Research Article - Biomedical Research (2017) Volume 28, Issue 19
Cerebral venous sinus thrombosis and acute subarachnoid hemorrhage: a retrospective study on diagnosis, treatment prognosis of 11 patients
Jianjun Gu1,2#, Bingbing Li2#, Jiebin Chen3#, Jingfang Hong2, Haibing Liu2 and Shousen Wang2*
1Stroke Center, Zhengzhou University People's Hospital, Henan Provincial People's Hospital, Zhengzhou, Henan, China
2Department of Neurosurgery, Fuzhou General Hospital, Xiamen University Medical College, 156 Xi’erhuanbei Road, Fuzhou 350025, Fujian, P.R. China
3Departments of Pediatrics, Taizhou People's Hospital Affiliated to Medical College of The Nantong University, Taizhou, Jiangsu 225300, P.R. China
#These authors contributed equally to this work
- *Corresponding Author:
- Shousen Wang
Department of Neurosurgery
Fuzhou General Hospital
Xiamen University Medical College
PR China
Accepted date: September 27, 2017
Abstract
Most nontraumatic subarachnoid hemorrhage (SAH) is resulted from ruptured aneurysm. Few cerebral venous sinus thrombosis (CVST) may appear after acute SAH with similar clinical manifestations as aneurysm bleeding, raising difficulties for SAH diagnosis in clinics. Retrospective analysis of 88 patients with CVST in our hospital from March 2008 to March 2015 was carried out. Among them, the radiologic characteristic, therapeutic strategies and prognosis of 11 patients with SAH as initial manifestation were analyzed. In total, 7 patients received pure heparin anticoagulant treatment and 4 patients received intrasinus thrombolytic treatment with urokinase. Non-enhancement CT or MRI proved that SAH localized at the parasagittal or dorsolateral cerebral convexity. Empty delta sign was present in 5 cases by enhanced CT. CVST involved superior sagittal sinus in 9 patients, lateral sinus in 5 patients, sigmoid sinus in 3 patients, straight sinus in 1 patients and cortical veins in 4 patients under magnetic resonance venography (MRV) or digital subtraction angiography(DSA). Complete recanalization of the occluded sinus or cortical veins was evident in 8 patients and partial recanalization was observed in 2 patients. Non- recanalization was still in 1 patient who received craniotomy hematoma craniectomy because of secondary intracranial hemorrhage after intrasinus thrombolytic therapy. All patients except one exhibited satisfactory outcome. CVST should be considered in patients with nonaneurysmal SAH, based on which early diagnosis and treatment can be applied. CT, MRI and MRV imaging are effective for early diagnosis and anticoagulants and endovascular treatment is safe and effective against CVST.
Keywords
Cerebral venous sinus thrombosis, Subarachnoid haemorrhage, Anticoagulants, Endovascular treatment
Introduction
Cerebral venous sinus thrombosis (CVST) is a rare and distinct cerebrovascular disorder with highly variable clinical presentations [1]. It occurs in 0.5%-3% of stroke [2] with incidence around 0.00035% and predominantly affecting younger people, particular children with incidence around 0.0007%. [3]. Hemorrhagic infarctions were evident in 40% patients under CT-scan and hemorrhagic transformation occasionally lead to intracerebral or subarachnoid hemorrhages (SAH) [4-6], however SAH is a rare clinical presentation. Diversity of clinical manifestations increases the difficulty for CVST diagnosis and leads to poor treatment on CVST patients with SAH [6].
Systemic studies of CVST-related SAH was lacking in the past. Up to now, only Boukobza [7] reported a study involving 22 CVST patients with SAH, in which they evaluated the incidence and radiological characteristics of CVST-associated SAH associated with CVS by using T2* with main focus on presence, location, and distribution of associated cortical venous thrombosis. In current study, we reported 11 CVST patients initially with acute SAH in our institution from March 2008 to March 2015 and characterized the relevant clinical characteristic, therapeutic strategies and prognosis.
Materials and Methods
Subjects
A total of 88 CVST patients in Fuzhou General Hospital from March 2008 to March 2015 were retrospectively analyzed, in which 11 patients with SAH-associated CVST were included in this study. Informed consent and a confidentiality agreement were obtained from all patients or their families. The study was approved in advance by the Medical Ethics Committee of Fuzhou General Hospital, Xiamen University Medical College. Of the 11 patients, 5 were males and 6 were females. Patients’ age ranged between 13 and 66 years with an average of 37.5 ± 14.6 years. Patients’ disease-history were as followed, two with long-term history of contraceptive usage, two with a history of trauma, one with nephrotic syndrome, one with puerperium, one in pregnancy, one with mastoiditis, one with Behcet's disease and two without evident risk factors. The clinical manifestations were headache in 9 (82%), epilepsy in 5 (45%), thunderclap headache in 1 (9%) and disturbance of consciousness in 1 (9%). The duration from the onset of symptoms to admission ranged between 1 and 12 days with a mean of 4.7 ± 3.6 days. Cerebrospinal fluid (CSF) sample of lumbar puncture showed bloody appearance in all patients and CSF opening pressure was between 240 and 350 mm H2O with an average pressure of 288.2 ± 27.7 mm H2O. Cerebral circulation time was between 9.7 to 12.5 s, with an average time of 10.9 ± 0.7 s.
Imaging procedures
All patients with SAH were diagnosed via non-enhancement CT-scan or MRI. The results proved that SAH localized at the parasagittal in 4 patients and at dorsolateral cerebral convexity in 8 patients and at interhemispheric fissure in 4 patients. Empty delta sign was present in 5(45%) patients under enhanced CT. The presence of superior sagittal sinus in 8 patients, lateral sinus in 4 patients, sigmoid sinus 2, straight sinus in 1 patient and cortical veins in 6 patients were confirmed by MRV or DSA.
Treatments
All patients firstly received systemic heparin anticoagulation after definite diagnosis. 7 patients showed good treatment efficacy upon solely heparin anticoagulant therapy, while 4 patients showed progressive aggravation or no efficacy within 24 h. Then these patients received further endovascular treatment with general anesthesia. 6 F vessel sheaths were inserted in the right femoral artery and femoral vein. Selected 5 F catheter was used to confirmed the location of thrombosis. A 6 F envoy support catheter (Cordis Neurovascular, USA) was placed in the sigmoid sinus under 0.35 loach guidewire. Affected sinus was confirmed with angiography, under which the loach guidewire was repeatly pumped down along the sinus in the row direction to cut the thrombus using the function of mechanical cutting action.
A Prowler 14 microcatheter (Cordis Neurovascular, USA) was directly placed in the distal end of the thrombus and 5 × 105 units of urokinase was administered. Support catheter was removed and the microcatheter was remained after thrombolysis. Four to six days after surgery, urokinase (5 × 104 units per hour) was administered daily via microcatheter. Heparinization was continued and the international normalized ratio (INR) between 1.5 to 2.5 and the activated partial thromboplastin time (APTT) between 18 to 30 S were calculated.
Results
Non-recanalization was carried out in one patient who received intracranial hematoma decompression craniectomy for intracranial hemorrhage after intrasinus thrombolytic treatment. The remaining ten patients exhibited favorable outcome. All patients received repeat DSA for seven days after treatment. Complete recanalization of the occluded sinus or cortical veins was confirmed in 8 and partial recanalization was observed in 2 cases. Cerebral circulation time was shortened, between 7.1 to 12.1 s with an average time of 8.75 ± 1.4 s. Onset of symptoms reduction or disappearing was between 12 h to 96 h after treatment with an average delay of 51.3 ± 25.2 h. Modified Rankin Scale scores improved three months after surgery in ten patients with a score of 0 and one patient with a score of 1. Follow-up lasted for 6-12 months and no recurrence was observed.
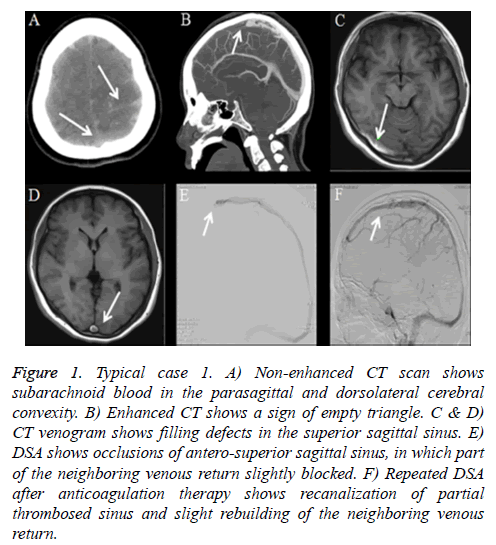
Typical case 1: A 48-year-old female with history of cesarean section postpartum for 22 days suffered sudden onset severe headache for 4 days. Headache located in the occipital without other symptoms. Except for neck pain, there was no other neurological abnormality or indirect signs of elevated intracranial pressure during clinical examination. CT of the brain at the referring hospital uncovered blood in interhemispheric fissure and adjacent sulci. The patient was then transferred to our institution with a diagnosis of SAH. Further brain CT revealed subarachnoid blood in the parasagittal and dorsolateral cerebral convexity (Figure 1A) and enhanced head CT scan revealed a sign of empty triangle (Figures 1B-1D). Cerebrospinal fluid (CSF) sample showed bloody appearance and CSF opening pressure was 240 mm H2O. DSA showed no sign of aneurysm, but occlusion of antero-superior sagittal sinus and slightly part of the neighbouring returning venous (Figure 1E). Cerebral arteriovenous circulation time was 11.3 s. The patient received systemic intravenous heparin for two weeks after which his clinical symptoms and signs of occlusion resolved completely. Recanalization of partial thrombosed sinus was confirmed with repeat DSA (Figure 1F) and cerebral circulation time was shortened to about 8.6 s. The patient continued to take warfarin as an outpatient for 6 months and the International Normalization ratio (INR) was targeted between 2-3. After one year, no recurrence was observed under MRV.
Figure 1: Typical case 1. A) Non-enhanced CT scan shows subarachnoid blood in the parasagittal and dorsolateral cerebral convexity. B) Enhanced CT shows a sign of empty triangle. C & D) CT venogram shows filling defects in the superior sagittal sinus. E) DSA shows occlusions of antero-superior sagittal sinus, in which part of the neighboring venous return slightly blocked. F) Repeated DSA after anticoagulation therapy shows recanalization of partial thrombosed sinus and slight rebuilding of the neighboring venous return.
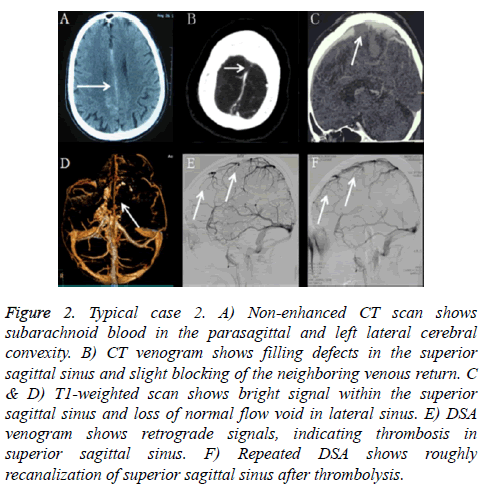
Typical case 2: A 54-year-old male with a 4-month history of repeated and gradually-developing headache over 9 days which was followed by a generalized seizure for twice was with past medical history as Behcet's disease. Emergency head CT reported the diagnosis as SAH in the interhemispheric fissure and convexities (Figure 2A). He was transferred to our institute for further treatment. The patient show normal level of consciousness. Head CT and CTA further confirmed SAH was most prominent along the interhemispheric fissure and convexities (Figure 2B). MRI and MRV revealed thrombosis in superior sagittal sinus and cortical veins and lateral sinus (Figures 2C and 2D).
Figure 2: Typical case 2. A) Non-enhanced CT scan shows subarachnoid blood in the parasagittal and left lateral cerebral convexity. B) CT venogram shows filling defects in the superior sagittal sinus and slight blocking of the neighboring venous return. C & D) T1-weighted scan shows bright signal within the superior sagittal sinus and loss of normal flow void in lateral sinus. E) DSA venogram shows retrograde signals, indicating thrombosis in superior sagittal sinus. F) Repeated DSA shows roughly recanalization of superior sagittal sinus after thrombolysis.
Fluoroscopic-guided lumbar puncture was positive for blood and CSF opening pressure was 290 mm H2O. DSA confirmed cerebral venous sinus thrombosis and cerebral arteriovenous circulation time was 11.2s. He was treated with full anticoagulation immediately. However, in the process of anticoagulation, he suffered progressive aggravation. Then he underwent thrombolysis with daily urokinase (5 × 104 units per hour) applied via a microcatheter in the superior sagittal sinus for 1 week. The blood flow of the superior sagittal sinus was fluent under DSA after thrombolysis (Figures 2E and 2F) and cerebral arteriovenous circulation time is 8.9 s, which was shorter than that before treatment. The lumbar puncture pressure was 180 mm H2O. Two weeks days later, he took warfarin orally. The patient was fully recovering and no relapse occurred during 6 months' follow-up.
Discussion
The most frequent risk factor of CVST is inherited or acquired thrombophilia. Inherited factors include homocysteinemia, factor V homozygous mutation of Leiden, G20210A prothrombin gene and protein C and S and anti-thrombin III deficiency, and positive anti-cardiolipin or anti-phospholipid reactions [8]. Acquired factors include pregnancy and puerperium, oral contraceptives, and intracranial infections and so on [9]. However about one third of CVST cases are etiology unknown. Clinical symptoms of CSVT are extraordinarily variable and hence they are more difficult to be diagnosed. Most of clinical presentation of CVST patients depends on intracranial pressure, the location and extension of the thrombosis and the extent of the venous occlusion [10]. Headache is the most common symptom of CSVT in almost 90% of patients. The most frequent type of headache is diffuse headache and often progressive in severity over days to weeks [11,12]. In 30-40% of patients, there are focal or generalized epileptic seizures as initial presentation [13]. In our observation, most patients presented severe headache as initial presentation and their CSF pressure exceeded normal range, which confirmed the existence of increased intracranial pressure.
Hemorrhagic infarctions were the most common symptoms in CVST patients under CT-scan [4], which is hard to be distinguished from a aneurysmal source or dural arteriovenous fistulas (DAVF). Oppenheim et al. [6] recommended that CVST should be considered if SAH localizes at the parasagittal or dorsolateral cerebral convexity and spares the basal cisterns and skull base. SAH in CVT can be attributed to oozing of blood into the subarachnoid space due to inflammation, localized venous hyperpressure with subsequent dilatation and eventually rupture resulted from extension of sinus thrombosis into cortical or cerebellar veins causes. Sinus thrombosis increases the venous pressure that can cause the dilatation and rupture of the adjacent fragile, thin-walled cortical veins which could lead to localized hemorrhage entering the subarachnoid space [6,14,15]. However Adaletli et al. [15] and Anderson et al. [16] respectively reported SAH was widely extended to those including basal cisterns, bilateral sylvian fissures, and anterior interhemispheric fissure, during which pathological mechanism seemed to be the rupture of superficial vein or veins since only extensive SAH was observed under CT [15].
Since SAH always localized in brain convexity and/or sulci, it could be easily overlooked under CT. According to previous reports, nonenhanced CT could report CVST in only 31% of the patients, while hyperdense thrombus in the occluded sinus, delta sign and cord sign can be revealed under NECT imaging. In addition, intraluminal thrombus with enhancement of the dural sinus wall and empty delta sign could be evident under enhanced CT imaging in 16-46% of cases [17]. Recent reports considered MRI is the most valuable method for diagnosis and localization of acute and subacute low-grade SAH and MRI/MRV can be applied as a technique of choice for definitive diagnosis in all phases and follow-up of CVST [18]. FLAIR is known to be extremely sensitive in diagnosing acute or subacute low-grade SAH whose typical presentation is hyperintensities in subarachnoid spaces. SWI sequence has been demonstrated to be very sensitive to small amounts of SAH. Therefore, SWI may provide complementary information to CT in SAH diagnosis [19]. However in the current studies, DSA was also thought to be the gold standard and the most preferred method to confirm the diagnosis of CVST.
Early recanalization, preventing thrombus aggravation, treating prothrombotic state and preventing recurrence were the aims of the present CVST treatment [3,20]. Hence, once CVST is confirmed, immediate initiation of full anticoagulation therapy is critical [11,21]. But in patients with SAH and CVST, other potential causes of rebleeding should be ruled out before anticoagulant therapy, since the management of the aneurysm and the CVST are absolutely different. Although anticoagulant treatment in patients with intracranial haemorrhages is stills controversial, several studies such as the International Study on Cerebral Vein and Dural Sinus Thrombosis (ISCVT) proved that anticoagulation increased the risk of new intracerebral haemorrhage or increased its severity but was safe to use in patients with either intracerebral haemorrhages or subarachnoid haemorrhages [22,23]. Coutinho et al. [24] thought withholding anticoagulation leaves patient at risk of ongoing occlusion of additional cortical veins and sinuses which can result in more extension of venous infarcts. Although the majority of patients recovered with anticoagulation therapy, only a small percentage of them have not responded to conventional anticoagulant therapy. So endovascular thrombolysis may be considered. Two studies with large samples showed endovascular thrombolysis with fibrinolytic agents could restored blood flow in majority of patients (71.4%) [25,26] and restored blood flow more quickly and efficiently than heparin, although carries the risk of haemorrhage [5].
In this study, all patients received full anticoagulation therapy once CVST was confirmed. During anticoagulation treatment, no increased bleeding or emergence of new bleeding lesion was observed. Our results proved anticoagulation is safe for patients with SAH and CVST. Following anticoagulation treatment, seven patients recovered significantly while four patients exhibited progressive aggravation or no recovery within 24 h who further received anticoagulation treatment combined with intrasinus thrombolysis. After thrombolysis treatment, immediate angiography showed complete recanalization of the occluded sinus or cortical veins in two cases and partial recanalization in one case. However, non-recanalization was still in one patient who exhibited increased bleeding and new bleeding lesions arising after intrasinus thrombolysis. The reason may be that the criminal venous sinus was not timely recanalization and cortical venous collateral was not established that resulted in intracranial hypertension.
Conclusion
Occurrence of CVST combined with SAH is rare. CVST should be firstly considered when SAH localizes at the parasagittal or dorsolateral cerebral convexity and spares the basal cisterns and skull base. But when SAH widely extended, etiology other than arterial bleeding, excluding CVST, needs to be considered as the cause of bleeding. Early diagnosis and full anticoagulation, including intravenous heparin injection or subcutaneous low molecular weight heparin for two to three weeks and then warfarin oral in-taking for three to six months are of great benefit for patients at early phase. Endovascular thrombolysis is an effective treatment for patients who have not responded to anticoagulant therapy. Direct thrombectomy of venous sinus is a further treatment if neither anticoagulation nor thrombolysis works.
References
- Algahtani HA, Aldarmahi AA. Cerebral venous sinus thrombosis. Neurosciences (Riyadh) 2014; 19: 11-16.
- Bousser MG, Ferro JM. Cerebral venous thrombosis: an update. Lancet Neurol 2007; 6: 162-170.
- Stam J. Thrombosis of the cerebral veins and sinuses. N Engl J Med 2005; 352: 1791-1798.
- Fischer C, Goldstein J, Edlow J. Cerebral venous sinus thrombosis in the emergency department: retrospective analysis of 17 cases and review of the literature. J Emerg Med 2010; 38: 140-147.
- Guenther G, Arauz A. Cerebral venous thrombosis: a diagnostic and treatment update. Neurologia 2011; 26: 488-498.
- Oppenheim C, Domigo V, Gauvrit JY, Lamy C, Mackowiak-Cordoliani MA, Pruvo JP, et al. Subarachnoid hemorrhage as the initial presentation of dural sinus thrombosis. AJNR Am J Neuroradiol 2005; 26: 614-617.
- Boukobza M, Crassard I, Bousser MG, Chabriat H. Radiological findings in cerebral venous thrombosis presenting as subarachnoid hemorrhage: a series of 22 cases. Neuroradiology 2016; 58: 11-16.
- Wysokinska EM, Wysokinski WE, Brown RD, Karnicki K, Gosk-Beirska I, Grill D, et al. Thrombophilia differences in cerebral venous sinus and lower extremity deep venous thrombosis. Neurology 2008; 70: 627-633.
- Ferro JM, Canhão P. Cerebral venous sinus thrombosis: update on diagnosis and management. Curr Cardiol Rep 2014; 16: 523.
- Rangel Guerra R. Avances recientes en el diagnóstico y tratamiento de la trombosis venosa cerebral. Medicina Universitaria 2002; 4: 15-27.
- Saposnik G, Barinagarrementeria F, Brown RD, Bushnell CD, Cucchiara B, Cushman M. Diagnosis and management of cerebral venous thrombosis: A statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2011; 42: 1158-1192.
- Sharma B, Satija V, Dubey P, Panagariya A. Subarchnoid haemorrhage with transient ischemic attack: another masquerade in cerebral venous thrombosis. Indian J Med Sci 2010; 64: 85-89.
- Weimar C. Diagnosis and treatment of cerebral venous and sinus thrombosis. Curr Neurol Neurosci Rep 2014; 14: 417.
- Sztajzel R, Coeytaux A, Dehdashti AR, Delavelle J, Sinnreich M. Subarachnoid hemorrhage: a rare presentation of cerebral venous thrombosis. Headache 2001; 41: 889-892.
- Adaletli I, Sirikci A, Kara B, Kurugoglu S, Ozer H, Bayram M. Cerebral venous sinus thrombosis presenting with excessive subarachnoid hemorrhage in a 14-year-old boy. Emerg Radiol 2005; 12: 57-59.
- Anderson B, Sabat S, Agarwal A, Thamburaj K. Diffuse Subarachnoid Hemorrhage Secondary to Cerebral Venous Sinus Thrombosis. Pol J Radiol 2015; 80: 286-289.
- Renowden S. Cerebral venous sinus thrombosis. Eur Radiol 2004; 14: 215-226.
- Sahin N, Solak A, Genc B, Bilgic N. Cerebral venous thrombosis as a rare cause of subarachnoid hemorrhage: case report and literature review. Clin Imaging 2014; 38: 373-379.
- Wu Z, Li S, Lei J, An D, Haacke EM. Evaluation of traumatic subarachnoid hemorrhage using susceptibility-weighted imaging. AJNR Am J Neuroradiol 2010; 31: 1302-1310.
- Einhäupl K, Stam J, Bousser MG, De Bruijn SF, Ferro JM, Martinelli I, Masuhr F; European Federation of Neurological Societies. EFNS guideline on the treatment of cerebral venous and sinus thrombosis in adult patients. Eur J Neurol 2010; 17: 1229-1235.
- Viegas LD, Stolz E, Canhão P, Ferro JM. Systemic thrombolysis for cerebral venous and dural sinus thrombosis: a systematic review. Cerebrovasc Dis 2014; 37: 43-50.
- Selim M. Cerebral venous thrombosis: another heparin controversy. Stroke 2014; 45: 8-9.
- Ghandehari K, Riasi HR, Noureddine A, Masoudinezhad S, Yazdani S, Mirzae MM. Safety assessment of anticoagulation therapy in patients with hemorrhagic cerebral venous thrombosis. Iran J Neurol 2013; 12: 87-91.
- Coutinho JM, Stam J. How to treat cerebral venous and sinus thrombosis. J Thromb Haemost 2010; 8: 877-883.
- Kim SY, Suh JH. Direct endovascular thrombolytic therapy for dural sinus thrombosis: Infusion of alteplase. AJNR Am J Neuroradiol 1997; 18: 639-645.
- Horowitz M, Purdy P, Unwin H, Carstens G 3rd, Greenlee R, Hise J, Kopitnik T, Batjer H, Rollins N, Samson D. Treatment of dural sinus thrombosis using selective catheterization and urokinase. Ann Neurol 1995; 38: 58-67.