Research Article - Biomedical Research (2017) Volume 28, Issue 19
A study on the antibacterial effect of Acanthopanax sessiliflorum on inflammatory diseases in the oral cavity
Young-Soon Choi1, Na-Hyun Kim2, Hyo-Jin Kim2, Su-Jin An2, Bit-Na Lee2, Min-Ju Jung2, Ji-Young Hwang2 and Seoul-Hee Nam2*
1Department of Nursing, College of Health Science, Kangwon National University, Samcheok-si, 25949, Republic of Korea
2Department of Dental Hygiene, College of Health Science, Kangwon National University, Samcheok-si, 25949, Republic of Korea
- *Corresponding Author:
- Seoul-Hee Nam
Department of Dental Hygiene
College of Health Science
Kangwon National University
Hwangjo-gil, Dogye-eup
Samcheok-si, Gangwon-do, 25949
Republic of Korea
Accepted date: September 23, 2017
Abstract
This study was conducted to investigate the antimicrobial effect of Acanthopanax sessiliflorum extract, a natural medicinal substance against Candida albicans (C. albicans), a common inflammatory disease in the oral cavity, and to utilize it in the prevention and treatment of inflammatory oral diseases. In studying the antimicrobial effect of 80% ethanol Acanthopanax sessiliflorum extract against C. albicans, disc diffusion method (paper disk) was used, while growth curve, minimum inhibitory concentration and minimum bactericidal concentration, and colony-forming unit were measured. As a parameter of the antimicrobial effect of Acanthopanax sessiliflorum extract, the clear zone of C. albicans was found to be 15 mm at 40 mg/ml, 14 mm at 45 mg/ml, 19 mm at 50 mg/ml, 20 mm at 55 mg/ml and 21 mm at 60 mg/ml, respectively. The minimal inhibitory concentration (MIC) of the extract against C. albicans was 20 mg/ml, and minimum bactericidal concentration (MBC), which is the concentration where bacteria do not exist, was found to be 50 mg/ml. As for the colony-forming unit (CFU), there was bactericidal effect at 20 mg/ml, while no bacteria were observed at 50 mg/ml. Thus, the antimicrobial effect against C. albicans was enhanced as the concentration of Acanthopanax sessiliflorum extract increased. Based on these results, Acanthopanax sessiliflorum extract can be considered as a suitable replacement for antimicrobial agents. Therefore, developing oral health care products, such as gargles or toothpastes using natural Acanthopanax sessiliflorum extract, would be beneficial in preventing inflammatory oral disease.
Keywords
Antimicrobial effect, Acanthopanax sessiliflorum, Candida albicans, Oral candidiasis, Oral diseases
Introduction
Oral disease, a disease that occurs in the mouth, including the teeth or gums, is not limited to the teeth or gums, but is closely related to human health in general [1]. Oral candidiasis, commonly called as thrush, is an infectious disease caused by fungi and is an infectious disease that frequently affects people these days [2]. Oral candidiasis, an oral mucosal disease that occurs in the tongue, cheek, mucous membrane, or gums in the mouth, is an opportunistic infection caused mostly by Candida albicans (C. albicans), residing on wet skin and mucous membranes, and migrates through skin contact [3]. The incidence of candidiasis was 42.9% among smoking, natural teeth males and 14.3% among those who do not smoke and regardless of gender; the infection rate was 50% among women and 36% among men. Also, in terms of dental condition, the infection rate was 70% among people who had dentures, and 39% among those who had natural teeth [4]. This indicates that the incidence of candidiasis is due to various circumstances. In general, it does not easily occur among healthy people, but it can easily be transmitted when the host's defensive function or immune system is impaired, or when they have poor oral hygiene [5,6]. Currently, although antibiotics, steroids, or oral disinfectants are mostly used to treat oral candidiasis, the use of such antibiotics has become a major social issue in modern society because of the various problems they pose and their side effects. Their long-term use can break down the balance of normal flora and cause teeth discoloration [7,8]. Many studies have been conducted to address these drawbacks, and most of these studies are focused on finding new antibiotic substances in natural products [9-11]. The interest and demand for natural extracts have increased in recent years, and the development and application of natural substances for various functions are becoming more significant [12]. Acanthopanax sessiliflorum refers to the roots, stems, and branches of Eleutherococcus sessiliflorus, or its congener plants that are members of the Araliaceae family [13]. Since the discovery of lignan (eleutheroside E) and acanthoside D, the main components of the Acanthopanax sessiliflorum extract, sesamine, phenolic glycoside, syringgaresinol diglucoside, B-sitosterol, isofraxidin, friedelin, syringin, and pimaric acid, which can also be found in the extract, have been studied in various research [14]. It has been reported that Acanthopanax sessiliflorum has been associated with immune enhancement, hypoglycemic, antiviral, anti-cancer, anti-aging, stress resistance, and endurance enhancement effects [15]. Although Acanthopanax sessiliflorum extract has been used in various ways, studies of its application on the oral cavity, especially regarding oral candidiasis, have been insufficient.
Therefore, this study was conducted to examine the antimicrobial activity of Acanthopanax sessiliflorum extract against C. albicans, the bacteria that cause inflammatory oral disease, in order to find biocompatible antibiotic substances that can prevent and treat oral diseases.
Materials and Methods
Sample extraction
For this experiment, 100 g of dried Acanthopanax sessiliflorum grown in Gyeongsan, Gyeongsangbuk, South Korea, was purchased from Sanaeherb Pharm Co., Ltd. After adding 80% methanol 10 times to 100 g of crushed Acanthopanax sessiliflorum, extraction was done in a heated mantle at 65°Ϲ for 12 h. The extract was filtered by using filter paper (Advantec No. 2, Toyo, Japan), and the Acanthopanax sessiliflorum extract was concentrated and lyophilized by using a rotary vacuum evaporator (N-1300E.V.S. EYELA Co., Japan).
Experimental strains and culture
The strain of the C. albicans (KCTC 7965/ATCC 10231) used in this experiment were grown in yeast mold broth (YM, Difco, USA) and cultivated overnight in liquid media that were incubated at 37°Ϲ.
Paper disk method
Antimicrobial activity was investigated by using the paper disk agar method [16]. The Acanthopanax sessiliflorum extract was completely dissolved in YM broth, and a sterilized filter paper disc (Advantec Toyo Kaisha, Ltd.) that was 8 mm in diameter was dissolved with Acanthopanax sessiliflorum extract (0 mg/ml, 20 mg/ml, 25 mg/ml, 30 mg/ml, 35 mg/ml, 40 mg/ml, 45 mg/ml, 50 mg/ml, 55 mg/ml, and 60 mg/ml).
Minimal inhibitory concentration and Minimum bactericidal concentration of the Acanthopanax sessiliflorum extract
To measure Minimal Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC), each sample was inoculated into a YM broth (10 ml) and then, 100 μl of cultured strain (2246 × 107 CFU/ml) was inoculated into a 1 ml vial containing the Acanthopanax sessiliflorum extract at each concentration (0, 20, 25, 30, 35, 40, 45, 50, 55, and 60 mg/ml). This vial was incubated at 37°Ϲ for 24 h, and then absorbance (OD) was measured at an absorption wavelength of 660 nm with an UV spectrophotometer (SHIMADZU UV-1280, Shimadzu Co., Kyoto, Japan) to evaluate the antimicrobial effect of the Acanthopanax sessiliflorum extract.
Measurement of the Colony-forming unit (CFU)
To measure the CFU of the Acanthopanax sessiliflorum extract, 1 ml of Acanthopanax sessiliflorum extract at each concentration (0, 20, 25, 30, 35, 40, 45, 50, 55, and 60 mg/ml) was uniformly smeared in a YM agar medium, and cultured at 37°C for 24 h.
Results
Measurement of antimicrobial activity via the paper disk method
The antibacterial effects of Acanthopanax sessiliflorum extract on C. albicans were examined by measuring the clear zone via the paper disk method, and were presented in Figure 1. The clear zone of C. albicans was found to be 15 mm at 40 mg/ml, 14 mm at 45 mg/ml, 19 mm at 50 mg/ml, 20 mm at 55 mg/ml and 21 mm at 60 mg/ml, respectively. Therefore, the antibacterial effects of Acanthopanax sessiliflorum extract against C. albicans that were shown through the diameter of the clear cones were enhanced as its concentration was increased (Figure 1).
MIC/MBC of Acanthopanax sessiliforum extract
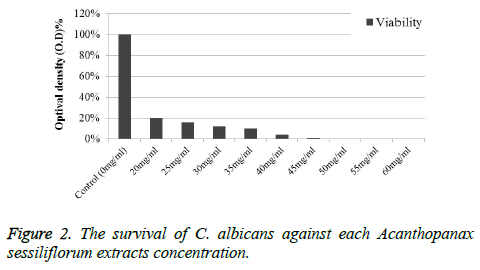
The antibacterial effects of Acanthopanax sessiliflorum extract on C. albicans were measured with the MIC and MBC, and were presented in Figure 2. The MIC of the extract against C. albicans was 20 mg/ml. The MBC, which is the concentration where bacteria do not exist, is 50 mg/ml.
The number of counted CFU
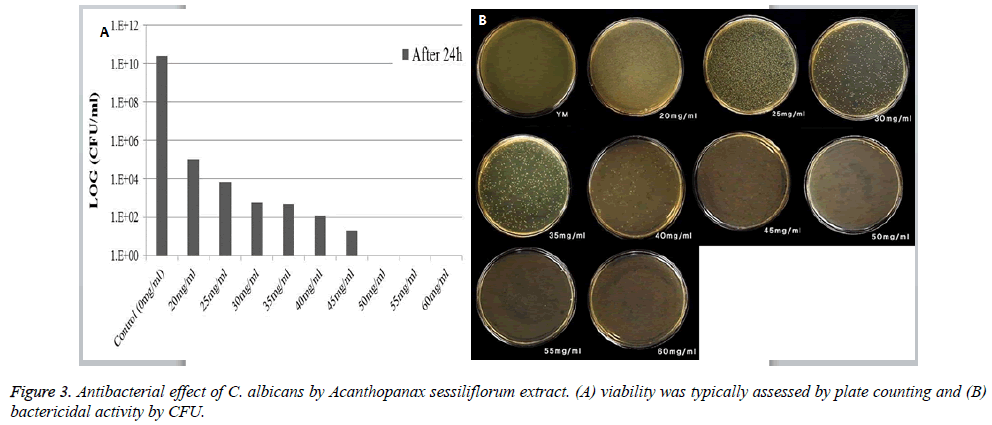
The bacterial survival rate of C. albicans slightly decreased at 20 mg/ml, and bacteria were not observed starting from 50 mg/ml (Figures 3A and 3B). As the concentration of Acanthopanax sessiliflorum extract was increased, a decrease in the number of bacteria was observed.
Discussion
Oral candidiasis, a common oral disease, is a common fungal infection in the mouth, and is commonly caused by the Candida species. This type of species resides in the human body without any symptoms or signs in about 60% of healthy adults and about 45-65% of healthy children. C. albicans, the most common type of this species, proliferates in the oral cavity and causes oral lesions in pathological conditions [17,18]. Candida species are susceptible to local and systemic antifungal agents, but antifungal agents can cause various side effects, such as nausea, vomiting, gastrointestinal effects, skin irritation, burning sensation, visual disturbances, fever, rash, and headache [19,20].
Health is gradually becoming a main subject of interest as a result of economic growth, increase in national income, and the change in values in the modern society, and substances that can be used in lieu of chemical agents that contain risks of side effects are being widely studied [21]. Therefore, to develop antimicrobial substances that are effective in preventing and treating oral candidiasis and cause fewer side effects and resistance, interest in natural extracts has been growing in many countries, and studies that confirm the antimicrobial effect of natural plant sources have been actively conducted. Acanthopanax sessiliflorum has been found in Korea, Japan, and China, and its therapeutic effects on diabetes, cancer, rhematoid arthritis, and osteoarthritis have been reported [22]. This organism has components that are known to have antiinflammatory, anti-hepatoxicity, anti-diabetic, and antiviral properties [23] and, therefore, this study was conducted to prove its antibacterial effect.
According to Jang Seon-young's research, antimicrobial activity against Porphyromonas gingivalis (P. gingivalis), which is a typical periodontal bacteria, has not been found in studies on the antimicrobial effect of Acanthopanax sessiliflorum and Angelica dahurica root [24]. However, it was confirmed that Acanthopanax sessiliflorum extract had excellent antimicrobial effect against C. albicans, a causative agent of oral candidiasis.
Moreover, according to a study by Kim et al. [25], the MIC value of fluconazole, an antifungal agent, for C. albicans was 32 mg/ml. However, in this experiment, the MIC of Acanthopanax sessiliflorum extract for C. albicans was 20 mg/ml, and it was confirmed that its antibacterial effect was better at a lower concentration than fluconazole. Furthermore, according to a study done by Kim et al. [26], C. albicans showed a clear zone of 3 mm at a concentration of 200 μg/ml of Rhizome extract. Compared to Acanthopanax sessiliflorum extract that was used in this study, Rhizome extract showed an antimicrobial effect at 200 mg/ml for C. albicans, whereas Acanthopanax sessiliflorum extract resulted in a wider clear zone of 15 mm at a much lower concentration, which is 40 mg/ml. Thus, it can be concluded that Acanthopanax sessiliflorum extract has excellent antimicrobial activity at a lower concentration compared to that of Rhizome extract.
As demonstrated by the results, Acanthopanax sessiliflorum extract has shown a distinctive antimicrobial effect at a lower concentration than other natural extracts as the MIC was at 20 mg/ml, and it can be seen that its antimicrobial effect against C. albicans is enhanced with its concentration, as its MBC, the concentration in which the bacteria do not exist, is 50 mg/ml. Thus, Acanthopanax sessiliflorum extract is considered as a suitable substitute for chemical-based antibiotics. Conclusively, Acanthopanax sessiliflorum extract was proven to possess antibacterial functions against C. albicans, which causes an oral inflammatory disease called oral candidiasis, and, therefore, developing gargles or toothpastes using natural Acanthopanax sessiliflorum extract would be beneficial in preventing and treating inflammatory oral disease.
Based on these results, it can be seen that Acanthopanax sessiliflorum extract has a distinctive antimicrobial effect at a lower concentration than other natural extracts, and the MIC of 20 mg/ml is shown. The concentration of MBC in the absence of bacteria is 50 mg/ml. Thus, its antimicrobial effect against C. albicans increased. This shows that Acanthopanax sessiliflorum extract is suitable as a replacement for antibiotics.
Therefore, it is considered that Acanthopanax sessiliflorum extract is effective against C. albicans, which is a causative agent of oral disease that can lead to an infectious disease in the oral cavity, and that it is possible to prevent and treat inflammatory diseases by developing a salicylate or an oral liquid with Acanthopanax sessiliflorum extract as a key natural ingredient.
Conclusion
Acanthopanax sessiliflorum extract was studied to demonstrate its antimicrobial effect against C. albicans, a common type of inflammatory bacteria, and the results showed that antimicrobial effects began at a low concentration of 20 mg/ml (MIC), and a definite antimicrobial effect was examined at 50 mg/ml (MBC). Therefore, Acanthopanax sessiliflorum extract performs an antimicrobial function at a low concentration, and is considered suitable as a natural substitute for conventional antibiotics.
Conflict of Interest
The authors report no conflicts of interest related to this study. Likewise, the authors do not have any financial interest in the companies whose materials were included in this study.
References
- Kim CS, Choi YK. Survey of adult’s perceptions of the association between chronic diseases and oral health. J Korean Dent Hyg Sci 2017; 17: 12-19.
- Mizugai H, Isogai E, Hirose K, Chiba I. Effect of denture wearing on occurrence of candida species in the oral cavity. J Appl Res 2007; 7: 250-254.
- Ashman RB, Farah CS, Wanasaengsakul S, Hu Y, Pang G, Clancy RL. Innate versus adaptive immunity in Candida albicans infection. Immunol Cell Biol 2004; 82: 196-204.
- Lee CG, Kim CH. A study on the prevenalence and intra-oral distribution of candida albicans. J Korean Acad Prosthodont 1986; 24: 91-103.
- Ellepola AN, Samaranayake LP. Oral candidal infections and antimycotics. Crit Rev Oral Biol Med 2000; 11: 172-198.
- McCullough MJ, Savage NW. Oral candidosis and the therapeutic use of antifungal agents in dentistry. Aust Dent J 2005; 50: S36-39.
- Loe H, Schiott CR, Karring G, Karring T. Two years oral use of chlorhexidine in man. I. General design and clinical effects. J Periodontal Res 1976; 11: 135-144.
- Ciancio SG, Mather ML, Bunnell HL. Clinical evaluation of a quaternary ammonium-containing mouthrinse. J Periodontol 1975; 46: 397-401.
- Ooshima T, Minami T, Matsumoto M, Fujiwara T, Sobue S, Hamada S. Comparison of the cariostatic effects between regimens to administer oolong tea polyphenols in SPF rats. Caries Res 1998; 32: 75-80.
- Tsuchiya H. Effects of green tea catechins on membrane fluidity. Pharmacology 1999; 59: 34-44.
- Yanagida A, Kanda T, Tanabe M, Matsudaira F, Oliveira Cordeiro JG. Inhibitory effects of apple polyphenols and related compounds on cariogenic factors of mutans streptococci. J Agric Food Chem 2000; 48: 5666-5671.
- Hwang HS, Park GD. A comparative study of anti-oxidant effect and cell proliferation effect based on extraction method of natural substances. J Kor Soc Cosm 2015; 21: 729-736.
- Kim GY, Kim KY, Jeong HW. Immunostimulating effects of Acanthopanax in mice following gamma-ray irradiation. Korean J Orient Physiol Pathol 2006; 20: 670-674.
- Kim YH, Bae DB, Park SO, Lee SJ, Joe OH, Lee OH. Method validation for the determination of eleutherosides and ß-glucan in Acanthopanax koreanum. J Korean Soc Food Sci Nutr 2013; 42: 1419-1425.
- Ham SH, Lim BL, Yu JH, Ka SO, Park BH. Fermentation increases antidiabetic effects of Acanthopanax senticosus. J Kor Orient Physiol Pathol 2008; 22: 340-345.
- Kim MS, Lee DC, Hong JE, Chang IS, Cho HY, Kwon YK, Kim HY. Antimicrobial effects of ethanol extracts from Korean and Indonesian plants. Korean J Food Sci Technol 2000; 32: 949-958
- Darwazeh AM, Hammad MM, Al-Jamaei AA. The relationship between oral hygiene and oral colonization with Candida species in health adult subjects. Int J Dent Hyg 2010; 8: 128-133.
- Navazesh M, Wood GJ, Brightman VJ. Relationship between salivary flow rates and Candida albicans counts. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1995; 80: 284-288.
- Taillandier J, Esnault Y, Alemanni M. A comparison of fluconazole oral suspension and amphotericin B oral suspension in older patients with oropharyngeal candidosis. Multicentre Study Group. Age Ageing 2000; 29: 117-123.
- Gothard P, Rogers TR. Voriconazole for serious fungal infections. Int J Clin Pract 2004; 58: 74-80.
- Kim HK. Na GM, Ye SH, Han HS. Extraction characteristics and antioxidative activity of Schiznadra chinensis extracts. J Korean Soc Food Cult 2004; 19: 484-490.
- Jung BS, Shin MK. Hyang Yak Dae Sajeon. 3rd ed. Young Lim Sa Publisher 2003: 432-435.
- Jung HJ, Nam JH, Choi J, Lee KT, Park HJ. Anti-inflammatory effects of chiisanoside and chiisanogenin obtained from the leaves of Acanthopanax chiisanensis in the carrageenan and Freund's complete adjuvant-induced rats. J Ethnopharmacol 2005; 97: 359-367.
- Jang SY, Park JW, Yoon SW, Ryu BH, Kim JS. Study on deodorizing effects of mume fructus, eriobotryae folium, acanthopanacis cortex and angelicae dahuricae radix for the development of a gargle solution. J Korean Oriental Med 2010; 31: 115-128.
- Kim YH, Lim JH, Kim DH, Kim SH, Kim HR, Kim YM. Synergistic antimicrobial effect of Sargassum serratifolium (C.Agardh) C.Agardh extract against human skin pathogens. Korean J Food Sci Technol 2016; 48: 241-246.
- Kim JY, Yi YS, Lim YH. Biological and antifungal activity of herbal plant extracts against candida species. Korean J Microbiol Biotechnol 2009: 37: 42-48.