Research Article - Current Pediatric Research (2017) Volume 21, Issue 3
Urinary interleukin-4 levels in nephrotic children with and without concomitant asthma
Amal A Al-Eisa, Maysoun Al-Rushood
Associate Professor, Department of Pediatrics, Faculty of Medicine, Kuwait University, Kuwait.
- *Corresponding Author:
- Amal A Al-Eisa
Associate Professor, Department of Pediatrics
Faculty of Medicine, Kuwait University P.O. Box 23924
Safat 13110, Kuwait City, Kuwait.
Tel: 965-25319486
Fax: 965-25338940
E-mail: amal@hsc.edu.kw
Accepted date: May 16, 2017
Abstract
Background and Aim: The role of cytokines in the immune-pathogenesis of idiopathic nephrotic Syndrome (INS) had been widely postulated. Some cytokines, most importantly IL-4, have been also involved in the pathogenesis of asthma. The majority of INS patients do exhibit a form of atopy including asthma, implying a common pathological background of both diseases. The aim of this study is to explore the role of IL-4 in the pathogenesis of a relapses in INS patients with Asthma compared to those without concomitant asthma. Patients and Methods: Demographic, biochemical data and mean urinary IL-4 levels were evaluated in 41 steroid-sensitive INS patients and 30 matched Asthmatic controls without INS or atopy. Mean urinary IL-4 levels in INS patients were compared to controls as well as in the 2 subgroups of INS patients: Group A (INS with Asthma, n=14) and Group B (INS patients without Asthma or atopy, n=27) to controls during relapse and remission. Results: Mean age of patients at study was 8.9 ± 3.5years (range: 49 months-15 years). Male: Female ratio was 28:13. Mean serum creatinine was 49 ± 10 μmol/l and mean serum albumin was 29 ± 10 g/L. Mean urinary IL-4/Cr in INS patients during relapse and remission was 12.170 ± 20.013 pg/μmol and 15.269 ± 41.275 pg/μmol, respectively (P=0.716). Both levels were not statistically different when compared to controls (24.78 ± 89.51 pg/μmol) (P=0.5431 and P=0.662, respectively). Mean urinary IL-4/Cr level was significantly higher in Group A vs. Group B during relapse (28.22 ± 26.12 pg/μmol vs. 2.73= ± 3.38 pg/μmol, P=0.003) and during remission (31.52 ± 57.13 pg/μmol vs. 1.04 ± 1.48 pg/μmol, P=0.004). Group A patients had a higher mean urinary protein excretion and a longer duration to remission than group B patients. Conclusion: INS patients with asthma have a higher IL-4 excretion which might increase the severity and duration of proteinuria. The coexistence of Asthma in INS patients requires an early and abrupt treatment of relapses.
Keywords
Idiopathic nephrotic syndrome (INS), Bronchial asthma, Interleukin 4 (IL-4), Relapse, Remission.
Introduction
Idiopathic nephrotic syndrome (INS) is a common glomerular disease in children. Many studies have suggested that it is a primary immune disease associated with immune-regulatory imbalance between T helper subtype 1 (Th1) and T helper subtype 2 (Th2) cytokines [1]. A decrease in T-regulatory cells and/or their cytokines were proposed as major players [2,3]. The incidence of asthma in INS patients varied from 34% up to 63% reported by different studies [4-6]. Many previous studies showed increased incidence of atopy and family history of atopy in patients with nephrotic syndrome [4,5,7-11].
Atopic nephrotic syndrome patients, especially frequent relapsers, were found to have increased levels of IgE and Interleukin-4 (IL-4), the levels of which were correlated with the degree of proteinuria [5]. Moreover, serum IgE level was found to be typically elevated in minimal change nephrotic syndrome (MCN), both in acute phase and during remission [12].
IL-4, which is produced by Th2-cells, is the key cytokine involved in the development of atopy and is absolutely required for class switching of B-cells to IgE production and also promoting eosinophil chemotaxis and adherence [13]. Many studies have reported conflicting results regarding the serum levels of IL-4 in INS patients and no previous studies correlated urinary IL-4 levels in INS during relapse and remission. Moreover, data on the effect of coexistence of Asthma with INS on the clinical pattern and course of the disease are lacking in the literature.
In this study we investigate the levels of urinary excretion of IL-4 in INS patients with and without Asthma exploring its role and possible effect on relapses of INS patients with and without asthma.
Patients and Methods
Children aged 1-12 years who were diagnosed with steroid-sensitive idiopathic nephrotic syndrome during the period January 2012 to December 2015 were recruited in the study. All were managed in the outpatient clinics of the department of Pediatrics at Mubarak university teaching hospital. All patients were either newly-diagnosed at their first presentation or known to have INS but were off steroids treatment for more than 2 months prior to inclusion. All patients were treated for their relapse with the standard daily dose of prednisolone (60 mg/M2/day). Patients (newly-diagnosed or the known INS) who did not show response to steroid within 4 weeks were excluded from the study. Previously diagnosed-INS patients who had a non-minimal change histopathology or had received second or third line immunosuppressive treatment within 6 months prior to the study were also excluded. Fourteen of included INS patients had a confirmed diagnosis of atopic bronchial asthma for more than 6 months with no other atopic symptoms. All had a well- controlled asthma for more than 2 months prior to inclusion and were not on long-term prophylactic medications or on steroid inhalers. The other 27 INS patients had no atopic history or asthma. Asthma was defined, as per American Academy of Allergy Asthma and Immunology (AAAI) as a chronic inflammatory condition of the airways characterized by 3 features including airway obstruction that is at least partially reversible by bronchodilators, airway hypersensitivity or hyper-responsiveness to a variety of external stimuli and chronic inflammation of the airways [14]. Atopy was defined as the genetic tendency to develop allergic diseases such as allergic rhinitis, asthma and atopic dermatitis which is associated with heightened immune responses to common allergens such as inhaled or food allergens [14]. Diagnosis of asthma and atopy were confirmed by expert allergists who examined all Asthmatic patients as well as controls. Diagnosis of INS was based on the presence of edema, nephrotic-range proteinuria and hypoalbuminemia [15,16]. Nephrotic-range proteinuria was defined as the presence of ≥ 40 mg/M2 body surface area/h or a Protein: Creatinine ratio on early morning urine of ≥ 2.0. Hypoalbuminemia was defined as a serum albumin level <25 g/L. Relapse of nephrotic syndrome was defined as the appearance of nephrotic range proteinuria or urine albumin dipstick of ≥ 2+ on 3 successive days. Remission was defined as disappearance or trace of proteinuria on albumin dipsticks for 3 successive days.
Thirty Asthmatic age and sex-matched children of the same ethnic background were included as controls and were evaluated by an experienced allergist to confirm the diagnosis of Asthma, the absence of other atopy or proteinuria. All controls had a well-controlled asthma with no exacerbations for more than 2 months prior to inclusion and were not on long-term prophylactic medications nor on steroid inhalers. All controls had a serum creatinine (within15-80 μmol/l) and normal serum albumin level (Range: 30-40 g/l).
Urinary samples for IL-4 were collected from INS patients (both group A and group B) at presentation with a relapse before the initiation of prednisolone therapy. A second sample was collected at day 7 of attaining a remission. All controls had their urinary IL-4 level at their visit to the clinic and after confirming their fulfillment of the inclusion criteria as controls.
Cytokines Assay
Freshly voided urine samples were collected and centrifuged at 1500=m at 40°C for 20 min. The supernatant was then collected, aliquoted and immediately frozen at -80°C until assessment. ELISA assay for IL-4 was carried out using commercial kits purchased from Biosource International (San Diego-California-USA). The minimum detection range of IL-4 cytokine was 20.8 pg/ml. Immunoassays for the quantitation of cytokine by Flowcytomix kits (Bender Medsystems GmbH, Vienna, Austria), according to manufacturer’s instructions.
Statistical Analysis
Statistical analysis and graphical presentation was performed using SPSS 22 on P.C. Mann-Whitney nonparametric test was used to compare the significant difference in the values between the groups. X2 or Fisher’s exact test was applied to evaluate differences in proportions. All p-values are two-sided and p<0.05 indicates the statistical significance.
Results
A total of 41 patients with INS were included. The mean age of patients at study was 8.9 ± 3.5 years (range: 49 months-15 years). They were 28 male and 13 female. A total of 14 INS patients had definite history of Asthma as per AAAI criteria (group A) while 27 had no Asthma or atopy (group B). Newly- diagnosed INS patients were 21 whereas the other 20 patients were frequent relapsers who had a new relapse while off any immunosuppressive drugs as per inclusion criteria. A total of 11 patients received cyclophosphamide and 8 had cyclosporine during the course of their disease but were off these drugs for more than 6 months prior to recruitment. All INS patients had normal serum creatinine with a mean of 49 ± 10 μmol/l and Mean serum albumin level during relapse was 20 ± 4 g/l. Table 1 summarizes the clinical and biochemical data of patients and controls.
| INS Patients (n=41) | Controls (n=30) | Normal Range | ||
|---|---|---|---|---|
| Mean Age (years) | 8.9 + 3.5 | 7.1 + 2.3 | ||
| Gender | Male | 28 | 19 | |
| Female | 13 | 11 | ||
| Mean Serum Creatinine (µmol/L) | 49 + 10 | 54 + 9 | (15-88) | |
| Mean Serum Protein (g/L) | 50 + 5 | 75 + 3 | (68-80) | |
| Mean Serum Albumin (g/L) | 20 + 4 | 35 + 3 | (30-40) | |
| Mean Serum Cholesterol (mmol/L) | 6.3 + 0.7 | 3.1 + 1.5 | (3.1-5.2) | |
| Mean UP: Cr ratio* (mg/mg) | 2.7 ± 0.6 | 0.02 ± 0.04 | (= 0.2) | |
*UP: Urine Protein; Cr: Creatinine.
Table 1. Clinical and laboratory data of patients with idiopathic nephrotic syndrome (INS) and controls at time of inclusion
Cytokines Measurement
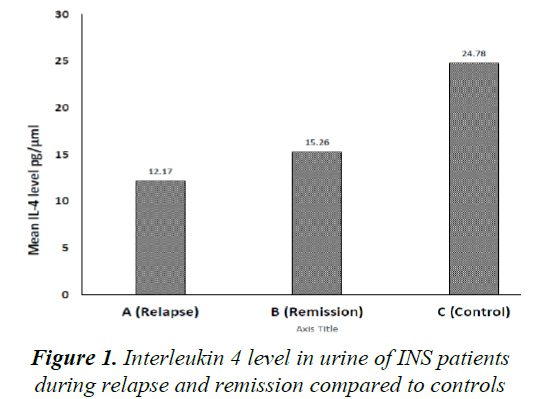
Mean urinary IL-4, corrected to urinary creatinine, in INS patients during relapse and remission was 12.170 ± 20.013 pg/μmol and 15.269 ± 41.275 pg/μmol, respectively (P=0.716). Both levels were not statistically different when compared to controls (24.78 ± 89.51 pg/μmol) (P=0.54 and P=0.66, respectively) (Figure 1).
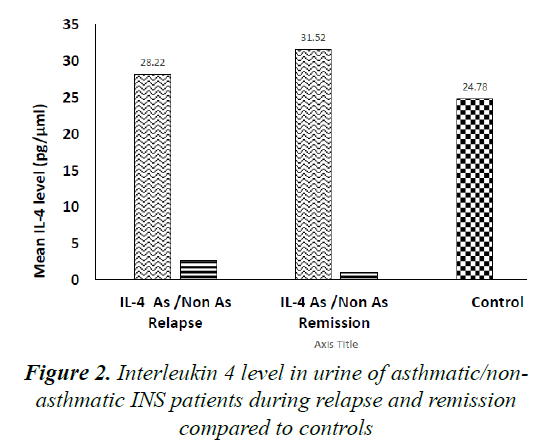
Comparing mean urinary IL-4 levels in Asthmatic (Group A) to Non-Asthmatic INS patients (Group B), levels were significantly higher in group A compared to group B during INS relapse (28.22 ± 26.12 pg/μmol vs. 2.73 ± 3.38 pg/μmol, P=0.03) as well as during remission (31.52 ± 57.13 pg/μmol vs. 1.04 ± 1.48 pg/μmol, P=0.04).
No significant difference was found comparing mean IL-4 level in group A to controls both during relapse as well as during remission (P=0.85 and P=0.76, respectively). Moreover, when comparing mean urinary IL-4 level in Group B to controls, levels were significantly lower in the former group during relapse both during relapse and remission (P=0.05 and P=0.04, respectively). Figure 2 illustrates these results.
Mean urinary protein: creatinine ratio was significantly higher during relapse in Group A (3.3 ± 0.2 mg/mg) compared to Group B (2.4 ± 0.3 mg/mg) (P<0.05). Mean duration from starting prednisolone treatment for relapse to remission was significantly longer in Group A patients (18 ± 4 days) compared to Group B patients (7 ± 3 days) (P<0.05).
Discussion
The association between idiopathic nephrotic syndrome and allergic phenomenon was reported decades ago as INS was triggered by allergic reactions to stings, poison ivy and pollen and food hypersensitivity [4,17-20]. Asthma and allergic dermatitis were found in about 34 to 63% of nephrotic syndrome patients [4,6,7]. Moreover, INS incidence was found to occur 3.3 folds greater in asthma cohort than non-Asthma cohorts [21].
IL-4 production by mast cells and Th2 is important in the pathophysiology of allergic diseases including asthma. In Studies on the role of IL-4 in nephrotic syndrome patients are conflicting. Levels of Th2-associated cytokines such as interleukin-4 and IL-13 were found to be elevated in the serum of relapsing nephrotic patients in some reports [7,23,24]. Other studies showed high serum levels of IL-4 in INS patients during relapse as well as during remission compared with controls [25-27]. Evidence of enhanced activity of IL-4 in MCN with increased serum levels, increased production by peripheral blood mononuclear cells in vitro and enhanced expression of IgE receptors were also reported in other studies [28,29]. On the contrary, Kimata et al. studied unstimulated production of cytokines by T-lymphocyte of MCNS patients and found a normal production of IL-4 in serum of such patients [23].
No previous reports studied levels of IL-4 excretion in urine of INS, Asthmatic or INS patients with concomitant Asthma. As our aim in this study was to explore the effect of IL-4 in relapse of INS with and without coexistent Asthma, we elected to measure urinary and not serum IL-4. Locally acting IL-4 in the kidneys would be more reflected by urinary IL-4 levels and its effect in situ on the glomerular capillary bed, and consequently on the protein leakage, is more related to its levels excreted in urine.
We found a significantly higher urinary excretion of IL-4 in INS patients with asthma during relapse and remission of INS compared to non-asthmatic INS patients. This finding supports the assumption of the role of IL-4 in acting in-situ on glomerular vasculature aggravating proteinuria in INS asthmatics during relapse. It is well known that nephrotic proteinuria is caused by various immunologic and non-immunologic factors leading to slitpore leakage of proteins. Nevertheless, our results support a possible role of IL-4 in exaggerating this leakage effect. Moreover, the prolonged duration to attain remission in INS patients with asthma in comparison with nonasthmatic nephrotic patients suggests a more sustained effect of IL-4 on glomeruli due to its slow clearance or probably prolonged excretion. This theory can explain the high excretory levels of IL-4 in Asthmatic INS patients even during remission. Moreover, the significantly lower urinary IL-4 levels in INS non-asthmatic patients support the assumption that the source of IL-4 in urine is most likely systemic rather than a local production in situ within the kidney tissues. Nevertheless, a large-scale study with more Asthmatic INS patients is warranted to prove or disprove this assumption.
However, comparing the mean urinary IL-4 levels in INS patients, as a whole, compared to controls, we found no difference between the two groups. This is most likely due to the small number of asthmatic INS patients included (n=14) compared to the majority of INS patients who were non asthmatics (n=27).
The relatively small number of asthmatic INS patients recruited in this study was one of the main limitations of the study. The strict inclusion criteria of INS patients with and without asthma as well as the selection of appropriate asthmatic control were also a limiting factor to recruit more subjects. Moreover, the reluctance of many patients to give blood samples after remission was an obstacle to do simultaneous measurements of IL-4 in serum and urine, which could have added much value to this work.
Conclusion
In conclusion, INS patients with asthma have a higher IL-4 excretion which might be a factor in increasing the severity and duration of proteinuria. The coexistence of Asthma in INS patients requires an early and more aggressive treatment of INS relapses. Targeting IL-4 with anti-cytokine therapy might be a future therapeutic option in managing INS relapses in patients with concomitant asthma.
Acknowledgement
We would like to thank Mrs. Asiyah Ebrahim for her technical help.
Ethical Approval
This study was approved by the Joint Committee for Protection of Human Subjects in Research of the Faculty of Medicine and Ministry of Health, Kuwait. Informed consent was obtained from the study subjects and/or their care givers as per regulations of the Ethics Committees.
References
- Mathieson P. Cytokine polymorphisms and nephrotic syndrome. Clin Sci 2002; 102: 513-514.
- Shalaby S, Al-Edressi H, El-Tarhouny S, et al. Type 1/type 2 cytokine serum levels and role of interleukin-18 in children with steroid-sensitive nephrotic syndrome. Arab J Nephrol Transplant 2013; 6: 83-88.
- Pereira W, Brito-Melo G, Guimaraes F et al. The role of the immune system in idiopathic nephrotic syndrome: A review of clinical and experimental studies. Inflamm Res 2014; 63: 1-12.
- Meadow S, Sarsfield J. Steroid-responsive nephrotic syndrome and allergy: Clinical studies. Arch Dis Child 1981; 56: 509-516.
- Yap H, Yip W, Lee B, et al. The incidence of atopy in steroid-responsive nephrotic syndrome: Clinical and immunological parameters. Ann Allergy 1983; 51: 590-594.
- Roy R, Islam R, Matin A, et al. Relationship of childhood idiopathic nephrotic syndrome with asthma, hypertension, complement C3, urine analysis. Bangl J Child Health 2011; 35:11-15.
- Meadow S, Sarsfield J, Scott D, Rajah S. Steroid-responsive nephrotic syndrome and allergy: Immunological studies. Arch Dis Child 1981; 56: 517-524.
- Sandberg D, McIntosh R, Bernstein C, et al. Severe steroid-responsive nephrosis associated with hypersensitivity. Lancet 1977; 1: 388-391.
- Arkwright P, Clark G. Infantile nephrotic syndrome and atopy. Ped Nephrol 1996; 10: 509-510.
- Lagrue G, Laurent J. Is lipoid nephrosis an ?allergic? disease? Transplant Proc 1982; 4: 485-488.
- Laurent J, Rostoker G, Robeva G, et al. Is adult idiopathic nephrotic syndrome a food allergy? Nephron 1987; 47: 7-11.
- Trompeter R, Barratt T, Kay R, et al. HLA, atopy and cyclophosphamide in steroid-responsive childhood nephrotic syndrome. Kidney Int 1980; 17: 113-117.
- Brown MA, Hural J. Functions of IL-4 and control of its expression. Crit Rev Immunol 1997; 17:1-32.
- Algorithm for the diagnosis and management of asthma: A practice parameter update. Ann Allergy 1998; 81: 415-420.
- International Study on Kidney Diseases in Children. Nephrotic syndrome in children: Prediction of histopathology from clinical and laboratory characteristics at time of diagnosis. A report of the International Study of Kidney Diseases in Children. Kidney Int 1978; 13: 159-165.
- International Study on Kidney Diseases in Children. The primary nephrotic syndrome in children. Identification of patients with minimal change nephrotic syndrome from initial response to prednisone. A report of the International Study of Kidney Diseases in Children. J Pediatr 1981; 98: 561-564.
- Fontana V, Spain W, Desanetis A. The role of allergy in nephrosis. NY state J Med 1956; 56: 3907-3910.
- Hardwicke J, Soothill J, Squire J, Holti G. Nephrotic syndrome with pollen hypersensitivity. Lancet 1959; 1: 500-502.
- Wittig H, Goldman A. Nephrotic syndrome associated with inhaled allergens. Lancet 1970; 1: 542-543.
- Reeves W, Cameron J, Johansson S, et al. Seasonal nephrotic syndrome; description and immunological findings. Clin Allergy 1975; 5: 121-137.
- Wei C, Lin C, Shen T, Li Y. Risk of idiopathic nephrotic syndrome among children with asthma: A nationwide, population-based cohort study. Pediatr Res 2015; 78: 212-217.
- Strait R, Morris S, Smiley K, et al. IL-4 exacerbates anaphylaxis. J Immunol 2003; 170: 3835-3842.
- Kimata H, Fujimoto M, Furusho K. Involvement of Interleukin IL-13 but not IL-4 in spontaneous IgE and IgG4 production in nephrotic syndrome. Eur J immunol 1995; 25: 1497-1501.
- Cho B, Yon S, Jang J, et al. Up-regulation of interleukin-4 and CD23/FcepsilonRII in minimal change nephrotic syndrome. Pediatr Nephrol 1999; 13: 199-204.
- Kanai T, Yamagata T, Momoi M. Macrophage inflammatory protein-1 beta and interleukin-8 associated with idiopathic steroid-sensitive nephrotic syndrome. Pediatr Int 2009; 51: 443-447.
- Youn Y, Lim H, Lee J. The clinical characteristics of steroid responsive nephrotic syndrome of children according to the serum immunoglobulin E levels and cytokines. Yonsei Med J 2012; 53: 715-722.
- Neuhaus T, Wadhwa M, Callard R, Barratt T. Increased IL-2, IL-4 and interferon-gamma (IFN-gamma) in steroid-sensitive nephrotic syndrome. Clin Exp Immunol 1995; 100: 475-479.
- Cho B, Lee C, Pyun K. Studies on the role of interleukin-4 and Fc-epsilonRII in the pathogenesis of minimal change nephrotic syndrome. J Korean Med Sci 1992; 7: 343-348.
- Kang J, Bai K, Wang B, et al. Increased production of interleukin-4 in children with simple idiopathic nephrotic syndrome. China Med J 1994; 107: 347-350.