Research Article - Biomedical Research (2017) Volume 28, Issue 9
Effect of TiO2 with 0.3 wt (%) Ag as an additive on Pseudomonas aeruginosa under different sources of light: a qualitative analysis
Hima Bindu M1,2*, Indu Kapur1, Vijayaraghavan R31Department of Microbiology, Mallareddy Institute of Medical Sciences, India
2Department of Research, Saveetha University, India
3Research Director, Saveetha University, India
- *Corresponding Author:
- Hima Bindu M
Department of Microbiology
Mallareddy Institute of Medical Sciences, India
Accepted date: February 11, 2017
Abstract
Background: Titanium (Ti) and Ti alloys are known to have good mechanical properties with high corrosion resistance and excellent biocompatibility. Doping of silver ions onto a titanium dioxide based surface resulted in the movement of the absorption to a longer wavelength due to a change in the electronic and optical properties of TiO2 and thereby increasing the antibacterial effect.
Aim: This work was done to investigate the effectivity of nanoparticles of TiO2 doped with 0.3% wt of silver prepared by sol-gel technique, as an anti-bacterial agent against Pseudomonas aeruginosa from clinical samples.
Materials and Methods: TiO2 with 0.3% silver as additive was systematically assessed on 25 Pseudomonas aeruginosa isolates from various clinical samples and incubated in dark, in visible light and under UV radiation for the antibacterial effect. After each hour of incubation, the isolates were inoculated onto Mueller Hinton agar.
Results: The viable colonies diminished within 2 h of incubation under UV irradiation, while under visible light, the number of viable colonies was highly reduced after 3 h. Under dark, the reduction was considerably slower, taking 6 h, while in all the light conditions the control, which had no exposure to nanoparticles, showed no reduction in the number of colonies.
Conclusion: TiO2 with Ag as an additive is highly effective as an antibacterial agent on many human pathogens including Pseudomonas aeruginosa, which is notorious in causing hospital acquired infections. TiO2 with 0.3 wt (%) Ag is most effective under UV irradiation and least in dark.
Keywords
Additive, Antibacterial effect, Nanoparticles, Pseudomonas, Silver, Titanium dioxide (TiO2).
Introduction
Microbial infections, especially nosocomial infections are one of the most notorious causes of morbidity and mortality in the health industries. The patient suffering from a hospital acquired infection has a longer hospital stay and as a result, there is an increase in health care costs [1]. Lowering of the immune status of the patients especially due to a surgical intervention further enhances the chances of an infection.
Pseudomonas aeruginosa is a gram negative, non-sporing, non-capsulated, straight or slightly curved rod shaped bacterium, which may occur singly, in pairs or sometimes in short chains [2]. They are intrinsically highly resistant organisms known to occur even in disinfectants, respiratory equipment, common areas such as on taps, mops, sinks etc. It is also one of the notorious organisms that are capable of forming biofilms leading to a variety of hospital acquired infections [3].
Infections due to P. aeruginosa normally evolve to a pattern of chronic and persistent infections. The organisms undergo phenotypic changes frequently characterized by the production of a polysaccharide called alginate [4]. This phenotype called mucoid phenotype is associated with a difficulty in eradicating the organism, thereby eliciting a major inflammatory response and poor prognosis [4-6].
Titanium (Ti) and Ti alloys are known to have good mechanical properties with high corrosion resistance and excellent biocompatibility and are commonly found in orthopaedic prostheses, orthodontics, joint replacement and so on [7-10]. Although titanium products are seen very often in the dental appliances, its use in biomedical appliances has not been well studied.
Silver (Ag) is a non- specific, noble metal nanoparticle whose antibacterial effect has been well documented and is known to be a broad spectrum anti-bactericidal as well as a fungicidal [11]. It has a good stability in the environment as a result difficult for the microorganisms to develop resistance against it [12-14]. There is documentation of the therapeutic window of Ag to be small and larger doses are required for a cytotoxic effect [15,16].
It has been proposed that the photoactivity of Ago/Ag2O deposited onto TiO2 lead to the photoexcitation of Ag2O rather than Ago and acts as active sites which are responsible for the enhancement of the photocatalytic effect. Ago is said to contribute to the stability if the molecule [17]. These silver ions are known to cause degradation of proteins present in the bacterial cell walls and slow down the bacterial growth, and as a result have found variety of applications in the fields of biodiagnostics, manufacture of optical fibres etc. [18].
The most remarkable mechanism of antibacterial activity of silver nanoparticles was found to be the formation of free radicals which take part in the oxidative damage of the cell membranes of the bacteria [19,20]. Doping of silver ions onto a titanium dioxide based surface resulted in the movement of the absorption to a longer wavelength due to a change in the electronic and optical properties of TiO2. Also TiO2 was a very good supporting material for silver nanoparticles as it had a small crystal size and high surface area.
Much of the work has been done on the efficacy of nanoparticles in cleaning of water and environment and there is very little literature on the effect of silver doped titanium dioxide on pathogens from clinical samples.
Hence, this work was done to investigate the effectivity of nanoparticles of TiO2 doped with 0.3% wt of silver prepared by sol-gel technique, as an anti-bacterial agent against Pseudomonas aeruginosa from clinical samples.
Materials and Methods
This experimental study was conducted at Mallareddy Institute of Medical sciences, Hyderabad over a period of 2 and half years, between March 2014 and September 2016.
Initial preparation
A glass slide was cut into 3 equal parts with a glass cutter so that 3 equal squares of the glass slides are formed. On this, deposition of thin films of TiO2 doped with 0.3% wt of Ag was done by sol-gel method (Courtesy Nano-Ram Technologies, Bangalore). 3 Plain glass slides of the same size with no nanoparticle coatings were taken as controls. All the glass slides were properly labelled and covered with tissue paper and then with silver foil individually. They were all then placed in separate petri plates and sterilized in hot air over at 160˚C for 1h.
Collection of samples
Various clinical samples like urine, pus, sputum, swabs from various sites, were inoculated with aseptic measures, onto MacConkey agar and Blood Agar as per the regular protocol. All these plates were labeled properly and incubated overnight at 37˚C. Next day the growth was identified by the colony morphology on MacConkey and Blood agar and various biochemical reactions and their antibiograms were put up on Mueller Hinton Agar. The drug sensitivity pattern was identified according to CLSI guidelines. 25 samples of Pseudomonas aeruginosa which were sensitive to all the drug groups like ampicillin, cephalosporins, quinolones, etc. were included into the study.
All gram positive organisms and all gram negative bacilli other than P. aeruginosa were excluded from the study. Drug resistant P. aeruginosa especially those resistant to cephalosporins and metallo-beta lactam producers, were also excluded from the study.
Processing of P. aeruginosa on TiO2 slide
One colony of P. aeruginosa was inoculated onto sterile peptone water and incubated overnight at 37˚C. Next day the peptone water was adjusted to 0.5 MacFarland’s standard to make the working broth.
The 6 Petri plates, 3 in which TiO2 was coated and the 3 plain slides were taken and the covering papers were removed. 100 l was taken from the working broth and added onto all the slides aseptically. 50 l was taken from this by microtitre pipette and added onto sterile Mueller Hinton agar each and a well inoculum was made. Primary, secondary, tertiary and tail streaks were made from here and incubated at 37˚C overnight.
All the 6 slides were placed back in the respective plates. Of the three nano particle coated and control slides, one plate each was covered with a black paper and incubated at 37˚C in the incubator. Of the other two plates, one was incubated at 37˚C in visible light and the other in the BOD incubator at 37˚C with the UV light on.
After 1 h, all the slides were taken and 50 l sterile distilled water was added to each and mixed thoroughly. 50 l was taken from this solution and added to another set of sterile MHA plates, streaked and incubated at 37˚C overnight. The slides were once again processed and incubated as above. This process of adding the distilled water onto TiO2 coated and washing was repeated at 2, 4, 6 and 8 h. 50 l from this was taken each time and inoculated onto MHA plate and incubated at 37˚C overnight.
The growth was observed the next day.
Results
There were 5 sets of plates from 6 slides-TiO2 coated slide and control incubated in light, in dark and in UV light at 37˚C.
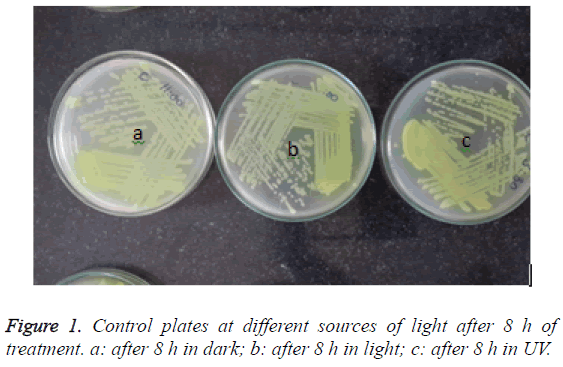
Out of them, the inoculum from the control slides showed confluent growth even up to the tertiary and tail of the streak lines, showing no reduction of the amount of viable bacteria over time (Figure 1). Even after 8 h of treatment at light or dark or under UV light, there was no reduction or difference in the growth pattern of the organism although there was a marginal reduction of growth in the plate which was incubated in the UV light.
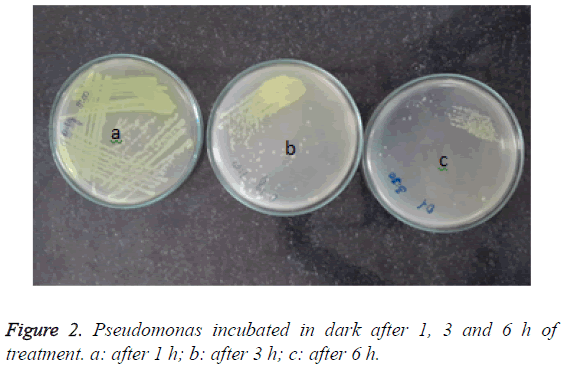
However, the reduction in the treatment of the organism with TiO2 doped with 0.3% wt Ag, remarkable reduction in the growth. After 3 h interval, growth was observed up to the tertiary streak lines in the plates with organisms incubated in the dark, however, there was slight growth in the well only after 6 h of treatment Figure 2.
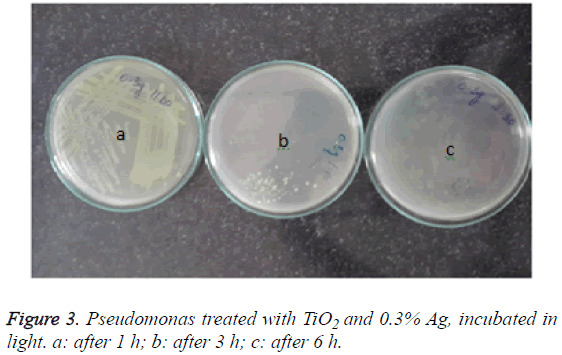
In the plates with nanoparticles incubated under visible light, less than 100 colonies were observed after 3 h of treatment itself and no growth was seen after 6 h (Figure 3).
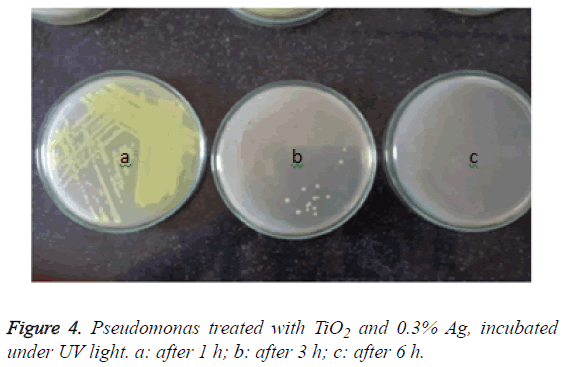
However, in the plates with nanoparticles incubated under ultraviolet light, no growth of the organisms were seen in the 3rd hour itself, while around 10 colonies were seen after the incubation of 2 h (Figure 4).
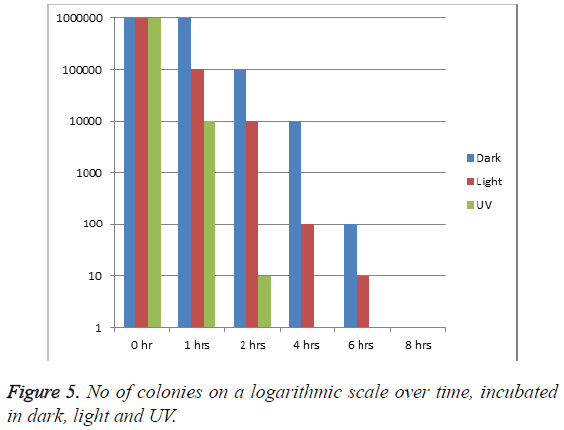
When the logarithmic scale of the organism was taken, it was observed that the number of colony forming units/ml at the start of the study in each condition of incubation was>106. However, after one hour of incubation, it was observed that the number of colonies reduced to one third (around 10000 cfu/ml) when incubated under UV, while it remained almost constant when incubated in dark. Within 2 h, the cfu/ml was reduced to 10 log while it took 6 h when incubated in light (Figure 5). 100% antibacterial effect was observed within 4 h when incubated in UV, while it took over 6 h when incubated in light, at which time there were double the number of organism in dark as compared to those in light.
Discussion
Implants made up of titanium dioxide have been highly used for some time now in many practices in medicine, such as knee implants, dental implants, screws for orthopaedic surgeries, pacemakers etc.
Nanoparticles are said to exhibit very strong inhibitory effect towards a large number of bacterial strains. Metal oxides according to many studies are believed to be carrying a positive charge while the cell walls of the microorganisms carry a negative charge. As a result, there is an electromagnetic effect between these two structures, leading to oxidization and ultimately the death of the organism [21].
Pseudomonas is associated with many hospital acquired infections including biofilms formations in conditions such as cystic fibrosis and in ventilators. Evaluation of effectivity of nanoparticles on resistant bacteria such as Pseudomonas, Staphylococcus aureus etc., is becoming very difficult and hence a reason for many studies [22].
The present study by treating with nanoparticles has shown a marked reduction of Pseudomonas aeruginosa under all the sources of light i.e. in dark, in visible light as well as under UV illumination within 6 h of treatment. In fact with UV illumination, there was no growth of viable bacteria within the 3rd hour of treatment itself, showing that the nanoparticles act better at UV light rather than under visible light and in dark.
Although it is estimated that light is necessary for the excitation of silver ions for antibacterial effect, it is observed that even under dark, the efficacy of the nanoparticles is maintained.
In a study by Sangchay et al. it was observed that E. coli was eliminated much faster by TiO2 doped with Ag under UV radiation [23]. More over as the exposure to UV radiation increased, so did the efficacy of the nanoparticles.
ATCC strains of water pollutants such as E. coli and Klebsiella spp were tested against TiO2 doped with ZnO under UV irradiation as well as under visible light. It was observed that there was considerable reduction in the growth of the microorganisms when incubated with TiO2 or in combination with ZnO in UV or in visible light which corresponded to the results of our study. This extent of the bacterial inhibition depended on the concentration of the ZnO particles and also the nature of the electromagnetic spectrum [24]. However in a study with Cu as the dopant, there was no difference in the number of viable counts of the fungal pathogens such as Candida with increase in the concentration of Cu, while there was a considerable decrease in the bacterial counts [25].
In yet another study by Prasad et al. Ag and its various concentrations on TiO2 showed high antibacterial effect against the bacterial and fungal water pathogens. The results were in accordance to our study on clinical pathogens.
The study of nanomedicine and its applications, although has been known for a very long time, has taken enormous proportions only recently. There has been a tremendous amount of work in the field of water sterilization and environmental cleaning. Silver ions have been used in dental implants for a very long time now with spectacular success, though only of later, the use of Ag in the form of nanoparticles has spiked the interest of the researchers.
Our study has used the clinical samples which are capable of causing infections in the human body at various sites and observed the effect of TiO2 with Ag as an additive. This present study has not been done on the metal implants in vivo, which can be the topic of interest in further studies in future.
Conclusion
TiO2 with Ag as an additive is highly effective as an antibacterial agent on many human pathogens including Pseudomonas aeruginosa, which is notorious in causing hospital acquired infections. TiO2 with 0.3 wt (%) Ag is most effective under UV irradiation and least in dark.
As today, most of the technology has become nano-oriented; we should also modulate our treatment to incorporate the new trends. Thus, more such tests on different surfaces and their effects in the human body must be carried out in future so that newer developments in science and medicine can be make our quality of life better.
References
- Burke JP. Infection control-a problem for patient safety. N Engl J Med 2003; 348: 651-656.
- Todar K. Todars online book of bacteriology: http://textbookof bacteriology 1.net pseudomonas.html 2011.
- Murray PR, Rosenthal KS, Kobayashi GS, Pfaller MA. Medical microbiology. Philadelphia Elsevier Mosby (4th Edn.) 2002; 278-303.
- Govan JR, Deretic V. Microbial pathogenesis in cystic fibrosis: mucoid Pseudomonas aeruginosa and Burkholderiacepacia. Microbiol Rev 1996; 60: 539-574.
- Nixon GM, Armstrong DS, Carzino R, Carlin JB, Olinsky A, Robertson CF. Clinical outcome after early Pseudomonas aeruginosa infection in cystic fibrosis. J Pediatr 2001; 138: 699-704.
- Henry RL, Mellis CM, Petrovic L. Mucoid Pseudomonas aeruginosa is a marker of poor survival in cystic fibrosis. Pediatr Pulmonol 1992; 12: 158-161.
- Geetha M, Singh AK, Asokamani R, Gogia AK. Ti based biomaterials, the ultimate choice for orthopaedicimplantsea review. Prog Mater Sci 2009; 54: 397-425.
- Niinomi M. Mechanical biocompatibilities of titanium alloys for biomedical applications. J Mech Behav Biomed Mater 2008; 1: 30-42.
- Puleo DA, Kissling RA, Sheu MS. A technique to immobilize bioactive proteins, including bone morphogenetic protein-4 (BMP-4), on titanium alloy. Biomaterials 2002; 23: 2079-2087.
- Cioffi M, Gilliland D, Ceccone G, Chiesa R, Cigada A. Electrochemical release testing of nickel-titanium orthodontic wires in artificial saliva using thin layer activation. Acta Biomater 2005; 1: 717.
- Burke JP. Infection control-a problem for patient safety. N Engl J Med 2003; 348: 651-656.
- Hardes J, Ahrens H, Gebert C, Streitbuerger A, Buerger H. Lack of toxicological side-effects in silver-coated megaprostheses in humans. Biomaterials 2007; 28: 2869-2875.
- Zhao LZ, Wang HR, Huo KF, Cui LY, Zhang WR, Ni HW. Antibacterial nano-structured titania coating incorporated with silver nanoparticles. Biomaterials 2011; 32: 57.
- Zheng Y, Li J, Liu X, Sun J. Antimicrobial and osteogenic effect of Ag-implanted titanium with a nanostructured surface. Int J Nanomedicine 2012; 7: 875-884.
- Greulich C, Braun D, Peetsch A, Diendorf J, Siebers B, Epple M. The toxic effect of silver ions and silver nanoparticles towards bacteria and human cells occurs in the same concentration range. RSC Adv 2012; 2: 6981.
- Chernousova S, Epple M. Silver as antibacterial agent: ion, nanoparticle, and metal. Angew Chem Int Ed Engl 2013; 52: 1636-1653.
- Zhang H, Wang G, Chen D, Lv X, Li J. Tuning photoelectrochemical performances of Ag-TiO2 nanocomposites via reduction/oxidation of Ag. Chem Mater 2008; 6543-6549.
- Pin-Ching M, Sharon S, Daniel MB, Zheng H, Edward JW, William AJ. Bactericidal activity of photocatalytic tio2 reaction: toward an understanding of its killing mechanism. Appl Environ Microbial 1999; 65: 4094-4098.
- Kaur P, Saxena M, Vadehra DV. Plasmid mediated resistance to silver ions in Escherichia coli. Indian J Med Res 1985; 82: 122-126.
- Hwang ET, Lee JH, Chae YJ, Kim YS, Kim BC, Sang BI, Gu MB. Analysis of the toxic mode of action of silver nanoparticles using stress-specific bioluminescent bacteria. Small 2008; 4: 746-750.
- Zhang H, Chen G. Potent antibacterial activities of Ag/TiO2 nanocomposites powders synthesized by a one-potsolgel method. Environ Sci Technol 2009; 34: 2905-2910.
- Livia V, Luigi DN, Carlo P, Lucio M, Alberto C, Marcello I, Carla RA. Titanium oxide antibacterial surfaces in biomedical Devices. Int J Artif Organs 2011; 34: 929-946.
- Sangchay W, Rattanakun T. Photocatalytic, antibacterial and self-cleaning properties of TiO2 and Ag doped TiO2 thin films using sol-gel method. RMUTP Res J 2016.
- Sophee SS, Prasad RGSV, Srinivas JV, Aparna RSL, Phani AR. Antibacterial activity of TiO2 and ZnO microparticles combination on water polluting bacteria. J Green Sci Technol 2012; 1: 1-7.
- Prasad RGSV, Basavaraju D, Rao KN, Naveen CS, Endrino J, Phani A. Nanostructured TiO2 and TiO2-Ag antimicrobial thin films. IEEE Nanosci Technol Soc Impl 2011.