- Biomedical Research (2006) Volume 17, Issue 1
The effect of Smilax calophylla on testicular 11?-hydroxysteroid dehydrogenase activity and plasma testosterone levels in rats
Nuraliza AS1*, Nwe KHH2, Morat PB3, Hamid A2 and Khalid BAK41Faculty of Medicine, University Technology MARA, 40450 Shah Alam, Selangor, Malaysia
2Faculty of Medicine and Health Sciences, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia
3Department of Biomedical Science, Faculty of Allied Health Science, Universiti Kebangsaan Malaysia, Jalan Raja Muda Abdul Aziz, 50300 Kuala Lumpur, Malaysia
4School of Medicine and Health Sciences, Monash University Malaysia Campus, 46150 Petaling Jaya, Selangor, Malaysia
- *Corresponding Author:
- A.S. Nuraliza
Faculty of Medicine, University Technology
MARA 40450 Shah Alam, Selangor, Malaysia
E-mail: nuraliza064 ( at ) salam.uitm.edu.my
Accepted date: November 10, 2005
Abstract
The present study documented the effect of Smilax calophylla “Akar dawai”, used tradition-ally in Malaysia as a male sexual tonic on the plasma testosterone (T) levels and testicular 11 β -hydroxysteroid dehydrogenase (11 β -HSD) activity in the adult male Wistar rats. The ac-tivity of testicular 11 β -HSD was determined as the percentage conversion of corticosterone to 11 -dehydrocorticosterone while plasma T levels were determined by using Coat-A-Count Diagnostic Kit. Administration of Akar dawai water extract (AD) at the dose of 8mg/kg body weight for seven consecutive days resulted in an attenuated testicular 11 β -HSD activ-ity (p<0.05) whereas the plasma T levels remained unchanged. Exogenous corticosterone (CORT), on the other hand, was found to reduce testicular 11 β -HSD activity (p<0.001) as well as plasma T levels (p<0.05) compared to that of the control. However, treatment of AD given to the CORT-primed rats evidently increased testicular 11 β -HSD activity (p<0.001) as well as plasma T levels (p<0.05) compared to rats treated with CORT alone. Since AD is found to be able to neutralize the CORT-associated reduction in testicular steroidogenesis, it is, therefore, proposed that the stress-induced androgen deficiency could possibly be recti-fied by AD.
Keywords
Smilax calophylla, testicular 11β-HSD, plasma testosterone levels
Introduction
Elevated glucocorticoid levels in stressful conditions ex-ert detrimental effects on male reproductive function, by lowering the levels of testosterone (T) [1,2]. This is partly attributable to the direct inhibitory effects of glucocorti-coids on Leydig cells steroidogenesis [3,4]. Leydig cells contain glucocorticoid receptors (GR), thus indicating that they could be the target cells for glucocorticoid [5]. Low T production following stress or excess glucocorticoids has also been found to be associated with the reduction in the testicular 11 β -hydroxysteroid dehydrogenase (11 β -HSD) activity [6].
11β-hydroxysteroid dehydrogenase is a microsomal en-zyme, whereby in the tissues it regulates the levels of glu-cocorticoids by catalyzing the inter conversion of active glucocorticoids to the inactive form [7]. This enzyme is found in the testis, liver and in many other rat tissues [8,9]. In the testis however, 11 β -HSD activity is pro-posed to be predominantly oxidative [10,11]. The inhibi-tion of testicular 11 β -HSD oxidative activities by carbe-noxolone increases the potency of glucocorticoids in re-ducing T biosynthesis [10]. In fact, in certain treatments, the 11 β -HSD activity and plasma T levels showed a positive correlation whereas suppression of 11 β -HSD activity reduces the plasma T levels or vise-versa [12]. Therefore, 11 β -HSD activity has been suggested to pro-tect testis from the deleterious effect of glucocorticoid [10,13].
Smilax calophylla “Akar dawai”, is an indigenous herb and the decoction of Akar dawai rhizome is traditionally consumed by local Malaysian folks as a male sexual tonic. Furthermore, extract of Akar dawai rhizome has been used as the treatment of gonorrhoea and oedema. The Smilax plants are reputed by Malays to have a potent aphrodisiac property [14] but the belief is still lacking of scientific supports. Studies on several Smilax plants re-vealed that the major components of the plant rhizomes are steroidal glycosides [15,16]. Moreover, some other Smilax species are shown to have an anti mutagenic [17], anti bacteria [18], anti mycotic [19] and hypoglycemic effects [20]. However, to date no scientific study has so far been carried out on Akar dawai.
Therefore, the aim of the present study was to determine the effects of Akar dawai rhizome water extract on the plasma levels of T as well as on the testicular 11 β -HSD activity in corticosterone-treated fertile male rats.
Materials and Methods
Chemicals
Corticosterone (CORT), 11-dehydrocorticosterone, olive oil, glucose, bovine serum albumin (BSA) and NADP were purchased from Sigma Chemical Co. St. Louis, USA. Dye reagent was obtained from Bio-Rad Laboratory, CA, USA. The commercial RIA kit, Coat-a-count was obtained from Diagnostic Products Corp., Los Angeles, CA. Water extract of Akar dawai rhizome (AD) was kindly supplied by Prof. Dr. Johari Mohd. Saad, Depart-ment of Biochemistry, Faculty of Medicine, University of Malaya.
Animals and treatments
Adult male Wistar rats of 200-250g body weight (BW) were used. Rats were randomly assigned into the treatment groups. Six to twelve rats were assigned into each group. Two to three control rats were housed together with their corresponding treated rats. Rats were housed at a temperature of 27-29°C with 12:12 hour dark- light cycle. Control rats received either intra muscular injection of 0.1ml olive oil or 0.5ml normal saline orally. Steroid was dissolved in olive oil and injected (0.1ml) intramuscularly daily, while the water extract of AD was gavaged (0.5ml) daily.
Rats were treated with either AD (8mg/kg BW) alone, CORT (2.4mg/kg BW) or the combination of AD (8mg/kg BW) and CORT (2.4mg/kg BW). All the treat-ments were given daily for seven consecutive days. The dosage and the duration of treatment for steroid used were scheduled according to previous studies [6]. Rats were sacrificed 24 hours following the last dose scheduled between 8.30 and 9.00 a.m. From the AD time and dose dependant graph, we have found that three days treatment of AD (8mg/kg BW) significantly increased the levels of plasma T [21]. Rats were also treated with AD (8mg/kg BW) or CORT (2.4mg/kg BW) alone for three consecutive days.
The Medical Research and Ethics Committee of National University of Malaysia (UKM) had approved the method-ology used in the present study.
Assay of 11 β -HSD enzyme activity
After the rats were sacrificed, testes were removed, dissected on ice, homogenized with Krebs solution containing glucose on ice, and 250 l homogenate was assayed for 11 β -HSD activity as described previously [22]. The assay was performed by incubating tissue homogenate with 12nM 3H-B and 200 M NADP at 37°C for 10 minutes. Then the reaction was stopped and the steroids were extracted by the addition of ethyl acetate. After thin layer chromatography, the steroids were identified under ultraviolet light and the radioactivity was determined in liquid scintillation counter.
The 11 β -HSD activities were expressed as percentage conversion of corticosterone to 11-dehydrocorticosterone. Incubation time and dose used in the enzyme assay were according to the previous study on time and dose depend-ant curves and most appropriate under that same circumstances that have been reported [23].
Radioimunoassay (RIA) for plasma testosterone levels
Five minutes before sacrifice, rats were given diethyl ether anesthesia and 3-4ml of blood was collected in heparinized tube at the time of sacrifice. Ether was used to minimize marked fluctuation of plasma T levels [24]. Plasma levels of T were estimated by using the commercial RIA kit (Coat-a-count, Diagnostic Products Corp. CA) [12].
Statistical analysis
The data for 11 β -HSD oxidative activities were expressed as mean ± standard error of mean (SEM) while plasma levels of T were expressed as mean ± 95% confidence interval (CI). Data were analyzed using the Statistix Programme. Differences between groups were analyzed by analysis of variance (ANOVA) and student t-test. The differences were considered significant at p<0.05 [25].
Results
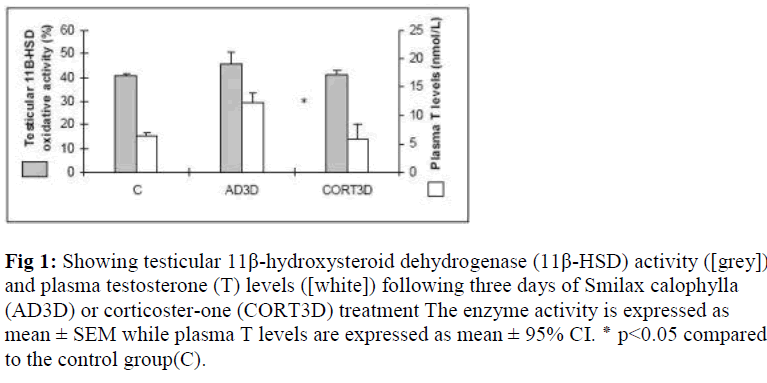
Testicular 11 β -HSD activity and plasma testosterone levels in rats treated with Smilax calophylla or corticosterone for three consecutive days From AD time- and dose-dependent study [21], it was found that AD (8mg/kg BW) treatment for three consecutive days (AD3D) significantly increased the plasma levels of T (p<0.05). Treatment of CORT for three consecu-tive days (CORT3D) was also introduced. However, we found no significant change in the enzyme activity as well as plasma levels of T (Fig. 1).
Figure 1: Showing testicular 11β-hydroxysteroid dehydrogenase (11β-HSD) activity ([grey]) and plasma testosterone (T) levels ([white]) following three days of Smilax calophylla (AD3D) or corticoster-one (CORT3D) treatment The enzyme activity is expressed as mean ± SEM while plasma T levels are expressed as mean ± 95% CI. ٭ p<0.05 compared to the control group(C).
Testicular 11 β -HSD oxidative activity
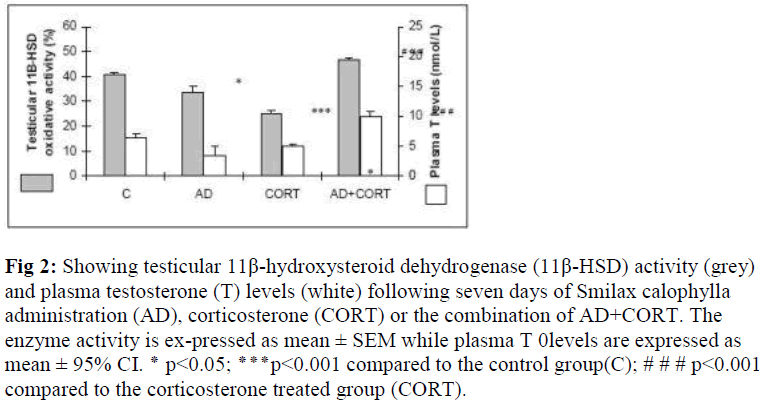
The oxidative activity of 11 β -HSD was significantly decreased (p<0.001) following CORT treatment for seven days compared to that of the control. Similarly, a signifi-cant reduction was noted on the enzyme activity (p<0.05) after seven days of AD (dose: 8mg/kg BW) treatment. However, a concomitant treatment of AD concurrently with CORT increased the 11β -HSD activity (p<0.001) compared to CORT treated group (Fig. 2).
Figure 2: Showing testicular 11β-hydroxysteroid dehydrogenase (11β-HSD) activity (grey) and plasma testosterone (T) levels (white) following seven days of Smilax calophylla administration (AD), corticosterone (CORT) or the combination of AD+CORT. The enzyme activity is ex-pressed as mean ± SEM while plasma T 0levels are expressed as mean ± 95% CI. ٭ p<0.05; ٭٭٭p<0.001 compared to the control group(C); # # # p<0.001 compared to the corticosterone treated group (CORT).
Plasma testosterone levels Corticosterone given for seven days significantly reduced plasma levels of T (p<0.05) compared to that of the control. Plasma levels of T did not differ significantly in the AD-treated rats compared to the control. Akar dawai water extract when given in combination with corticosterone Testicular 11 β -HSD oxidative activity (AD+CORT), a significant increase in plasma T levels was recorded compared to that of the CORT- treated group (p<0.001) (Fig.2).
Discussion
Chronic exposure to high glucocorticoid levels following stress suppresses plasma levels of T [2] either by its direct inhibitory effect on T production or by suppressing luteinizing hormone (LH) levels [26]. In fact, the degree of inhibitory effect of glucocorticoid on the testicular steroidogenesis is determined by the 11β-HSD oxidative activities, intra cellular glucocorticoid concentration and the number of GR [27]. It has been documented that an excess CORT inhibits LH-stimulated steroidogenesis by decreasing the number of LH receptors in the testis. The effect of CORT on T biosynthesis in Leydig cell is found to be a receptor-mediated mechanism [3]. Lowering of plasma levels of T in stressed and CORT-treated rats could also be associated with the reduction in testicular 11β-HSD activities [6]. Previous studies also showed that CORT inhibits the testicular 11β-HSD activity [28] as well as plasma levels of T [9,27]. However, the degree of inhibitory effect of CORT on steroidogenesis increases with the level of suppression of the 11β-HSD activity [11].
Testicular 11β-HSD activities have a significant correla-tion with the plasma levels of T in some treatments where the suppression of 11β-HSD activity leads to a reduction in plasma levels of T [12]. In fact, studies using rat tissues showed that testicular Leydig cells of the rats contain the highest 11β-HSD activity compared to other tissues [23]. Thus, by modulating glucocorticoid concentration in the testis, 11β-HSD is proposed to play a pivotal role in protecting the testis from the deleterious effects of glucocorticoid especially on steroidogenesis [9,11]. We therefore, decided to look only at the oxidative activity of this en-zyme.
The decoction of Akar dawai rhizomes consumed tradi-tionally as a male sexual tonic, claimed to have aphrodisiac effect [14]. Most probably the claimed effect is due to an increase in plasma levels of T. From the AD time and dose dependant graph, it has been found that at the dose of 8mg/kg BW for three consecutive days, AD significantly increases plasma levels of T with no significant change in 11β-HSD activity [21]. In the present study however, AD (8mg/kg BW) for seven consecutive days showed low 11β-HSD activity with no significant change in the plasma levels of T compared to that of the control. Similarly, CORT treatment for seven consecutive days caused a significant reduction on the 11β-HSD activity [28] and plasma levels of T [22] compared to that of the control, unlike which was found when CORT was treated for three consecutive days in either of the parameters studied. Hence, the comparative experiments were carried out after seven days of treatment as the reduction in 11β-HSD activity and plasma levels of T after seven days of CORT treatment, is very much in agreement with the reported findings of others [22]. A multiple reports confirm that high levels of CORT are associated with low testicular 11β-HSD activity and T too [6,29]. Moreover, high dose of CORT (2.5-7.5 mg/100g BW) leads to apoptosis of the Leydig cells [30]. Since CORT fails to alter 11β-HSD activity in adrenalectomized rats testis, it is unlikely that CORT exerts a competitive inhibition on the 11β-HSD activity [6,28].
In the present study, AD was given in combination with CORT for seven days. Interestingly, it was recorded that AD could overcome the suppressive effect of exogenous CORT (2.4mg/kg BW) in rats. The effect could be the result of a competitive interaction between the two on the same receptors in the testis. We, therefore, suggest that AD possibly acts as a partial glucocorticoid receptor ago-nist because AD treatment alone for seven consecutive days also showed the same effect as CORT where it reduced testicular 11β-HSD activity. Based on the above findings, we are tempted to believe that AD might be use-ful in counteracting the adverse effect of stress on normal testicular functioning.
Acknowledgements
We wish to thank Prof. Dr. Johari Mohd. Saad, Faculty of Medicine, University of Malaya for the supply of Smilax calophylla water extract. The present work was supported by IRPA research grant 06-02-02-0081. A critical review of this manuscript by Professor Dr Amar Chatterjee, Fac-ulty of Medicine, UiTM, is highly appreciated.
References
- Cumming DC, Quigley ME, Yen SSC. Acute suppression of circulating testosterone levels in men. Journal of Clinical Endocrinology 1983; 57: 671-673.
- Wingfield JC, Sapolsky RM. Reproduction and resistance to stress: When and how. Journal of Neuroendocrinology. 2003; 15: 711-724.
- Bambino TH, Hsueh AJW. Direct inhibitory effects of glucocoticoids upon testicular luteinizing hormone receptor and steroidogenesis in vivo and in vitro. Endo-crinology. 1981; 108: 2142-2147.
- Fenske M. Role of cortisol in the ACTH-induced suppression of testicular steroidogenesis in guinea pig. Journal of Endocrinology. 1997; 154: 407-141.
- Stalker A, Hermo L, Antakly T. Covalen affinity labeling, radiography and immunochemistry localize the glucocorticoid receptors in rat testicular leydig cells. The American Journal of Anatomy 1989; 186: 369-377.
- Nwe KHH, Norhazlina AW, Hamid A, Morat PB, Khalid BAK. In vivo effects of stress, ACTH and corticosterone on testicular 11β-hydroxysteroid dehydro-genase oxidative activity in rats and the possible mechanism of action. Experimental Clinical Endocrinology Diabetes, 2000a; 106: 109.
- Walker BR, Edwards CRW. 11β-hydroxysteroid dehydrogenase and enzyme mediated receptor protection : Life after liquorice. Clinical Endocrinology. 1991; 35: 281-289.
- Koerner FR. 11β-hydroxysteroid dehydrogenase of lung and testis. Endocrinology.1966; 79 : 935-938.
- Phillips DM, Lakshmi V, Monder C. Corticosteroid 11β-dehydrogenase in rat testis. Endocrinology. 1989; 125 : 209-216.
- Stahl K, Lichtenstein I, Siebe H, Hierholzer K. Interac-tion of 11β- hydroxysteroid-oxido reductase in different organs of various mammalian species. Kidney Interna-tional. 1996; 49: 156-159.
- Ge RS, Gao HB, Nacharaju VL, Gunsalus GL, Hardy MP. Identification of a kinetically distinct activity of 11β-hydroxysteroid dehydrogenase in rat Leydig cells. Endocrinology. 1997; 138: 2435-3442.
- Nwe KHH, Hamid A, Norhazlina AW., Khalid BAK, Morat, P.B. The relationship between plasma testosterone levels and testicular or hepatic 11β- hydroxysteroid dehydrogenase activity in normal rats with various treatments. Journal of Asean Federation of Endocrine Societies. 2000b; 18: 6-13.
- Ge RS, Dong Q, Niu E, Sottas CM, Hardy DO, Catter-all JF, Latif SA, Morris DJ, Hardy MP. 11β-hydroxy-steroid dehydrogenase 2 in rat Leydig cells : Its role in blunting glucocorticoid action at physiological levels of substrate. Endocrinology. 2005; 146: 2657-2664.
- Gimlette JD. A dictionary of Malayan medicine, Ox-ford University Press, Kuala Lumpur. 1971.
- Jia ZHJ, Ju Y. Steroidal saponins from Smilax lebrunii. Phytochemistry. 1992; 31: 3173-3175
- Bernardo RR, Pinto AV, Parente JP. A steroidal saponins from Smilax officinalis. Phytochemistry. 1996; 43: 465-469.
- Lee H, Lin JY. Antimutagenic activity of extract from anticancer drugs in Chinese medicine. Mutation Research. 1988; 204: 229-234.
- Caceres A, Cano O, Samayoa B, Aguilar L. Plants used in Guatemala for the treatment of gastrointestinal disorders.1. Screening of 84 plants against enterobacteria. Journal of Ethnopharmacology. 1990; 30: 55-73.
- Caceres A, Lopez BR, Giron MA, Longemann H. Plants used in Guatemala for the treatment of dermatophytic infections. 1. Screening for antimycotic activity of 44 plants extracts. Journal of Ethnopharmacology. 1991; 31: 263-276.
- Fukunaga T, Miura T, Furuta K, Kato A. Hypoglycemic effect of the rhizomes of Smilax glabra in normal and diabetic mice. Biological & Pharmaceutical Bulle- tin. 1997; 20: 44-46.
- Nuraliza AS, Khatiza HA, Morat PB, Hamid A, Khalid BAK. Effect of Smilax calophylla on testicular 11β-hydroxysteroid dehydrogenase oxidative activity and plasma testosterone levels of rats. Proceeding of The Annual Workshop of National Science Fellowship. 2002; 357-360.
- Nwe KHH, Morat PB, Khalid BAK. Opposite effects of sex steroids on 11β- hydroxysteroid dehydrogenase activity in the normal and adrenalectomized rat testis. General Pharmacology. 1997; 28: 661-664.
- Nwe KHH, Hamid A, Morat PB, Khalid BAK. Differential regulation of the oxidative 11β-hydroxysteroid dehydrogenase activity in testis and liver. Steroids. 2000c; 65: 40-45.
- Lamming GE. Marshall’s Physiology of Reproduction, Fourth Edition,Churchill Livingstone., Longman Gro-up, United Kingdom. 1990.
- Pipkin FB. Medical Statistics Made Easy, Churchill Livingstone, Edinburgh, UK. 1984.
- Maric D, Kostic T, Kovacevic R. Effects of acute and chronic immobilization stress on rat Leydig cell steroidogenesis. Journal Steroid Biochemistry Molecular Bi-ology. 1996; 58: 351-355.
- Hardy MP, Ganjam VK. Stress, 11β-HSD, and Leydig cell function. Journal of Andrology. 1997; 18: 475-479.
- Nwe KHH, Morat PB, Khalid BAK. The effects of corticosteroids on the activity of 11β-hydroxysteroid dehydrogenase in rat testis. The 10th Asia-Oceania Congress of Endocrinology.1994; p 170.
- Sankar BR, Maran RR, Sudha S, Govindarajulu P, Balasubramaniam K. Chronic corticosterone treatment impairs Leydig cell 11β-hydroxysteroid dehydrogenase activity and LH-stimulated testosterone production. Hormone & Metabolic Research. 2000; 32: 142-146.
- Gao HB, Tong MH, Hu YQ, Guo QS, Ge RS, Hardy MP. Glucocorticoid induces apoptosis in rat Leydig cells. Endocrinology.2002; 143: 130-138.