Research Article - Journal of Oral Medicine and Surgery (2022) Volume 5, Issue 1
Assessment of bone regeneration in mandibular defect with hyaluronic acid and rosemary to nanoparticles hydroxyapatite in rabbits.
Noor A. Sulaiman*, Israa A. Abdulhameed, Huda A. Salim
Department of Oral and Maxillofacial Surgery, College of Dentistry, Mosul University, Mosul, Iraq
- Corresponding Author:
- Noor A. Sulaiman
Department of Oral and Maxillofacial Surgery
Mosul University, Mosul Iraq
Tel: 009647704818554
E-mail: noorabdullah@uomosul.edu.iq
Received: 16-oct-2021, Manuscript No. aaomt-22-44690; Editor assigned: 19-Oct-2021, PreQC No. aaomt-22-44690 (PQ); Reviewed: 03-Dec-2021, QC No. aaomt-22-44690; Revised: 27-Dec-2021, Manuscript No. aaomt-22-44690 (R); Published: 03-Jan-2022, DOI:10.35841/aaomt-5.1.101
Citation:Noor Sulaiman A, Israa Abdulhameed A, Huda Salim A. Assessment of bone regeneration in mandibular defect with hyaluronic acid and rosemary to nanoparticles hydroxyapatite in rabbits. J Oral Med Surg. 2022;5(1):101.
Abstract
This study was conducted to assess the effectiveness of a newly formulated novel paste of natural herbs in bone defect regeneration in rabbits. A total of 42 adult male rabbits were included in this study and randomized into three groups according to healing periods and bone defect induced. The bone defects were treated with different materials added to hydroxyapatite which is made from seashells which served as a control, these are Hyaluronic acid, Rosemary with Hyaluronic acid. At week one, the control group presented with an inflammatory reaction score of 2 that increased gradually to a mean of scoring 3.85 at 1 month, these results were also seen in granulation tissues and blood vessel formation. New bone, osteoblast, and osteoclast formation revealed the same results with a mean of 0.42 at 1 week and 1.42 in 2 weeks later, which increased to 2.85 at 1 month for both new bone formation and osteoblast, while osteoclast was 3 after 1 month. In a group treated with (hydroxyapatite and hyaluronic acid), the risk of inflammatory reactions and blood vessel formation increased, while decreasing in means of granulation tissue formation than the control group during the study period, both new bone and osteoblast formation showed the same mean with 1.42 at 1 week, and 2.14 at 2 weeks then increased to 3 after 1 month, there were no osteoclast cells present. The last group showed an inflammatory reaction mean of 4 through the study period, granulation tissue formation mean was 2 that increased to 3; blood vessels means was 3 that decreased to 1 after 1 month, new bone and osteoblast formation revealed similar results with a mean of 3 with no osteoclast cells. Rosemary has an anti-inflammatory effect and can accelerate bone formation by increasing the differentiation of mesenchymal cells to osteoblast cells.
Keywords
Bone regeneration, Hyaluronic acid, Hydroxyapatite nanoparticles, Nanotechnology, Rosemary.
Introduction
Bone is a dynamic tissue that has considerable potential for healing, especially with small defects that can heal spontaneously [1]. However, critical size defects can be extremely challenging to be restored in clinical practice, especially in the maxillofacial area as it is a highly demanding region both esthetically and functionally [2]. Trauma, resections due to cancer, orthognathic surgeries, infections, or developmental deformities all lead to defects in a facial bone that need reconstruction [3]. Bone grafts used for reconstruction are divided according to their regenerative properties into autografts, allografts, xenografts, and phalloplasty, they either have osteogenic activity (comprise osteoblast cells), osteoinductive (contain bone inducing substances), or they are osteoconductive (serve as a scaffold for bone formation).
The most effective grafting materials are autograft and allograft as they have more capacities for bone regeneration [4]. Yet, due to these grafts being associated with many limitations like donor site morbidity, inadequate supply, costly autographs, and risk of disease transmission and rejection of allografts, another possibility such as phalloplasty has been used. An alloplastic is a synthetic or inert foreign body material that has osteoconductive capacity. It can be manufactured in unrestricted quantities, and no risk of disease transmission can be detected. Allopasts nowadays used are coralline hydroxyapatite, collagen-based matrices, calcium sulfate, and tri-calcium phosphat. Marine skeletons are standard bioresources own special constructions which offer structural support and multiple actions suitable for human tissue reformation and rehabilitation [5]. Calcium phosphates, particularly hydroxyapatite (Hap) and tri-calcium phosphate (TCP) can turn up from naturalistic materials consist of calcium carbonates such as sea coral, mussels, eggshells, and nacre Venus Verrucosa for biomedical applications. Many processes have been used for the reconstruction of (Hap) comprise precipitation, mechanochemical, and hydrothermal processes. Biocompatibility (Hap) has many properties, including biocompatibility, bioactivity, low solubility in water, and the ability to replace toxic ions, so it is widely used in biomaterials [6]. Compared to traditional hydroxyapatite, an interesting formulation of Nanophase hydroxyapatite has mineral composition parallel to that of bone with higher osteoplastic activity, bioactive constancy, and mechanical architectural leading to the formation of malleable, strong, and liable bone graft, nanotechnology introduced by Richard Feynman in 1959 [7]. Nanotechnology uses in bone healing have the possibility of success due to the structure of bone is made from nanoscopic collagen fibres, applications of nanotechnology in tissue and organ rejuvenation enlarged over time, it provides motivating properties in the field of regenerative medicine [8]. In this study, we used nanoparticle hydroxyapatite derived from seashells by the mechanochemical procedure.
Hyaluronic acid (HA) known as hyaluronan or hyaluronate is a major component of the extracellular matrix almost in all living tissues. It has a key role in the function of both extracellular mineralized and non-mineralized matrices, which comprise cell migration, proliferation, and differentiation with tissue hydrodynamics. The anti-inflammatory property of HA recently revealed that it increases osteoblastic bone formation in vitro by accelerating mesenchymal cell differentiation and migration [9].
Rosemary (Rosmarinus officinalis L.) is a woody plant of the Lamiaceae group that grows in the Mediterranean region. It has been used as a spice and zest flavour in food. It has been shown that rosemary has various physiological roles like memory enhancement, anti-inflammation, antioxidant, and osteoporosis suppressant. Rosemary extracts have been used as herbal remedies in old popular medicine, and its oil possesses numerous health advantages to include inhibition of bronchial asthma, atherosclerosis, ischemic heart diseases, and anticarcinogen [10,11]. Many studies revealed that rosemary had an anti-osteoporotic effect as it contains erucic acid that potentiates the differentiation of MSCs into osteoblasts [12,13].
As there are many previous studies evaluated uses of materials, like hydroxyapatite, hyaluronic acid, and rosemary, as a substitute for bone reconstruction separately. In this study, we hypothesized that a combination of these materials [hydroxyapatite, hyaluronic acid, and rosemary] together may have a significantly higher effect on bone regeneration and accelerate bone defect reconstruction. This study aimed to assess the effect of the new combination of hyaluronic acid and rosemary oil extract added to nanoparticles hydroxylapatite on bone regeneration of mandibular defect in rabbits.
Materials and Methods
Preparation of materials
Preparation and characterization of Hydroxyapatite nanoparticles from seashell: 100 g of seashell cleaned by distilled water thoroughly, boiled for 10 minutes to remove any debris or pathogens, then the internal protein layer from shells was removed and dried. After that shells grounded to produce powder, decarbonized by heating at 900°C to form whitish colour powder of calcium oxide (CAO) according to Alkhalidi EF et al 2015, each 100 g of seashell produce 64 g of (CAO). To create principle hydroxyapatite, 30 ml of phosphoric acid added to 50 g of (CAO) purchased from a seashell, vapour and heated under the control of PH and reactant concentration, the resulted powder dried at 150°C for 2 hours according to the method produced by Amer A. Taqa et al 2014 [14]. Efficient parts in newly created hydroxyapatite products were checked by Alfa Bruker instrument Infrared Spectroscopy (FTIR), Germany. Nanoparticles were formed by the mechanical attrition method to produce refined nano-Hydroxyapatite (nHAp) from a previously prepared powder of seashell which fragmented and crushed to minute pieces with a size of 20-35 nm [15,16]. The structural characterization of nHAp powder manufactured samples were measured in the laboratory of Al-Nahrain University using a Philips machine (Model CM10, Holland) Scanning Electron Microscope (SEM).
Preparation of Rosemary extract
Rosemary oil extract was obtained according to the method explained by Jennifer et.al, 2017, desiccated rosemary leaves from Alrayan Medical Herbal Center, Erbil, Iraq, got from Morocco, reigning vegetal verification as Rosmarinus officinalis L. used to obtain abstract washed with water them hydrodistelized for 3 hours in a Clevenger apparatus to produce 5 g of oil extract that stored in a sterile basin at –10°C until to use [17].
Therapeutic effective components made from; 0.25 ml of hyaluronic acid (HA) presented as hydrogel in a sterilized syringe of 1.2 ml size commercially available for dental uses (Hyadent®, BioScience GmbH, Dümmer, Germany), 25 μl oil of rosemary abstract (Rosemary Officinalis L.) and hydroxyapatite as nanoparticles prepared from a seashell. The main ingredients were integrated and thoroughly merged to produce homogenized paste five minutes before treatment. Concentrations of active elements were documented by preceding outcomes of original in vitro studies (not presented). Each bone defect was treated according to the plan established for it in this study. One of these defects was treated daily with hydroxyapatite (Hap) nanoparticles powder, other one received 0.25 ml of hyaluronic acid (HA) (Hyadent®, BioScience GmbH, Dümmer, Germany) added to hydroxyapatite (Hap), last one treated by daily 25 μl of rosemary extract oil (Rosemary Officinalis L.) (Morocco) got from (Alrayan Medical Herbal Center, Erbil, Iraq) with 0.25 ml of hyaluronic acid (HA) and (Hap). All of the measured active ingredients were added under continuous stirring and mixed very well to provide daily treated dose/ defect according to the study design.
The strategies and protocol of this study had been accepted by the Scientific Committee of the Oral and Maxillofacial Department of College of Dentistry and College of Dentistry/ Mosul University Council, number of session 381 in 14-5-2019. Animal operations were performed causing no harshness on empirical animals.
Animals
42 healthy household male rabbits of 7-8 months old, weighing 2 ± 0.5 kg were taken. All of them were kept in a chamber under a stable temperature of 24 ± 2 Co and normal light cycle, fed individually with a natural diet (corn, vegetables, and cereals) and water. Prior to treatment, the rabbits were assessed for 1 week to observe their general health and be sure that they did not have any contagious disorders.
Surgical Procedure
Rabbits were arbitrarily separated into 3 equivalent sets according to the time of bone healing to 1, 2 weeks, and 1 month, each group containing 14 animals, 3 artificial defects were made in the mandible of each rabbit by the researcher. Each rabbit was given a mixture of xylazine 4 mg/kg and 40 mg/kg ketamine Paknejad et al., 2007, Kilic, 2004, injection in the rabbit thigh muscle, through operation additional sedation was given as necessary. Complete anaesthesia had been attained within 5 min, the surgical field was shaved, cleaned, and disinfected with povidone-iodine solution and covered with a sterile towel. In the surgical area, local anaesthesia (1 ml of 2% lidocaine with epinephrine 1:80,000, New Stetic com, Colombia) was given, three defects of 0.5 cm length and 0.5 cm depth has been created by round bur in the mandible of each rabbit, a distance of about 2 cm was left between one defect and another, after reflection of the mucoperiosteal flap gently by blunt dissector, one of the defects filled by seashell hydroxyapatite only to serve as a control. The middle defect was treated with a paste of hyaluronic acid gel+ seashell powder. While the final defect was treated with a paste of hyaluronic acid gel and Rosemary extract oil added to the seashell powder. Animals were sacrificed at 1 week, 2 weeks, and 1 month after an intervention, biopsies from all animals of each group were taken and histopathological examination was done [18,19].
Histopathological examination and scoring system
Tissue specimens were fixed in 10% formalin, then desiccated over ascending gradient sequences of xylene and ethanol prior to establishing in paraffin wax. Slices of 4-5 μm density were sheared off and discoloured with hematoxylin and eosin. Inspection of slides was done by a binocular light microscope (Optika, Ponteranica BG- Italy) which linked to a digital camera 8 megapixel (Aiptek, Germany) to take pictures of each group's histologic segments and saved on a personal computer. The histopathological examination was accomplished by 2 professional pathologists employing the double-blind screening method. All slides were assessed under 4 X and 10 X magnification low power field (LPF) to evaluate the wound healing [20,21].
The histopathological scoring used was that of Sultana and Solchagal with small modifications, as the follows:
A. Inflammatory infiltrate: 1. Plenty. / 2.Moderate. / 3.Few. / 4.Absent.
B. Amount of granulation tissue: 1. Profound./ 2.Moderate. / 3.Scant. / 4.Absent.
C. De novo formation of blood vessels:
0. Absent. / 1.Present peripherally. / 2.Present centrally. / 3.Present centrally and peripherally.
D. New bone formation: 0. Absent/ 1. Present at the periphery. / 2. Present centrally. / 3. Present centrally and the periphery.
E. Presence of osteoblasts: 0. Absent. / 1.Present peripherally./ 2.Present centrally./ 3.Present centrally and peripherally.
F. Presence of osteoclasts: 0. Absent./ 1.Present peripherally./ 2.Present centrally./ 3.Present centrally and peripherally.
Anthropometric measurements viz, height, weight, waist and hip circumference were taken according to standard protocols, for all the adults among selected households. Anthropometric indices such as Body Mass Index (BMI) and Waist Hip Ratio (WHR) were calculated to classify based on WHO standards.
Data analysis
The data of each group presented as mean separately with standard deviation SD. IBM SPSS Statistics version 25.0 statistical program was used for the analysis of data. The comparison of non-numeric data of unrelated samples among the group was done by the Kruskal-Wallis test. Differences were considered significant at p ≤ 0.05.
Results
All animals were clinically healthy at the time of sacrifice. All specimens in the study showed no signs of infection, dehiscence, or any abnormalities on observation. The average inflammatory reaction scores after 1 week were 2 for the control group and 3.71 for a second while the third group was 4 and there were significant differences in favour to the third group than control with P value 0.00, at 2 weeks control group score was 3.14 and 4 for both other groups which were significant than control one and P-value was 0.00, at 1 month the results were close for all groups with no significances.
Granulation tissue scores at 1 week were 1.71 for both control and second groups and 2 for the third group with no significance, at 2 weeks control group score was1.71 with 2.28 for the second group and 3 for the third group with significance in favour to third than the control group and P-value was 0.000, 1-month scores were 2.28 for control with 2.57 for second and 3 for the third group and there is significant P-value 0.015 in favour to the third group.
New blood vessels formation at 1-week scores were 1.42 for control, 2.14 for the second group and 3 for the third group and there were significant differences in favour to the third group with P value 0.000, at 2 weeks control group score was 2.71 while both other groups show the same score 3, at 1 month the score of control was 3, 1.85 for second and 1 for the third group with significant P value 0.000.
Both new bone formation and osteoblasts presences scores were the same, at 1-week score were 0.42 for the control group, 1.42 for second and 3 for the third group with significance in favour to the third group than control and second groups with P-value 0.000, 2 weeks scores were 1.42 for control, 2.14 for the second group and 3 for the third group with significant P value 0.000 in favour of the third group than the control group, 1-month show scores of 2.85 for the control group and 3 for both second and third groups.
The presence of osteoclast scores for the control group was are at 1 week 0.42, 2 weeks 1.42 and 3 at 1 month while for the second and third group were 0 during the period of the study and there were significant differences in favour to third and second groups to control group and P-value was 0.001 at 1 week and 0.000 at both 2 week and 1-month durations.
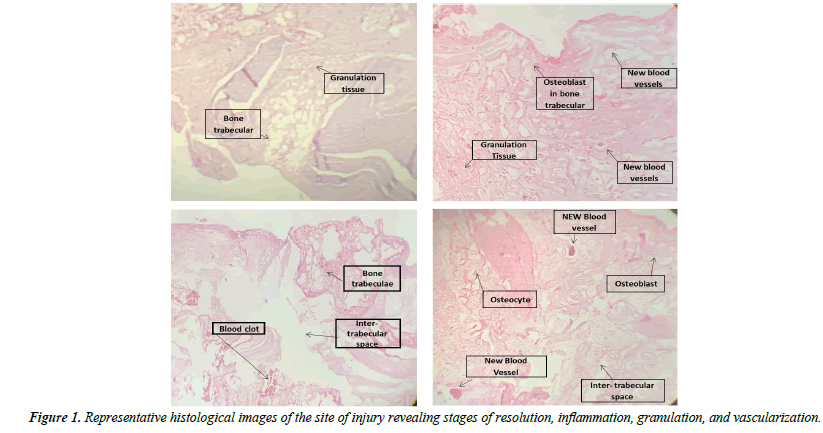
Differences in parameter means among the three groups are summarized in Tables 1-6 and represented in Figure 1.
| Period | Mean ±SD for a con. hydroxy Gp. | Mean ±SD for hydroxy +H.A. Gp. | Mean ±SD for Hydroxy +H.A.+R.M. Gp. | P. Value |
|---|---|---|---|---|
| 1 Week | 2.00±.000 | 3.71±.726 | 4.00±.000 | 0.001* |
| 2 Week | 3.14±.363 | 4.00±.000 | 4.00±.000 | 0.001* |
| 1 Month | 3.85±.363 | 4.00±.000 | 4.00±.000 | 0.129 |
Data presented as means with standard deviation SD, *p value ≤ 0.05 is significant.
Table 1. Means and comparison of inflammatory reaction among groups by Kruskal-Wallis test.
| Period | Mean ±SD for a con. hydroxy Gp. | Mean ±SD for hydroxy +H.A. Gp. | Mean ±SD for hydroxy +H.A.+R.M. Gp. | P. Value |
|---|---|---|---|---|
| 1 Week | 1.71±.913 | 1.71±.468 | 2.00±.000 | 0.212 |
| 2 Week | 1.71±.913 | 2.28±.468 | 3.00±.000 | 0.000* |
| 1 Month | 2.85±.363 | 2.57±.513 | 3.00±.000 | 0.015* |
Data presented as means with standard deviation SD, *p value ≤ 0.05 is significant.
Table 2. Means and comparison of Granulation tissue variable among groups by Kruskal-Wallis test.
| Period | Mean ±SD for a con. hydroxy Gp. | Mean ±SD for Hydroxy +H.A. Gp. |
Mean ±SD for hydroxy +H.A.+R.M. Gp. | P value |
|---|---|---|---|---|
| 1 Week | 1.42±.755 | 2.14±1.027 | 3.00±.000 | 0.001* |
| 2 Week | 2.71±.726 | 3.00±.000 | 3.00±.000 | 0.129 |
| 1 Month | 3.00±.000 | 1.85±1.027 | 1.00±.000 | 0.001* |
Data presented as means with standard deviation SD, *p value ≤ 0.05 is significant.
Table 3. Means and comparison of new blood vessel formation variable among groups by Kruskal-Wallis test.
| Period | Mean ±SD for a con. hydroxy Gp. | Mean ±SD for Hydroxy +H.A. Gp. |
Mean ±SD for hydroxy +H.A.+R.M. Gp. | P value |
|---|---|---|---|---|
| 1 Week | 0.42±.513 | 1.42±1.089 | 3.00±.000 | 0.001* |
| 2 Week | 1.42±.755 | 2.14±1.027 | 3.00±.000 | 0.001* |
| 1 Month | 2.85±.363 | 3.00±.000 | 3.00±.000 | 0.129 |
Data presented as means with standard deviation SD, *p value ≤ 0.05 is significant
Table 4. Means and comparison of new bone variable among groups by Kruskal-Wallis test.
| Period | Mean ±SD for a con. hydroxy Gp. | Mean ±SD for Hydroxy +H.A. Gp. |
Mean ±SD for hydroxy +H.A.+R.M. Gp. | P value |
|---|---|---|---|---|
| 1 Week | 0.42±.513 | 1.42±1.089 | 3.00±.000 | 0.001* |
| 2 Week | 1.42±.755 | 2.14±1.027 | 3.00±.000 | 0.001* |
| 1 Month | 2.85±.363 | 3.00±.000 | 3.00±.000 | 0.129 |
Data presented as means with standard deviation SD, *p value ≤ 0.05 is significant
Table 5. Means and comparison of osteoblast cells variable among groups by Kruskal-Wallis test.
Discussion
Quite a knowledge of the physiology of bone regeneration and repair is crucial for the innovation of original materials that can be used in the treatment of bone diseases, fractures, and other abnormalities. In the current study, we use a new mixture of many materials that have the ability to accelerate bone regeneration when applied separately and evaluate its bone rejuvenation efficacy when applied together as a new formulation mixture.
Our results of an inflammatory reaction at 1 week showed few inflammatory cells infiltration that disappears after 2 weeks and 1 month in the group treated by adding HA to hydroxyapatite which is significantly decreased than a control group, this due to the anti-inflammatory and anti-bacterial effect of the HA and this agreed with other studies which revealed that Hyaluronic acid has anti-inflammatory, anti-bacterial effects so has been used in the management of the inflammatory process in numerous fields like orthopaedics, dermatology, and ophthalmology [22]. Another study stated that hyaluronan acts as a scavenger drain prostaglandin, metalloproteinase, and other bio-active molecules and has an osmotic activity that is associated with the anti-edematous effect [23].
There is no inflammatory reaction observed in the group treated by both HA and Rosemary during the study period this may be related to an anti-inflammatory effect of both Rosemary as it has several acids which suppress inflammatory cytokines, in addition to HA this agreed with the previous study that revealed rosemary contains PPARγ agonists such as carnosic acid, rosemary acid, and carnosol, which are proposed to aid in glucose-lowering or anti-inflammatory effects of rosemary [24]. Another study found that rosemary inhibits cytokines release due to its Eucalyptol (1,8-cineole) component that aid in the anti-inflammatory and antioxidant effect of rosemary [25].
In the current study, granulation tissue formation for the control group was profound at 1 and 2 weeks that decreased to become moderate after 1 month, the second group shows a similar result to the control group with profound granulation tissue at 1 week that decreased to moderate after 2 weeks and still so to the end of the study duration. In the third group treated with rosemary granulation tissue at first week was moderate that decrease obviously became scant at 2 weeks and 1 month with significant differences than control and second groups. As there is no previous information or researches related to this part so we depend on our analysis, granulation tissue related to the inflammatory process as inflammatory cells infiltration include microphage which stimulates fibroblast differentiation and organize its activity to form granulation tissue so an increase in an inflammatory reaction in control group lead to profound granulation tissue formation, while decreased inflammatory process in the second group during study lead to decrease in granulation tissue formation. As there is no inflammation in groups treated with rosemary, so the granulation tissue was scanty.
The histological finding showed new blood vessels at the periphery of the specimens of the control group at 1 week that become more centrally at 2 weeks and diffused centrally and peripherally after 1 month, while the second group at 1 week showed highly neovascularization centrally that increased after 2 weeks to become centrally and peripherally without any significant differences relative to the first group, after 1month there is a regression of blood vessels to the periphery of the defect as the osteoblast differentiation increased. There is an increase in new blood vessels form in the specimens of (Hydroxy with H.A. and Rosemary) group that appear centrally and peripherally at 1 and 2 weeks then recede peripherally as new bone formation initiated, the results came highly significant than other two groups particularly at 1 week and after 1 month so it goes with a preceding study which compares the effect of 5% rosemary extract to povidone-iodine and normal saline in wound healing and found that new vessels formation is highly detected in rosemary and povidone-iodine solution treated groups on day 10 of study [26].
This study showed non-significant differences in new bone formation in the second group than the control group with osteoblast cells present peripherally then infiltrated centrally through 1 and 2 weeks and increased to occupy central and peripheral sides of all specimens after 1 month. This goes with a previous study that used hyaluronic acid as an aqueous binder of the BCP bone graft granules and found that new bone formation was non-significant in the HA group than BCP alone group and HA permitted more granules deposition [27]. In contrast, another study stated that the addition of HA to the autogenous bone in the extraction sockets increases bone trabecular formation and percentage of mean area filling than the group treated by autogenous bone alone [28]. Also, our study disagrees with two earlier studies, one used hyaluronic acid gel in a mandibular bone cavity in rabbits, while other compare group treated by BCP alone to a group treated by BCP and HA graft in immediate dental implant placement, these studies showed that new bone formation increased in the group treated by HA in comparison to other groups. In our study, we found a highly significant increase in osteoblast cell differentiation and new bone formation that diffuse both peripherally and centrally throughout the whole-slide of the group treated with (Hydroxy with H.A. and Rosemary) from the first week to the end of the study than control and second group this probably due to presence of erucic acid component in rosemary that has the ability to differentiate mesenchymal cells to osteoblast and this agreement with a prior study revealed that innovative erucic acid element of rosemary inhibit PPARγ transcriptional activity in rosemary and stimulate the differentiation of MSCs into osteoblasts rather than adipocytes by enhancing expression of osteoblast marker genes and suppression expression of adipocyte marker genes [29,30].
Our study revealed that there is no osteoclast formation in both the second and third groups with highly significant differences than the control group during the whole time of the study the obtained results agree with the findings of a preceding study which uses essential oils of many herbs including rosemary to evaluate its anti-osteoporotic effect and found that the monoterpenes and essential oil extracts act directly on bone cells by suppression of mevalonate path and the crenulation of small G-proteins such as Rac and Rho to prevent bone resorption.
Conclusion
Hyaluronic acid has an anti-inflammatory effect, adding it to hydroxyapatite increases bone formation slightly. The use of rosemary extract oil in this mixture (hydroxyapatite and hyaluronic acid) leads to decreased inflammation and accelerates the differentiation of osteoblast cells with new bone formation.
List of abbreviation
Con.=control, Gp.=group, H.A.=Hyaluronic acid, Hydroxy = Hydroxyapatite, R.M.= Rosemary
Funding
This research does not supply funding from any source.
Conflict of Interest
All authors declare no conflict of interest.
Acknowledgement
The authors acknowledge all the members of the College Of Dentistry of our institution for their unlimited supports.
Availability of Data and Materials
The data supporting the findings of the article is available when requested from authors.
References
- Isaksson S, Alberius P. Comparison of regenerative capacity elicited by demineralized bone matrix of different embryonic origins. J Cranio-Maxillo-Fac Surg. 1992;20(2):73-80.
- Bosch C, Melsen B, Vargervik K. Importance of the critical-size bone defect in testing bone-regenerating materials. J Craniofac Surg. 1998;9(4):310-6.
- Schortinghuis J, Ruben JL, Raghoebar GM, et al. Ultrasound to stimulate mandibular bone defect healing: a placebo-controlled single-blind study in rats. J Oral Maxillofac Surg. 2004;62(2):194-201.
- Segari WA, Radwan DA, Abd El Hamid MA. The effect of adding hyaluronic acid to calcium phosphate on periapical tissue healing following periradicular surgery in dogs. Tanta Dent J. 2014;11(2):122-9.
- Macha IJ, Ben-Nissan B. Marine skeletons: towards hard tissue repair and regeneration. Mar Drugs. 2018;16(7):225.
- Fadli A, Akbar F, Putri P, et al. Hydroxyapatite Powder Prepared by Low Temperature Hydrothermal Method from Sea Shells. Sci Eng. 2014;19.
- Brannigan k, Griffin M. An Update into the Application of Nanotechnology in Bone Healing, Review article. The Open Ortho J. 2016;10:808-823.
- Sullivan MP, McHale KJ, Parvizi J, et al. Nanotechnology: current concepts in orthopaedic surgery and future directions. Bone Jt J. 2014;96(5):569-73.
- Rink JS, Plebanek MP, Tripathy S, et al. Update on current and potential nanoparticle cancer therapies. Curr Opin Oncol. 2013;25(6):646.
- Inoue KI, Takano H, Shiga A, et al. Effects of volatile constituents of rosemary extract on lung inflammation induced by diesel exhaust particles. Basic Clin Pharmacol T. 2006;99(1):52-7.
- Al-Sereiti MR, Abu-Amer KM, Sena P. Pharmacology of rosemary (Rosmarinus officinalis Linn.) and its therapeutic potentials.
- Elbahnasawy AS, Valeeva ER, El-Sayed EM, et al. The impact of thyme and rosemary on prevention of osteoporosis in rats. J Nutr Metab. 2019.
- Mühlbauer RC, Lozano A, Palacio S, et al. Common herbs, essential oils, and monoterpenes potently modulate bone metabolism. Bone. 2003;32(4):372-80.
- Alkhalidi EF, Alsalman TH, Taqa AA. Mechanical properties of new calcium based cement prepared from egg Shell. Int J Enhanc Res sci Tec Eng. 2014;3:70-6.
- Alkhalidi EF, Alsalman TH, Taqa AA. Antibacterial properties of new calcium based cement prepared from egg shell. Edorium J Dentistry. 2015;2:21-8.
- DeCastro CL, Mitchell BS. Nanoparticles from mechanical attrition. Synthesis, functionalization, and surface treatment of nanoparticles. 2002;5.
- De Araujo JT, De Oliveira Pantoja F, Sá PS, et al. Effect of Essential Oil of Rosmarinus officinalis L.(Rosemary) on the Healing of Cutaneous Lesions in Mice. J Chem Pharm Res. 2017;9(5):381-6.
- Paknejad M, Rokn AR, Eslami B, et al. Evaluation of three bone substitute materials in the treatment of experimentally induced defects in rabbit calvaria. Frontiers in Dentistry. 2007;171-6.
- Kiliç N. A comparison between medetomidine-ketamine and xylazine-ketamine anaesthesia in rabbits. Turkish J Vet Anim Sci. 2004;28(5):921-6.
- Sultana J, Molla MR, Kamal M, et al. Histological differences in wound healing in maxillofacial region in patients with or without risk factors. Bangladesh J Pathol. 2009;24(1):3-8.
- Solchaga LA, Yoo JU, Lundberg M, et al. Hyaluronan?based polymers in the treatment of osteochondral defects. J Orthop Res. 2000;18(5):773-80.
- Dahiya P, Kamal R. Hyaluronic acid: a boon in periodontal therapy. Nor Amer J of Med Sci. 2013;5(5):309.
- Abdulhameed BS, Ibraheem L. Periodontal effect of 8% Hyaluronan as an Adjunct to Scaling and Root Planning in the Treatment of Chronic Periodontitis (Comparative Study). J Dent Med Sci. 2014;13:76-81.
- Takahashi A, Dohi H, Egashira Y, et al. Erucic acid derived from rosemary regulates differentiation of mesenchymal stem cells into osteoblasts/adipocytes via suppression of peroxisome proliferator?activated receptor γ transcriptional activity. Phytother Res. 2020;34(6):1358-66.
- Juergens LJ, Racké K, Tuleta I, et al. Anti-inflammatory effects of 1, 8-cineole (eucalyptol) improve glucocorticoid effects in vitro: a novel approach of steroid-sparing add-on therapy for COPD and asthma?. Synergy. 2017;5:1-8.
- Yilmaz R, Ozyildiz Z, Temmaogullari F, Hayat A. The Effects of Rosemary (Rosemarinus officinalis) Extract on Wound Healing in Rabbits. FÜS a? Bil Vet Derg. 2012;26(2):105-9.
- Aguado E, Pascaretti-Grizon F, Gaudin-Audrain C, et al. β-TCP granules mixed with reticulated hyaluronic acid induce an increase in bone apposition. Biomed Mat. 2013;9(1):015001.
- Taman RA, Fahmy MH, Karam SS, et al. Post-Extraction Socket Preservation With Autogenous Bone Graft And Hyaluronic Acid Followed By Delayed Implant Placement. Alexandria Dental Journal. 2017 1;42(2):170-6.
- Radhi IH, Al-Ghaban NM. Evaluation the effect of hyaluronic acid on bone healing process in rabbits (Immunohistochemical study for TGF-β). J Baghdad college Dentistry. 2015;325(2218):1-3.
- Shamma MM, Ayad SS, El-dibany RM, et al. Evaluation of the effect of hyaluronic acid mixed with biphasic calcium phosphate on bone healing around dental implants (experimental study). Alexandria Dental J. 2017;42(1):104-7.
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref