Research Article - Biomedical Research (2017) Volume 28, Issue 12
Diagnosis and treatment of severe acute radiation pneumonitis
Jia-Long Tao#, Feng-yun Zhong#, Si-jia Chen, Hui Wang, Xue Chu and Yu-Song Zhang*
Department of Oncology, the Second Affiliated Hospital of Soochow University, Suzhou, Jiangsu, PR China
#These authors contributed equally to this work
- *Corresponding Author:
- Yu-song Zhang
Department of Oncology
The Second Affiliated Hospital of Soochow University, PR China
Accepted on May 4, 2017
Abstract
Concurrent radiotherapy and chemotherapy is the main treatment for locally advanced non-small cell lung cancer. Acute Radiation Pneumonitis (ARP) is one of the most important complications of radiotherapy for lung cancer and is difficult to treat and causes high mortality rates. In this study, we reported a patient with grade IV ARP who underwent 2 months of radiotherapy. This patient showed significantly different clinical features from common ARP, he had early symptoms of illness without evident signs and imaging manifestations, rapid short-term illness progression, wide range of lesions, and bystander effect. We described our experience in the diagnosis and treatment of ARP, and discussed the importance of early prevention of ARP during radiotherapy.
Keywords
Radiotherapy, Lung cancer, Acute radiation pneumonitis, Diagnosis, Treatment.
Introduction
Concurrent radiotherapy and chemotherapy is the main treatment for locally advanced non-small cell lung cancer. Acute Radiation Pneumonitis (ARP) is one of the most important complications of radiotherapy for lung cancer and is also considered the major dose-limiting factor. Grades I-III ARP is commonly observed in clinical setting, while grade IV ARP is rarely detected but causes serious respiratory malfunction, incomplete oxygen inhalation, or assisted ventilation [1]. ARP is difficult to treat and causes high mortality rates.
In this study, we reported a patient with grade IV ARP who underwent 2 months of radiotherapy. This patient showed significantly different clinical features from common ARP, he had early symptoms of illness without evident signs and imaging manifestations, rapid short-term illness progression, wide range of lesions, and bystander effect. We discussed our experience in the diagnosis and treatment of ARP.
Subject and Methods
Subject
This study was approved by Institution Ethics Committee and the patient provided signed consent. The patient was a 78 y old male who experienced recurring cough in January 2014 and manifested blood in the sputum in March 2014. He had no diabetes history but had hypertension for 10 y. He was taking anti-hypertensive drugs, and his blood pressure was kept at ideal levels. He had history of Chronic Obstructive Pulmonary Disease (COPD) and smoking but no history of recurrent attacks and allergy.
Diagnosis and treatment
X-CT and biopsy were performed for the diagnosis of lung cancer. Paclitaxel+carboplatin (TC regimen) chemotherapy was administered twice on April 19 and May 18, 2014. From 2014 April 22 to June 10, radiotherapy of lung lesions and specific X-ray 3DCRT DT 7000cGy/35f/49d were performed, and chemotherapy was continued. The patient was followed by X-CT, physical examination and blood gas analysis, and received corresponding treatment.
Results
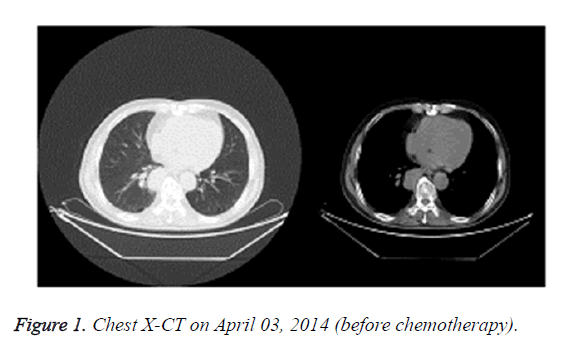
In April 2014, X-CT showed that the right lower lung lobe contained lesions with hilar lymph node metastasis (Figure 1). Endoscopic biopsy of trachea and bronchus was then performed and pathological analysis revealed squamous cell carcinoma. The patient was diagnosed with squamous cell carcinoma of the right lung (stage IIIA cT3N1M0).
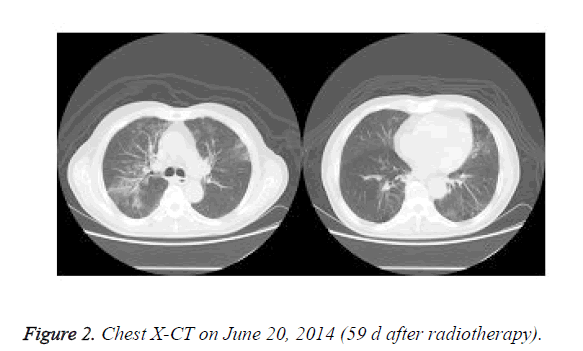
Paclitaxel+carboplatin (TC regimen) chemotherapy was administered twice on April 19 and May 18, 2014. From 2014 April 22 to June 10, radiotherapy of lung lesions and specific X-ray 3DCRT DT 7000cGy/35f/49d were performed. After the radiotherapy, the patient manifested frequent dry cough, fever, and occasional bloody sputum. In June 2014, X-CT showed enlarged right lung and mediastinal lymph nodes, fusion and calcification of the right hilum, pneumonia, emphysema, and pulmonary multiple bullae (Figure 2). Lung cancer was significantly reduced after radiotherapy and chemotherapy.
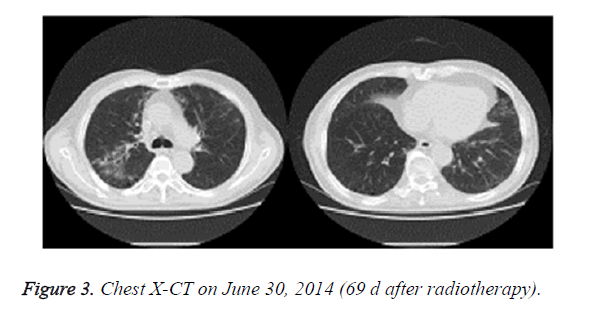
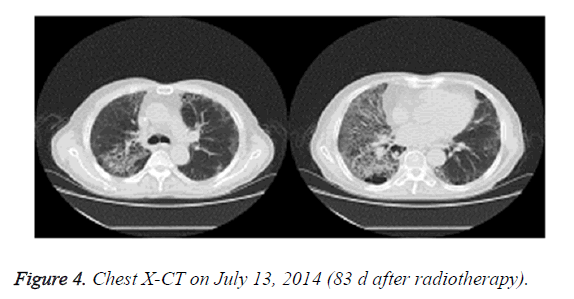
On June 30, 2014, the patient showed apparent chest tightness, shortness of breath, and limited activity, as well cough, less phlegm, thick sound of the two lungs when breathing, and Velcro rales were detected at the bottom of the two lungs. Chest X-CT showed slight inflammation of the two lungs (Figure 3). After 2 weeks of anti-inflammatory treatment, the condition of the patient did not significantly improve and the symptoms gradually worsened. On July 13, 2014, chest X-CT showed a large area of lung inflammation (interstitial based) (Figure 4). Blood gas analysis results were consistent with type I respiratory failure. No abnormality was observed in ECG and cardiac ultrasound examination. BNP and quantitative myocardial infarction were normal. Sputum culture and sputum smear examination showed no abnormality. Lung function analysis showed moderate and restrictive ventilation dysfunction. Based on these, the patient was diagnosed with grade IV ARP and type I respiratory failure, which occurred after radiotherapy and chemotherapy for lung cancer.
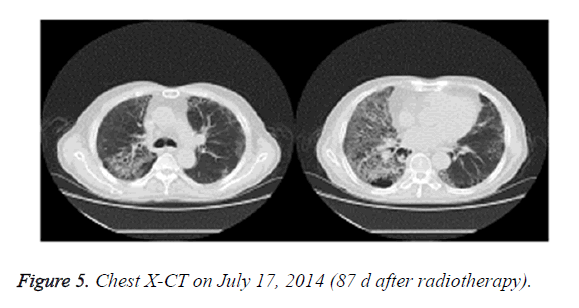
On July 14, 2014, the patient received 40 mg urbason for shock therapy, 100 ml ofloxacin for anti-inflammation therapy, and noninvasive assisted ventilation. On July 17, 2014, the patient complained of dyspnea symptoms, and chest X-CT showed that the right lung lesions were enlarged and the left lung disease did not improve (Figure 5). The patient was then treated with 5 mg dexamethasone and high doses of hormones, and showed thickened tongue for about a week. Meanwhile, examination of sputum culture, as well as sputum and urine fungal smear, showed no bacterial and fungal infection. After radiotherapy and chemotherapy, the patient presented low immune function and was given large doses of hormone therapy. The patient was given 4% sodium bicarbonate gargle and oral nystatin prophylaxis to prevent fungal infection. As large doses of hormone may induce adverse side effects, the patient was given decreased dosage of urbason (40 mg) and dexamethasone (5 mg).
In July 27, 2014, the patient presented progressive cough, white foam sputum, and wide range of Velcro rales from the two lungs. Blood gas analysis (oxygen concentration 33%) results were as follows: oxygen partial pressure, 75%; carbon dioxide partial pressure and oxygen partial pressure/oxygen concentration, 2.3 mmHg; oxygen saturation, 95%. These results showed no significant changes compared to those at the time of admission. Examination of sputum culture, as well as sputum and urine fungal smear, showed no bacterial and fungal infection. Therefore, the patient was given piperacillin tazobactam, instead of ofloxacin, as anti-inflammatory treatment.
On August 1, 2014, the patient manifested a fever of 39°C and worsened dyspnea symptoms. Sputum culture results were positive for Staphylococcus saprophyticus, Acinetobacter and Bacillus. Blood culture results demonstrated no bacterial growth. Since pulmonary infection worsened we recommended biapenem for anti-inflammatory treatment. The patient was advised to stay in the ICU for further treatment, but his family refused. The patient died of pulmonary infection and respiratory failure on the same day at 9:00 p.m.
Discussion
The patient underwent concurrent radiotherapy and chemotherapy for lung cancer for 2 months. The patient had a history of COPD and showed evident symptoms of dyspnea and Velcro rales from the two lungs. CT indicated that the quality of the large area was interstitially changed, consistent with the typical clinical manifestations of radiation pneumonitis. Eliminating pathogens that caused pulmonary infection is therefore necessary. The patient presented with cough and sputum, chest tightness and shortness of breath, but had no fever and sputum culture was negative. Thus we ruled out pulmonary infection and anti-infection treatment was considered ineffective.
Ionizing radiation is known to induce bystander effect [2,3]. In present study the patient had two pulmonary diffused interstitial changes after radiotherapy. The symptoms of the patient did not match those of radiotherapy radiation pneumonitis, which usually occurs in the radiation field. In particular, the left lung was not subjected to radiation exposure but showed diffuse interstitial damage. The patient presented no other possible factors that may cause interstitial pneumonia. Thus, we should consider the bystander effect induced by ionizing radiation. Ionizing radiation from field-emission radiotherapy causes injury in lung tissues, but the related mechanism through which this process induces similar damages to non-irradiated parts of the lung tissue remains unclear.
ARP is a kind of lymphocytic inflammatory reaction caused by direct injury and is generally regarded as irreversible. According to the evaluation criteria of RTOG, lung injury in lung cancer that occurs within 90 d after the radiotherapy is called ARP, whereas injury that manifested beyond 90 d is called advanced damage. Clinical factors associated with ARP include the age, gender, ECOG score, chronic obstructive lung disease history, smoking history, tumor site and clinical stage. Several studies showed that female, concurrent chemoradiotherapy, ECOG score>2, poor lung function, COPD, age, primary tumor located in the lower lobe of the lung, and smoking increased the risk of ARP occurrence [4-8]. Jin et al. retrospectively analysed ARP risk factors of 576 patients with NSCLC and revealed that a long history of smoking was a valuable clinical factor [9]. Therefore, patients with lung cancer and ARP associated risk factors should be subjected to carefully optimized radiotherapy plan and effective prevention measures.
Administering chemotherapy drugs and radiation therapy at the same time increases the radiation sensitivity of normal tissues. Previous studies showed that the use of cyclophosphamide and gemcitabine increased the incidence of RP. Robnett et al. reported that paclitaxel-based chemotherapy did not increase the risk of RP and had no significant relationship to RP [10]. It is suggested that the risk of radiation-induced lung injury can be increased by the frequency of concurrent chemotherapy, and chemotherapy cycles higher than 5 could be correlated with ARP. Leprieur et al. suggested that the use of gemcitabine before radiation should be avoided to decrease the risk of ARP [11]. Nevertheless, no correlation exists between the dose of chemotherapeutic drugs and the occurrence of ARP. In current study, the patient was treated with synchronous radiotherapy and chemotherapy, but paclitaxel and carboplatin did not increase the incidence of ARP. However, patients who underwent radiotherapy with sequential chemotherapy may present reduced incidence of ARP. Therefore, patients with high risk factors of ARP may endeavor sequential treatment with chemotherapy cycles less than five.
The mechanism of the bystander effect of radiation-induced lung injury remains unclear although Reactive Oxygen Species (ROS), soluble cellular factors and gap junction intercellular communication may be involved [12,13]. In this study, the bystander effect occurred in lung tissues adjacent to irradiated tissues and in remote areas of the patient, suggesting the involvement of all the above mechanisms. Further studies are needed to understand the mechanisms responsible for bystander effect of radiation to develop prevention and early treatment regimens for radiation-induced lung injury.
The most important step for the prevention of ARP is to control radiation dose. Permanent radiation damage of normal lung tissues will appear after irradiation with 2,000 Gy, and acute exudation inflammation will occur after irradiation with 3,000-4,000 Gy. According to chest X-ray or CT images, the patient was diagnosed with ARP in the irradiation site, and the radiation field was consistent with the blurred shadow or a patchy shadow in the lung. The patient showed significantly worsened dyspnea one month after radiotherapy. We prescribed large-dose hormone therapy and treated the condition as common pneumonia. After 2 weeks, CT image showed a large area of a rope-like shadow, although the diagnosis of ARP remained inconclusive. The optimal treatment time was therefore missed. During radiotherapy, the symptoms such as cough and shortness of breath should be closely observed. Once these symptoms appear, lung CT scan should be performed. Hormone treatment should be immediately terminated when a slight glass shadow appears near the pleural and replaced with glucocorticoid. Otherwise the disease will continue to progress, eventually leading to respiratory failure.
RP is mainly treated by using large doses of hormones, and the treatment course can take two months or more. Long-term use of corticosteroids can cause metabolism disorders and affect the function of adrenal cortex. Symptoms such as edema, low blood potassium, hypertension, diabetes, skin thinning, full moon face, buffalo back, central obesity, hairy, acne, muscle weakness, and muscle atrophy generally do not require special treatment. Long-term use of corticosteroids can also cause fungal infection, and sodium bicarbonate gargle should be employed to prevent fungal infection. Prompt treatment with anti-fungal agents is necessary after the detection of fungal infection. Long-term use of corticosteroids can further cause stomach ulcers and bleeding, thus proton pump inhibitors should be used to protect gastric mucosa, and fecal occult blood should be monitored to detect chronic hemorrhage. Osteoporosis and vertebral compression fractures are serious complications observed in elder patients with glucocorticoid treatment. Vitamin D, calcium salt, and protein assimilation hormones should be given as appropriate supplements for the prevention and treatment of osteoporosis.
In conclusion, it is important for the early diagnosis of ARP. Patients with severe ARP and respiratory insufficiency/ continuous oxygen inhalation or assisted ventilation should undergo blood gas analysis to detect respiratory failure. For patients with a large amount of sputum secretion from pulmonary infection prone to asphyxia, tracheotomy-assisted ventilation should be performed to relieve ARP.
Acknowledgments
This study was supported by grants from Suzhou Science and Technology Program (No. SYS201345), Bureau of Public Health of Jiangsu Province (No. H201413), the second affiliated hospital of Soochow university clinical discipline group project funding (No. XKQ 2015008) and the second affiliated hospital of Soochow university pre-research project (No. SDFEYGJ1609).
Conflict of Interest
None
References
- Provatopoulou X, Athanasiou E, Gounaris A. Predictive markers of radiation pneumonitis. Anticancer Res 2008; 28: 2421-2432.
- Wang H, Yu KN, Hou J, Liu Q, Han W. Radiation-induced bystander effect: early process and rapid assessment. Cancer Lett 2015; 356: 137-144.
- Nagasawa H, Little JB. Induction of sister chromatid exchanges by extremely low doses of alpha-particles. Cancer Res 1992; 52: 6394-6396.
- Moreno M, Aristu J, Ramos LI, Arbea L, Lopez-Picazo JM, Cambeiro M, Martínez-Monge R. Predictive factors for radiation-induced pulmonarytoxicity after three-dimensional conformal chemoradiation in locally advanced non-small-cell lung cancer. Clin Transl Oncol 2007; 9: 596-602.
- Hope AJ, Lindsay PE, El Naqa I, Alaly JR, Vicic M, Bradley JD, Deasy JO. Modeling radiation pneumonitis risk with clinical, dosimetric, and spatial parameters. Int J Radiat Oncol Biol Phys 2006; 65: 112-124.
- Cui Z, Tian Y, He B, Li H, Li D, Liu J, Cai H, Lou J, Jiang H, Shen X, Peng K. Associated factors of radiation pneumonitis induced by precise radiotherapy in 186 elderly patients with esophageal cancer. Int J Clin Exp Med 2015; 8: 16646-16651.
- Kong FM, Hayman JA, Griffith KA, Kalemkerian GP, Arenberg D, Lyons S, Turrisi A, Lichter A, Fraass B, Eisbruch A, Lawrence TS, Ten Haken RK. Final toxicity results of a radiation-dose escalation study in patients with non-small-cell lung cancer (NSCLC): predictors for radiation pneumonitis and fibrosis. Int J Radiat Oncol Biol Phys 2006; 65: 1075-1086.
- Dang J, Li G, Lu X, Yao L, Zhang S, Yu Z. Analysis of related factors associated with radiation pneumonitis in patients with locally advanced non-small-cell lung cancer treated with three-dimensional conformal radiotherapy. J Cancer Res Clin Oncol 2010; 136: 116 9-1178.
- Jin H, Tucker SL, Liu HH, Wei X, Yom SS, Wang S, Komaki R, Chen Y, Martel MK, Mohan R, Cox JD, Liao Z. Dose-volume thresholds and smoking status for the risk of treatment-related pneumonitis in inoperable non-small cell lung cancer treated with definitive radiotherapy. Radiother Oncol 2009; 91: 427-432.
- Robnett TJ, Machtay M, Vines EF, McKenna MG, Algazy KM, McKenna WG. Factors predicting severe radiation pneumonitis in patients receiving definitive chemoradiation for lung cancer. Int J Radiat Oncol Biol Phys 2000; 48: 89-94.
- Leprieur EG, Fernandez D, Chatellier G, Klotz S, Giraud P, Durdux C. Acute radiation pneumonitis after conformational radiotherapy for nonsmall cell lung cancer: clinical, dosimetric, and associated-treatment risk factors. J Cancer Res Ther 2013; 9: 447-451.
- Wang X, Zhang J, Fu J, Wang J, Ye S. Role of ROS-mediated autophagy in radiation-induced bystander effect of hepatoma cells. Int J Radiat Biol 2015; 91: 452-458.
- Hattori Y, Suzuki M, Funayama T, Kobayashi Y, Yokoya A, Watanabe R. A mathematical model of radiation-induced responses in a cellular population including cell-to-cell communications. Radiat Prot Dosimetry 2015; 166: 142-147.