Research Article - Biomedical Research (2017) Volume 28, Issue 21
Correlation analysis of ADAMTS-4, VCAM-1, and TAK1 expression in cartilage tissue from osteoarthritis patients
Xiaodong Yu1, Hua Yin2, Ning Dong2, Huanli Zhao1, Qian Li1 and Jingjun Wei3*
1Department of Emergency, Linyi People's Hospital, Linyi, Shandong, China
2Department of Neurosurgery, Linyi Traditional Chinese Medicine Hospital, Linyi, Shandong, China
3Department of Medical Examination, Linyi People's Hospital, Linyi, Shandong, China
Accepted on September 13, 2017
Abstract
Objective: Osteoarthritis (OA) is a chronic progressive lesions caused by multiple factors. Previous finding showed that the expressions of ADAMTS-4, VCAM-1, and TAK1 were increased in the cartilage tissue from OA compared with normal tissue. ADAMTS-4 was positively correlated with TAK1 level. VCAM-1 expression was related to severity of OA. It was found that ADAMTS-4 and VCAM-1 can influence the dynamic balance of ECM, while TAK1 may participate in OA through MAPKs and NF-κB signaling pathways. However, the correlation of their expressions in cartilage tissue from patients with OA is still unclear. This study intends to analyse their correlation in OA cartilage through the detection of expressions in OA and normal cartilage.
Patients and methods: A total of 72 OA patients were enrolled and divided into two groups: severe OA and mild-to-moderate OA groups. Another 22 healthy individuals were selected as normal control. Western blot was applied to detect ADAMTS-4, VCAM-1, and TAK1 expression in cartilage tissue. Their correlations were then analysed.
Results: There were 35 patients in severe group with mean age at 58.64 ± 4.32 y old, while 37 subjects in mild-to-moderate group with average age at 57.32 ± 5.21 y old, the ages of which were no significant different. The levels of ADAMTS-4, VCAM-1, and TAK1 in severe group were obviously higher than that in mild-to-moderate group and normal control. The levels of these three genes presented positive correlation with each other.
Conclusions: The expressions of ADAMTS-4, VCAM-1, and TAK1 were elevated and exhibited positive correlation in cartilage tissues of patients with OA, which provides academic basis for the future biomarkers in the therapy of OA.
Keywords
Osteoarthritis, ADAMTS-4, TAK1, Cartilage tissue, VCAM-1.
Introduction
With the growth of aging population in China, the incidence of Osteoarthritis (OA) keeps on increasing, which seriously influence the patient's psychological health, physical health, and normal life [1]. As a type of chronic progressive joint cartilage degenerative lesions, OA occurrence is accompanied by a series of physiological and pathological processes including inflammation and immune reaction. It is mainly featured as cartilage Extracellular Matrix (ECM) degradation and articular cartilage loss in clinic, of which the dynamic imbalance of bone ECM metabolism is the major cause [2].
It was showed that a disintegrin and metalloproteinase with a thrombospondin (ADAMTS) can promote the degradation of ECM and participate in the maintenance of ECM dynamic balance. However, the aberrant expression of ADAMTS may directly induce imbalance of ECM. Moreover, it was found that ADAMTS-4 was overexpressed in OA compared with normal people [3-6].
Vascular Cell Adhesion Molecule 1 (VCAM-1) is a kind of important cell surface glycoprotein and is characterized as regulating cell adhesion between cells. It was revealed that VCAM-1 level was significantly higher in serum and cartilage tissue of patients with OA than that in normal ones. Meanwhile, VCAM-1 expression showed certain correlation with the severity of OA and could be treated as one of the predictors of the onset risk of OA [7-9].
Accumulative studies demonstrated that as one of the member of Mitogen-Activated Protein Kinase (MAPKs) family, transforming growth factor β activated kinase 1 (TAK1) was not only related to the immune and inflammation, but also involved in regulation of cell proliferation, differentiation, and apoptosis. Furthermore, it participates in the regulation of a variety of signaling pathways, such as MAPKs and NF-κB. Experimental data demonstrated that TAK1 was highly expressed in the cartilage tissue of OA patients, thus related to the course of OA [10-13].
As the expressions of ADAMTS-4, VCAM-1 and TAK1 showed certain association with OA occurrence and development, this study aimed to clarify the interaction and correlation among the three proteins by detecting their expressions in the cartilage tissue from patients with OA, thus to provide effective biomarkers for the early diagnosis of OA.
Materials and Methods
Materials
Goat anti-human monoclonal antibodies for TAK1, ADAMTS-4 and VCAM-1 were bought from Xinlebio. Mouse anti-goat secondary antibody was purchased from Santa Cruz. EP tube was from Axygen. The washing buffer for Western blot was from Beyotime. ECL reagent was from Thermo fisher. Transfer buffer was from Bio-Rad. Lysis buffer was from Shanghai Yili biochemical reagent co., Ltd. SDS-PAGE automatic exposure meter was from ImageQuant™ LAS 500 integration imaging.
Clinical information
A total of 72 OA patients in Linyi People's Hospital from October 2015 to October 2016 were enrolled. Exclusion criteria: patients with tumor, acute or chronic arthritis, tuberculosis, diabetes, autoimmune diseases such as rheumatoid arthritis and ankylosing spondylitis, obvious narrowed knee joint space, significant joint changes such as advanced joint osseous stiffness and intra-articular fracture, in pregnancy and lactation, allergic constitution, or other reactive arthritis, etc. Another 22 patients with normal cartilage received amputation were selected as normal control.
This study has been pre-approved by the ethical committee of Linyi People's Hospital. All subjects have signed the consent forms before recruitment in this study.
Mankin scoring
The cartilage in normal group presents moderate chondrocytes number with normal cell structure under the microscope. The surface is slightly uneven but the structure is complete. Some cells appear mild hyperplasia but arrange in alignment. Staining results show normal or light dyeing is considered as 0-1 point. More malacia, fracture, erosion, and ulceration appeared on cartilage surface with local surface fibrosis is evaluated as 2-14 points. According to the score, 0-1 was considered as normal, 2-9 was treated as mild-to-moderate, while ≥ 10 was evaluated as severe. The OA patients were divided into severe and mild-to-moderate groups. There were 35 patients in severe group with mean age at 58.64 ± 4.32 y old, while 37 subjects in mild-to-moderate group with average age at 57.32 ± 5.21 y old. No statistical difference of age was observed among groups.
Cartilage tissue isolation and extraction
The cartilage tissue was isolated and kept on ice. The tissue with size of around 1 mm3 was moved to 1.5 ml EP tube every 0.1 g tissue and stored at -80°C. The stored sample was rewarmed on ice for 10 min and then treated by the mixture of 490 μl lysis and 10 μl protease inhibitor for 30 min. Next, the tissue was grinded and centrifuged at 10 min at low temperature. The supernatant was moved to a new EP tube and boiled for 15 min together with 50 μl 10X loading buffer.
HE staining
The slice was dewaxed by xylene for 20 min and then gradient dehydrated by ethanol (100%, 95%, 85% and 70%). After being washed by ddH2O, the slice was stained by hematoxylin for 5 min. Next, the slice was treated by 1% hydrochloricalcohol solution for 15 s. After stained by eosin for 5 min, the slice was observed under the microscope to analyse the cartilage tissue morphology.
Western blot
A total of 50 μg protein was separated by 8% SDS-PAGE at 60 V for 45 min followed by 125 V for 1.5-2 h. Then the protein was transferred to membrane at 35 V for 40 min using the semidry method. Next, the membrane was incubated in primary antibody at 1:100 at 4°C overnight and secondary antibody at 1:500 at room temperature for 2 h. At last, the membrane was treated by ECL and detected by Bio-Rad automatic detector. Protein expression was normalized to β- actin and the expressional folds were determined.
Statistical analysis
All statistical analysis was performed using SPSS 13.0 software. The data was depicted as mean ± standard deviation and analysed by Spearman rank correlation analysis. A statistical difference was presented as P<0.05.
Results
Morphologic observation and scoring of OA cartilage tissue
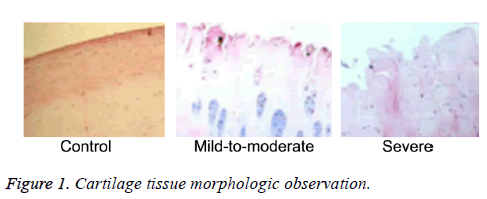
The cartilage tissue was stained by HE for microscopic observation according to Mankin criteria. As shown in Figure 1, normal articular cartilage exhibited smooth surface, more cell number in alignment, uniform color, clear and visible cartilage cell nucleus, and pink cartilage matrix. Comparatively, the cartilage in OA patients presented coarse surface, collagen fibrosis and crack formation in shallow cartilage matrix layer, degenerate and hypertrophic chondrocytes in fascicular alignment, and irregular cartilage matrix staining. Additionally, uneven surface, decreasing cell number, irregular shape, interstitial fibrosis, and crack phenomenon were observed in mild-to-moderate group. The disappearance of cartilage surface and extreme reduction of cell number were found in severe group.
Protein expression in cartilage tissue
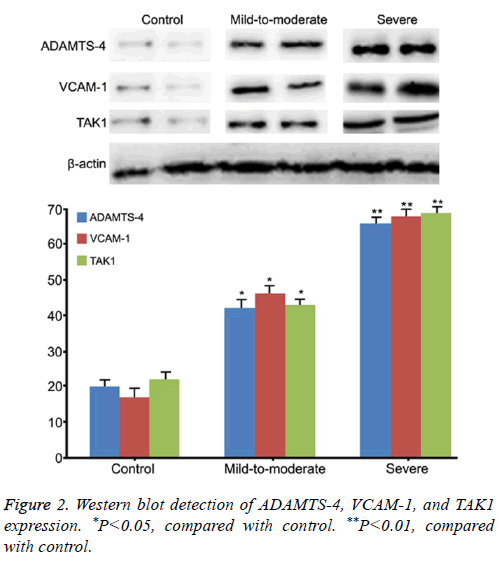
Western blot demonstrated that the expressions of ADAMTS-4, VCAM-1 and TAK1 protein in cartilage tissue from OA patients were statistically higher than that in normal control (P<0.05, Figure 2). Moreover, their levels in severe group were significantly higher than that in mild-to-moderate group (P<0.05). It suggested that TAK1, ADAMTS-4, and VCAM-1 changes in cartilage tissue of OA were positively related to OA process.
Correlation analysis of ADAMTS-4, VCAM-1 and TAK1 expression
Pairwise comparison was performed to analyse the correlation of ADAMTS-4, VCAM-1, and TAK1 expressions in cartilage tissue. Our data showed positive correlation among the levels of these three genes in OA patients. The levels of ADAMTS-4, VCAM-1, and TAK1 exhibited significant difference between mild-to-moderate group and normal control (P=0.032, P=0.041, P=0.026, respectively), severe group and normal control (P=0.014, P=0.0092, P=0.0054, respectively), and severe group and mild-to-moderate group (P=0.043, P=0.045, P=0.036, respectively).
Discussion
OA is a kind of progressive disease, the causes of which are complicated. It was reported that the environmental factors and aberrant expressions of genes were both directly involved in the occurrence and development of OA. Hysteretic treatment due to insensitivity of early diagnosis leads to the degeneration of articular cartilage and even cartilage loss, which needs artificial joint replacement and causes serious harm to the patients. At present, there is still lack of effective prediction, diagnosis for the timely treatment of OA. Thus, it is of great significance to determine novel biomarkers for the diagnosis, prevention and control of OA [1-5].
In clinic, it was confirmed that cartilage degeneration was the main pathological features of OA, while ECM homeostatic imbalance was an important reason. ADAMTS-4 shows potent effect to degrade aggrecanse and its elevation can induce ECM imbalance, resulting in cartilage degeneration and OA. Therefore the upregulation of ADAMTS-4 was proposed to be treated as a predictor for OA [14,15]. Of note, as an important cell adhesion factor, VCAM-1 is involved in regulating cellcell interaction and cell surface glycoprotein. The elevation of VCAM-1 may inhibit local tissue degradation [16,17]. TAK1 plays a critical role in MAPKs and NF-κB signaling pathways. It was revealed to participate in the pathological process of OA. Similarly, our clinical data demonstrated that the expressions of ADAMTS-4, VCAM-1, TAK1 in cartilage tissue from OA patients were altered, which were consistent with the previous findings [18-20].
Intriguingly, the levels of ADAMTS-4, VCAM-1 and TAK1 from patients were gradually enhanced as the OA aggravated. Correlation analysis demonstrated that ADAMTS-4, VCAM-1, and TAK1 expressions had significant correlation in both severe group and mild-to-moderate groups, indicating that the expressions of ADAMTS-4, VCAM-1 and TAK1 could be used as predictor for the OA diagnosis and evaluation of OA severity.
Conclusion
The expressions of ADAMTS-4, VCAM-1 and TAK1 were increased in cartilage tissue of patients with OA and exhibited positive correlation, which could be further used as indicators in the prediction of occurrence and development of OA.
Disclosure of Conflict of Interest
None
Acknowledgments
This work was supported by Shandong medical and health issues (No.2013WSA13014).
References
- Perez-Garcia S, Carrion M, Gutierrez-Canas I, Gonzalez-Alvaro I, Gomariz RP, Juarranz Y. VIP and CRF reduce ADAMTS expression and function in osteoarthritis synovial fibroblasts. J Cell Mol Med 2016; 20: 678-687.
- Yaykasli KO, Hatipoglu OF, Yaykasli E, Yildirim K, Kaya E, Ozsahin M, Uslu M, Gunduz E. Leptin induces ADAMTS-4, ADAMTS-5, and ADAMTS-9 genes expression by mitogen-activated protein kinases and NF-kB signaling pathways in human chondrocytes. Cell Biol Int 2015; 39: 104-112.
- Roberts S, Evans H, Wright K, van Niekerk L, Caterson B, Richardson JB, Kumar KH, Kuiper JH. ADAMTS-4 activity in synovial fluid as a biomarker of inflammation and effusion. Osteoarthritis Cartilage 2015; 23: 1622-1626.
- Larkin J, Lohr TA, Elefante L, Shearin J, Matico R, Su JL, Xue Y, Liu F, Genell C, Miller RE, Tran PB, Malfait AM, Maier CC, Matheny CJ. Translational development of an ADAMTS-5 antibody for osteoarthritis disease modification. Osteoarthritis Cartilage 2015; 23: 1254-1266.
- Dubail J, Apte SS. Insights on ADAMTS proteases and ADAMTS-like proteins from mammalian genetics. Matrix Biol 2015; 44-46: 24-37.
- Alper M, Aydemir AT, Kockar F. Induction of human ADAMTS-2 gene expression by IL-1alpha is mediated by a multiple crosstalk of MEK/JNK and PI3K pathways in osteoblast like cells. Gene 2015; 573: 321-327.
- Peng S, Zheng Q, Zhang X, Dai L, Zhu J, Pi Y, Hu X, Cheng W, Zhou C, Sha Y, Ao Y. Detection of ADAMTS-4 activity using a fluorogenic peptide-conjugated Au nanoparticle probe in human knee synovial fluid. ACS Appl Mater Interfaces 2013; 5: 6089-6096.
- Conde J, Scotece M, Lopez V, Gomez R, Lago F, Pino J, Gomez-Reino JJ, Gualillo O. Adiponectin and leptin induce VCAM-1 expression in human and murine chondrocytes. PLoS One 2012; 7: 52533.
- van Beuningen HM, de Vries-van Melle ML, Vitters EL, Schreurs W, van den Berg WB, van Osch GJ, van der Kraan PM. Inhibition of TAK1 and/or JAK can rescue impaired chondrogenic differentiation of human mesenchymal stem cells in osteoarthritis-like conditions. Tissue Eng Part A 2014; 20: 2243-2252.
- Green YA, Ben-Yaakov K, Adir O, Pollack A, Dvashi Z. TAK1 is involved in the autophagy process in retinal pigment epithelial cells. Biochem Cell Biol 2016; 94: 188-196.
- Zhang Y, Lin J, Wei F. The Function and Roles of ADAMTS-7 in Inflammatory Diseases. Mediators Inflamm 2015; 2015: 801546.
- Ogura Y, Hindi SM, Sato S, Xiong G, Akira S, Kumar A. TAK1 modulates satellite stem cell homeostasis and skeletal muscle repair. Nat Commun 2015; 6: 10123.
- Farias R, Rousseau S. The TAK1 → IKKbeta → TPL2 → MKK1/MKK2 signaling cascade regulates IL-33 expression in cystic fibrosis airway epithelial cells following infection by Pseudomonas aeruginosa. Front Cell Dev Biol 2015; 3: 87.
- Ding Y, Okeefe H, DeLorey JL, Israel DI, Messer JA, Chiu CH, Skinner SR, Matico RE, Murray-Thompson MF, Li F, Clark MA, Cuozzo JW, Arico-Muendel C, Morgan BA. Discovery of potent and selective inhibitors for ADAMTS-4 through DNA-Encoded Library Technology (ELT). ACS Med Chem Lett 2015; 6: 888-893.
- Zhou S, Thornhill TS, Meng F. Influence of osteoarthritis grade on molecular signature of human cartilage. J Orthop Res 2016; 34: 454-462.
- Cao H, Lu J, Du J, Xia F, Wei S. TAK1 inhibition prevents the development of autoimmune diabetes in NOD mice. Sci Rep 2015; 5: 14593.
- Appleton CT, Usmani SE, Pest MA, Pitelka V, Mort JS, Beier F. Reduction in disease progression by inhibition of transforming growth factor alpha-CCL2 signaling in experimental posttraumatic osteoarthritis. Arthritis Rheumatol 2015; 67: 2691-2701.
- Buckland J. Osteoarthritis: positive feedback between ADAMTS-7 and TNF in OA. Nat Rev Rheumatol 2013; 9: 566.
- Li TF, Gao L, Sheu TJ, Sampson ER, Flick LM, Konttinen YT, Chen D, Schwarz EM, Zuscik MJ, Jonason JH, OKeefe RJ. Aberrant hypertrophy in Smad3-deficient murine chondrocytes is rescued by restoring transforming growth factor beta-activated kinase 1/activating transcription factor 2 signaling: a potential clinical implication for osteoarthritis. Arthritis Rheum 2010; 62: 2359-2369.
- Meier T, Kharofa J. Magnetic resonance imaging-guided high-dose rate brachytherapy for cervical cancer. Semin Roentgenol 2016; 51: 106-111.