- Biomedical Research (2012) Volume 23, Issue 4
Association of serum leptin with various indices of bone turn over in renal transplant patients
Mahmoud Rafieian-kopaei1, Azar Baradaran2, Hamid Nasri3*, Soleiman Kheiri1, Houshang Sanadgol4, Mohamadreza Tamadon51Medical Plants Research Center, Shahrekord University of Medical Sciences, Shahrekord, Iran.
2Department of Clinical Pathology, Isfahan University of Medical Sciences, Isfahan, Iran.
3 Department of Nephrology, Shahrekord University of Medical Sciences, Shahrekord, Iran.
4Department of Nephrology, Semnan University of Medical Sciences, Semnan, Iran.
5Department of Nephrology, Zahedan University of Medical Sciences, Zahedan, Iran.
- Corresponding Author:
- Hamid Nasri
Department of Nephrology
Shahrekord University of Medical Sciences, Shahrekord, Iran.
Accepted date: August 22 2012
Abstract
Leptin is a small peptide hormone that is mainly produced in adipose tissues. Leptin plays an important role in regulating appetite and energy expenditure and may be involved in modulating bone mineralization. The relationship between leptin and bone mineral density and bone biochemical parameters in not completely understood. This study was designed to examine the relationship between some biochemical parameters such as alkaline phosphatase (ALP), phosphorus (P), intact parathormone (iPTH) and calcium (Ca) with serum leptin level in renal transplant recipients. We studied 72 renal transplant patients (47 men and 25 women) with mean ages of 44 (± 12) years and mean body mass index (BMI) of 24.2 ± 3.9Kg/m² .Serum Ca, P, ALP, iPTH and serum leptin were measured. In this study a significant difference of serum leptin between males and females with higher values in females was seen. In patients a significant positive correlation of serum leptin with serum iPTH was found. There was not relationship between serum leptin with ALP, P, Ca, BMI, age and creatinine clearance. A negative relationship between serum leptin and the duration of kidney transplantation was found. Positive relationship of serum leptin with serum parathormone suggests that leptin may affect bone metabolism in renal transplant patients.
Keywords
Leptin, Parathormone, Kidney, Transplantation, Renal function test
Introduction
Renal transplantation (RT) is a treatment of choice for patients with end-stage kidney diseases [1-5]. Kidney transplantation corrects most of the metabolic disorders [4,5]. Post-transplant bone disease is a heterogeneous disease with various effects of age, gender, persistent hyperparathyroidism, corticosteroids and therapy with calcineurin inhibitors [4-10]. Leptin is released from fat tissues and circulating levels of leptin have been shown to be positively associated with fat mass in both lean and obese individuals [10,11]. Previous studies have shown elevated serum leptin levels in renal failure patients [12]. Leptin level is increased before transplantation, and decreased in the first year of RT [13]. Increase in serum leptin level may be seen thereafter and may be due to increase in fat mass, insulin resistance or steroid use in kidney transplant patients [14]. Leptin first emerged as a component of a regulatory fat mass and energy expenditure but recent evidence showed that leptin might be a potential mediator of the protective effects of fat mass on bone tissues [14,15]. Both a peripheral and a central action of leptin on bone have been proposed. Peripherally, leptin is found to exert positive effects on bone formation, whereas it is thought to reduce bone formation through a central control mechanism when binds to its specific receptors located on the hypothalamic nuclei [16-19]. However, the exact role of leptin on bone metabolism has not yet been clarified. It seems that leptin has the ability to affect directly the bone remodeling by modulating both osteoblast and osteoclast activities [17-19]. This study was desgined to evaluate the association between various indices of bone turn-over with serum leptin level in kidney transplant patients.
Material and Methods
Patients
This cross-sectional study was conducted on a group of stable kidney transplant patients, referred to the clinic of nephrologyto continue their treatment. All patients signed the consent form for participation in this study. The etiologies of renal failure were diabetic nephropathy, hypertension, various glomerular diseases, autosomal dominant poly cystic kidney disease (ADPKD) or urinary tract infections [20-27]. Exclusion criteria included presence of acute rejection, any active or chronic infection, taking antibiotics during the past two months, and taking medications such as non steroidal anti-inflammatory drugs. After admission, all patients were examined for blood pressure (BP) and body mass index. Patients' histories concerning the length of the time that they were undergone kidney transplantation and their treatments were also obtained.Body mass index (BMI) was calculated using the standard formula (weight in kilograms/height in square meters) [28].
The basic immunosuppressive regimen of the recipients consisted of a combination of Cyclosporine at a mean dosage of 190 ± 60mg/day (median=200 mg/day), Prednisolon 7.5 mg/day for all of the patients and Mycophenolate mofetile in 46% of the patients at a dosage of 1500 ± 500 mg/day (median=1500 mg/day)/or Azathioprine at a dosage of 50 to 100 mg/day in the rest of the patients. Also patients were under treatment of Calcium carbonate and active vitamin D3 (Calcitriol; Rocaltrol) at various doses.
Laboratory methods
Serum leptin was measured as follows; blood samples were drawn after an overnight fasting, and centrifuged within 15 minutes of drawing. The levels were measured Serum leptin was measured as follows; blood samples were drawn after an overnight fasting, and centrifuged within 15 minutes of drawing. The levels were measured by an enzyme-linked immunosorbent assay (ELISA) method using DRG kit (DRG diagnostic, Berline, Germany). Serum leptin normal rangefor males was considered to be 3.84 (± 1.79) and for females 7.36 (± 3.73) ng/mL. Intact parathormone (iPTH) was measured by radioimmunoassay method (normal range, 10-65 pg/mL). After an overnight fast peripheral venous blood samples were collected for biochemical analysis including serum creatinine (Cr), blood urea nitrogen (BUN), albumin (Alb), serum magnesium (Mg), phosphorus (P), calcium (Ca), alkaline phosphatase (ALP) and serum albumin (Alb) levels, using standard kits. Creatinine clearance was evaluated from serum creatinine, considering age and body weight [29].
Statistical Analysis
The results were expressed as the mean ± SD and median values. A statistical correlation was assessed using a partial correlation test. Comparison between groups were done using student’s t test. For normalization of the iPTH and leptin data, their second square were used. All statistical analyses were performed with the SPSS statistical package (version 11.500 for Windows; SPSS, Chicago, USA). Statistical significance was determined at a Pvalue < 0.05.
Results
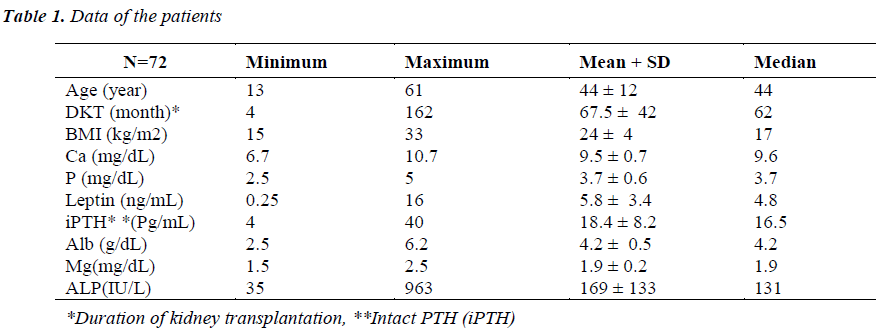
A total of 72 patients were enrolled in this study, including 47 men, 25 women, in whom, 11 patients had diabetes mellitus. The mean patients’ age was 44 (± 12) years. The mean length of the time that they had received a transplant kidney was 67.5 (± 42) months (median: 62 years). The patients data are shown in table 1. In this study there was a significant difference of serum leptin between females and males (r< 0.001; Figure 1). There was no significant difference of serum leptin between diabetic and non diabetic patients. In this study, there was a negative relationship between serum iPTH and creatinine clearance (r=- 0.44 P< 0.001). There was not any correlation between the duration of renal transplantation and serum iPTH (P>0.05).
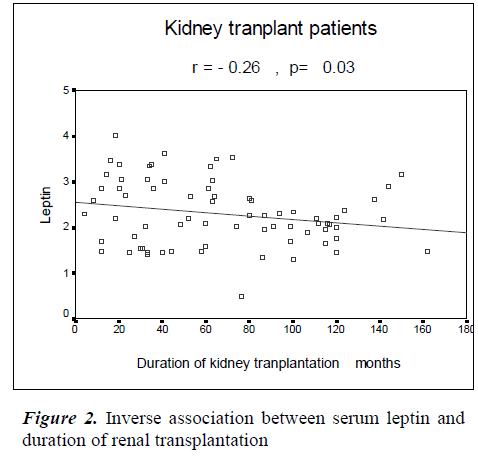
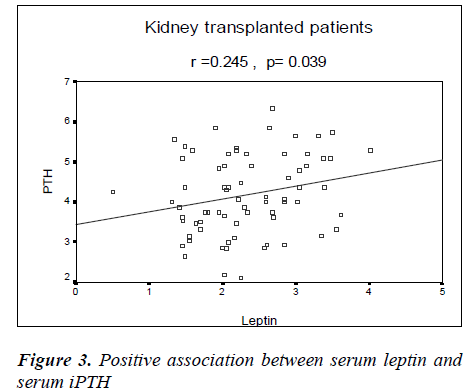
There were inverse correlations of serum ALP with age (r=-0.35 P= 0.02) and duration of kidney transplantation (r=-0.29, P= 0.01). In this study, there was an inverse association between serum leptin and duration of renal transplantation (r=-0.26, P= 0.03; Figure 2). There was not any significant association between serum leptin and creatinine clearance, BMI or ages of the patients (P> 0.05). In this study, we also found a positive association between serum leptin and serum iPTH (r=0.24, P= 0.03; Figure 3). The association of serum leptin with serum Ca, P and ALP was not significant (P> 0.05).
Discussion
In the current study we found a significant difference of serum leptin between females and males, while there was no significant difference of serum leptin between diabetic and non-diabetic patients. There was a negative relationship between serum iPTH and creatinine clearance, however, there was not any correlation between the duration of renal transplantation and serum iPTH. There was inverse correlation between the serum ALP and age of the patients or duration of kidney transplantation. There was an inverse association between serum leptin and duration of renal transplantation. There were not any significant association between serum leptin and creatinine clearance or age of the patients. This study showed also a positive association between serum leptin and serum iPTH and finally the association of serum leptin with serum Ca, P and ALP was not significant.
This study used the association between parathyroid hormone and serum leptin to define bone turn over in kidney transplant patients. Evenepoel et al. studied 1332 kidney allograft recipients, transplant between 1989 and 2000 [30]. They found that 4.1% of their patients had persistent hyperparathyroidism and needed parathyroidectomy after a first successful kidney transplantation. They concluded that persistent hyperparathyroidism requiring parathyroidectomy after successful kidney transplantation is a common clinical problem [30]. While in the present study our results are different, the patients’ mean iPTH being 18.4 (± 8.2) pg/mL (median: 16.5 pg/mL) means there was not any persistent hyperparathyroidism. Studies have shown that parallel changes in body weight and bone mass can be partially mediated via circulating leptin. Among the hormones involved in bone and mineral metabolism, such as oestrogens, testosterone and parathormone, leptin has recently become a subject of considerable interest [31-35]. It is thought that in kidney transplant recipients, serum leptin levels are often elevated and bone mineral density (BMD) decreased [31-35]. Souza et al. carried out a study on thirty-two RT patients and found no correlation between leptin and renal function in their study. They showed also that during the first post transplant year, serum leptin levels decreased significantly in relation to pre-transplant period [13]. Published data ex plaining leptin effects on bone metabolism remain conflicting, with positive and negative effects [14,15,31-35]. It has been shown that increased fat is accompanied by greater serum leptin concentrations, bone mineral mass and a lower risk of osteoporosis [36-42]. Malnutrition reduces serum leptin concentrations and bone mineralization in men [43-50]. It was also found that both serum leptin concentrations and bone mineral are increased at the initiation of puberty [45-54]. These findings show that leptin may modulate bone mineral. Steppan et al. showed in young ob/ob mice that treatment with leptin elevates bone size, bone mineral content, and density, indicating that leptin is needed for bone growth and modeling [55].
Previously we conducted a study on a group of hemodialysis patients. We found a significant positive correlation of serum leptin with iPTH. We concluded that in hemodialysis patients serum leptin may affect bone activity [56]. Agras et al. found that elevated leptin level is associated with increased bone mass at lumbar sites in renal transplant recipients. This finding showed that increased leptin might have a bone-sparing effect, especially in the lumbar region, in RT patients [57]. In a study on 27 kidney allograft recipients, Malyszko et al. studied markers of bone turnover and leptin [58]. They found that leptin was correlated with the percentage of body fat, serum creatinine, and urea. They concluded that leptin levels were associated with graft function and body fat in kidney allograft recipients and leptin was not related to nutritional status or bone metabolism in kidney allograft recipients, but it was associated with the current dosage of immunosuppressant and the serum calcium [58]. Recently, in a study on a group of healthy women, Maetani et al. observed strong positive association between leptin and PTH levels [59]. In the present study, we tried to consider the interrelationship between serum leptin and bone turn over or parathyroid activity in a group of kidney transplant patients. Further studies are needed to clarify significance of our findings.
Acknowledgements
This study was supported by a grant from Shahrekord University of Medical Sciences.
Ethical Issues.This study was approved by the ethical committee of Shahrekord University of Medical Sciences.
Conflict of Interest. The authors declare that they have no conflict of interest.
Authors' Contributions. HN and MRK defined the aims of research and the design of experiment and carried out the experiments. AB and SK participated in the design of the study and performed the statistical analysis. HS and MRT reviewed the paper. All authors read and approved the final manuscript.
References
- Lehmann G, Ott U, Stein G, Steiner T, Wolf G. Renal osteodystrophy after successful renal transplantation: a histomorphometric analysis in 57 patients. Transplant Proc. 2007; 39(10): 3153-3158.
- Tayebi Khosroshahi H. Short history about renal transplantation program in Iran and the world: Special focus on world kidney day 2012. J Nephropathology. 2012; 1 (1): 5-10.
- Einollahi B. Are acquired cystic kidney disease and autosomal dominant polycystic kidney disease risk factors for renal cell carcinoma in kidney transplant patients? J Nephropathology. 2012; 1(2): 65-68.
- Rolla D. Effects of kidney transplantation on bone: osteoporosis or other? G Ital Nefrol. 2008; 25 (4): 432- 440.
- Assadi F. The epidemic of pediatric chronic kidney disease the danger of skepticism. J Nephropathology. 2012; 1 (2): 61-64.
- Roe SD, Porter CJ, Godber IM, Hosking DJ, Cassidy MJ. Reduced bone mineral density in male renal transplant recipients: Evidence for persisting hyperparathyroidism. Osteoporos Int 2005; 16(2):142-148.
- Tolou-Ghamari Z. Nephro and neurotoxicity, mechanisms of rejection: A review on Tacrolimus and Cyclosporin in organ transplantation. J Nephropathology. 2012; 1 (1): 23-30.
- Gheissari A, Mehrasa P, Merrikhi A, Madihi Y. Acute kidney injury: A pediatric experience over 10 years at a tertiary care center. J Nephropathology 2012; 1 (2): 101-108.
- Mubarak M, Kazi JI, Kulsoom U, Ishaque M. Detection of immunoglobulins and complement components in formalin fixed and paraffin embedded renal biopsy material by immunoflourescence technique. J Nephropathology. 2012; 1 (2): 91-100.
- Nasri H. Inverse association ofserum Leptin with serum C-reactive protein (CRP) in regular hemodialysis patients. Rawal Med J 2006; 31(1): 10-13.
- Nasri H. Positive correlation of serum homocysteine with Leptin in maintenance hemodialysis patients. Arch Med Sci 2006; 3: 185-189.
- El Haggan W, Chauveau P, Barthe N, Merville P, Potaux L, Aparicio M. Serum leptin, body fat, and nutritional markers during the six months post-kidney transplantation. Metabolism. 2004; 53 (5): 614-619.
- Souza GC, Costa CA, Gonçalves LF, Manfro RC. Leptin serum levels in the first year post-renal transplantation. Transplant Proc. 2007; 39(2): 439-440.
- Kayacan SM, Yildiz A, Kazancioglu R, Sahin S, Sever MS, Ark E. The changes in serum leptin, body fat mass and insulin resistance after renal transplantation. Clin Transplant. 2003; 17(1): 63-68.
- Sato M, Takeda N, Sarui H, Takami R, Takami K, Hayashi M, Sasaki A, Kawachi S, Yoshino K, Yasuda K. Association between serum leptin concentrations and bone mineral density, and biochemical markers of bone turnover in adult men. J Clin Endocrinol Metab. 2001; 86(11): 5273-5376.
- Karsenty G. Leptin controls bone formation through a hypothalamic relay. Recent Progress in Hormone Research 2001; 56; 401-415.
- Thomas T, Gori F, Khosla S, Jensen MD, Burguera B & Riggs BL. Leptin acts on human marrow stromal cells to enhance differentiation to osteoblasts and to inhibit differentiation to adipocytes. Endocrinology 1999; 140: 1630-1638.
- Holloway WR, Collier FM, Aitken CJ, Myers DE, Hodge JM, Malakellis M, Gough TJ, Collier GR & Ni cholson GC. Leptin inhibits osteoclast generation. J Bone and Mineral Res 2002 17: 200-209.
- Martin A, de Vittoris R, David V, Moraes R, Begeot M, Lafage-Proust MH, Alexandre C, Vico L & Thomas T. Leptin modulates both resorption and formation while preventing disuse-induced bone loss in tailsuspended rats. Endocrinology 2005; 146: 3652-3659.
- Ghorbani A, Ehsanpour A, Roshanzamir N, Omidvar B. Alterations in antibiotic susceptibility of urinary tract infection pathogens. J Nephropathology. 2012; 1 (1): 43-48.
- Nasri H, Mortazavi M, Ghorbani A, Shahbazian H, Kheiri S, Baradaran A, et al. Oxford-MEST classification in IgA nephropathy patients: A report from Iran. J Nephropathology. 2012; 1 (1): 31-42.
- Mubarak M. Oxford classification of IgA nephropathy: Broadening the scope of the classification. J Nephropathology. 2012; 1(1): 13-16.
- Mohammadi Torbati P. Focal segmental glomerulosclerosis; collapsing variant. J Nephropathology. 2012; 1 (2): 87-90.
- Mubarak M, Collapsing focal segmental glomerulosclerosis: increasing the awareness. J Nephropathology. 2012; 1 (2): 77-80.
- Ardalan MR, Samadifar Z, Vahedi A. Creatine monohydrate supplement induced interstitial nephritis. J Nephropathology. 2012; 1(2): 117-120.
- Khajehdehi P. Turmeric: Reemerging of a neglected Asian traditional remedy. J Nephropathology. 2012; 1(1): 17-22.
- Karimifar M. Deep vein thrombosis in combination with granulomatosis with polyangiitis (Wegener's). J Nephropathology 2012; 1 (1): 57-58.
- Baradaran A, Behradmanesh S, Nasri H.Association of body mass index and serum vitamin D level in healthy Iranian adolescents.Endokrynol Pol 2012; 63 (1): 29- 33.
- Cockcroft DW, Gault MH. Prediction of creatinin clearnce from serum creatinine nephron 1976; 16: 31- 41.
- Evenepoel P, Kuypers D, Maes B, Messiaen T, Vanrenterghem Y. Persistent hyperparathyroidism after kidney transplantation requiring parathyroidectomy. Acta Otorhinolaryngol Belg. 2001; 55(2): 177-186.
- Ormarsdottir S, Ljunggren O, Mallmin H, Olofsson H, Blum WF, Loof L. Inverse relationship between circulating levels of leptin and bone mineral density in chronic liver disease. J Gastroenterol Hepatol. 2001; 16: 1409-1414.
- Blain H, Vuillemin A, Guillemin F, Durant R, Hanesse B, de Talance N, Doucet B, Jeandel C. Serum leptin level is a predictor of bone mineral density in postmenopausal women. J Clin Endocrinol Metab 2002; 87: 1030-1035.
- Odabasi E, Ozata M, Turan M, Bingol N, Yonem A, Cakir B, Kutlu M, Ozdemir IC. Plasma leptin concentrations in postmenopausal women with osteoporosis. Eur J Endocrinol 2000; 142: 170-173.
- Pasco JA, Henry MJ, Kotowicz MA, Collier GR, Ball MJ, Ugoni AM, Nicholson GC. Serum leptin levels are associated with bone mass in nonobese women. J Clin Endocrinol Metab 2001; 86: 1884-1887.
- Thomas T, Burguera B, Melton 3rd LJ, Atkinson EJ, O’Fallon WM, Riggs BL, Khosla S. Role of serum leptin, insulin, and estrogen levels as potential mediators of the relationship between fat mass and bone mineral density in men versus women. Bone 2001; 29: 114- 120.
- Martini G, Valenti R, Giovani S, Franci B, Campagna S, Nuti R. Influence of insulin-like growth factor-1 and leptin on bone mass in healthy postmenopausal women. Bone 2001; 28: 113-117.
- Kontogianni MD, Dafni UG, Routsias JG, Skopouli FN. Blood leptin and adiponectin as possible mediators of the relation between fat mass and BMD in perimenopausal women. J Bone Miner Res 2004; 19: 546- 551.
- Ruhl CE, Everhart JE . Relationship of serum leptin concentration with bone mineral density in the United States population. J Bone Miner Res 2002; 17: 1896- 1903.
- Yamauchi M, Sugimoto T, Yamaguchi T, Nakaoka D, Kanzawa M, Yano S, Ozuru R, Sugishita T, Chihara K. Plasma leptin concentrations are associated with bone mineral density and the presence of vertebral fractures in postmenopausal women. Clin Endocrinol (Oxf). 2001; 55: 341-347.
- Roemmich JN, Clark PA, Berr SS, Mai V, Mantzoros CS, Flier JS, Weltman A, Rogol AD. Gender differences in leptin levels during puberty are related to the subcutaneous fat depot and sex steroids. Am J Physiol. 2001; 275: E543-E551.
- Horlick MB, Rosenbaum M, Nicolson M, Levine LS, Fedun B, Wang J, Pierson JR RN, Leibel RL Effect of puberty on the relationship between circulating leptin and body composition. J Clin Endocrinol Metab. 2000; 85: 2509-2518.
- Reid IR, Ames R, Evans MC, Sharpe S, Gamble G, France JT, Lim TM, Cundy TF. Determinants of total body and regional bone mineral density in normal postmenopausal women-a key role for fat mass. J Clin Endocrinol Metab 1992; 75: 45-51.
- Khosla S, Atkinson EJ, Riggs BL, Melton 3rd LJ. Relationship between body composition and bone mass in women. J Bone Miner Res 1996; 11: 857-863.
- Young D, Hopper JL, Macinnis RJ, Nowson CA, Hoang NH, Wark JD. Changes in body composition as determinants of longitudinal changes in bone mineral measures in 8- to 26-year-old female twins. Osteoporos Int 2001; 12: 506-515.
- Tremollieres FA, Pouilles JM, Ribot C 1993 Vertebral postmenopausal bone loss is reduced in overweight women: a longitudinal study in 155 early postmenopausal women. J Clin Endocrinol Metab 1993; 77: 683-686.
- Kirchengast S, Peterson B, Hauser G, Knogler W. Body composition characteristics are associated with the bone density of the proximal femur end in middleand old-aged women and men. Maturitas. 2001; 39: 133-145.
- Dubuc GR, Phinney SD, Stern JS, Havel PJ. Changes of serum leptin and endocrine and metabolic parameters after 7 days of energy restriction in men and women. Metabolism 1998; 47: 429-434.
- Argente J, Barrios V, Chowen JA, Sinha MK, Considine RV. Leptin plasma levels in healthy Spanish children and adolescents, children with obesity, and adolescents with anorexia nervosa and bulimia nervosa. J Pediatr. 1997; 131:833-838.
- Soyka LA, Grinspoon S, Levitsky LL, Herzog DB, Klibanski A. The effects of anorexia nervosa on bone metabolism in female adolescents. J Clin Endocrinol Metab. 1999; 84: 4489-4496.
- Karlsson MK, Weigall SJ, Duan Y, Seeman E. Bone size and volumetric density in women with anorexia nervosa receiving estrogen replacement therapy and in women recovered from anorexia nervosa. J Clin Endocrinol Metab. 2000; 85: 3177-3182.
- Clayton PE, Gill MS, Hall CM, Tillmann V, Whatmore AJ, Price DA. Serum leptin through childhood and adolescence. Clin Endocrinol (Oxf). 1997; 46:727–733.
- Lu PW, Cowell CT, Loyd-Jones SAL, Briody JN, Howman-Giles R. Volumetric bone mineral density in normal subjects, aged 5–27 years. J Clin Endocrinol Metab. 1996; 81: 1586-1590.
- Mantzoros CS, Flier JS, Rogol AD. A longitudinal assessment of hormonal and physical alterations during normal puberty in boys. V. Rising leptin levels may signal the onset of puberty. J Clin Endocrinol Metab. 1997; 82: 1066-1070.
- Ibanez L, Potau N, Ong K, Dunger DB, De Zegher F. Increased bone mineral density and serum leptin in non-obese girls with precocious pubarche: relation to low birth weight and hyperinsulinism. Horm Res 2000; 54: 192-197.
- Steppan CM, Crawford DT, Chidsey-Frink KL, Ke H, Swick AG. Leptin is a potent stimulator of bone growth in ob/ob mice. Regul Pept. 2000; 92: 73-78. 56.
- Nasri H, Association of Serm Leptin with Various Biochmical Parameters of Bone turnover in Maintenance Hemodialysis Patients. Pakistan Journal of Nutrition 2006; 5 (2): 180-184.
- Agras P I; Baskin E; Saatci U; Colak T; Cengiz N; Kinik S T; Isiklar I; Haberal A; Mert I; Haberal M. Relationship between leptin and bone mineral density in renal transplant recipients.Transplantation proceedings 2005; 37(7): 3106-3108.
- Malyszko J, Malyszko JS, Pawlak K, Konstantynowicz J, Wolczynski S, Kaczmarski M, Mysliwiec M. Correlations between leptin, body composition, bone mineral density, and bone metabolism in kidney transplant recipients. Transplant Proc. 2005 Jun; 37(5): 2151-2153.
- Maetani M, Maskarinec G, Franke AA, Cooney RV. Association of leptin, 25-hydroxyvitamin D, and parathyroid hormone in women. Nutr Cancer. 2009; 61 (2): 225-231.