Research Article - Biomedical Research (2017) Volume 28, Issue 1
Total flavonoid from Ba lotus leaf protected the reserpine-induced gastric ulcer in mice
Xin Zhao1-5#, Kai Zhu2-5#, Ruokun Yi2-5, Deguang Peng1*, Jia-Le Song61Chongqing Enterprise Engineering Research Center of Ba-lotus Breeding and Deep Processing, Chongqing 400041, PR China
2Department of Biological and Chemical Engineering, Chongqing University of Education, Chongqing 400067, China
3Chongqing Collaborative Innovation Center of Functional Food, Chongqing University of Education, Chongqing 400067, PR China
4Chongqing Engineering Technology Research Center of Functional Food, Chongqing University of Education, Chongqing 400067, PR China
5Chongqing Research and Development Engineering Laboratory of Functional Food, Chongqing University of Education, Chongqing 400067, PR China
6Department of Nutrition and Food Hygiene, School of Public Health, Guilin Medical University, Guilin, Guangxi 541004, PR China
#These authors contributed equally to this work
- *Corresponding Author:
- Deguang Peng
Chongqing Enterprise Engineering Research Center of Balotus Breeding and Deep Processing
Chongqing
PR. China
Accepted date: June 06, 2016
Abstract
The study was to investigate the protective effect of total flavonoid from Ba lotus leaf (BLLF) on reserpine (10 mg/kg)-induced gastric ulcer in KM (Kunming) mice. BLLF was given by gavage in mice, the serum and gastric tissue levels of MOT, SP, VIP, SS, SOD (T-SOD), GSH-Px and MDA were tested by experiment kits, the cytokine levels of TNF-α, IFN-γ, IL-6, and IL-12 were also checked by kits. The mRNA expression in gastric tissue was measured by RT-PCR assay. BLLF significantly reduced reserpine-induced gastric juice secretion and increased the pH of gastric juice. BLLF treatment also increased the levels of VIP and SS and reduced MOT, SP and inflammatory cytokines in serum. In addition, BLLF treatment was able to increase the gastric T-SOD, GSH-Px and reduced the MDA productions in gastric ulcer mice. It also modulated the gastric expression of NF-κB, IκB-α, Mn-SOD, Cu/Zn-SOD, GSH-Px, EGF and EGFR in ulcer mice by RT-PCR assay. These results propound that BLLF exhibits a protective effect against reserpine-induced gastric ulcers in KM mice by reducing gastric juice secretion, modulating serum neuropeptide levels, attenuating serum inflammatory cytokines, increased the gastric antioxidant activity and regulating gastric levels of NF-κB, IκB-α, EGF and EGFR.
Keywords
Ba lotus leaf, Total flavonoid, Neuropeptides, Inflammation, Antioxidant, Gastric ulcer.
Abbreviations
VIP: Vasoactive Intestinal Peptide; SS: Somatostatin; MOT: Motilin; SP: Substance P; T-SOD: Total Superoxide Dismutase; GSH-Px: Glutathione Peroxidase; MDA: Malondialdehyde; NF-κB: Nuclear Factor kappaB; IκB-α: Inhibitor kappaB-α; Mn-SOD: Mn-Superoxide Dismutase; Cu/Zn-SOD: Cu/Zn-SOD-Superoxide Dismutase; EGF: Epidermal Growth Factor; EGFR: Epidermal Growth Factor Receptor; BLLF: Ba lotus Leaf Flavones.
Introduction
The prevalence of chronic peptic ulcer disease, especially the gastric ulcer is increasing and associated with gastric carcinoma in modern society [1]. It is generally known that some unhealthy lifestyle factors (such as alcohol, smoking, and stress) caused the imbalance between offensive (acid, pepsin, and Helicobacter pylori infection) and defensive factors (mucin, prostaglandin, bicarbonate, and growth factors) plays an important role in the pathological process of chronic gastric ulcer and also influence the gastric carcinogenesis [1]. Long-term intake of antihypertensive reserpine could induce the excessive secretion of gastric acid through an increasing cholinergic activity, leading to excessive secretion of gastric acid and gastric ulceration and have been widely accepted as important causes in the pathogenesis of chronic gastric ulcer [2].
Neuropeptides, a type of small protein-like molecules, are widely distributed within the gastrointestinal tract, such as the vasoactive intestinal peptide (VIP), substance P (SP), somatostatin (SS), and motilin (MOT) which showed activities to regulate the gastric juice secretion, motility, contractions, and modulate the immune reaction in gastrointestinal tract [3]. Long et al. [4] have reported the abnormal levels of neuropeptides were associated with the pathological process in patients with gastrointestinal mucosal lesions and hemorrhage. In addition, a serious inflammatory response such as high levels of inflammatory cytokines interleukin (IL)-6, IL-12, tumor necrosis factor (TNF)-α and interferon (IFN)-γ was also observed in gastritis patients [5].
Some chemical reagents with an activity to reduce the secretion of gastric juice, such as histamine-2 receptor antagonist cimetidine and proton-pump inhibitor lansoprazole were used to treat a gastric ulcer in the clinic [6]. Long-term intake of these drugs may cause serious side-effects, especially the severity of liver disease and increased mortality in patients with cirrhosis [7]. Nature-source polyphenols and flavonoids have traditionally used for gastric ulcer treatment and preventing the gastric carcinoma [8]. Therefore, the development of more effective herb materials for treating gastric ulcers has become essential.
Lotus (Nelumbo Nucifera Gaertn., Nelumbonaceae) leaves are traditionally used as a material with homology of medicine and food to disperse the summer heat in Chinese traditional medicine. Several scientific investigations have reported that lotus leaves exhibited many health benefits and pharmacological activities, such as antioxidant [9], antidiarrheal [10], antiviral [11], antiobesity [12], and hepatoprotective [13]. In this study, we investigated the potential gastric protective activity of total flavonoids from Ba lotus, a new variety of Nelumbo Nucifera Gaertn yield in Chongqing, Southwest state of China, and to elucidate the mechanisms underlying its protective effects in reserpineinduced gastric ulcers in mice.
Materials and Methods
Chemical reagents
Trizol reagent, OligodT18 primer, murine Moloney leukemia virus (MMLV) reverse transcriptase, RNase inhibitor, ethidium bromide (EtBr), and agarose were purchased from Invitrogen Life Technologies (Carlsbad, CA, USA). Reserpine and ranitidine were purchased from Sigma Chemical Co. (St. Louis, MO, USA). All reagents were of analytical grade.
Extraction of Ba lotus leaf total flavonoid
Fresh Ba lotus leaves were purchased from Chongqing Enterprise Engineering Research Center of Ba-lotus Breeding and Deep Processing (Chongqing City, China), were freeze-dried and then ground into fine powder. Powdered Ba lotus leaves (1 kg) were added to 70% ethanol solution (10 L) and extracted at 70°C. After filtering, the Ba lotus leaf extraction solution was loaded into an ADS-17 macroporous resin column (5 cm × 50 cm). First elution was eluted with distilled water to remove water-soluble impurities and again eluted with 90% ethanol solution at 3 mL/min. The ethanol eluant was collected and then evaporated till dryness by SENCO R-201 vacuum rotary evaporator (Shanghai Shensheng Biotech Co., Shanghai, China) at 60°C, freeze-dried and stored at -80°C until further study [9]. The total flavonoid content of Ba lotus leaf was 0.627% by spectrophotometry.
Animal study and gastric ulcer induction
All of the KM mice (male, 6-wk-old) were purchased from Experimental Animal Center of Chongqing Medical University (Chongqing, China), which accommodated with a standard 12- h light-dark cycle at room temperature, and had access to complete nutrition granulated forage and water ad libitum. BLLF was dissolved at the concentrations of 50 and 100 mg/kg by distilled water. After 7 days, mice were randomly divided into five groups of seven mice each: group 1, mice were treated with 0.9% normal saline; group 2, reserpine -treated mice; groups 3, reserpine -treated mice were administered 50 mg/kg BLLF; groups 4, reserpine -treated mice were administered 100 mg/kg BLLF; groups 5, reserpine -treated mice were administered 50 mg/kg ranitidine [14] by intragastric route for 28 d. After treated with BLLF, gastric ulcer was induced by intraperitoneal (i.p.) administration of reserpine (10 mg/kg) according to the method described by Garattini et al. [15] at day 29. After reserpine inducing gastric ulcer for 24 h, all of the mice were sacrificed. Blood samples were collected from the heart using a vacuum blood collection tube (BD Biosciences, San Jose, CA, USA), then centrifuged (3,000 xg for 10 min at 4°C) and stored at -80°C until further study. The animal protocol used in this study was reviewed by the Animal Ethics Committee of Chongqing Medical University.
Evaluation of antiulcer activity
Gastric secretion volume of each mouse was collected using a 1mL syringe after the mouse was sacrificed, and the volume of each mouse was measured with a 10 mL measuring cylinder, and the pH of gastric juice was determined by using a SevenEasy pH meter (Mettler Toledo, Schwerzenbach, Switzerland). The isolated stomachs were inflated by injecting 10 mL of 1% formalin solution for 10 minutes to fix the tissues and opened along the greater curvature. The area (mm2) of hemorrhagic lesions that had developed in the stomach was measured under a Leica MZ7.5 dissecting microscope (Leica, Bensheim, Germany) with a square grid and the area was determined using ImageJ 1.44 software, the inhibitory rate was determined by the formula: inhibitory rate (%) = (control mice ulcer area - sample mice ulcer area) / control mice ulcer area × 100%.
Measurement of serum gastric neuropeptide levels
Serum levels of MOT, SP, VIP, and SS were measured by radioimmunoassay kits (Beijing Puer Weiye Biotechnology Co., Ltd., Beijing, China) according to the manufacture’s protocols.
Measurement of serum pro-inflammatory cytokine levels
Serum levels of TNF-α, IFN-γ, IL-6, and IL-12 were measured by a commercial ELISA kit (ELISA MAX, Biolegend, San Diego, CA, USA) according to the manufacturer’s protocol.
Gastric levels of total SOD (T-SOD), GSH-Px and MDA
Gastric tissue (100 mg) was first washed by cold phosphate-buffered saline (PBS) (pH 7.4, 0.1 M), and homogenized at 4°C. The total protein levels were determined using a Bio-Rad bicinchoninic acid (BCA) assay kit. Gastric levels of T-SOD, GSH-Px and MDA were measured by a commercial ELISA kit (Beyotime Institute of Biotechnology, Jiangsu, China) according to the manufacturer’s protocol.
Reverse transcription-polymerase chain reaction (RTPCR) assay
Gastric mRNA expression of NF-κB, IκB-α, Mn-SOD, Cu/Zn- SOD, GSH-Px, EGF, and EGFR was measured by an RT-PCR assay. Total RNA was isolated from 100 mg of gastric tissue using Trizol reagent according to the manufacture’s recommendations and centrifuged at 12,000 xg for 15 min at 25°C following the addition of chloroform. Isopropanol was added to the supernatant at a 1:1 ratio and the RNA was piloted by centrifugation (12,000 xg for 15 min at 4°C). After washing with ethanol, the RNA was solubilized in diethyl pyrocarbonate-treated RNase-free water and quantified by measuring the absorbance at 260 nm using a UV-1750 spectrophotometer (Shimadzu, Kyoto, Japan).
Equal amounts of RNA (1 μg) was reverse transcribed in a master mix containing 1x reverse transcriptase buffer, dNTPs (1 mM), oligodT18 primers (500 ng), MMLV reverse transcriptase (140 U), and RNase inhibitor (40 U) for 45 min at 42°C. PCR was then carried out in an automatic thermocycler (Bioneer, Daejeon, South Korea) for 30-35 cycles (94°C for 30 s, 55°C for 40 s, and 72°C for 40 s) followed by an 8-min extension at 75°C.
The PCR products were separated on 2% agarose gels and visualized by EtBr staining. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used for normalization [14].
Statistical analysis
All experiments were repeated three times, and the data were presented as the mean ± SD. Differences between the mean values for individual group was assessed by a one-way ANOVA with Duncan’s multiple range test. P<0.05 was considered to indicate a statistically significant difference. The SAS v9.1 statistical software package (SAS Institute Inc., Cary, NC, USA) was used for the analysis.
Results
BLLF attenuated reserpine-induced gastric ulcer
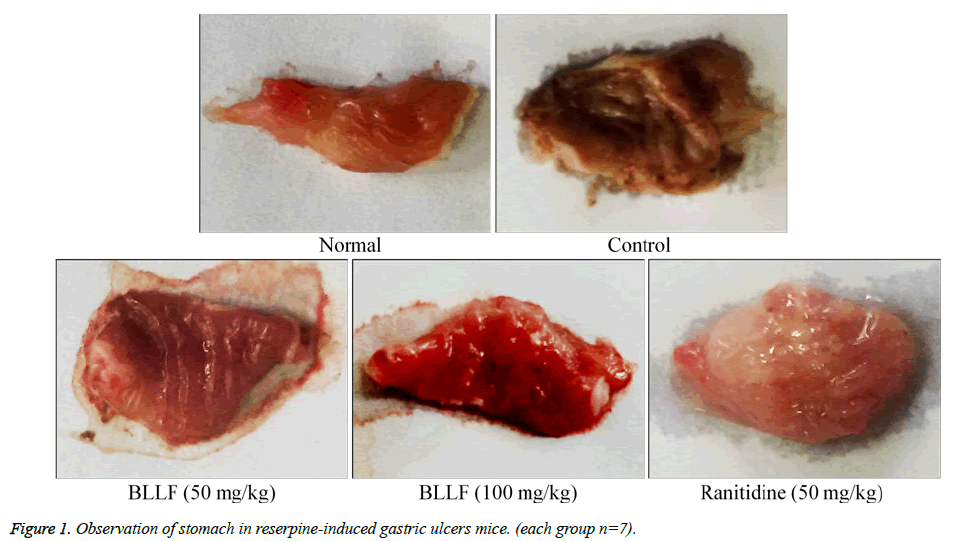
As presented in Table 1 and Figure 1, a dose of 10 mg/kg reserpine significantly induced gastric ulcers formation in mice (gastric ulcer area, 6.93 ± 0.56 mm2). Following 28 days of administration with 50 and 100 mg/kg of BLLF, the gastric ulcer level inhibition was 50.1% (gastric ulcer area, 3.46 ± 0.61 mm2) and 72.8% (gastric ulcer area, 1.88 ± 0.58 mm2), respectively. However, the anti-gastric ulcer activity of BLLF was weaker than that in ranitidine (50 mg/kg)-treated mice (gastric ulcer area 0.72 ± 0.22 mm2; inhibition rate 89.6%). As shown in Table 2, reserpine also significantly (P<0.05) increased the gastric juice secretion (0.91 ± 0.18 mL) compared to normal mice (0.30 ± 0.04 mL). Administration of 50 and 100 mg/kg of BLLF for 28 days, significantly reduced gastric juice secretion (P<0.05) to 0.65 ± 0.08 and 0.54 ± 0.07 mL, respectively. In addition, BLLF also significantly increased the pH of gastric juice in gastric ulcer mice treated with reserpine (10 mg/kg) significantly. None of the mice died during the experimental period.
| Group | Gastric ulceration | ||
|---|---|---|---|
| Gastric ulcer area (mm2) | Inhibitory rate (%) | ||
| Normal | 0.0 ± 0.0e | 100 ± 0.0a | |
| Control | 6.93 ± 0.56a | 0.0 ± 0.0e | |
| BLLF | 50 mg/kg | 3.46 ± 0.61b | 50.1 ± 8.8d |
| 100 mg/kg | 1.88 ± 0.58c | 72.8 ± 8.4c | |
| Ranitidine (50 mg/kg) | 0.72 ± 0.22d | 89.6 ± 3.2b | |
| a-eMean values with different letters in the same column are significantly different (P<0.05) according to Duncan’s multiple-range test. | |||
Table 1: Stomach appearance of Ba lotus leaf flavones (BLLF) treated reserpine-induced gastric ulcer mice (each group n=7).
| Group | Gastric secretion volume (mL) | pH of the gastric juice | |
|---|---|---|---|
| Normal | 0.30 ± 0.04e | 3.7 ± 0.3a | |
| Control | 0.91 ± 0.18a | 1.2 ± 0.4e | |
| BLLF | 50 mg/kg | 0.65 ± 0.08b | 2.2 ± 0.5d |
| 100 mg/kg | 0.54 ± 0.07c | 3.2 ± 0.2c | |
| Ranitidine (50mg/kg) | 0.42 ± 0.07d | 3.4 ± 0.3b | |
| a-eMean values with different letters in the same column are significantly different (P<0.05) according to Duncan’s multiple-range test. | |||
Table 2: Gastric secretion volume and pH of gastric juice of Ba lotus leaf flavones (BLLF) treated reserpine-induced gastric ulcer mice (each group n=7).
Effect of BLLF on serum neuropeptide levels in reserpine-induced gastric ulcer mice
As shown in Table 3, reserpine treatment significantly increased the serum levels of MOT (98.3 ± 6.2 μg/L) and SP (119.8 ± 5.3 μg/L) compared to levels in normal mice (MOT: 46.2 ± 2.2 μg/L and SP: 70.4 ± 3.2 μg/L). Treatment with 50 and 100 mg/kg of BLLF significantly attenuated the reserpine-induced SP (81.1 ± 4.9 and 65.2 ± 4. 2 μg/L) and MOT (97.6 ± 3.7 and 85.3 ± 2.9 μg/L) generation (P<0.05). BLLF also increased the levels of SS (82.7 ± 3.6 and 94.1 ± 4.0 μg/L) and VIP (74.2 ± 2.2 and 82.5 ± 3.1 μg/L) compared to that in reserpine-treated mice (77.6 ± 4.3 and 67.3 ± 2.8 μg/L, respectively). However, ranitidine showed a stronger activity to modulate the neuropeptide levels than BLLF.
| Group | MOT (μg/L) | SP (μg/L) | SS (μg/L) | VIP (μg/L) | |
|---|---|---|---|---|---|
| Normal | 46.2 ± 2.2e | 70.3 ± 3.2e | 117.8 ± 12.6a | 92.2 ± 4.5a | |
| Control | 98.3 ± 6.2a | 119.8 ± 5.3a | 77.6 ± 4.3e | 67.3 ± 2.8e | |
| BLLF | 50 mg/kg | 97.6 ± 3.7b | 81.1 ± 4.9d | 82.7 ± 3.6d | 74.2 ± 2.2d |
| 100 mg/kg | 85.3 ± 2.9c | 65.2 ± 4.2c | 94.1 ± 4.0c | 82.5 ± 3.1c | |
| Ranitidine (50 mg/kg) | 54.1 ± 2.5d | 78.5 ± 2.4d | 104.4 ± 3.8b | 88.7 ± 1.5b | |
| a-eMean values with different letters in the same column are significantly different (P<0.05) according to Duncan’s multiple-range test. | |||||
Table 3: Serum SP, VIP, MOT and SS levels of Ba lotus leaf flavones (BLLF) treated reserpine-induced gastric ulcer mice (each group n=7).
Effect of BLLF on gastric levels of T-SOD, GSH-Px and MDA in reserpine-induced gastric ulcer mice
As shown in Table 4, reserpine treatment significantly reduced the activity of T-SOD (61%) and GSH-Px (48%), and significantly induced MDA generation (4.5-fold) than that in gastric tissue of normal mice, respectively. In contrast, BLLF treatment significantly improved the activity of endogenous antioxidant enzymes in reserpine-treated control mice. 100 mg/kg of BLLF significantly enhanced about T-SOD (1.5-fold) and GSH-Px (1.7-fold) compared to that in control mice. In addition, 100 mg/kg of BLLF treatment significantly reduced MDA generation (62%) than in control mice. However, BLLF showed a weaker activity in regulating the antioxidant enzyme activity than ranitidine.
| Group | T-SOD (KU/mg protein) |
GSH-Px (mmoL/mg protein) |
MDA (μmoL/mg protein) |
|
|---|---|---|---|---|
| Normal | 31.26 ± 2.47a | 4.02 ± 0.13a | 16.17 ± 2.86e | |
| Control | 18.76 ± 2.62e | 1.89 ± 0.25e | 72.62 ± 7.44a | |
| BLLF | 50 mg/kg | 23.56 ± 3.32d | 2.41 ± 0.36d | 56.32 ± 4.43b |
| 100 mg/kg | 26.81 ± 2.80c | 3.17 ± 0.32c | 27.71 ± 3.08c | |
| Ranitidine (50 mg/kg) | 29.12 ± 2.91b | 3.72 ± 0.17b | 21.08 ± 2.17d | |
| a-eMean values with different letters in the same column are significantly different (P<0.05) according to Duncan’s multiple-range test. | ||||
Table 4: Gastric tissue total SOD (T-SOD), GSH-Px and MDA activities of Ba lotus leaf flavones (BLLF) treated reserpine-induced gastric ulcer mice (each group n=7).
Effect of BLLF on TNF-α, IFN-γ, IL-6 and IL-12 in reserpine-induced gastric ulcer mice
Reserpine treatment significantly increased the serum levels of TNF-α, IFN-γ, IL-6 and IL-12 than that in normal mice (Table 5). Both 50 and 100 mg/kg of BLLF treatment significantly reduced the generation of these inflammatory cytokines in reserpine-treated gastric ulcer mice. In addition, ranitidine treatment showed a stronger activity to reduce the reserpine-induced generation of these cytokines in BLLF treated gastric ulcer mice.
| Group | TNF-α (pg/mL) | IFN-γ (pg/mL) | IL-6 (pg/mL) | IL-12 (pg/mL) | |
|---|---|---|---|---|---|
| Normal | 45.1 ± 3.0e | 40.6 ± 2.7e | 47.3 ± 3.5e | 412.7 ± 33.9e | |
| Control | 103.8 ± 6.2a | 95.2 ± 5.6a | 118.7 ± 7.1a | 955.4 ± 51.2a | |
| BLLF | 50 mg/kg | 88.1 ± 5.7b | 80.1 ± 3.6b | 91.0 ± 5.4b | 782.3 ± 33.4b |
| 100mg/kg | 64.6 ± 4.8c | 55.1 ± 2.7c | 68.3 ± 3.1c | 624.1 ± 37.8c | |
| Ranitidine (50 mg/kg) | 53.1 ± 4.2d | 48.6 ± 2.1d | 55.7 ± 2.9d | 533.6 ± 27.5d | |
| a-eMean values with different letters in the same column are significantly different (P<0.05) according to Duncan’s multiple-range test. | |||||
Table 5: Cytokine IL-6, IL-12, TNF-α and IFN-γ levels of Ba lotus leaf flavones (BLLF) treated reserpine-induced gastric ulcer mice (each group n=7).
Effect of BLLF on gastric expressions of NF-κB, and IκB-α in reserpine-induced gastric ulcer mice
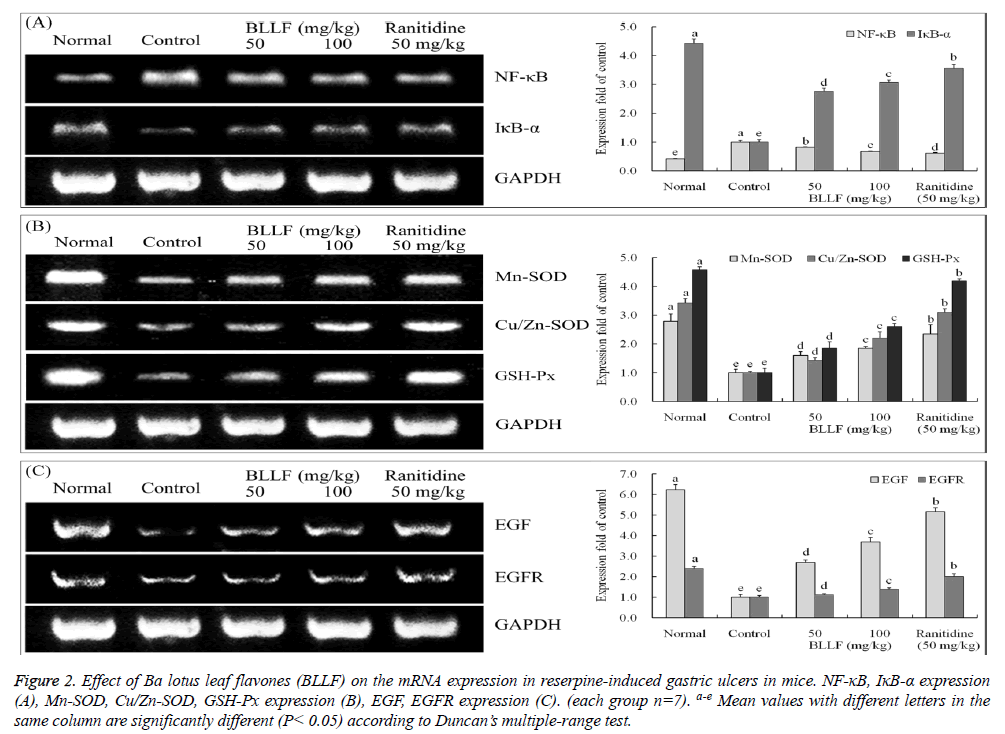
As shown in Figure 2A, reserpine significantly induced the mRNA expressions of NF-κB in gastric tissue than that in normal mice (0.41 folds of control). Administration of BLLF significantly reduced the mRNA levels of NF-κB (0.81 folds of control for 50 mg/kg BLLF and 0.67 folds of control for 100 mg/kg BLLF) in gastric tissue of ulcer mice. In addition, BLLF effectively increased gastric IκB-α (2.76 folds of control for 50 mg/kg BLLF and 3.07 folds of control for 100 mg/kg BLLF) expression in reserpine-treated gastric ulcer mice. However, BLLF exhibited a weaker activity in modulating the expressions of NF-κB, and IκB-α than ranitidine (0.61 and 3.57 folds of control).
Figure 2: Effect of Ba lotus leaf flavones (BLLF) on the mRNA expression in reserpine-induced gastric ulcers in mice. NF-κB, IκB-α expression (A), Mn-SOD, Cu/Zn-SOD, GSH-Px expression (B), EGF, EGFR expression (C). (each group n=7). a-e Mean values with different letters in the same column are significantly different (P< 0.05) according to Duncan’s multiple-range test.
Effect of BLLF on gastric expressions of Mn-SOD, Cu/Zn-SOD and GSH-Px in reserpine-induced gastric ulcer mice
Reserpine treatment significantly decreased the expressions of Mn-SOD, Cu/Zn-SOD and GSH-Px in gastric tissue than that in normal mice (2.79, 3.43 and 4.58 folds of control) (Figure 2B). BLLF treatment increased the mRNA levels of these antioxidant enzymes (1.59, 1.43, 1.86 folds of control for 50 mg/kg BLLF and 1.86, 2.21, 2.62 folds of control for 100 mg/kg BLLF) in gastric tissue of gastric ulcer mice. However, BLLF exhibited a weaker activity in increasing the expressions of Mn-SOD, Cu/Zn-SOD, and GSH-Px than ranitidine (2.35, 3.10 and 4.19 folds of control).
Effect of BLLF on gastric expressions of EGF and EGFR in reserpine-induced gastric ulcer mice
The gastric mRNA level of EGF and its receptor EGFR were reduced by reserpine in gastric ulcer mice (Figure 2C). Administration of BLLF, at the highest dose of 100 mg/kg increased the gastric mRNA levels of EGF (3.69 folds of control) and EGFR (1.39 folds of control) in gastric ulcer mice. However, ranitidine treatment showed greater improvement in EGF (5.16 folds of control) and EGFR (2.00 folds of control) expression compared to BLLF.
Discussion
It is well known that the excess secretion of gastric juice is an important risk factor to promote the gastric ulcer [16]. Reducing the secretion of gastric juice is a useful strategy to treat gastric ulcer1. We observed the BLLF treatment reduced the reserpine-induced gastric juice secretion and increased the pH of gastric juice in reserpine-treated mice. However, maintaining a higher pH condition in gastric juice was able to reduce the activity of pepsin (a risk factor for gastric ulcer) and improve the healing of the gastric mucosa [17].
Neuropeptides, which include SP, MOT, SS, and VIP, are not only involved in the regulation of gastric acid secretion but also play an important role in gastric tissue repair in normal physiological conditions [18]. The increased generation of SP results in a delayed gastric emptying and aggravates the gastric ulcer in gastritis patients [19], due to SP stimulates gastric juice secretion and decreases gastric contraction [20]. In addition, MOT is also a powerful inducer of gastrointestinal motor activity [21], intestinal contractility and stimulation of human gastric pepsin secretion [22]. In this study, administration of 50 and 100 mg/kg of BLLF effectively decreased the serum levels of SP and MOT in gastric ulcer mice. On the other hand, we observed that administration of 50 and 100 mg/kg of BLLF also significantly increased the serum levels of SS and VIP in gastric ulcer mice. SS is a peptide distributed in the brain and periphery, which is able to inhibit gastric acid secretion stimulated by pentagastrin, bethanechol, or histamine in rats through activation of somatostatin receptor type-2 receptors. It is well known that the gastric juice secretion is regulated by the central nervous system, in particularly the vagal nerve system has reported the peripheral injection of VIP was able to decrease gastric acid secretion through reducing the activation of vagal nerve in rats [23]. These results indicated that BLLF may influence the gastric juice secretion in reserpine-induced gastric ulcers by modulating the serum neuropeptides.
The excessive harmful chemicals induced oxidative stress to play an important role in the pathogenesis of gastric ulcer, and other chronic gastric disease [24]. The oxidative stress induced the lipid peroxidation in gastric mucosa [25]. Enhancing the activity of endogenous antioxidant enzymes including SOD, catalase and GSH-Px is able to decrease the gastric oxidative stress and against it induced gastric mucosal damage in gastric ulcer and gastric carcinoma [26]. Sumbul et al. [27] have reported that administration of some herb extracts and/or phenolic compounds with a high antioxidant activity were effectively increased the SOD activity and reduced chemically gastric ulcer in vivo. In this study, BLLF treatment increased the gastric SOD and GSH-Px activity and their mRNA expressions in reserpine-induced gastric ulcer mice. Itoh and Guth [28] has reported the administration of SOD was significantly reduced the hemorrhagic shock-induced gastric lesions in rats. In addition, treatment with SOD and catalase were able to reduce gastric lipid peroxidation levels in indomethacin-induced rats gastric ulcer [24]. The levels of MDA, final products of lipid peroxidation is an important indicator to evaluate various stress-induced tissue damage [29]. We found that administration of 50 and 100 mg/kg of BLLF significantly decreased the reserpine-induced gastric MDA generations in gastric tissue compared to reserpine-treated control mice.
By RT-PCR study, we also found the BLLF treatment was able to reduce the gastric mRNA levels of NF-κB, and increased the mRNA levels of IκB-α in gastric ulcer mice. NF-κB is a classic inflammatory factor to activate pro-inflammatory cytokines TNF-α and IL-6, and promote inflammatory disease and cancer [30]. As a nature inhibitor of NF-κB, enhancement of the IκB- α activity was able to reduce the inflammatory reaction and carcinogenesis in gastric disease [31]. TNF-α has been indicated as a key mediator in regulating the IL-6 in stomach disease [32]. However, administration of BLLF has significantly reduced the reserpine-induced generation of these inflammatory (TNF-α, IFN-γ, IL-6 and 12) in gastric ulcer mice. Reduction of these inflammatory cytokines was able to attenuate the inflammatory reaction of chronic gastric ulcer [5].
EGF is a growth factor that stimulates cell growth, proliferation, and differentiation by binding to its receptor EGFR, which is secreted by a salivary gland, duodenum Brunner gland, and pancreas [33]. The normal physiological functions of EGF was including inhibited gastric acid secretion, reduced the activity of gastric protease, promoted theIn this study, we demonstrated that BLLF exhibits a great protective effect against reserpine-induced gastric ulcers in KM mice. BLLF administration was able to attenuate reserpine-induced gastric acid secretion and increased the pH of gastric juice. In addition, BLLF administration also modulated serum levels of the neuropeptides, and decreased the serum levels of inflammatory cytokines, as well as increased the antioxidant enzymes to reduce the lipid peroxidation in gastric tissue of gastric ulcer mice. BLLF also reduced the expression of NF-κB and increased the activation of IκB-α to reduce the inflammatory reaction in gastric ulcer mice. In addition, BLLF treatment also increased the gastric levels of EGF, EGFR and antioxidant enzymes in ulcer mice. The results from this study indicate the potential mechanism of the protective effects of BLLF involves reduction of gastric juice secretion, modulation of neuropeptides, reducing the inflammatory reactions, and enhancing the antioxidant enzyme activity against reserpine-induced gastric ulcers. It is indicated that Ba lotus leaf total flavonoid represents a potential beneficial agent for the prevention of reserpine-induced gastric ulcer in vivo. gastric epithelial cells proliferation and prevented the ulcer formation [34]. We observed the BLLF treatment significantly increased the gastric mRNA levels of EGF and EGFR in ulcer mice.
References
- de Martel C, Forman D, Plummer M. Gastric cancer: epidemiology and risk factors. Gastroenterol Clin North Am 2013; 42: 219-240.
- Phillipson M, Atuma C, Henriksnäs J, Holm L. The importance of mucus layers and bicarbonate transport in the preservation of gastric juxtamucosal pH. Am J Physiol Gastrointest Liver Physiol 2002; 282: G211-G219.
- Gyires K. Neuropeptides and gastric mucosal homeostasis. Curr Top Med Chem 2004; 4: 63-73.
- Long DM, Leonard AS, Story J, French LA. Hypothalamus and gastric ulceration: II. Production of gastrointestinal ulceration by chronic hypothalamic stimulation. Arch Neurol 1962; 7: 176-183.
- Malfertheiner P, Chan FKL, Mccoll KEL. Peptic ulcer disease. Lancet 2009; 374: 1449-1461.
- Parsons ME, Keeling DJ. Novel approaches the pharmacological blockade of gastric acid secretion. Expert Opin Investig Drugs 2005; 14: 411-421.
- Thomson AB, Sauve MD, Kassam N, Kamitakahara H. Safety of the long-term use of proton pump inhibitors. World J Gastroenterol 2010; 16: 2323-2330.
- Borrelli F, Izzo AA. The plant kingdom as a source of anti-ulcer remedies. Phytother Res 2000; 14: 581-591.
- Lin HY, Kuo YH, Lin YL, Chiang W. Antioxidative effect and active components from leaves of Lotus (Nelumbo nucifera). J Agric Food Chem 2009; 57: 6623-6629.
- Talukder MJ, Nessa J. Effect of Nelumbo nucifera Rhizome Extract on the Gastrointestinal Tract of Rat. Bangladesh Med Res Counc Bull 1998; 24: 6-9.
- Kuo YC, Lin YL, Liu CP, Tsai WJ. Herpes simplex virus types 1 propagation in HeLa cells interrupted by Nelumbo nucifera. J Biomed Sci 2005; 12: 1021-1034.
- Ono Y, Hattori E, Fukaya Y, Imai S, Ohizumi Y. Anti-obesity effect of Nelumbo nucifera leaves extract in mice and rats. J Ethnopharmacol 2006; 106: 238-244.
- Sohn DH, Kim YC, Oh SH, Park EJ, Li X, Lee BH. Hepatoprotective and free radical scavenging effects of Nelumbo nucifera. Phytomedicine 2003; 10: 165-169.
- Li GJ, Sun P, Wang R, Zhou YL, Qian Y, Zhao X. Preventive effect of the polysaccharide of Larimichthys crocea swim bladder on a reserpine-induced gastric ulcer in ICR mice. Korean J Physiol Pharmacol 2014; 18: 183-190.
- Garattini S, Giachetti A, Joria A, Pieri L, Valzelli L. Effect of imipramine, amitriptyline and their monomethyl derivatives on reserpine activity. J Pharm Pharmacol 1962; 14: 509-514.
- Guth PH. Pathogenesis of gastric mucosal injury. Annu Rev Med 1982; 33: 183-196.
- Venables C. Mucus, pepsin, and peptic ulcer. Gut 1986; 27: 233-238.
- Holzer P. Neural emergency system in the stomach. Gastroenterology 1998; 114: 823-839.
- Geoghegan J, Pappas T. Central peptidergic control of gastric acid secretion. Gut 1997; 40: 164-166.
- Tache Y, Garrick T, Raybould H. Central nervous system action of peptides to influence gastrointestinal motor function. Gastroenterology 1990; 98: 517-528.
- Brown J, Parke C. Effect on fundic pouch motor activity of stimulatory and inhibitory fractions separated from pancreozymins. Gastroenterology 1967; 53: 731-736.
- Schubert ML, Peura DA. Control of gastric acid secretion in health and disease. Gastroenterology 2008; 134: 1842-1860.
- Sandor A, Kidd M, Lawton GP, Miu K, Tang LH, Modlin IM. Neurohormonal modulation of rat enterochromaffin-like cell histamine secretion. Gastroenterology 1996; 110: 1084-1092.
- Yoshikawa T, Naito Y, Ueda S, Takahashi S, Oyamada H, Morita Y. The role of oxygen-derived free radicals in the pathogenesis of gastric mucosal lesions in rats. J Clin Gastroenterol 1990; 12: S65-S71.
- Bhattacharyya A, Chattopadhyay R, Mitra S, Crowe SE. Oxidative stress: an essential factor in the pathogenesis of gastrointestinal mucosal diseases. Physiol Rev 2014; 94: 329-354.
- Tandon R, Khanna HD, Dorababu M, Goel RK. Oxidative stress and antioxidants status in peptic ulcer and gastric carcinoma. Indian J Physiol Pharmacol 2004; 48: 115-118.
- Sumbul S, Ahmad MA, Mohd A, Mohd A. Role of phenolic compounds in peptic ulcer: An overview. J Pharm Bioallied Sci 2011; 3: 361-367.
- Itoh M, Guth PH. The role of oxygen-derived free radicals in hemorrhagic shock-induced gastric lesions in the rat. Gastroenterology 1985; 88: 1162-1167.
- Jung HK, Lee KE, Chu SH, Yi SY. Reactive oxygen species activity, mucosal lipoperoxidation, and glutathione in Helicobacter pylori-infected gastric mucosa. J Gastroenterol Hepatol 2001; 16: 1336-1340.
- Gambhir S, Vyas D, Hollis M, Vyas A. Nuclear factor kappa B role in inflammation-associated gastrointestinal malignancies. World J Gastroenterol 2015; 21: 3174-3184.
- Yanai A, Maeda S, Shibata W, Hikiba Y, Sakamoto K, Nakagawa H, Ohmae T, Hirata Y, Ogura K, Muto S, Itai A, Omata M. Activation of IκB kinase β and NF-κB is essential for Helicobacter pylori-induced chronic gastritis in Mongolian gerbils. Infect Immun 2008; 76: 781-787.
- Abdollahi H, Shams S, Zahedi MJ, Darvish Moghadam S, Hayatbakhsh MM, Jafarzadeh A. IL-10, TNF-α and IFN-γ levels in serum and stomach mucosa of Helicobacter pylori-infected patients. Iran J Allergy Asthma Immunol 2011; 10: 267-271.
- Konturek JW, Bielanski W, Konturek SJ, Bogdal J, Oleksy J. Distribution and release of epidermal growth factor in man. Gut 1989; 30: 1194-1200.
- Reeves JR, Richards RC, Cooke T. The effects of intracolonic EGF on mucosal growth and experimental carcinogenesis. Br J Cancer 1991; 63: 223-226.