Research Article - Journal of RNA and Genomics (2017) Volume 13, Issue 1
Cloning and Interspecific Altered Expression of TPS Gene in Two Invasive Apple Snails, Pomacea canaliculata and Pomacea maculata (Gastropoda: Ampullariidae), Under Different Temperature
Guangfu Liu1,2*, Qianqian Yang2?Xiaona Chu3 and Xiaoping Yu2*
1Anhui Agricultural University, Hefei, China
2Zhejiang Provincial Key Laboratory of Biometrology and Inspection and Quarantine, China Jiliang University, China
3Qingdao Inspection Station of Feed and Veterinary drug, Qingdao, China
- *Corresponding Authors:
- Xiaoping Yu
Email: yxp@cjlu.edu.cn
Tel: +86-571-86836006
Fax: +86-571-86836079 - Guangfu Liu
Email: lgf@cjlu.edu.cn
Tel: +86-571-86836006
Fax: +86-571-86836079
Received: 22 September 2017; Revised: 21 November 2017; Accepted: 23 November 2017; Published: 30 November 2017
©Copyright The Author(s). First Published by Allied Academies. This is an open access article, published under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0). This license permits non-commercial use, distribution and reproduction of the article, provided the original work is appropriately acknowledged with correct citation details
Abstract
Studies have demonstrated differences in temperature tolerance between two Pomacea species, Pomacea canaliculata and Pomacea maculata. To determine whether the trehalose-6-phosphate synthase (TPS) genes in the two species exhibited different expression profiles at different temperatures, we cloned the full-length cDNA of TPS in the two Pomacea species and analyzed the expression profile of TPS across a temperature gradient through real-time quantitative analysis. The TPS gene of both P. canaliculata (denoted by PcTPS) and P. maculata (denoted by PmTPS) contained 1908 bp and 1914 ORFs encoding 635 and 637 amino acids, respectively. The genes have a classical polyadenylation signal sequence AATAAA and poly(A), as well as two highly conserved motifs of the TPS protein family in deduced amino-acid sequences. Across the temperature gradient (from 3°C to 42°C), the onset temperature (Ton) or maximal temperature (Tmax) for inducing TPS expression in P. maculata was 3°C lower than that in P. canaliculata, and Ton was highly consistent with the upper temperature limits of the range of the two Pomacea species. All results revealed that the cloned genes were inducible TPS gene. Moreover, in terms of gene-expression level, P. maculata was more susceptible to cold temperature than P. canaliculata. The Ton (or Tmax) of TPS could represent the differences in temperature tolerance of the two Pomacea species. The study also provided useful molecular information on the ecological adaptability of invasive apple snails against extreme environmental stress.
Keywords
Trehalose-6-phosphate synthase, Pomacea canaliculata, Pomacea maculata, Temperature gradient
Introduction
Golden apple snails are freshwater snails that are native to South America (Naylor, 1996). The snails were introduced to East and Southeast Asia in the 1980s for commercial production and as a dietary protein supplement (Cowie et al, 2006; Hayes et al, 2008; Mochida, 1991). However, commercial markets failed, and the discarded or escaped snails invaded rice paddies, thus, these snails have become an important rice pest in numerous countries (Hayes et al, 2008; Wada, 2004). Molecular biology renders more precise identification of apple snails (Rawlings et al, 2007; Hayes et al, 2008). Mitochondrial DNA analysis of samples collected from various countries in Asia and South America revealed four species of South American apple snails in Asia: Pomacea canaliculata, P. maculata (Perry, 1810) (previously known as P. insularum; Hayes et al, 2012), P. scalaris, and P. diffusa (Hayes et al, 2008). Among these species, P. canaliculata and P. maculata are widely distributed in rice ecosystems in Southeast and East Asia. These two species possess morphologically similar characteristics and have become a serious threat to rice production and ecosystems in Asia (Yoshida, 2014; Hayes et al, 2008). These species are distinct according to mitochondrial DNA (COI) analysis (Rawlings et al, 2007; Hayes et al, 2008; Matsukura et al, 2008), as well as shell anatomy and egg morphology (Hayes et al, 2012).
Trehalose is a non-reducing disaccharide and a main energy source in prokaryotes, yeasts, plants, and invertebrates. In addition, trehalose is an important protectant of protein integrity and performs an important function in physiological and biochemical adaptations under extreme environmental conditions (Goyal et al, 2005; Zhang et al, 2012). Trehalose synthesis is the most widely distributed pathway, which involves the transfer of glucose from UDP-glucose to glucose- 6-phosphate to form trehalose-6-phosphate and UDP via the trehalose-6-phosphate synthase (TPS) (Elbein et al, 2003). TPS, a protein in the biosynthesis of trehalose, is a glycosyltransferase that catalyzes the synthesis of α,α,α-1-trehalose-6-phosphate from glucose-6-phosphate by using a UDP-glucose donor (Elbein et al, 2003). TPS is noted in insects as a fused gene that codes two functional domains in tandem, namely, TPS, a homolog of Ost A of Escherichia coli, and trehalose-6-phophate phosphatase (TPP), a homolog of Ost B of E. coli. TPS and TPP are key proteins in trehalose biosynthesis (Becker et al, 1996; Chen et al, 2002). TPS is generally regarded as the key enzyme for catalyzing the first step in trehalose synthesis. In the long period of natural domestication, increase in trehalose content in insects may be due to the increasing expression of TPS gene (Clark et al, 2009). Trehalose has also been implicated as a key protective solute in a range of environmental stress responses, including low temperature (Lee, 2011) and hypoxia (Chen and Haddad, 2004), presumably by stabilizing cell membranes and proteins during periods of osmotic imbalance (Elbein et al, 2003). Trehalose appears to be particularly important during periods of extreme dehydration; for example, most invertebrates that are capable of anhydrobiosis use trehalose as the primary osmoprotectant (Clegg, 2001). This phenomenon implies that the TPS gene may perform an important function under environmental stressors.
Numerous studies have investigated the effect of temperature on apple snails (Matsukura et al, 2009; Wada and Matsukura, 2007). The activity of apple snails increases with increasing water temperature, and their crawling velocity is positively correlated with temperature (Song et al, 2014). Water temperature in the living habitat of snails also exhibits a critical effect on the growth, survival, development, and reproduction of apple snails. Yoshida (2014) found that different populations of apple snails exhibit varied resistance responses to low temperature. Therefore, temperature performs a critical function in the geographic distribution and spread of snails. In addition, Pomacea apple snails can resist pH 4.5-9.4 acid-based erosion and various types of chemical, bacterial, and parasitic threats. Thus, an increased understanding of the molecular mechanisms behind their invasion phenomenon is crucial.
P. canaliculata and P. maculata are widely distributed in more than 11 provinces in Southern China. These species may have expanded northward because of environmental adaptation and climate change, causing serious damage to humans and the environment (Li et al, 2009; Lv, et al, 2011; Song et al, 2014). Both species are tolerant to variable temperatures, salinities, and humidity, making them eminently suitable for successful dispersal (Cowie, 2002). However, the physiological mechanism of their distribution that extends further toward northern China has not been investigated in detail.
Numerous TPS genes have been cloned from animals, plants, bacteria, and fungi to better understand the structure and function of TPS at the molecular level. However, few data on molecular information of TPS in golden apple snails and their expression responses against environmental stressors are available. This study is the first to identify and characterize the TPS gene in apple snails. Quantitative polymerase chain reaction (PCR) was performed to analyze the differential expression of TPS in both P. canaliculata and P. maculata under different temperature treatments. The findings of this study reveal that the different temperature resistances of P. canaliculata and P. maculata result in different patterns of TPS expression, indicating that species-related differences likely exist in the temperature resistance of Pomacea apple snails. These results will provide valuable molecular information on increased tolerances against environmental stressors in P. canaliculata and P. maculata, as well as understanding the functional complexity of TPS in mollusks.
Materials and Methods
Snails used
Golden apple snails were collected from a field of wild rice, Zizania latifolia, in Yuyao City (30?02′N, 121?10′E), Zhejiang Province. Snails were identified as P. canaliculata and P. maculata based on mitochondrial COI gene sequences, as described in previous studies (Rawlings et al, 2007; Hayes et al, 2008; Matsukura, et al 2008).
Rearing and temperature treatments
These snails were reared in a 40 cm × 25 cm × 28 cm tank that contained 10 L of freshwater at 26 ± 1°C. A dead cabbage was provided as basic food, and egg masses were obtained from above snails. Hatched juveniles were reared in an aquarium in a rearing room under a 16 h light: 8 h dark photoperiod at 26 ± 1°C. Juvenile snails with a shell height of 7.5-15.0 mm (about a month after hatching) were selected for experiments. For the temperature gradient treatment (3, 6, 9, 12, 15, 26, 30, 33, 36, 39, and 42°C), six P. canaliculata and six P. maculate individuals were selected and exposed to each temperature for 2 h, and then snails were returned to acclimation temperature (26°C) for 1 h before the expression test. For the temporal expression experiment (2, 6, 12, 24, 48, 72, 96, and 120 h), six P. canaliculata and six P. maculate individuals were sampled and decapitated in each group after the same temperature treatment (12°C).
Cloning the full-length cDNA of TPSs
Total RNA was extracted from 10 mg foot muscle of each sample using TRIzol© reagent (Invitrogen, USA), and 1 μg of the total RNA was used to generate cDNAs. Degenerate primers (Table 1) were used to amplify the partial segments of TPSs, and then 5′ and 3′ RACE were applied to obtain full cDNA lengths following the manufacturer’s instructions (Smart™ RACE cDNA Amplification Kit, BD, Biosciences Clontech). Products were analyzed on a 1.2% agarose gel, and the objective bands were selected and purified with a gel extraction kit (Omega, Norcross, GA). The DNA fragment was then subcloned into the pMD18-T vector (TaKaRa, Tokyo, Japan). Following transfection into E. coli DH5α-competent cells, recombinants were identified using blue and white spot selection. Positive clones were sequenced by Sangon Biological Engineering Technology & Services Co., Ltd (Shanghai, China).
| Primers | Sequence (5'-3') | Technique |
|---|---|---|
| TPS-F | TCCACgaytaycayyt | Amplification of TPS core region |
| TPS-R | CCTTGGCCACCAGGttcatnccrtc | Amplification of TPS core region |
| TPS-3-F | CGCTCAGGTCGTAGTAGCAGAAGGTC | 3' RACE |
| TPS-3-R | TACCGTCGTTCCACTAGTGATTT | 3' RACE |
| TPS-5-F | GGCTTCTTTCTTCACATTCCGTTCCC | 5' RACE |
| TPS-5-R | CATGGCTACATGCTGACAGCCTA | 5' RACE |
| 18s-F | CCGTCCCTTTTGGTGACTCTG | Real-time PCR |
| 18s-R | GGATGTGGTAGCCGTTTCTC | Real-time PCR |
| TPS-qF | GCTCGTGAAACCGATGATGTCGCAT | Real-time PCR |
| TPS-qR | ACTGCGTTTTGACTTACCCTAACG | Real-time PCR |
Table 1 Name, sequence, and technique for PCR primers used in the cloning and expression of TPS genes from Pomacea snails.
Searches for nucleotide and amino-acid sequence similarities were performed with BLAST programs on the NCBI website (http://www.ncbi.nlm.gov/BLAST/). The open reading frame (ORF) was identified using the ORF Finder (http://www.ncbi. nlm.nih.gov/gorf/gorf.html), and the motif sequences were searched with the InterPro software (Hunter et al 2009). The resulting amino-acid sequence was predicted for theoretical molecular weight and isoelectric point using the ProtParam software (Gasteiger et al, 2005). Multiple sequence alignments and phylogenetic analysis were performed on amino-acid sequences of known TPS from other species using CLUSTALW and MEGA 6.0 (Tamura et al, 2013). The amino-acid sequences of TPS used in the phylogenetic study included TPS sequences of A. simplex (KJ560557.1), C. sapidus (EU679406.1), D. melanogaster (NM_134983.3), S. exiguas (FJ792706.1), P. haitanensis (KF658272.1), F. chinensis (EU555435.1), P. freudenreichii (DQ356268.1), C. elegans (AJ811574.1), C. bairdi (EU910084.1), B. antarctica (JX462664.1), P. savastanoi (AY308798.1), E. coli (FJ895834.1), L. migratoria (EU131894.1), and H. armigera (DQ086235).
Quantitative real-time PCR analysis of TPS mRNA expression
The mRNA expression levels of the TPS gene from the foot muscle of each sampled individual were measured by using fluorescent quantitative PCR (qPCR). Amplifications were performed in a 25 μL reaction volume including 12.5 of 2X SYBR Premix Ex Taq™ master mix (TaKaRa, Japan), 1 μL each of gene-specific primers (Table 1) and 1 μL of 1:29 diluted cDNA. qPCR analysis was carried out on IQTM 5 Multicolor Real-Time PCR detection system (Bio-Rad, USA). PCR conditions were as follows: 95°C for 1 min, followed by 45 cycles at 95°C for 10 s, 63°C for 25 s, and a plate read. At the end of each qPCR, melting curve analysis of amplification products was performed, and qPCR data were collected as Ct value. 18S RNA gene was cloned in both P. canaliculata and P. maculate and used as internal control. The relative expression levels of genes were calculated by 2−ΔΔCt method (Livak and Schmittgen, 2001). All data were expressed as the mean ± standard deviation (SD), and significant difference between the two Pomacea species at the same temperature or at the same time point was identified by an unpaired Student t-test. P < 0.05 (*) indicates significance, and P < 0.01 (**) was considered highly significant.
Results
Molecular characteristics of the TPS gene
The full-length cDNAs of TPS in the two Pomacea species, namely, P. canaliculata and P. maculate, were obtained. The cDNAs contained 1908 bp and 1914 ORFs encoding 635 and 637 amino acids (Figures 1A and 1B), respectively. Both Pomacea TPS genes contain a classical polyadenylation signal sequence AATAAA and poly (A). PcTPS contained a 5′-UTR of 151 bp, a 3′-UTR of 95 bp, while PmTPS contained a 5′-UTR of 167 bp and a 3′-UTR of 102 bp. The molecular weights are 73842 and 73954 Da for P. canaliculata and P. maculate, respectively. The PcTPS aa sequence is highly homologous (99% identity) to that of PmTPS, and high shared identities with other TPSs were also observed: 93% to C. elegans, 85% to L. migratoria, 70% to A. simplex, 61% to C. sapidus, 78% to D. melanogaster, 60% to S. exiguas, 71% to P. haitanensis, 60% to F. chinensis, 65% to P. freudenreichii, 61% to C. bairdi, 59% to B. antarctica, 53% to P. savastanoi, 55% to E. coli, and 61% to H. armigera. The alignment of TPSs from insects, bacteria, yeast, fungi, nematodes, and plants also showed that two conserved motifs, namely, HDYHL and DGMNLV, were found in both PmTPS and PcTPS. These motifs may be the signature sequences of TPSs and putative catalytic domains (Figures 1A and 1B).
Figure 1: A: Nucleotide and deduced amino-acid sequences of P. canaliculata PcTPS. B:Nucleotide and deduced amino-acid sequences of P. maculata PmTPS. Both initiation codon and termination codon were indicated in bold font and italics. Motifs (or signature motifs) that are unique to trehalose-6-phosphate synthase (TPS) are underlined and bold.
Phylogenetic analysis
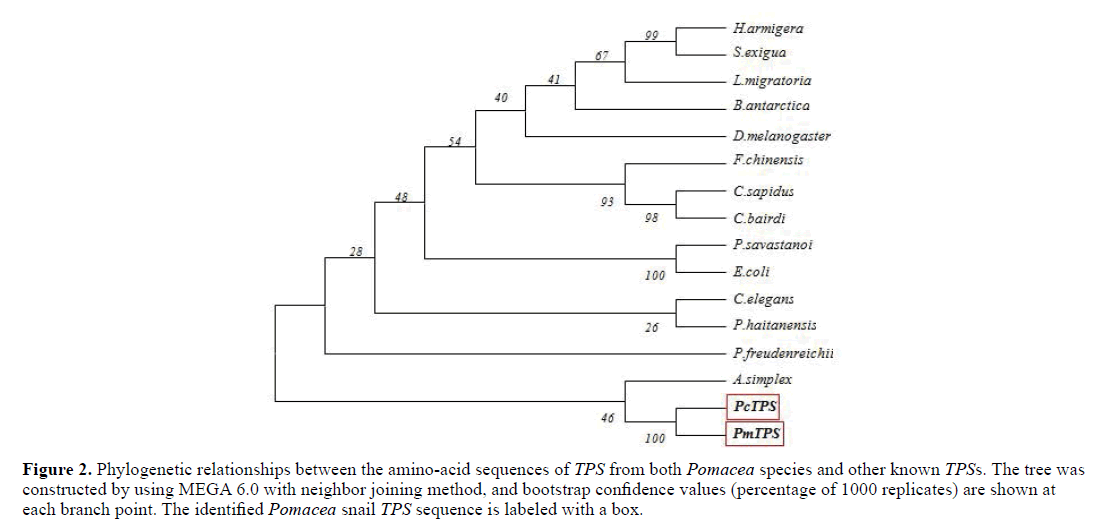
Based on the amino-acid sequences of Pomacea TPSs, a phylogenetic tree was constructed by using MEGA 6.0 using neighbor joining method. Fourteen TPSs family members were selected for analysis. Sequences of PcTPS and PmTPS were clustered into a large group with A. simplex. The phylogenetic relationships displayed in the phylogenic tree were consistent with the traditional classification. (Figure 2)
Figure 2: Phylogenetic relationships between the amino-acid sequences of TPS from both Pomacea species and other known TPSs. The tree was constructed by using MEGA 6.0 with neighbor joining method, and bootstrap confidence values (percentage of 1000 replicates) are shown at each branch point. The identified Pomacea snail TPS sequence is labeled with abox.
Expression of TPS mRNA in response to temperature gradient shock stress
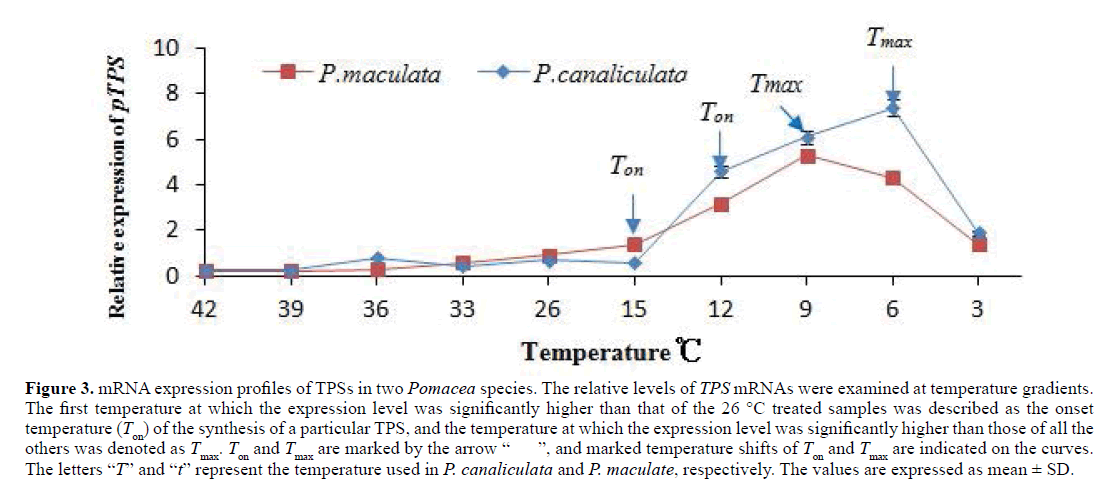
The relative mRNA levels of the two TPSs were quantified by real-time quantitative PCR at temperature gradients from 3°C to 42°C. Results revealed that the two TPS transcript levels were slightly decreased under high temperature (30°C, 33°C, 36°C, 39°C, and 42°C) compared with these at 26°C. Under cold treatment (3°C, 6°C, 9°C, 12°C, and 15°C), significant upregulation of the two TPSs were observed. The increase in PmTPS expression with temperature for P. maculata was less steep in compared with that in PcTPS for P. canaliculata. The highest expression level that was observed at 6°C was tenfold that at 26°C in P. canaliculata, whereas that at 9°C was six fold that 26°C in P. maculata. The maximal induction of PcTPS was significantly higher compared with PmTPS. After maximal induction, the expression level of the two TPSs significantly decreased in both Pomacea species (Figure 3).
Figure 3: mRNA expression profiles of TPSs in two Pomacea species. The relative levels of TPS mRNAs were examined at temperature gradients. The first temperature at which the expression level was significantly higher than that of the 26 °C treated samples was described as the onset temperature (Ton) of the synthesis of a particular TPS, and the temperature at which the expression level was significantly higher than those of all the others was denoted as Tmax. Ton and Tmax are marked by the arrow “ ”, and marked temperature shifts of Ton and Tmax are indicated on the curves. The letters “T” and “t” represent the temperature used in P. canaliculata and P. maculate, respectively. The values are expressed as mean ± SD.
Interspecific differences of TPS gene expression
For determining the temperature for onset (Ton) and maximal (Tmax) induction of TPS expression, the relative mRNA levels of TPSs were compared with each other, and an interspecific difference was observed in most comparisons. TPS started to be induced at 12°C in P. canaliculata and peaked at 6°C, whereas Ton and Tmax in P. maculate were 15°C and 9°C, respectively. Both Ton and Tmax shifted by 3°C between the two Pomacea species under cold treatments. Under heat stress conditions, the TPS relative expression levels in both Pomacea species were slightly decreased in the foot muscle in comparison with that at normal temperature, and the species effect was not significant. (Figure 3)
Temporal expression profiles of TPS transcripts after a thermal challenge
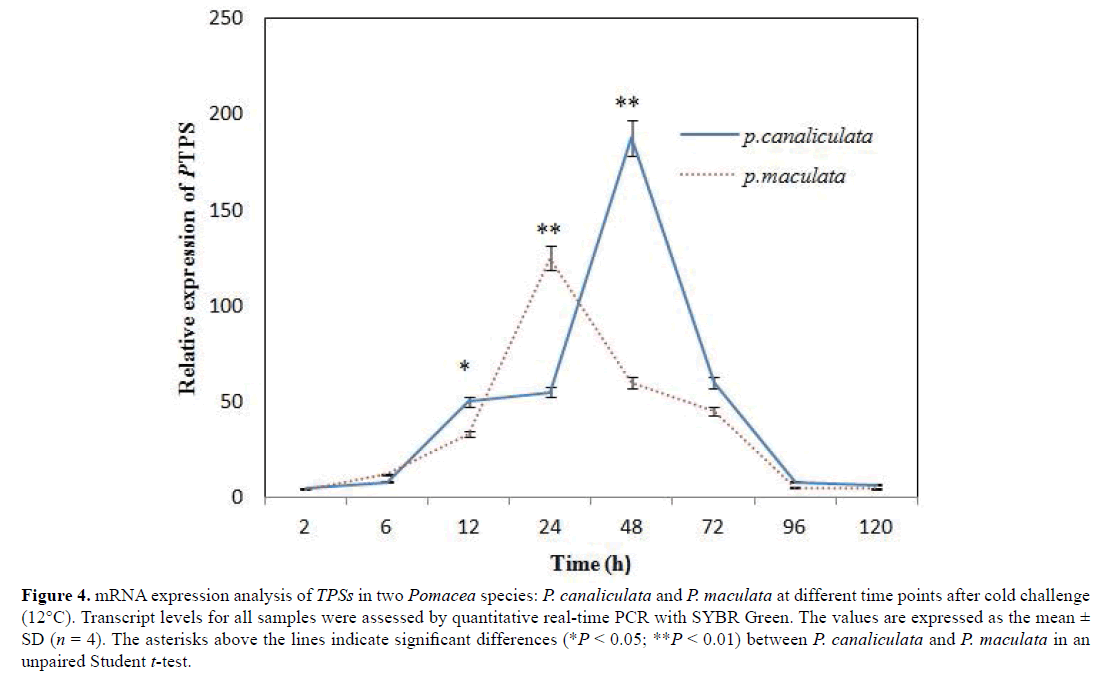
RT-PCR was conducted to examine the time-dependent expression pattern of the two TPS genes in the foot muscle of P. canaliculata and P. maculata at 2, 6, 12, 24, 48, 72, 96, and 120 h after cold treatment (12°C), A clear time-dependent expression pattern of TPS was observed (Figure 4) in two Pomacea species. After cold treatment, the expression of the PcTPS gene was upregulated and reached the highest level at 48 h in P. canaliculata. However, the expression gradually decreased after 72 h and then returned to its baseline value after 96 h. The expression of PmTPS was induced after 2 h in P. maculata and peaked at 24 h. The expression significantly decreased after 48 h and then returned to its baseline level after 96 h. The highest expression level was markedly higher in P. canaliculata compared with P. maculata (Figure 4). The TPS expression levels at 12, 24, 48, and 72 h were significantly different between the two Pomacea species.
Figure 4: mRNA expression analysis of TPSs in two Pomacea species: P. canaliculata and P. maculata at different time points after cold challenge (12°C). Transcript levels for all samples were assessed by quantitative real-time PCR with SYBR Green. The values are expressed as the mean ± SD (n = 4). The asterisks above the lines indicate significant differences (*P < 0.05; **P < 0.01) between P. canaliculata and P. maculata in an unpaired Student t-test
Discussion
Golden apple snails of the genus Pomacea (Mollusca: Ampullariidae) are some of the largest freshwater snails. These snails damage various crops, with rice as the major host, through eating rice seedlings during the first three weeks after transplanting. Golden apple snails have been ranked in the top 100 of the worst invasive species worldwide (Lowe et al, 2000). Enormous snail populations have developed an ecologically adaptive mechanism in response to all types of stresses during invasion, and temperature adaptability of species is speculated to be one of the most important attributes (Yusa et al, 2006). Trehalose is a non-reducing disaccharide that is a primary energy source for prokaryotes, yeasts, plants, and invertebrates. In addition, trehalose is an important protectant of protein integrity, performing an important function in physiological and biochemical adaptations under extreme environmental conditions (Goyal et al, 2005; Zhang et al, 2012). TPS, which is a protein in the biosynthesis of trehalose, is regarded as the key enzyme to catalyze the first step in trehalose synthesis. Increasing evidence has demonstrated the importance of trehalose as molecular protectant in resistance to heat, cold, and a range of other biotic and abiotic stressors. Numerous TPS genes have been reported, including S. exiguas (Tang et al, 2010), A. aegypti (GenBank: XM_001657763), D. melanogaster (GenBank: NM_134983), H. armigera (GenBank: DQ086235), and F. chinensis (Zhang et al, 2012).
The TPS mRNA expression in both Pomacea species significantly increased under cold treatments but slightly decreased under heat treatments. This finding suggests that a specific amount of TPS mRNA is present in foot muscle tissues under both stressed and non-stressed conditions, which may perform an important function in physiological processes and the prevention of cellular damage under temperature stressors. However, the expression level of TPS genes under heat stress were not significantly changed in comparison with that under normal temperature in both Pomacea apple snails. The same expression pattern was demonstrated in maize (Jiang et al, 2010), Chilo suppressalis (Zhang, 2007), and Harmonia axyridis (Qin et al, 2012). TPS may perform a lesser role in heat resistance. In the present study, we found that, under short-term cold treatment, TPS expression was markedly increased with decreasing temperature. Under long-term cold treatment, the TPS expression level initially increased and then decreased to baseline levels after 96 h in both Pomacea species. This result may be attributed to the conversion of trehalose to low molecular weight compounds, such as glycerol and amino acids, which could enhance cold hardiness in winter. This phenomenon is similar to those in insects.
The onset or maximal induction temperatures of TPS expression were used to interpret temperature tolerance. Under heat treatments, TPS could not be induced either in P. canaliculata or P. maculata at 26°C-42°C, revealing that no positive correlation was found between TPS expression levels and thermal tolerance. This result suggests that trehalose synthesis is not required for the induction of heat tolerance for Pomacea apple snails. Under cold treatment, the expression levels of TPS gene transcripts significantly increased in both Pomacea species and peaked at 6°C in P. canaliculata and 9°C in P. maculata, thereby suggesting that P. maculata was more susceptible to cold conditions than P. canaliculata. Previous studies revealed that a positive relationship exists between TPS expression levels and low temperature tolerance in organisms (Tang et al, 2010; Jiang et al, 2010; Zhang et al, 2012). However, further investigation is necessary to confirm this hypothesis because only a limited number of populations were examined in the present study. In particular, research that separates the influence of habitats on environmental stress from the influence of hybridization is required. High expression levels of the TPS gene enables organisms to survive during cold stress by repairing denatured proteins, and organisms could adapt to a wide range of temperature changes and resist cold stress. The maximal induction of TPS expression was 1.4 and 1.5 times higher in P. canaliculata compared with P. maculata under different cold temperature and long-term cold treatment, respectively. This finding indicates that P. canaliculata may be more resistant to cold than P. maculata, as was consistent with observations that the P. maculata population may be less tolerant to cold weather than P. canaliculata. Thus, P. canaliculata snails may possess better ability to colonize Asian paddy ecosystems than P. maculata (Yoshida et al, 2014). Apple snail populations may have suffered short-term cold weather in winter, which may limit the expansion of apple snails.
Previous studies showed that different species and populations of apple snails exhibit varied resistances in response to low temperature (Yoshida et al, 2014; Matsukura et al, 2008). Thus, cold stress resistance is a complex trait. Numerous differentially expressed genes are assumed to be involved in the difference in cold resistance in P. canaliculata and P. maculata. Thus, identifying differentially expressed genes under cold conditions is necessary for further understanding the molecular mechanism underlying the cold resistance difference in both Pomacea snails.
Acknowledgements
This work was supported by the National High Technology Research and Development Program of China (2012AA021601) and the National Basic Research Development Program of China (973 Program) (2012CB114100).
References
- Becker A, Schloder P, Steele JE, et al. 1996. The regulation of trehalose metabolism in insects. Experiential, 52, 433-439.
- Chen QF, Ma E, Behar KL, et al. 2002. Role of trehalose phosphate synthase in anoxia tolerance and development in Drosophila melanogaster. J Biol Chem, 277, 3274-3279.
- Chen QF and Haddad GG. 2004. Role of trehalose phosphate synthase and trehalose during hypoxia: from flies to mammals. J Exp Biol, 207, 3125-3129.
- Clark MS, Thorne MAS, Pura? J, et al. 2009. Surviving the cold: molecular analyses of insect cryoprotective dehydration in the Arctic springtail Magaphorura arctica (Tullberg). BMC Genomics, 10, 1-19.
- Clegg JS. 2001. Cryptobiosis-a peculiar state of biological organization. Comp Biochem Physiol B Biochem Mol Biol, 128, 613-624.
- Cowie RH. 2002. Apple snails (Ampullariidae ) as agricultural pests: their biology, impacts and management. In: Barker GM (ed) Molluscs as crop pests, CAB International, New York. Cowie RH, Hayes KA and Thiengo SC. 2006. What are apple snails? Confused taxonomy and some preliminary resolution. In: Joshi RC, Sebastian LS (eds) Global advances in ecology and management of golden apple snails. Philippine Rice Research Institute, Nueva Ecija, pp 3-23.
- Elbein AD, Pan YT, Pastuszak I, et al. 2003. New insights on trehalose: a multifunctional molecule, Glycobiology, 13, 17R-27R.
- Gasteiger E, Hoogland C, Gattiker A, et al. 2005. In: Walker JM (ed) The proteomics protocols handbook. Humana Press, New Jersey.
- Goyal K, Browne JA, Burnell AM, et al. 2005. Dehydrationinduced tps gene transcripts from an anhydrobiotic nematode contain novel spliced leaders and encode atypical GT-20 family proteins. Biochemie, 87, 565-574.
- Hayes KA, Joshi RC, Thiengo SC, et al. 2008. Out of South America: multiple origins of non-native apple snails in Asia. Diversity and Distributions, 14, 701-712.
- Hayes KA, Cowie RH, Thiengo SC, et al. 2012. Comparing apples with apples: clarifying the identities of two highly invasive Neotropical Ampullariidae (Caenogastropoda). Zool J Linn Soc, 166, 723-753.
- Hunter S, Apweiler R, Attwood TK, et al. 2009. InterPro: the integrative protein signature database. Nucleic Acids Res, 37, D211-D215.
- Jiang W, Fu FL, Zhang SZ, et al. 2010. Cloning and Characterization of Functional Trehalose-6-Phosphate Synthase Gene in Maize. J Plant Biol, 53, 134-141.
- Lee RE. 2011. A primer on insect cold tolerance. In: Denlinger DL,Lee RE Jr (eds) Low temperature biology of insects. Cambridge University Press, Cambridge, pp 3-34.
- Li X, Hu Y, Song H, et al. 2009. Invasion and monitoring methods of Pomacea canaliculata in China. Chin Agric Sci Bull, 25, 229-232.
- Livak KJ and Schmittgen TD. 2001. Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods, 25, 402-408.
- Lowe S, Browne M, Boudjelas S, et al. 2000. 100 of the world’s worst invasive alien species: a selection from the global invasive species database, Invasive Species Specialist Group Auckland, New Zealand.
- Lv S, Zhang Y, Steinmann P, et al. 2011. The emergence of angiostrongyliasis in the People’s Republic of China: the interplay between invasive snails, climate change and transmission dynamics. Freshw Biol, 56, 717-734.
- Matsukura K, Okuda M, Kubota K, et al. 2008. Genetic divergence of the genus Pomacea (Gastropoda: Ampullariidae) distributed in Japan and a simple molecular method to distinguish P. canaliculata and P. insularum. Appl Entomol Zool, 43, 535-540.
- Matsukura K, Tsumuki H, Izumi Y, et al. 2009. Physiological response to low temperature in the freshwater apple snail, Pomacea canaliculata (Gastropoda: Ampullariidae). J Exp Biol, 212, 2558-2563.
- Matsukura K, Tsumuki H, Izumi Y, et al. 2008. Changes in chemical components in the freshwater apple snail, Pomacea canaliculata (Gastropoda: Ampullariidae) in relation to the development of its cold hardiness. Cryobiology, 56, 131-137.
- Mochida O. 1991. Spread of freshwater Pomacea snails (Pilidae, Mollusca) from Argentina to Asia. Micronesica, 3, 51-62.
- Naylor R. 1996. Invasions in agriculture: assessing the cost of the golden apple snail in Asia. Ambio, 24, 443-448.
- Qin Z, Wang S, Wei P, et al. 2012. Molecular cloning and coldinduced expression of trehalose-6-phosphate synthase gene in Harmonia axyridis (Coleoptera: Coccinellidae). Acta Entomol Sin, 55, 651-658.
- Rawlings TA and Hayes KA. 2007. The identity, distribution, and impacts of non-native apple snails in the continental United States. BMC Evol Biol, 7, 97.
- Song H, Mu X, Gu D, et al. 2014. Molecular characteristics of the HSP70 gene and its differential expression in female and male golden apple snails (Pomacea canaliculata) under temperature stimulation. Cell Stress Chaperones, 19, 579-589.
- Tang B, Chen J, Yao Q, et al. 2010. Characterization of a trehalose-6-phosphate synthase gene from Spodoptera exigua and its function identification through RNA interference. J Insect Physiol, 56, 813-821.
- Tamura K, Stecher G, Peterson D, et al. 2013. MEGA 6: Molecular Evolutionary Genetics Analysis Version 6.0. Mol Biol Evol, 30, 2725-2729.
- Wada T. 1997. Introduction of the apple snail Pomacea canaliculata and its impact on rice agriculture. In: Proceedings of international workshop on biologica invasions of ecosystems by pests and beneficial organisms (E. Yano et al, eds), pp. 170-180. National Institute of Agro-Environmental Sciences, Tsukuba, Japan.
- Wada T. 2004. Strategies for controlling the apple snail, Pomacea canaliculata (Lamarck)(Gastropoda: Ampullariidae) in Japanese direct sown paddy fields. Jpn Agric Res Q, 38, 75-80.
- Wada T and Matsukura K. 2007. Seasonal changes in cold hardiness of the invasive freshwater apple snail, Pomacea canaliculata ( Lamarck) ( Gastropoda: Ampullariidae). Malacologia, 49, 383-392.
- Yoshida K, Matsukura, K, Cazzaniga NJ, et al. 2014. Tolerance to low temperature and desiccation in two invasive apple snails, Pomacea canaliculata and P. Maculata (Caenogastropoda: ampullariidae), collected in their original distribution area (Northern and central Argentina). J Molluscan Stud, 80, 62-66.
- Yusa Y, Wada T and Takahashi S. 2006. Effects of dormant duration, body size, self-burial and water condition on the long-term survival of the apple snail, Pomacea canaliculata (Gastropoda: Ampullariidae). Appl Entomol Zool, 41, 627-32.
- Zhang J, Wang J, Li F, et al. 2012. A trehalose-6-phosphate synthase gene from Chinese shrimp, Fenneropenaeus chinensis. Mol Bio Rep, 39, 10219-10225.
- Zhang Y. 2007. Studies on the Cold Hardiness and Mechanism of Over Vintering Larva of Chilo supperssalis (Walker), Wuhan: Huazhong Agricultural University.