- Biomedical Research (2013) Volume 24, Issue 1
Case Report: Potential Speciation in Humans Involving Robertsonian Translocations.
Bo Wang1, Yanzhi Xia2, Jieping Song1, Weipeng Wang1 and Yanping Tang31Genetics Laboratory, Hubei Maternal and Child Health Hospital, Wuhan, Hubei, PR China
2Department of Systems Biology, College of Life Science and Technology, Huazhong University of Science and Technology, Wuhan, Hubei, PR China
3Department of Medical Genetics, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, Hubei, PR China
- *Corresponding Author:
- Yanping Tang
Department of Medical Genetics
Tongji Medical College
Huazhong University of Science and Technology
Wuhan 430030, Hubei, PR China
Accepted date: November 29 2012
Abstract
Approximately one person in 1,000 is a Robertsonian translocation carrier. This type of translocation most likely arises during egg (or more rarely sperm) formation. Most Robertsonian translocation carriers are healthy and have a normal lifespan, but do have an increased risk of pregnancy loss and children with trisomies. We have found a Robertsonian translocation family, one of them who came from a consanguinous marriage has the previously undescribed balanced human karyotype 44,XY,der(14;15)(q10;q10), der(14;15)(q10;q10). Many such rearrangements in natural populations of different species are known. It is well-known that the fitness of rob translocation carries is reduced, but rob translocation can provide material for evolution.
Keywords
Chromosome, Robertsonian translocation homozygosity, evolution
Introduction
The frequency of Robertsonian translocation in newborn babies is approximately one in 1,000. Robertsonian translocation is an unusual type of chromosome rearrangement caused by two particular chromosomes joining together. In humans, it occurs in the five acrocentric chromosomes, 13, 14, 15, 21, and 22. During a Robertsonian translocation, the participating chromosomes break at their centromeres and the long arms fuse to form a single chromosome with a single centromere. The short arms also join to form a reciprocal product, which, in the acrocentric chromosomes, typically contains nonessential genes and repetitive sequences such as nucleolar organizing regions, and is usually lost within a few cell divisions.
Since Robertsonian translocation carriers have a balanced chromosomal complement, they are healthy and have a normal lifespan, and may be unaware of their unusual chromosome rearrangement. A Robertsonian translocation can be transmitted for many generations without detection. Robertsonian translocation carriers produce six kinds of gamete. One sixth of the gametes are normal, and lead to offspring with normal chromosomes. One sixth of the gametes contain the Robertsonian translocation, which is passed to the offspring. The remaining 4 types of gametes lead to offspring with monosomy or trisomy. Because of this, Robertsonian translocation carriers do have an increased risk of pregnancy loss. While a few babies with trisomy 13 or 21 will survive, those with trisomy 14, 15, or 22 usually miscarry in the first twelve weeks.
Case report
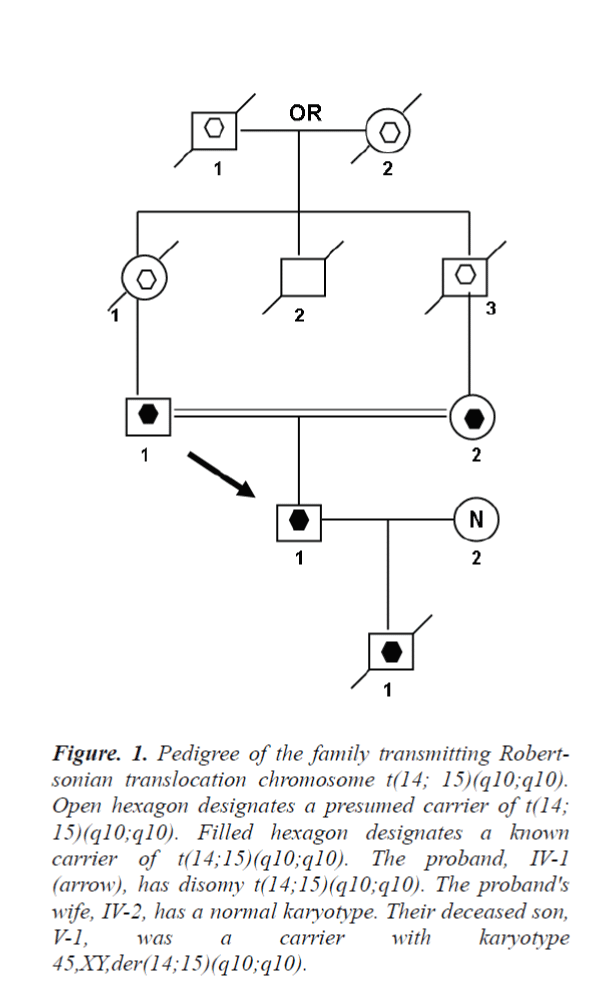
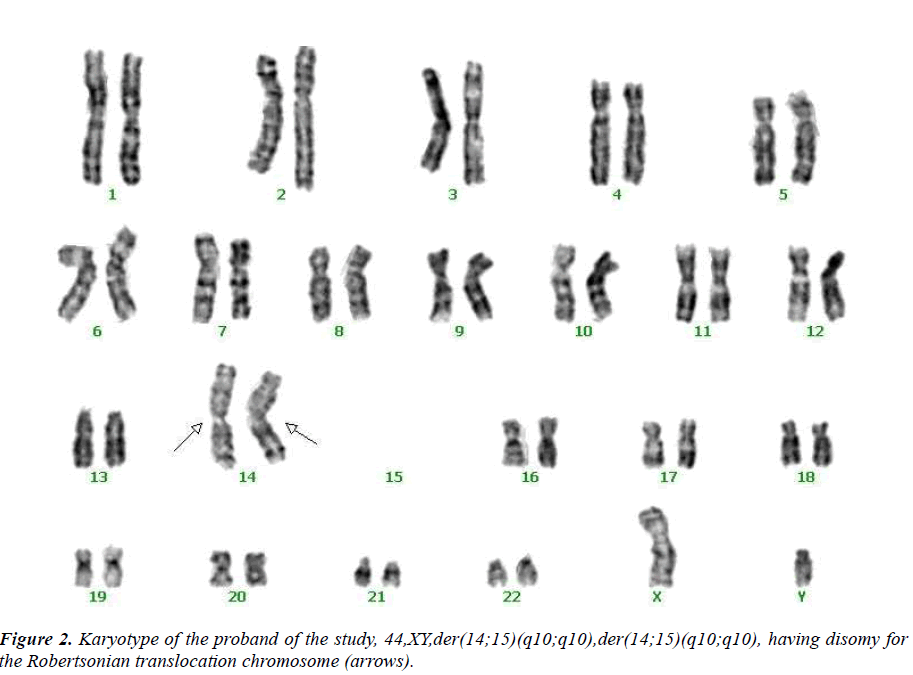
This case report was occasioned by the ascertainment of a 25-year-old Chinese man (IV-1) married to a nonconsanguineous woman with normal chromosomes (IV- 2). This couple had had a son who died at age 6 months, was buried without an autopsy, but had had a chromosome study because of cerebral palsy. The karyotyping analysis of IV-1 is Authenticated by the Chinese Academic Committee of the state key laboratory of medical genetics with the previously undescribed balanced human karyotype 44,XY,der(14;15)(q10;q10),der(14;15)(q10;q10).
The parents of the propositus are phenotypically normal first consins, each a carrier of the same Robertsonian translocation (ı-1, ı-2). Their parents, a mutual uncle, and both grandparents are deceased, thus it is not possible to determine whether I-1 or I-2 was the carrier of the translocation.
The respective probabilities of ı-1 and ı-2 to produce as follow (Fig. 3).
Figure 1: Pedigree of the family transmitting Robertsonian translocation chromosome t(14; 15)(q10;q10). Open hexagon designates a presumed carrier of t(14; 15)(q10;q10). Filled hexagon designates a known carrier of t(14;15)(q10;q10). The proband, IV-1 (arrow), has disomy t(14;15)(q10;q10). The proband's wife, IV-2, has a normal karyotype. Their deceased son, V-1, was a carrier with karyotype 45,XY,der(14;15)(q10;q10).
• Normal gametes is 1/6
• Balanced translocation gametes: 1/6
• Unbalanced gametes is 4/6; and their probability of producing
• A normal karyotyping child is 1/36
• A child with a balanced Rob chromosome is 6/36
• A child with a disomy Rob chromosome is also 1/36
• A genetically unbalanced zygote is 28/36, will increase pregnancy loss in early stage
Discussion
It is well-known that the fitness of rob translocation carries is reduced [2]; in some rob translocations the risk of unbalanced offspring may exceed 20%. The homozygous state of such a rearrangment presumably leads to infertility [3]. The concept of human aneuploidy syndromes as failed “attemps” at speciation was discussed by Neri and Opitz [4] in the context of trisomy “21”.
Rob translocation carries are phenotypically normal since they involve loss only of short arm material. Many such rearrangments in natural populations of different species are known, most famously in the house mouse, Mus poschiavinus (the “tobacco mouse”) being considered a distinct species [5], the genus Muntiacus, the cotton rats (Sigmodon) of North Amecica [6], and even rams with the Massey rob fusion [7]. What is clear from these and many other studies over the last 70-80 years is that rob rearrangments can increase polymorphisms in a species, provide material for natural selection and even lead to speciation [8-10]. John [11] has made the point that the types of chromosome rearrangments that occur as polymorphisms or as fixed permanent heterozygotes invariably involve meta- or submetacentric chromosomes. Those that distinguish species and serve to isolate these species involve telocentric or acrocentric chromosomes, which are selfsterilizing.
Robertsonian rearrangements are common chromosomal changes that can lead to rapid and efficient reproductive isolation between karyotypically similar populations, especially when many rob metacentric chromosomes display monobrachial homologies [12]. In the case of the Muntjac, little or no measurable genetic or morphological differences have been found [13].
Homozygosity for Robertsonian translocations in man has been described before. A fetus with two t(14;21) chromosomes was found by Dallapiccola et al. [14]. The related parents were heterozygous for the same translocation. Martinez et al. [15] described three adult sibs homozygous for t(13;14). Their parents were firstcousins and both were heterozygous carriers. Rajangam et al. [16] found a unique DS karyotype 45, XY, der(14;21)pat, der(14;21)mat, +21mat. Whereas translocation heterozygosity is associated with meiotic disturbances that cause infertility and subfecundity, translocation homozygosity should not have any effect on meiosis-at least in theory [15].
The propositus (IV-1) is healthy and has a balanced chromosomal complement. Assessment of a semen sample from the propositus (IV-1) showed normal sperm number and morphology. We were not able to analyse a sperm karyotype, but given his karyotype of 44,XY,der(14;15)(q10;q10),der(14;15)(q10;q10), we assume his sperm karyotype to be consistently 22,X and 22,Y. Since the propositus is phenotypically normal with normal fertility, we consider his chromosomal rearrangement to be a balanced polymorphism [11]. The aberration can provide material for evolution.
The establishment of a new human subspecies with a diploid complement of 44 chromosomes could occur if a small population with the karyotype of the propositus underwent long-term reproductive isolation.
Conclusion
Although a Robertsonian translocation carrier has a full genetic complement, their fitness is reduced due to high probability of genetically imbalanced gametes. However, this type of translocation can provide material for human evolution. Long term isolation of a group of individuals who are homozygous for a particular Robertsonian translocation chromosome could theoretically lead to the establishment of a new human subspecies having a full genetic complement in 44 chromosomes.
Acknowledgments
We thank Barry Starr and John M. Opitz for their help.
References
- Mateuca R, Lombaert N, Aka PV, Decordier I, Kirsch-Volders M, Chromosomal changes: induction, detection methods and applicability in human biomonitoring. Biochimie. 2006; 88: 1515-1531.
- Stene J. Stengel-Rutkowski S, Genetic risks of familial reciprocal and Robertsonian translocation carriers. Ch.1. In Daniel AR (ed). The cytogenetics of mammalian autosomal rearrangements. New York, Alan R. Liss; 1988: 3–72.
- Gropp A, Winking H, Robertsonian translocations: Cytology, meiosis, segregation patterns and biological consequences of heterozygosity. Symp Zool Soc London,1981 47:141-181.
- Neri G, Opitz JM, Down syndrome: Comments and reflections on the 50th anniversary of Lejeune’s discovery. Am J Med Genet Part A. 2009; 149A: 2647–2654.
- Redi CA., Capanna, E, Robertsonian heterozygotes in the house mouse and the fate of their germ cells. Ch.7. In: Daniel, A. (ed): The cytogenetics of mammalian autosomal rearrangements. New York, Alan R. Liss; 1988: 315–359.
- Elder FFB, Hsu TC, Tandem fusions in the evolution of mammalian chromosomes. Ch.14. In: Sandberg AA (ed) The cytogenetics of mammalian autosomal rearrangements. New York, Alan R. Liss; 1988: 481–506.
- Searle JB, Selection and Robertsonian variation in nature: The case of the common shrew. In: A. Daniel (ed): The cytogenetics of mammalian autosomal rearrangements. New York, Alan R. Liss; 1988: 507–531.
- Baker RJ, Bickham JW, Speciation by monobrachial centric fusions. Proc Natl Acad Sci U S A. 1986; 83:8245-8248.
- Corti M, Capanna E, Estabrook G, Microevolutionary sequences in house mouse chromosomal speciation. Syst Zool. 1986; 35:163–175.
- Vorontsov NN, On the methodology of morphology and levels of morphologic analysis. Zh Obshch Biol. 1989; 50:737-745.
- John B, Chromosome change and evolutionary change: A critique. In: Atchley WR, Woodruff DS, editors. Evolution and specification: Essays in honor of M.J.D. White, Chapter 2. London, 1981, New York: Cambridge University.
- Britton-Davidian J, Catalan J, da Draca Ramalhinho M, et al. Rapid chromosomal evolution in island mice. Nature. 2000; 403:158.
- Wang W, Lan H, Rapid and parallel chromosomal number reductions in muntjac deer inferred from mitochondrial DNA phylogeny. Mol Biol Evol. 2000; 17:1326–1333.
- Dallapiccola B, Ferranti G, Altissimi D, Colloridi F, Paesano R, First-trimester prenatal diagnosis of homozygous (14;21) translocation in a fetus with 44 chromosomes. Prenat Diagn. 1989; 9:555-8.
- P. Martinez-Castro, M. C. Ramos, J. A. Rey, et al. Homozygosity for a Robertsonian-translocation (13q14q) in three offspring of heterozygous parents. Cytogenet. Cell Genet. 1984; 38:310-312.
- Sayee Rajangam, Ron C. Michaelis, GopalRao V.N. Velagaleti, Shavanthi Lincoln, Sridevi Hegde, Sanjeev Lewin, Jack Tarleton, Irene M. Thomas, Avirachan T. Tharapel, Down Syndrome With Biparental Inheritance of der(14q21q) and Maternally Derived Trisomy 21: Confirmation by Fluorescent In Situ Hybridization and Microsatellite Polymorphism Analysis. Am J Med Genet Part A. 1997; 70:43-47.