- Biomedical Research (2014) Volume 25, Issue 3
Cardiovascular autonomic neuropathy in diabetics: Correlation with duration of diabetes.
Bandi Hari Krishna1*, Mallikarjuna Reddy N1, Sharan B, Singh M1, Sasi Kala P1, Kiran Kumar C H1, Uma B V1, Vijetha P1, Shravya Keerthi G21Department of Physiology, Narayana Medical College (NMC), Nellore, Andhra Pradesh, India
2Department of Physiology, Jawaharlal Institute of Postgraduate Medical Education and Research (JIPMER), Puducherry, India
- *Corresponding Author:
- Bandi Hari Krishna
Department of Physiology
Narayana Medical College
Nellore,India
Accepted March 17 2014
Abstract
Cardiovascular Autonomic Neuropathy (CAN) is one of the most overlooked complications of DM and is clinically important because of its life threatening consequences. CAN results from damage to the autonomic nerve fibers that innervate the heart and blood vessels and it causes abnormalities in control of heart rate and vascular dynamics .The present study was designed to assess the cardiovascular autonomic function in type 2 DM. The cardiovascular autonomic function have been conducted in 30 cases of type II DM and 30 controls, aged between 30-50 years, Including both genders, by using Deep breathing test (DBT), Valsalva Maneuvre (VM), Cold Pressor Test (CPT), Lying to standing test (LST) and its correlation with duration of diabetes. The duration of DM was negatively correlated with delta heart rate (-0.908), valsalva ratio (-0.926), rise in diastolic pressure during hand grip test (– 0.747) and cold pressor test (-0.888). From this study it is concluded that, the duration of diabetes has more effect of CAN.
Keywords
Diabetes mellitus, Cardiovascular autonomic neuropathy, Autonomic function tests.
Introduction
Diabetes mellitus (DM), is now taking its place as one of the main threats to human health in the 21st century [1]. The global figure of people with diabetes is set to rise from the current estimate of 150 million to 22 million in 2010 and 300 million in 2025 [2]. Developing countries such as India have had the maximum in the last few years. The current prevalence of type 2 DM is 2.4% in the rural population and 11.6% in the urban population of India. It has been estimated that by 2025 India would be the leading cause of death in adults [2].
Neuropathy is one of the most common complications of diabetes. About half of all people with diabetes have some degree of neuropathy, which can be polyneuropathy, mononeuropthy and/or autonomic neuropathy. Autonomic neuropathy can involve multiple systems including cardiovascular, gastrointestinal, genitourinary, motor and endocrine systems [3]. Diabetic autonomic Neuropathy (DAN) is a stealthy complication of DM developing slowly over the years and quietly robbing diabetic patients of their ability to sense when they are becoming hypoglycemic or having heart attack. DAN is among least recognized and understood complication of DM, despite its significant negative impact on survival and quality of life in people with DM [4,5] it impairs the ability to conduct activities of daily living and increases the risk of death. It also accounts for a large portion of the cost of care [6].
Cardiovascular Autonomic Neuropathy (CAN) is one of the most overlooked complications of DM and is clinically important because of its life threatening consequences [7]. CAN results from damage to the autonomic nerve fibers that innervate the heart and blood vessels and it causes abnormalities in control of heart rate and vascular dynamics [8].
The present study was designed to assess the cardiovascular autonomic function in type 2 DM by using Deep breathing test (DBT), Valsalva Maneuver (VM), Cold Pressor Test (CPT), Lying to standing test (LST) and its correlation with duration of diabetes.
Material and Methods
Patients and controls
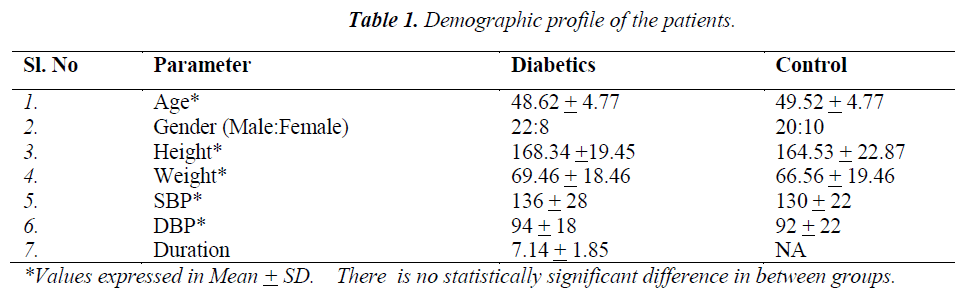
Thirty cases of controlled type II diabetes mellitus and thirty non-diabetic age matched controls, including both males and females, were assessed for cardiovascular autonomic status after obtaining written and informed consent.
The following criteria were followed while selecting the patients as cases:
1. The individuals of controlled (i.e. fasting blood sugar level ≤126 mg/dl and post prandial blood sugar level ≤180 mg/dl) type II diabetes mellitus attending medicine O.P.D. for regular check up.
2. Age between 30-50 years.
3. Not suffering from any other disease or complications.
All the healthy subjects (controls) and patients (cases) were subjected to general and physical examination. Cardiovascular autonomic function tests were carried out in the morning, after familiarizing the subjects with the testing procedures.
Laboratory setting
All experiments were performed at the cardiac autonomic function research laboratory in Dept of Physiology, Narayana Medical College (NMC), Nellore. The patients were asked to refrain from heavy physical activity for 24 hours and from consumption of alcohol and caffeinated beverages for 12 hours prior to the measurements. The temperature of the laboratory was kept between 25o C - 28o C and lights subdued. The patients were asked to void urine before testing and made to sit in the lab comfortably to accustom to the new environment. First heart rate and ascultatory blood pressure was measured after subject had been sitting quietly for 10mins.The mean of three consecutive measurements with a maximum variation of 4mm Hg of both systolic and diastolic blood pressures was accepted [13]
Tests for assessment of cardiovascular autonomic status
The test for the assessment of CAN was done as per standard protocols published in literature [9,10,11,12].
Deep breathing test
The recording of heart rate was done from the ECG recodings on the ECG machine (Cardiowin system, PC based 12 channel simultaneous digital ECG, Genesis Media System Pvt. Ltd, India). A baseline recording of ECG was taken for 30 seconds. The subject was asked to take slow and deep inspiration followed by slow and deep expiration such that each breathing cycle lasted for 10 seconds. Calculation was done from the tracing of ECG. The changes in the heart rate between inspiration and expiration were averaged over 6 cycles.
Valsalva maneuver
It was done in sitting position. The patient was instructed to blow into a mouth piece attached to sphygmomanometer. The expiratory pressure was kept at 40 mmHg for 15 seconds. At the end of 15 seconds the subject was asked to release the pressure. Valsalva Ratio was calculated from the longest RR interval during phase IV and shortest RR interval during phase II.
Handgrip test
The baseline blood pressure was recorded. The subject was asked to press a handgrip dynamometer at 30% of maximum voluntary contraction for 4 minutes. The blood pressure was recorded at 1st, 2nd and 4th minute of contraction. The rise in the diastolic pressure above the baseline was noted.
Cold pressor test
The baseline blood pressure was recorded. The subject was instructed to immerse the right hand in the cold water (8 degree Celsius) for 1 minute up -to the wrist. The blood pressure was measured at the end of one minute. The rise in the diastolic pressure over baseline was noted.
Results
The results of the above tests were compared between the cases (type II DM) and healthy age matched controls and correlated with the duration of diabetes. Demographic profile of the patients and the controls were depicted in Table 1.
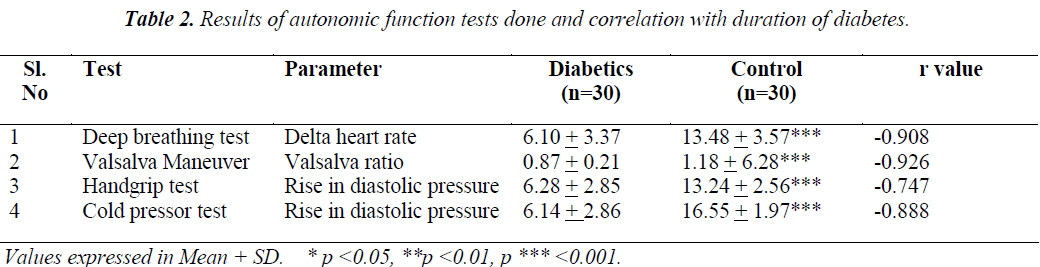
As shown in Table 2, the delta heart rate in deep breathing test, valsalva ratio, rise in diastolic pressure during hand grip test and cold pressor test. The duration of DM was negatively correlated with delta heart rate (-0.908), valsalva ratio (-0.926), rise in diastolic pressure during hand grip test (– 0.747) and cold pressor test (-0.888).
Discussion
The Prevalence of DM is rising rapidly because of increasing obesity and reduced physical activity levels; as countries become more industrialized. Although, the most common neurological disturbance in diabetes is the involvement of peripheral nerves; the concomitant involvement of autonomic nervous system (ANS) has long been known. When ANS is affected, it can lead to variety of symptoms such as tachycardia, orthostatic hypotension, gastro-paresis, fecal incontinence, bladder dysfunction; thus significantly affecting the quality of life of a diabetic patient. Furthermore, the presence of autonomic neuropathy carries a significantly increased risk of cardiovascular mortality, i.e. sudden cardiac arrest. Clinical symptoms of autonomic neuropathy generally do not occur until long after the onset of diabetes. Sub-clinical autonomic dysfunction, can however, occur within a year of diagnosis in type II diabetic patients [14,15] Cardiovascular autonomic neuropathy (CAN) is clinically important form of diabetic autonomic neuropathy (DAN) which is associated with an increased risk of silent myocardial ischemia and mortality. In this study, we tried to assess cardiovascular sympathetic function in cases of controlled type II diabetes mellitus by measuring the delta heart rate in deep breathing test, valsalva ratio, rise in diastolic pressure during hand grip test and cold pressor test, fall in systolic pressure during lying to standing. These results in cases were compared with age matched healthy non diabetic controls. The results in cases were, also analyzed for correlation with the duration of diabetes mellitus.
Normally the heart rate varies continually but this depends on this depends on an intact parasympathetic nerve supply. During deep breathing in DM patients with CAN, may have a noticeable reduction in, and sometimes complete absence of variation. In our study DM patients the delta heart rate is 6.10 + 3.37 in DM and 13.48 + 3.57 in controls which is highly significant (p<0.001) and it is negatively correlated (-0.908) with duration of DM which means that, as duration of DM increases the delta heart rate decreased.
During the strain period of valsalva maneuver the blood pressure drops and the heart rate rises. After release the blood pressure rises, overshooting its resting value and the heart slows. In patients with CAN the blood pressure slowly falls during strain and slowly returns to normal after release, with no overshoot rise in blood pressure and no change in heart rate. In this study the DM patients the valsalva ratio was 0.87 + 0.21 and controls 1.18 + 6.28 which was significantly lower in DM patients (p<0.001) when compared to controls and it is negatively correlated with duration of DM (-0.926) which indicates the duration of DM is the cause for worsening of valsalva ratio.
During sustained handgrip a sharp rise in blood pressure occurs, due to a heart rate dependent increase in cardiac output with unchanged peripheral resistance should the normal reflex pathways be damaged, as in diabetics with extensive peripheral sympathetic abnormalities, the rise in blood pressure is abnormally small. In our patients the rise in diastolic pressure in response to handgrip test was 6.28 + 2.85 whereas in controls it is 13.24 + 2.56 which is highly significant and negatively correlated with the duration of diabetes (-0.747) which clearly shows the effect of duration of DM on CAN.
Under conditions of stress of either physical or psychological origin, there is activation of SNS. The cold pressor response which consists of placing the hand in cold water acts as a painful stimulus and has been used to study the autonomic response. In cold-pressor test, an increase in the SBP by 10-20 mm of Hg and DBP by 10mm ofHg is considered as a normal response. The afferent fibers for this response are the somatic fibers carrying the pain sensation, which are stimulated by placing the hand in cold water, and efferent are the sympathetic fibers. In our study the difference in SBP and DBP values is less in diabetics as compared with controls. The difference in DBP difference values are statistically significant .The possible explanation for this pattern of autonomic nervous dysfunction is the presence of long axons in cardiac parasympathetic systems, which are relatively more sensitive to the metabolic derangement as compared to the relatively short axons in cardiac sympathetic nerves.
Hypotheses concerning the multiple etiologies of DAN include a metabolic insult to nerve fibers, neurovascular insufficiency, autoimmune damage, and, neurohumoral growth factors. Hyperglycemic activation of the polyol pathway leading to accumulation of sorbitol and potential changes in the NAD: NADH ratio may cause direct neuronal damage and/or decreased nerve blood flow. Activation of protein kinase c induces vasoconstriction and reduces neuronal blood flow. Increased oxidative stress, with increased free radical production, causes vascular endothelium damage and reduces nitric oxide bioavailability. Alternatively, excess nitric oxide production may result in formation of peroxynitrite and damage to endothelium and neurons, a process referred to as nitrosative stress. In a subpopulation of individuals with neuropathy, immune mechanisms may also be involved. Reduction in neurotrophic growth factors, deficiency of essential fatty acids, and formation of advanced glycosylation end factors (localized in endoneurial blood vessels) also result in reduced endoneural blood flow and nerve hypoxia with altered nerve function. The result of this multi fractional process may be activation of polyl ADP ribosylation depletion of ATP, resulting in cell necrosis and activation of genes involved in neuronal damage which leads to CAN and this damage is more when the duration of DM increases [16]. From this study it is concluded that, the duration of diabetes has more effect of CAN. So, it is recommended that testing for CAN should be done as a routine investigation of DM patients.
The procedures for performing tests for detection of CAN are safe, non-invasive and require minimal infrastructure.
Limitations and future perspectives
The major limitation of the present study is less sample size. Also, we have not studied the newly diagnosed diabetics. In our future studies, we plan to assess the CAN by in newly diagnosed patients also. The autonomic activity measured in this study is not very accurate. Therefore, future studies should include more accurate methods of assessment of sympathetic activity such as estimation of plasma catecholamine or metabolites of catecholamine in urine like Vanillylamandelic acid (VMA), Metanephrine, and Normetanephrine.
References
- Zimmet P. Globalization, coca-colonization and the chronic disease epidemic: can the Doomsday scenario be averted? J Intern Med. 2000; 247(3): 301-310.
- King H, Aubert R, Herman W. Global burden of dia- betes, 1995-2025, Prevalence, numerical estimates and projections. Diabetes Care. 1998; 21: 1414-1431.
- Chen YD, Reaven GM. Insulin resistance and athero- sclerosis. Diabetes Rev. 1997; 5: 331-343.
- Vinik AI, Erbas T. Recognizing and treating diabetic autonomic neuropathy. Cleve Clin J Med. 2001; 68(11): 928-930.
- Freeman R. The peripheral nervous system and disease. Joslins Diabetes Mellit Weir G Khan R King G 14th Ed. Lippincott, Philadelphia; 2002. p. 794–816.
- Vinik AL, Mitchell BD, Leicher SB, Wagner AL, O’Brain JT, Georges LP. Epidemiology of complica-tions of diabetes. Leslie RDG Robbins DC Diabetes Clin Sci Pract. 1st Ed Cambridge: Cambridge university press1995; 221-287.
- S. Aggarwal, Tonpay PS, Trikha S, Bhansal A. Preva- lence of Autonomic Neuropathy in Diabetes Mellitus. Curr Neurobiol. 2011; 2 (2): 101-105.
- Schummer MP, Joyner SA, Pfeifer MA. Cardiovascular autonomic neuropathy testing in patients with diabetes. Diabetes Spectr 1998; 11: 227-231.
- Mathias CJ and Bannister R. Investigation of auto- nomic disorders. In: Bannister SR and Mathias CJ, edi- tors. Autonomic Failure. A text book of clinical disor- ders of Autonomic Nervous System. 3rd ed. New York: Oxford University Press 1992; 253-290.
- Low PA. Laboratory Evaluation of Autonomic Func- tion. In: Low PA, editor. Clinical Autonomic Disor- ders. 2nd ed. New York: Lippincott-Raven Publishers; 1997; 179-208.
- Oribe E and Appenzeller O. Evaluation of autonomic reflexes. In: Mohr JP and Gautier JG, editors. Guide to Clinical Neurology. 1st ed. New York: Churchil Liv- ingstone Inc; 1995; 213-218.
- Malik M. Clinical Guide to Cardiac Autonomic Tests. Netherlands: Kluwer Academic publishers; 1998.
- Chobanian AV, Bakris GL, Black HR, et al. Joint Na- tional Committee on Prevention, Detection, Evalua- tion, and Treatment of High Blood Pressure. National Hear, Lung, and Blood Institute; National High Blood Pressure Education Program Coordinating Committee. Seventh report of the Joint National Committee on Pre- vention. detection, evaluation and treatment of high blood pressure. Hypertension. 2003; 42: 1206-1252.
- Aaron I. Vinik, Raelene E. Maser and Roy Free- man. Diabetic Autonomic Neuropathy. Diabetes Care 2003; 26: 1553-1579.
- Raelene E. Maser and M. James Lenhard. Cardio- vascular Autonomic Neuropathy due to Diabetes Melli- tus: Clinical Manifestations, Consequences and Treat- ment. Journal of Clinical Endocrinology and Me- tabolism 2005; 90: 5896-903.
- Vinik AI, Maser RE, Mitchell BD, Freeman R. Dia- betic autonomic neuropathy. Diabetes care. 2003; 26 (5): 1553-1579.