- Biomedical Research (2016) Volume 27, Issue 3
Comparison of myocardial bridging prevalence using 64-slice versus 256-slice computed tomography scanners: What has changed with recent innovations in CT?
Cemile Ay?e Görmeli1*, Jülide Ya?mur2, Ramazan Özdemir2, Zeynep Mara? Özdemir1, Ay?egül Sa??r Kahraman1, Cemil Çolak3
1Department of Radiology, Inonu University School of Medicine, Turkey
2Department of Cardiology, Inonu University School of Medicine, Turkey
3Department of Biostatistics, Inonu University School of Medicine, Turkey
Accepted date: March 30, 2016
Abstract
Objective: To compare the incidence and appearance of myocardial bridging (MB) by computed tomography (CT) angiography using 64-slice versus 256-slice CT.
Methods: In total, 775 consecutive patients who underwent coronary CT angiography were evaluated using a 64-slice or a 128x2-slice dual-source scanner.
Results: The prevalence of MB was 13.9% using 64-slice-CT and 31.5% using 256-slice-CT. The superficial type of MB segments was detected three times more frequently by 256-slice-CT.
Conclusions: A higher prevalence of MB and the superficial type of bridging were revealed with the 256- slice-CT scanner. Advances in CT technology may enable detection and potentially prevent the unwanted effects of MB.
Keywords
Myocardial bridging, Computed tomography, Coronary artery
Introduction
Myocardial bridging (MB) is a congenital anatomical variant where a portion of the coronary artery courses under the myocardial fibers rather than running epicardially [1-4]. Although MB is traditionally considered a benign condition, arrhythmias, myocardial ischemia, myocardial infarction, ventricular fibrillation, exercise-induced ischemia, or sudden cardiac death have been reported in association with this condition [4-7]. Furthermore, during coronary artery surgery, difficulties and complications, such as localization of the coronary artery and perforation of the right ventricle, may occur because of MB [8]. Still, the real prevalence of this condition remains unknown. In autopsies, MB was found to range from 5% to 86%; however, this rate was as low as 0.8% to 4.9% in conventional angiographic series [9,10]. Due to recent improvements in computed tomography (CT) technology, coronary arteries can be imaged in various planes, and the relationship between artery walls and myocardium can be evaluated by coronary CT angiography. Additionally, recent studies have proposed that CT angiography is the most powerful technique for the assessment of MB [1,11-15]. However, the prevalence of MB evaluated by CT angiography varies between 3.5 to 39%, which may be attributed to differences in temporal and spatial resolution between scanners [1]. The aim of this study was to compare the incidence and appearance of MB by CT angiography using 64-slice versus 256-slice CT in a successive series of 775 patients. Furthermore, to the best of our knowledge, this is the first comparative study on the prevalence of MB detection between 64-slice and 256-slice CT scanners.
Material and Methods
Study population
In total, 775 consecutive patients who were referred to our radiology department and who underwent coronary CT angiography for suspected or known coronary artery disease between June 2014 and February 2016 were retrospectively reviewed. Seven patients were excluded from the study due to poor image quality.
CT scanners
Coronary CT angiography scans were performed using two different multislice scanners: a 64-slice scanner (Aquilion 64, Toshiba Medical Systems, Tochigi, Japan) and a 128 x 2-slice dual-source scanner (Somatom Definition Flash, Siemens Healthcare, Forchheim, Germany).
CT protocol for the first scanner (64-slice): All examinations were performed at 120 kV, with 400 mAs, 0.625 mm detector collimation and 400 ms tube rotation time, using retrospective electrocardiographic (ECG) gating in the 64-slice single-source scanner (Aquilion 64, Toshiba Medical Systems, Tochigi, Japan). Patients with a heart rate greater than 70 beats/min were intravenously administered a beta-blocker drug (5 to 15 mg of metoprolol) immediately prior to the scan. A volume of 80 to 100 mL of non-ionic contrast medium, such as 350 mg/ml iobitridol (Xenetix®, Guerbet, Aulnay, France), 350 mg/ml iohexol (Omnipaque®, Schering, Berlin, Germany) or 400 mg/mL of iodine (Iomeron®, Bracco, Milan, Italy), was intravenously given with an automatic injector system using an antecubital 18-gauge catheter at a speed of 4-5 mL/s, followed by 30 ml of saline at 4 ml/s. Using the automated bolus tracking technique, scan time was defined according to the region of interest (ROI) over the descending aorta, and the threshold was set to 150 HU. Images were routinely reconstructed at 40% and 75% phases of the R-R interval. If it was necessary to obtain the highest image quality, then reconstructions were performed in 10% steps of the R-R interval period.
CT protocol for the second scanner (128 x 2-slice dualsource): All examinations were performed at 100-120 kV, with 300-420 mAs (topogram-based automatic modulation of the tube current - CareDose 4D, Siemens Medical Solutions, Forchheim, Germany), 0.6 mm detector collimation and 280 ms tube rotation time, in the 256-slice dual source scanner (Somatom Definition Flash, Siemens Healthcare, Forscheim, Germany). Image acquisition and reconstruction were irrespective of the patient’s heart rate due to the scanner’s features. No medication was used to lower or control the heart rate. Coronary arteries were scanned with prospective or retrospective ECG gating, depending on the patient’s heart rate (ranged between 60 to 150 beats/min). A 70-90 ml volume of non-ionic contrast medium, such as 350 mg/mL iobitridol (Xenetix®, Guerbet, Aulnay, France), 350 mg/mL iohexol (Omnipaque®, Schering, Berlin, Germany) or 400 mg/mL of iodine (Iomeron®, Bracco, Milan, Italy), was given intravenously with an automatic injector system using an antecubital 18-gauge catheter at a speed of 5-6 mL/s, followed by 30 mL of saline at 5 mL/s. Using the automated bolus tracking technique, scan time was defined according to the ROI over the left atrium, and the threshold was set to 110 HU. Images were routinely reconstructed at the best systole and the best diastole phases (automatic detection of the optimal cardiac phase for motion-free reconstruction - Cardio BestPhase, Siemens Healthcare, Forscheim, Germany) of the R-R interval. If it was necessary to obtain the highest image quality, then images were reconstructed in 10% steps of the R-R interval period.
Image analysis
The raw data generated from the first scanner were reconstructed with a slice thickness of 0.5 mm and an increment of 0.3 mm using a smooth kernel (B25f) and then sent to a Vitrea 2 workstation (Vital Images, Plymouth, Minn., USA). In addition, all data sets generated from the second scanner were reconstructed using a medium-smooth tissue convolution kernel (B26f) with a slice thickness of 0.75 mm and an increment of 0.4 mm and then transferred to a dedicated workstation (Syngo.via, Siemens Medical Solutions, Forscheim, Germany) for evaluation of the coronary arteries.
At first, all images were examined in axial projection; thereafter, coronary arteries were assessed with multiplanar reconstructions (MPR), curved multiplanar reconstructions, maximum intensity projections, and the volume-rendering technique. The images were analyzed by the same radiologist (CAG), who had 3 years of experience in the field of cardiovascular imaging and was blinded to the patients’ history and past conventional angiographic findings.
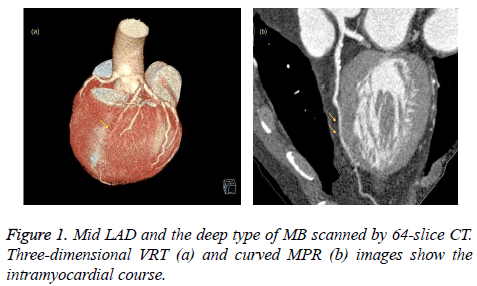
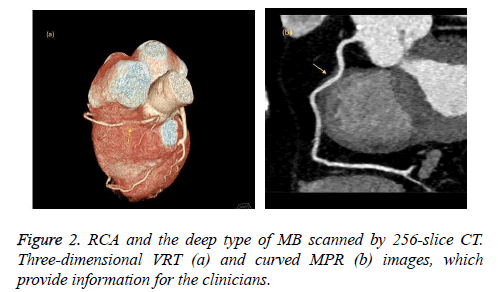
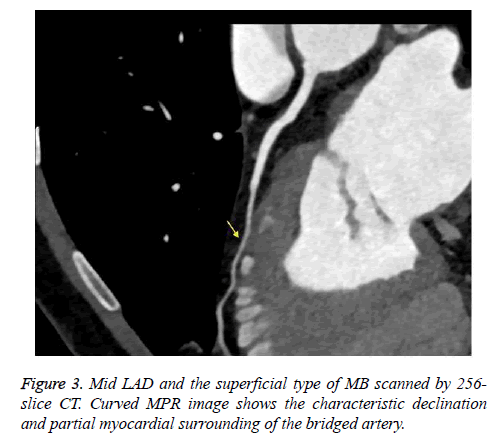
When the intramuscular course of a coronary artery segment was shown on axial and MPR images, it was defined as MB. Moreover, due to the possibility for compression during the systole phase, coronary artery segments extending in a deep gorge but coated by only fibrous-fatty tissue or a fine sheet of muscle were also evaluated as MB [14,16]. The following parameters were used: the name and segment of the coronary artery that showed MB and the length and depth of the intramuscular segment. The coronary artery segments were categorized according to the American Heart Association (AHA) guidelines. The length and depth of the MB were evaluated in axial and sagittal plane MPR images. Moreover, based on the depth of the bridged segment, it was classified as superficial (≤ 1 mm) or deep (>1 mm) MB, as previously reported [14].
Statistical analysis
The data were expressed as the mean (standard deviation, SD) depending on the overall variable distribution. Normality was assessed using the Shapiro Wilk test. The normally distributed data were analyzed with independent samples t tests. Qualitative data were analyzed with the Pearson chi-square test, Fisher’s exact test or Yates’ corrected chi-square test where appropriate. The associations of the groups with the depth type of MB segments were analyzed using multiple logistic regression analysis. Intraclass correlation coefficients (ICC) were calculated for quantitative variables, and Cohenated for q) coefficient was estimated for qualitative variables; the 95% confidence intervals (CI) were also calculated [17]. P<0.05 was considered significant. All qualitative and quantitative parameters were evaluated with at least 2 weeks between analyses for the intraobserver agreement calculation. IBM SPSS statistics version 23.0 for Windows was used for statistical analyses.
Results
We retrospectively analyzed 768 consecutive patients (7 patients were excluded due to poor image quality) who underwent coronary CT angiography using 64-slice CT (447 patients) or 256-slice CT (321 patients) at our radiology department between June 2014 and February 2016. No complications occurred related to the premedication, the contrast agent or the CT scanning. The patients who were scanned with the 64-slice CT scanner (group 1) and who were scanned with the 256-slice CT scanner (group 2) had similar demographic data, such as gender and age. A total of 62 patients (13.9%) in group 1 and 101 patients (31.5%) in group 2 were found to have MB (Figures 1-3). The frequency of MB between groups 1 and 2 was significantly different (P<0.001). These findings are summarized in Table 1. Most of the MB segments were detected in the mid left anterior descending coronary artery (LAD) for both group 1 (67.7%) and group 2 (63.4%). For 10 patients in group 1 and 29 patients in group 2, more than 1 bridged segment was defined. The distribution of MB was represented in Table 2.
| 64-slice CT scanner patients n (%) | MB in patients of 64-slice CT scanner n (%) | 256-slice CT scanner patients n (%) | MB in patients of 256-slice CT scanner n (%) | |
|---|---|---|---|---|
| Patients | 447 (100%)a | 62 (14%)b | 321 (100%)a | 101 (31,4%)b |
| Female | 214 (47,9%)c | 27 (43,5%)d | 148 (46,1%)e | 42 (41,6%)f |
| Male | 233 (52,1%)c | 35 (56,5%)d | 173 (53,9%)e | 59 (58,4%)f |
| Age (mean ± SD) | 51,9 ± 12,1g | 49,7 ± 12,4 g | 51 ± 11,4 g | 51,1 ± 11,6g |
a,c-g No significant differences were found between the groups (P>0,05). b Significant differences were found between the groups (P<0,0001). Abbreviations: CT: Computed Tomography; MB: Myocardial Bridging; SD: Standard Deviation.
Table 1: Patients characteristics.
| MB in patients of 64-slice CT scanner n (%) | MB in patients of 256-slice CT scanner n (%) | P value | |
|---|---|---|---|
| Proksimal LAD | 1 (1,6%) | 2 (2%) | ns |
| Mid LAD | 42 (67,7%) | 64 (63,4%) | ns |
| Distal LAD | 15 (24,2%) | 34 (33,6%) | ns |
| Cx | 1 (1,6%) | 4 (4%) | ns |
| OM | 7 (11,2%) | 14 (13,9%) | ns |
| RCA | 1 (1,6%) | 5 (4,9%) | ns |
| Ramus intermedius branch | 1 (1,6%) | 4 (3,9%) | ns |
| Diagonal branch | 1 (1,6%) | 5 (4,9%) | ns |
*No significant differences were found in coronary artery segments between all of the groups. Abbreviations: CT: Computed Tomography; MB: Myocardial Bridging; LAD: Left Anterior Descending Coronary Artery; Cx: Circumflex Coronary Artery; OM: Obtuse MarginalisBranch; RCA: Right coronary artery, ns; nonsignificant.
Table 2: The distribution of MB depending on localization.
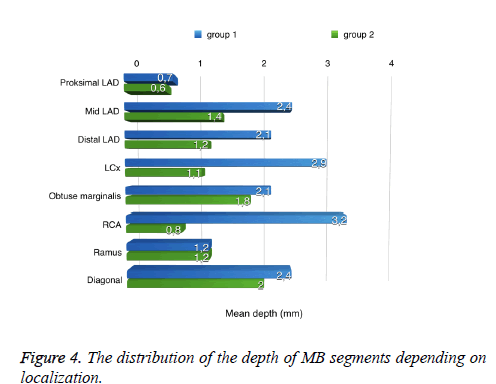
The mean length of bridged segments was 15.8 ± 7.6 mm in group 1 and 13.1 ± 11 mm in group 2. In group 1, the average depth of the bridged segments was 2.3 ± 11 mm, and in group 2, the average depth was 1.5 ± 11 mm. Although there was no significant difference (P>0.05) in length, significantly increased values (P<0.001) were shown in the depth of MB in group 2 compared to group 1 (Table 3 and Figure 4). The depth type of MB was superficial in 2.7% and deep in 11.4% of patients in group 1; however, the prevalence was 16.2% superficial and 15.2% deep in group 2. There was no significant difference (P>0.05) in depth type of MB between groups 1 and 2; however, significantly larger results (P<0.001) were shown for the superficial type of MB in group 2. Furthermore, using multiple logistic regression analysis, superficial MB segments were detected three times more often in group 2 than in group 1 (P<0.05).
| Group 1 | Group 2 | |
| MB length (mean ± SD) mm | 15,8 ± 7,6a | 13,1 ± 11a |
| MB depth (mean ± SD) mm | 2,3 ± 1,2b | 1,5 ± 1,2b |
a No significant differences were found between the groups (P=0.063). b Significant differences were found between groups 1 and 2 (P<0.0001). Abbreviations: MB; Myocardial Bridging, SD; Standard Deviation.
Table 3: Length and depth quantification of MB segments related to CT scanners.
The ICC for the MB length measurements was 0.998 (95% CI: 0.996-0.999), indicating nearly perfect agreement. The ICC for the MB depth measurements was 0.990 (95% CI: 0,986-0,993), which also showed excellent agreement. The following Cohenol k values (P<0.001) were obtained for the qualitative detection of MB segments depending on localization: k=1 for the proximal LAD and the circumflex coronary artery (Cx); k=0.838 (0.045; SE-standard error) for the mid LAD; k=0.937 (0.031; SE) for the distal LAD; k=0.880 (0.058; SE) for the obtuse marginalis branch (OM); and k=0.906 (0.093; SE) for the Right Coronary Artery (RCA), ramus intermedius and diagonal branch. Thus, the data did not provide variance components.
Discussion
MB is a frequent anatomical condition that develops in the coronary arteries in humans. It is usually considered a benign condition but can cause severe ischemia, arrhythmias, myocardial infarction, exercise-induced ventricular tachycardia, or sudden cardiac death [4-7,18]. Additionally, during bypass surgery, complications may occur due to MB [8]. While MB has been reported in autopsy series, studies using conventional angiography or CT angiography have shown marked differences in the prevalence of MB [19-21]. Recent innovations in CT technology cause high temporal and spatial resolution, which has enabled radiologists to more easily evaluate MB. To the best of our knowledge, this is the first study comparing the incidence and appearance of MB by CT angiography using 64-slice versus 256-slice CT. In our study, MB was detected with a 13.9% prevalence in group 1 and a 31.5% prevalence in group 2, with a significant difference between groups. Based on our findings, the 256- slice CT scan offers higher precision for evaluating MB due to its higher temporal and spatial resolution.
In concordance with previous reports [11-15], most of the MB segments were revealed in the mid LAD with no significant difference between groups 1 and 2. There was no significant difference (P>0.05) in length measures and in depth type of MB between groups 1 and 2. However, 2.7% of MB cases in group 1 and 16.2% of MB cases in group 2 were noted as the superficial type. Most notably, a significantly higher proportion (P<0.001) of the superficial type of MB was reported in group 2 compared to group 1. Moreover, using multiple logistic regression analysis, we revealed that MB segments with a depth ≤ 1 mm could be identified three times more often with the 256-slice CT scanner. Most importantly, the superficial MB segments that went unnoticed with the 64-slice CT scanner were detected with the 256-slice CT scanner. One important limitation of this study is that there was no comparison of our CT evaluations with other modalities, such as conventional angiography or cardiac surgery.
Conclusions
In conclusion, the detection rate of MB was significantly higher in patients scanned with the 256-slice CT scanner than with the 64-slice CT scanner. Additionally, a high prevalence was observed for the superficial type of MB with the 256-slice CT scanner. The differences between the two scanners can likely be attributed to the higher spatial and temporal resolution of the 256-slice CT scanner. Advances in CT technology may reveal a higher MB prevalence using radiologic methods, which may reach the presentation ratios revealed in autopsy series. Ultimately, an increased detection frequency of MB may provide protection from the unwanted effects of this condition and may help to avoid surgical complications.
References
- Konen E, Goitein O, Di Segni E. Myocardial bridging, a common anatomical variant rather than a congenital anomaly. Semin Ultrasound CT MR 2008; 29: 195-203.
- Ma ES, Ma GL, Yu HW, Wu W, Li K. Assessment of myocardial bridge and mural coronary artery using ECG-gated 256-slice CT angiography: a retrospective study. Scientific World J 2013; 2013: 947876.
- La Grutta L, Runza G, Galia M, Maffei E, Lo Re G, Grassedonio E, Tedeschi C, Cademartiri F, Midiri M. Atherosclerotic pattern of coronary myocardial bridging assessed with CT coronary angiography. Int J Cardiovasc Imaging 2012;28:405-14.
- Mohlenkamp S, Hort W, Ge J, Erbel R. Update on myocardial bridging. Circulation 2002;106:2616-2622.
- Alegria JR, Herrmann J, Holmes DR Jr, Lerman A, Rihal CS. Myocardial bridging. Eur Heart J 2005;26:1159-1168.
- Tauth J, Sullebarger T. Myocardial infarction associated with myocardialbridging: case history and review of the literature. CathetCardiovascdiagn 1997; 40:364-367.
- Yetman AT, McCrindle BW, MacDonald C, Freedom RM, Gow R. Myocardial bridging in children with hypertrophic cardiomyopathy-a risk factor for sudden death. N Engl J Med 1998;339:1201-1209.
- Broderick TM, Kereiakes DJ, Whang DD, Toltzis RJ, Abbottsmith CW. Myocardial bridging may predispose to coronary perforation during rotational atherectomy. J Invasive Cardiol 1996;8:161-163.
- Ferreira AG Jr, Trotter SE, K├ā┬Čnig B Jr, D├ā┬®court LV, Fox K, Olsen EG. Myocardial bridges: morphological and functional aspects. Br Heart J 1991;66: 364-367.
- Kosinski A, Grzybiak M. Myocardial bridges in the human heart: morphological aspects. Folia Morphol (Warsz) 2001;60: 65-68.
- Canyigit M, Hazirolan T, Karcaaltincaba M, Dagoglu MG, Akata D, Aytemir K, Oto A, Balkanci F,Akpinar E, Besim A. Myocardial bridging as evaluated by 16 row MDCT. Eur J Radiol 2009;69:156-164.
- Kantarci M, Duran C, Durur I, Alper F, Onbas O, Gulbaran M, Okur A. Detection of myocardial bridging with ECG-gated MDCT and multiplanar reconstruction. AJR Am J Roentgenol 2006;186:391-394.
- Ko SM, Choi JS, Nam CW, Hur SH. Incidence and clinical significance of myocardial bridging with ECG-gated 16-row MDCT coronary angiography. Int J Cardiovasc Imaging 2008;24:445-452.
- Konen E, Goitein O, Sternik L, Eshet Y, Shemesh J, Di Segni E. The prevalence and anatomical patterns of intramuscular coronary arteries: a coronary computed tomography angiographic study. J Am CollCardiol 2007;49:587-593.
- Zeina AR, Odeh M, Blinder J, Rosenschein U, Barmeir E. Myocardial bridge: evaluation on MDCT. AJR Am J Roentgenol 2007;188:1069-1073.
- M├ā┬Čhlenkamp S, Hort W, Ge J, Erbel R. Update on myocardial bridging. Circulation 2002;106:2616-2622.
- Bland JM, Altman DG. Measuring agreement in method comparison studies. Stat Methods Med Res 1999;8:135-160.
- Yano K, Yoshino H, Taniuchi M, Kachi E, Shimizu H, Watanuki A, Ishikawa K. Myocardial bridging of the left anterior descending coronary artery in acute inferior wall myocardial infarction. ClinCardiol 2001;24:202-208.
- Goitein O, Lacomis JM. Myocardial bridging: noninvasive diagnosis with multidetector CT. J Comput Assist Tomogr 2005;29:238-240.
- Johansen C, Kirsch J, Araoz P, Williamson E. Detection of myocardial bridging by 64-row computed tomography angiography of the coronaries. J Comput Assist Tomogr 2008;32:448-451.
- Alegria JR, Herrmann J, Holmes DR, Lerman A, Rihal CS. Myocardial bridging. Eur Heart J 2005; 26: 1159-1168.