.
- Price DD, McGrath PA, Rafii A, Buckingham B. The validation of visual analogue scales as ratio scale measures for chronic and experimental pain. Pain 1983; 17: 45-56.
- Pilgaard L, Lund P, Rasmussen JG, Fink T, Zachar V. Comparative analysis of highly defined proteases for the isolation of adipose tissue-derived stem cells. Regen Med 2008; 3: 705-715.
- Pagano C, Calcagno A, Giacomelli L, Poletti A, Macchi V, Vettor R, De Caro R, Federspil G. Molecular and morphometric description of adipose tissue during weight changes: a quantitative tool for assessment of tissue texture. Int J Mol Med 2004; 14: 897-902.