Research Article - Biomedical Research (2017) Volume 28, Issue 3
The effects of gender, unhealthy lifestyle and ALDH2 mutations on patients diagnosed with coronary artery diseases
Qiao-Yan Li1#, Song-Tao An2#, Ning-Min Zhao1, Hong-Fei Duan1, Yong-Cheng Ma1, Wei Zhang1, Yu-Hua Qin1* and Hong-Wei Zhao1*1Department of Pharmacy, Henan Provincial People’s Hospital, Zhengzhou University, Zhengzhou, Henan Province, China
2Department of Cardiology, Henan Provincial People’s Hospital, Zhengzhou University, Zhengzhou, Henan Province, China
#These authors equally contributed to this paper
- *Corresponding Author:
- Yu-Hua Qin
Department of Pharmacy
Henan Provincial People’s Hospital, China
Hong-Wei Zhao
Department of Pharmacy
Henan Provincial People’s Hospital, China
Accepted date: July 28, 2016
Abstract
Background: The potential associations between gender, lifestyles (only smoking and drinking habits considered), ALDH2 genotypes and Coronary Artery Diseases (CAD) have been sparingly reported. However, these previous researches did not provide a panoramic view on the inherent relationship between these negative factors and CAD.
Methods: Initially, 800 CAD patients and 247 healthy people were involved in this study. All the female patients involved, who formed group 1, had no smoking or drinking history. And the male patients were divided into three groups (Group 2 ~ 4): Group 2 without smoking or drinking habits, Group 3 with only smoking habits and Group 4 bearing both smoking and drinking habits.
Results: Based on the comparison between Group 2 and other groups, it was found that: first, the female patients suffered from significantly different symptoms of dyslipidaemia; second, it is obvious that patients bearing more bad habits were found more possible to get hyper TG. The frequencies of ALDH2 mutant genotypes in Group 1, Group 2 and Group 3 were higher than that of the healthy subjects. Also, it was found that subjects with the mutant genotypes suffered from hypo HDL-C more commonly, but hypertension was less prevalent among them, compared with patients taking wild genotype of ALDH2.
Conclusion: Gender, unhealthy lifestyle and ALDH2 mutations all had significantly effects on the progress of CAD by affecting the prevalence of dyslipidaemia or/and hypertension. The results of this research will help the treatment of CAD.
Keywords
ALDH2 genotypes, Unhealthy lifestyle, Coronary artery disease (CAD), Dyslipidaemia, Hypertension.
Introduction
Alcohol consumption and smoking have been discovered to play an important role in Coronary Artery Disease (CAD). ALDH2, the primary enzyme involved in alcohol metabolism, contributes significantly to the ethanol-derived acetaldehyde and environmental aldehyde (acrolein) conversion [1,2]. Likewise, the mutant or inactive ALDH2*2 gene cause a significant accumulation of these harmful aldehydes. Therefore, the ALDH2*2 variant has been unanimously identified to be a risk factor for diseases related with smoking and drinking, such as CAD [1,3-9].
Over the past years quite a lot of efforts have been paid to illustrate the mechanisms how ALDH2 polymorphism influences the progress of CAD, focusing on the regulation of alcohol consumption, oxidative stress, asymmetric dimethyl arginine, the prevalence of hypertension and hyperglycaemia [4-6,8,9]. Also, smoking or/and drinking reportedly play roles in prevalence of dyslipidaemia and hypertension in CAD patients [10-13]. However, the synergetic effects of lifestyles and ALDH2 genotypes on dyslipidaemia and hypertension were not unified and even contentious. Albeit, most researches in East Asians reported that the genetic variation of ALDH2 could influence HDL-C levels, independent of or in company with alcohol consumption [14,15]. A study on Japanese population reported that after adjusting for other variables including drinking status carrying the mutant genotypes of ALDH2 was a significant and positive factor related to hyper- LDL-C [16]. Another study on Chinese male population disclosed that ALDH2 genotypes were significantly associated with serum TG levels in drinkers [17]. In addition, some studies unveiled that the wild-type ALDH2*1 allele was a risk factor for elevated blood pressure independently of alcohol consumption [18,19]. However, contradictorily, some studies proposed that the presence of ALDH2*1 gene enhance the risk of hypertension by affecting alcohol drinking behaviours [20]. Thus, based on this point, it was critical to conduct a comprehensive research on the potential relationship between these possible factors and CAD. This research was the first case that illuminated the independent and combined effects of gender, lifestyles and ALDH2 genotypes on the progress of CAD. And in our studies, CAD patients were recruited instead of healthy people, aiming to an improved risk management of CAD and precise therapies.
Methods
Subjects
This study approved by the ethical committees of Henan Provincial People’s Hospital, was conducted from September 2012 to December 2014 in Henan Provincial People’s Hospital, Henan province, China. The CAD was diagnosed by angiography. Patients with excessive smoking (>20 g tobacco/ day) or excessive drinking (>100 g liquors/day) were excluded. And 800 CAD patients (579 males, 221 females) and 247 healthy people (186 males, 61 females) were registered in this study. Written informed consents were obtained from all patients. Subjects were divided into four groups based on the smoking or/and drinking status. All the involved female patients had no smoking or drinking history (Group 1, n=221). The male patients were categorized into three groups: Group 2 without smoking or drinking habits (n=346), Group 3 bearing only smoking habits (≤ 20 g tobacco/day, n=73) and Group 4having both smoking and drinking habits (≤ 20 g tobacco/day and ≤ 100 g liquors/day, n=160). Notably, few subjects having only drinking habits without smoking history were excluded.
Clinical data
Information about tobacco smoking, alcohol consumption and disease history was collected from the face-to-face interviews. Hypertension was defined as a systolic blood pressure larger than 140 mmHg and/or diastolic blood pressure larger than 90 mm Hg or history of hypertension. Dyslipidaemia was defined as TG levels higher than 1.7 mmol/L, TC levels higher than 5.2 mmol/L, LDL-D levels higher than 3.4 mmol/L, and HDL-C levels less than 1.0 mmmol/L or history of dyslipidaemia. Information on ages when they began to suffer from or diagnosed with CAD and they participated in this research was systematically recorded.
Experimental determinations
Venous blood samples were drawn and collected in Ethylenediaminetetraacetic Acid (EDTA) tubes. DNA was obtained by using a whole blood DNA extraction and purification kit (BaiO, Shanghai, China) according to the manufacturer’s instructions. Samples were coded to allow blinding of the investigators who carried out the genotyping. The ALDH2 genotypes was determined by PCR- Genotyping microarray analysis of the two important allelic variants (ALDH2*1 and ALDH2*2) by BaiO gene detection kit (Genotyping microarray method, BaiO, Shanghai, China).
Statistical methods
The effects of ALDH2 genotypes on prevalence of dyslipidaemia and hypertension were investigated using Paired Samples T test. Chi-Square test was applied to determine the influences of gender, tobacco smoking and alcohol consumption in the cross-comparison between every two groups. A P value of <0.05 (two-tailed) was considered to be statistically significant.
Results
The influence of gender on CAD patients
The clinical characteristics of the participants are shown in Table 1. No significant differences were observed in BMI and prevalence of hypertension and diabetes mellitus between Group 2 and Group 1. Compared with Group 2, the patients from Group 1 had higher prevalence of hyper-TC and hyper- LDL-C (P<0.001 and p=0.01 respectively), but female patients had lower prevalence of hypo-HDL-C than the male patients (P<0.001). Different gender had rather different features of dyslipidaemia.
| Group 1 | Group 2 | Group 3 | Group 4 | |
|---|---|---|---|---|
| n | 221 | 346 | 73 | 160 |
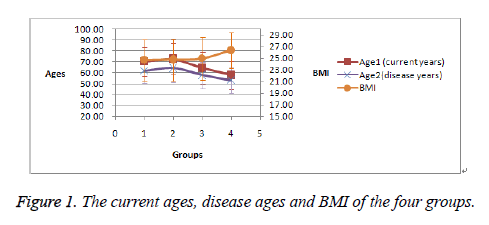
| Age-1 (current ages) | 70.3 ± 13.0 | 72.8 ± 14.0 | 64.3 ± 14.7 | 57.9 ± 12.9 |
| Age-2 (disease ages) | 62.1 ± 11.2 | 64.3 ± 12.7 | 58.0 ± 12.1 | 53.2 ± 11.7 |
| BMI (kg/m2) | 24.7 ± 3.5 | 24.8 ± 3.6 | 25.0 ± 3.7 | 26.4 ± 3.1 |
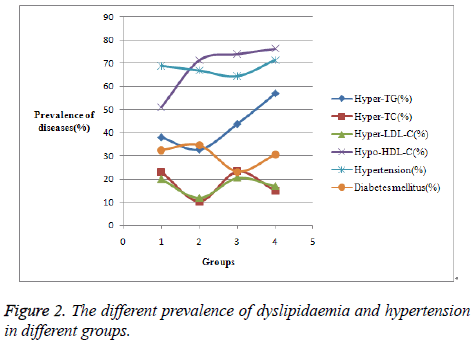
| Hyper-TG (%) | 38.0 | 32.7 | 43.8 | 56.9 |
| Hyper-TC (%) | 23.1 | 10.4 | 23.3 | 15.0 |
| Hyper-LDL-C (%) | 19.9 | 11.8 | 20.5 | 16.9 |
| Hypo-HDL-C (%) | 51.1 | 71.4 | 74.0 | 76.3 |
| Hypertension (%) | 68.8 | 66.8 | 64.4 | 71.3 |
| Diabetes mellitus (%) | 32.6 | 34.7 | 23.3 | 30.6 |
Table 1. Clinical characteristics of participants.
The influence of lifestyle on CAD patients
It was found that it was easier for the male patients with more bad habits (from Group 2 to Group 4) to get overweight (BMI>25) and get CAD at younger age, evidenced by Figure 1. It can be obvious in Figure 2 that the prevalence of hyper- TG becomes higher and higher from Group 2 to Group 4. No significant differences were shown in the prevalence of hypertension and diabetes mellitus among the male patients.
The influence of ALDH2 genotypes on CAD patients
First, the ALDH2 genotypes distribution in the different groups was shown in Table 2. It was noteworthy that patients in Group 1, Group 2 and Group 3 bore higher frequencies of ALDH2*1/*2 and *2/*2 alleles than the controls with P=0.040, 0.096 and 0.071, respectively. But patients from Group 4 had comparable frequencies of ALDH2*1/*2 and *2/*2 alleles with the controls.
| Group 1 | Group 2 | Group 3 | Group 4 | Controls | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| ALDH2 genotypes | *1/*1 | *1/*2 and *2/*2 | *1/*1 | *1/*2 and *2/*2 | *1/*1 | *1/*2 and *2/*2 | *1/*1 | *1/*2 and *2/*2 | *1/*1 | *1/*2 and *2/*2 |
| n | 148 | 73 | 240 | 106 | 47 | 26 | 128 | 32 | 187 | 60 |
| % | 67.0 | 33.0 | 69.4 | 30.6 | 64.4 | 35.6 | 80.0 | 20.0 | 75.7 | 24.3 |
| P | 0.04 | 0.096 | 0.071 | 0.334 | --- | |||||
Table 2. Distribution of ALDH2 genotype in the CAD patients and controls.
Then, the above four groups were further divided into eight subgroups, according to the ALDH2 genotypes. The clinical characters of different subgroups were shown in Table 3. Based on the comparison of the average values of the current ages, the disease ages and the BMI by paired T test, no significant differences were found between the subgroups with wild allele and the subgroups with mutant alleles. But the frequency of hyper-HDL-C in the mutant alleles subgroups was higher than that of the wild allele subgroups (P=0.03). On the contrast, the frequencies of hypertension in the wild allele subgroups were higher than that of the mutant alleles subgroups (P=0.02). The patients with mutant alleles of ALDH2 suffered from higher risk in getting diabetes mellitus than patients with wild genotype of ALDH2, though the difference was not significant.
| Group 1 | Group 2 | Group 3 | Group 4 | |||||
|---|---|---|---|---|---|---|---|---|
| *1/*1 | *1/*2 and *2/*2 | *1/*1 | *1/*2 and *2/*2 | *1/*1 | *1/*2 and *2/*2 | *1/*1 | *1/*2 and *2/*2 | |
| Years (Current ages) |
70.3 ± 13.6 | 74.2 ± 10.9 | 73.4 ± 13.9 | 71.3 ± 14.2 | 66.1 ± 14.4 | 61.2 ± 14.9 | 58.3 ± 13.0 | 56.3 ± 12.3 |
| Years (Disease ages) |
62.3 ± 11.9 | 63.8 ± 10.1 | 65.1 ± 12.8 | 62.7 ± 12.3 | 59.6 ± 11.9 | 55.2 ± 12.0 | 53.1 ± 11.8 | 53.2 ± 11.2 |
| BMI(kg/m2) | 24.9 ± 3.6 | 24.4 ± 3.3 | 24.6 ± 3.2 | 25.3 ± 4.2 | 25.3 ± 4.2 | 24.5 ± 2.5 | 26.3 ± 3.2 | 26.7 ± 2.7 |
| Hyper-TG (%) | 36.5 | 41.1 | 32.9 | 32.1 | 44.7 | 42.3 | 56.3 | 59.4 |
| Hyper-TC (%) | 23.6 | 21.9 | 10.8 | 9.4 | 21.3 | 26.9 | 16.4 | 9.4 |
| Hyper-LDL-C (%) | 20.3 | 19.2 | 11.7 | 12.3 | 19.1 | 23.1 | 18.0 | 12.5 |
| Hypo-HDL-C (%) | 48.0 | 57.5 | 70.8 | 72.6 | 70.2 | 80.8 | 74.2 | 84.4 |
| Hypertension (%) | 70.9 | 64.4 | 69.6 | 60.4 | 70.2 | 53.8 | 74.2 | 59.4 |
| Diabetes mellitus (%) | 29.7 | 38.4 | 35.0 | 34.0 | 27.7 | 15.4 | 28.9 | 37.5 |
Table 3. The clinical characters of the subgroups with different ALDH2 genotypes.
The combined influence of gender, lifestyle and ALDH2 genotypes on CAD patients
As to the influence of lifestyle, it was discovered that the male patients with more bad habits were more inclined to obtain hyper-TG, hyper-TC and hyper-LDL-C. From Table 3, it is observed that it is easier for male patients from group 3 and group 4 with the mutant alleles of ALDH2 to get hyper-TG than those with wild ALDH2 genotype; in contrast, patients with wild ALDH2 genotypes were more inclined to get hyper- TC and hyper-LDL-C than those with mutant alleles of ALDH2. Furthermore, ALDH2 mutant alleles had been found as risk factors for hypo-HDL-C and diabetes mellitus, but the effects were significant only in female patients from Group 3 and Group 4. However, the effects of ALDH2 *1/*1 genotype on the prevalence of hypertension was independent of genders and lifestyles.
Discussion
Despite that ALDH2 genotypes has been paid much attention to on the progress of CAD [1-9], lifestyles (smoking and drinking) had more important effects on the progress of CAD and serves as the only factor that affects the age when people get CAD, more bad habits the people had at younger age they would get CAD as shown in Figure 1. If so, it seems reasonable that the effect of ALDH2 mutant genotypes on CAD prevalence was more significant in Group 1, Group 2 and Group 3 (which bore higher frequencies of ALDH2*1/*2 and *2/*2 alleles) rather than Group 4. However, the effect of ALDH2 genotypes was dimmed by the effects of smoking combined with drinking in Group 4. ALDH2 genotypes and lifestyle had synergetic effects on the prevalence of dyslipidaemia. Notably the effects of ALDH2 genotypes were multiple. For example, in Group 3 and Group 4 ALDH2*1/*1 genotype had preventive effects on hyper-TG and hypo-HDLC, but it was a risk factor on hyper-TC and hyper-LDL-C. Moreover, the prevalence of hypertension was only affected by ALDH2 genotypes with nothing to do with gender and lifestyle. However, the prevalence of hypo-HDL-C and diabetes mellitus was affected by the combination of gender, lifestyle and ALDH2 genotypes.
In conclusion, this research reported the independent and combined effects of gender, lifestyles and ALDH2 genotypes on the progress of CAD. It was expected to benefit the treatment of CAD. Abandoning these bad habits for patients with smoking or drinking histories or improving the HDL-C levels for patients with mutant ALDH2 genotypes can help reduce the TG levels and then slow down the progression of CAD. Notably, different therapeutic regimen can be considered for different genders, due to different lipid profiles between genders.
References
- Che-Hong C, Lihan S, Daria MR. Mitochondrial aldehyde dehydrogenase and cardiac diseases. Cardiovasc Res 2010; 88: 51-57.
- Belem YS, Jose S, Rodriguez Z. Differences in susceptibility to inactivation of human aldehyde dehydrogenases by lipid peroxidation byproducts. Chem Res in Toxicol 2012; 25: 722-729.
- Che HC, Julio CBF, Eric RG. Targeting aldehyde dehydrogenase 2: new therapeutic opportunities. Physiol Rev 2014; 94: 1-34.
- Shuichi T, Naoharu I, Ryoko Y. Aldehyde dehydrogenase 2 gene is a risk factor for myocardial infarction in Japanese men. Hypertens Res 2002; 5: 677-681.
- Yuan B, Yu GC, Feng X. The polymorphism in aldehyde dehydrogenase-2 gene is associated with elevated plasma levels of high-sensitivity C-reactive protein in the early phase of myocardial infarction. Tohoku J Exp Med 2010; 2: 107-112.
- Antje DE, Kazuki K, Ping L. Characterization of the molecular mechanisms underlying increased ischemic damage in the aldehyde dehydrogenase 2 genetic polymorphism using a HUAM induced pluripotent stem cell model system. Sci transl med 2014; 6: 255ra130.
- Yuefei W, Fengxia D, Hongye Z. Synergistic association between two alcohol metabolism relevant genes and coronary artery disease among Chinese hypertensive patients. PLoS One 2014; 7: 103161.
- Feng X, Yaning S, Rui S. The Glu504Lys polymorphism of aldehyde dehydrogenase 2 contributes to development of coronary artery disease. Tohoku J Exp Med 2014; 2: 143-150.
- Guo YJ, Chen L, Ben YP. The ALDH2 Glu504Lys polymorphism is associated with coronary artery disease in Han Chinese-Relation with endothelial ADMA levels. Atheroscl 2010; 2: 545-540.
- Sandra NS, Jana VO, Judith MV. Associations between smoking, components of metabolic syndrome and lipoprotein particle size. BMC Med 2013; 11: 195-209.
- Sandra NS, Jana VO, Judith MV. Combined effects of smoking and alcohol on metabolic syndrome-The life lines cohort study. PLoS One 2014; 4: e96406.
- Guo ZS, Zhao L, Liang G. High prevalence of dyslipidaemia and associated risk factors among rural Chinese adults. Lipids Health Dis 2014; 13: 189-199.
- John BW, Andrew CH, Pamela AFM. Metabolic and biochemical effects of low-to-moderate alcohol consumption. Alcohol Clin Exp Res 2013; 4: 575-586.
- Manabu W, Makoto D, Mitsuru E. Genetic association between aldehyde dehydrogenase 2 (aldh2) variation and high-density lipoprotein cholesterol (hdl-c) among non-drinkers in two large population samples in Japan. J Atheroscler Thromb 2008; 15: 179-184.
- Nakamura Y, Amamoto K, Tamaki S. Genetic variation in aldehyde dehydrogenase 2 and the effect of alcohol consumption on cholesterol levels. Atherosclerosis 2002; 1: 171-177.
- Kotani K, Sakane N, Yamada T. Association of an aldehyde dehydrogenase 2 (ALDH2) gene polymorphism with hyper-low-density lipoprotein cholesterolemia in a japanese population. Ethn Dis 2012; 3: 324-328.
- Aihua T, Jielin S, Ning X. A genome-wide association and gene–environment interaction study for serum triglycerides levels in a healthy Chinese male population. Hum Mol Genet 2012; 7: 1658-1664.
- Zhang SY, Chan SW, Zhou X. Meta-analysis of association between aldh2 rs671 polymorphism and essential hypertension in Asian populations. Herz 2014.
- Peng H, Tomohiro N, Akihiko M. Common single nucleotide polymorphisms in Japanese patients with essential hypertension: aldehyde dehydrogenase 2 gene as a risk factor independent of alcohol consumption. Hypertens Res 2007; 7: 585-592.
- Shuichi T, Shunroku B, Naoharu I. The aldehyde dehydrogenase 2 gene is a risk factor for hypertension in Japanese but does not alter the sensitivity to press effects of alcohol the suit study. Hypertens Res 2001; 4: 365-370.