Research Article - Biomedical Research (2017) Volume 28, Issue 8
The curative effect of modified Wuqi decoction against non-steroidal antiinflammatory drugs-induced gastric ulcer by regulating the pathway of COX2/PGE2
Lanting Zou1*, Weimin Lu2 and Liang Zheng11The Second Affiliated Hospital of Nanjing University of Traditional Chinese Medicine, Nanjing, PR China
2The First Affiliated Hospital of Nanjing University of Traditional Chinese Medicine, Nanjing, PR China
- *Corresponding Author:
- Lanting Zou
The Second Affiliated Hospital of Nanjing University of Traditional Chinese Medicine, PR China
Accepted date: December 29, 2016
Abstract
This study aims to observe the curative effect of modified Wuqi decoction (MWQD) on non-steroidal anti-inflammatory drugs-induced gastric ulcer (NSAIDU) and explore the mechanism. 80 NSAIDU subjects were enrolled into the study according to inclusion criteria and divided into the treatment group (CM, n=40) and the control group (CON, n=40) using double-blindly randomized method. The patients in the CM group were administered with Pantoprazole Sodium Enteric-coated Capsules together with MWQD, while the patients in CON group were administered with Pantoprazole Sodium Enteric-coated Capsules and control granules. The treatment lasted for 2 months. All subjects went through Carbon 13 urea breath test to detect Helicobacter Pylori (Hp), and the following procedures were conducted before and after the treatment: (a) the executors of this study blindly filled in the scale assessment of positive symptoms of subjects; (b) 3 ml venous blood was collected for the PGE2 and IL-1 test via ELISA and the expression of TNF-α; (c) the subjects were examined via gastroscopy to observe the gross morphology of gastric mucosa, then the gastric mucosa was received HE staining after pathologic biopsy; and moreover, the COX2 expressions of all the gastric mucosa specimens before and after treatment were tested via immunohistochemistry. The subjects with positive Hp were treated with radical treatment in combination with this therapy protocol. Before treatment, the inflammatory factor, endoscopic grades and gastric biopsy assessment have no significant difference (Ρ>0.05) between CM and CON groups, i.e., they exhibit the comparability. After treatment, the overall effective rates of CM and CON groups are 100% and 90%, respectively; immunohistochemical results illustrate that COX2 protein expression of CM group is higher than that of CON group (Ρ<0.05), while IL-1 and TNF-α expressions are lower than those of CON group (Ρ<0.05); PGE2 expression increases (Ρ<0.05). MWQD is proved to be effective in relieving inflammatory reactions by inhibiting inflammatory factors such as IL-1 and TNF-α, upregulating the expression of PGE2/COX2 to strengthen the protective and self-healing functions of gastric mucosa, thereby enhancing the clinical cure rate and lowering the recurrence rate of NDGU. This study can provide a therapy protocol for long-term Aspirin-applied NSAIDs.
Keywords
Non-steroid anti-inflammatory drugs-induced gastric ulcer (NSAIDU), Modified Wuqi decoction (MWQD), IL-1, TNF-α, PGE2/COX2
Introduction
Non-steroid anti-inflammatory drugs (NSAIDs), known as Aspirin-like drugs or anti-inflammatory drugs, have important therapeutic effects on relieving pain, fever, inflammation and etc. [1]. Like the other drugs, NSAIDs have extensive side effects, especially the toxic and side effects on gastrointestinal tract. Previous studies illustrate that NSAIDs can injure gastric mucosa via inhibiting gastric mucosa prostaglandins, and thus the rick factor of the patents who take NSAIDs is four times higher than that of the patient who does not take these drugs [2]. Therefore, the use of NSAIDs is a common cause of gastrointestinal ulcers, and the resulting ulcer is called NSAIDs-induced ulcer (NSAIDU) [3].
Though some studies prove that the additional use of PPI, prostaglandin analogues and radical elimination of Helicobacter Pylori (Hp) can prevent NSAIDU, some patients with high risk factors such as the patients who have gastric medical histories and received the combined therapy with the anticoagulant and the aged also have relatively high morbidities and hospitalization rates [4]. The use of NSAIDs is the most common cause of peptic ulcer. The number of people who preventatively use NSAIDs is increasing, and approximately 5% of the patients suffer from gastric mucosa injuries after the use of two weeks, i.e., the appearance of erosions or ulcers. The proportion will increase to 10% after continuous use of such therapy for four weeks or longer. Concomitant infection of Hp can increase NSAIDU risk. There exist especially high risks of ulcers for the patients who are older than 60 years, have the previous history of ulcers, use hormones at the same time, take large doses of NSAIDs, or with severe comorbidities [5]. For the patients with severe complications to anticoagulant drugs, such as warfarin, the use of NSAIDU, is likely to cause life-threatening gastrointestinal bleeding. Some patients with gastric mucosa lesion have high perception threshold, and NSAIDU of asymptomotic ulcer is usually observed under endoscope, thereby leading to high risks, for example, bleeding, perforation and other adverse events, etc. However, the patients with cardiovascular or other systemic diseases are more likely to exhibit side effects when stopping the use of NSAIDs drugs. Accordingly, how to shorten ulcer healing time, decrease the hospitalization rate and recurrence rate during the use of NSAIDs becomes an important issue [6,7].
Given that patients with anticoagulants such as warfarin or serious complications, NSAIDU is more likely to cause lifethreatening upper gastrointestinal bleeding. Therefore, the inclusion criteria for clinical research mainly focus on A2 and H1 of patients with gastric ulcer to avoid the risk of serious complications threatening lives. Additionally, because previous studies have found that many NSAIDs patients were asymptomatic, it may be associated with older age or the upregulation of stomach nerve receptor threshold [6]. The conclusion is consistent with patient symptoms scale results that were filled by our group members. Therefore, the symptom criteria were not listed in efficacy criteria, only based on the endoscopic ulcer healing condition. In line with the characteristics of the disease, it was also prompted that, for the initial therapy for the patients with NSAIDs were, even if they were asymptomatic, it should be undergone with endoscopy at the beginning of two weeks of treatment, or began taking PPI, Chinese medicine and other preventive treatment. In addition, the concurrent infection of Hp increased the risk for NSAIDU, and the previous studies regarding whether they can delay ulcer healing process still remain controversial [8,9]. But recent studies have confirmed that Hp infection may exacerbate the gastric mucosal damages to the patients with long-term use of NSAIDs [10]. In this study, carbon 13 urea breath tests were conducted on all the patients to detect Hp, and the Hp-positive patients were treated with anti-Hp therapy to minimize the interference of confounding factors. Finally, whether to disable NSAIDs is still controversial after it appeared. However, recent clinical studies found that after stopping the use, since the patients who are taking these drugs are with severe cardiovascular diseases or other diseases, the risk of suffering from severe complications or even deaths after the stop was significantly higher than that caused by the side effect of the use of NSAIDs on gastrointestinal tracts [11]. Therefore, the patients in the study were continuously administrated with low-dose aspirin, but the longest time not more than 2 months.
Aspirin is one of the earliest synthesized drugs, and is a common oral-administration drug worldwide, as well as the most common OTC. However, the initial anti-inflammatory functions have been replaced by other drugs. Nowadays, aspirin is mainly applied as a major cardiovascular drug worldwide since it can continuously inhibit platelet COX-1 and thus reduce the role of the coagulation. Aspirin was selected as a representative of NSAIDs, mainly because of its user universality and dosage controllability, which can reduce the gastric mucosa injuries caused by drug or dosage difference. As part of the platelet are regenerated by the bone marrow every day, the inhibitory effect of aspirin will be offset, and the minimum daily dose (75 mg, q.d) is required for inhibition of platelet function. In the meantime, the patients with myocardial infarction and other cardiovascular problems can gain the benefits [12]. Epoxidase (COXs) are the targets of NSAIDs drugs [1]. It has two subtypes, COX-1 and COX-2. It was found that COX-1 plays an indispensable role in the regulation of gastric homeostasis, including protection, hemostatic effect and the regulation of inflammatory cells in the gastric stomach cells [13]. The role of COX-2 is more diverse and controversial. Some studies show that COX-2 plays a leading role in the pathogenesis of inflammation and tumor formation, which is the theoretical basis of the selective COX-2 inhibitor development [14]. Although these drugs have been demonstrated to reduce gastrointestinal side effects on the stomach, but other study found that COX-2 knockout rats had more severe inflammation. COX-2 may play a significant role in recognition of inflammation and ulcer healing. In the experiment for which both COX-1 and COX-2 are inhibited, ulcers may appear and it was confirmed that reduction of the overall level of PGE2 is more important than a particular COX enzyme in production inhibition [15-17].
Both IL-1 and TNF-α are proinflammatory cytokines, and the generation and development of IL-1 are controlled by inflammasomes. Under the limited and acute inflammatory conditions, such as gastric mucosal damage caused by NSAIDs drugs stimulation, the macrophages of gastric tissue can release the inflammatory cytokines IL-1 and TNF-α. Modified Wuqi pulvis (MWQD, Astragalus Menbranceus Soil, Dioscorea Oppostita Thunb, Schisandra Chinensis Turcz Baill, Panax Notoginseng, Bletilla Striata, Calcined Cuttlefish Bone, 2 g per one, were grinded into powder and mix well) is made by Schisandra Chinensis Turcz Baill, Panax Notoginseng and the drugs that can strengthen spleen, reinforcing qi, convergence wound and promote tissue regeneration. We found that MWQD can increase the clinical effects of some NSAIDU patients in the clinical application. Related articles also reported that some Chinese traditional medicines can modulate the expression of PGE2/COX2, exhibiting important effects on mucosal cell proliferation [18]. Hence, this study uses random control to observe the therapeutic effects of MWQD so as to illustrate the functions of MWQD in inhibiting IL-1, TNF-α inflammatory factors, upregulating the expression of PGE2/COX2 and thus contribute to the recovery of gastric mucosa. Therefore, based on the clinical observation findings that MWQD can be effective, it is proposed that MWQD may regulate COX-2, thus promoting ulcer healing, increasing PGE2 and inhibiting inflammatory cytokines assumptions IL-1 and TNF-α's.
Patients and Methods
Case selection
A data of 80 subjects were collected from January 2011 to January 2016 in the Out-patient Department of the Second Affiliated Hospital of Nanjing University of Traditional Chinese Medicine University of Traditional Chinese Medicine. All subjects met inclusion criteria and exclusion criteria, and signed the informed consents. By reference to the diagnostic criteria of peptic ulcer on "Lancet" [3], the present inclusion and exclusion criteria are as follows. Inclusion criteria: 1. Male or female patients aged between 18 to 80 years old; 2. Patients who have a history of specific Aspirin drug; 3. Patients who are diagnosed with gastric ulcer or duodenal ulcer through gastroscopy results; 4. Fecal occult blood test (o-methyl benzidine method) was negative; 5. Patients who agree to sign the Informed Consent Form. Exclusion criteria: 1. Patients who disagree to sign the Informed Consent Form; 2. Patients during the pregnancy or lactation period; 3. Patients together with vomiting, melena and other syndromes, or active bleeding observed under the microscope, together with weight loss, in appetence and anemia, etc.; 4. Patients who receive the combined therapy of anticoagulant drugs, hormones and other drugs; 5. Patients suffering from digestive tract tumors or other cancer patients.
The study was approved by the Ethics Committee of Ethics Committee of the Second Affiliated Hospital of Nanjing University of Traditional Chinese Medicine University of Traditional Chinese Medicine, and strictly followed the guide of the Ethics Committee. No adverse drug reactions were observed in the study. According to endoscopy results and medication history, the study members selected the patients who met the inclusion criteria for the study, and then the other members (knowing nothing about the subjects’ specific diseases) divided the subjects into two groups, Chinese medicines (CM) group and the control group (CON), by reviewing the random number table. All included patients underwent endoscopy before the study. No related treatments were applied during gastroscopy examination. The patents who were diagnosed as the gastric ulcer A2 or H1 would be included in the experiment until no malignant lesions were reported in the pathological results of gastric mucosa. Group members filled in the medical history, medication history, symptoms, signs, scales and other related information of all subjects before and after treatment.
Drug preparation
MWQD was from the Department of Pharmacy, the Second Affiliated Hospital of Nanjing University of Traditional Chinese Medicine, with the dosage form of granules; placebo was also from the Pharmacy Department of our hospital. Both two drugs adopted the same packages and unified codes. The codes were under the unified management of the third-party pharmacy staffs, and research members and patients used the blind method. Pantoprazole enteric-coated capsules were purchased from the Pharmacy Department of our hospital, and produced by Shandong Renhetang Pharmaceutical Co., Ltd.
Observation Indexes
Sera diagnosis
Fasting venous bloods were collected before and after treatment (the morning within 24 h). Bloods was allowed to stand at room temperature for 10-20 minutes, centrifuged for 20 min (at the rate of 2000 rpm) and then the supernatants were sampled. The expressions of PGE2, IL-1, TNF-α was detected. Human Elisa kits were purchased from Shanghai Changjin Biological Co., Ltd. The experimental procedure was executed with reference to the instructions of kit. Then, the absorbance values of the sample (OD values) were determined by an ELIASA, and the concentrations of PGE2, IL-1 and TNF-α were calculated through the standard curves.
Protein detection
Expression of COX2 was detected by the immunohistochemistry and COX2 polyclonal antibody was purchased from Wuhan Boster Biological Engineering Co., Ltd, and the immunohistochemical tests followed the instructions of kits. Expressions of COX2 were assessed by referring to the results of immunohistochemical integral in the relevant literatures [19].
Gastric mucous tissues Hp and the pathologic and morphological observations
Gastroscopy was conducted, and two gastric mucosa specimens were selected by the pliers and then sent to the Department of Pathology, the Second Affiliated Hospital of Nanjing, University of Traditional Chinese Medicine Hospital, for pathologic and morphological examinations. One collected by the group members was placed into 4% paraformaldehyde (Guangzhou Jing Xin Biological Technology Co., Ltd., China) to be fixed. After paraffin-embedding, HE staining and immunohistochemistry were conducted on the specimens for the detection of COX2 expression. The same method was adopted after 2 months.
Treatment
Carbon 13 urea breath test was conducted on all the subjects. The patients with positive Hp were given Hp treatment for 10 days [pantoprazole enteric-coated capsules (40 mg qd, ac) + furazolidone (0.1 g, 3-4 times daily) + levofloxacin (0.1 g, 2-3 times daily)]. After the anti-Hp treatment, the patients in the CM group were treated with Pantoprazole enteric-coated capsules and MWQD (10 g, 120-150 milliliter warm boiled water was added, mix up and drink immediately, taken night and morning); and the patients in the CON group were treated with Pantoprazole enteric-coated capsules and control granules. The patients with negative Hp were treated with group treatment. The total therapy time lasted for 2 months. The dosage of Aspirin was consistent as lowest 75 mg q.d. The patients were followed up by the study executors one month after the treatment, and the drug numbers, dosages, symptom improvements and side effects were recorded.
Standard criteria of ulcer therapeutic effect under gastroscope
With reference to the standards in The Clinical Guidance for New Traditional Chinese Medicines (Trials) [20], according to the basic situations of the included experimental subjects, the following therapeutical effect criteria were established. Clinical cure: ulcer was healed up, with no obvious scars; Conspicuous effect: ulcer was healed up, with red scare (S1) or white scars (S2) under gastroscope; Effective: Ulcer was shrunk, with significant H2, or the ulcer was mitigated by a level; Invalid: no significant alleviation under endoscope.
Statistical method
The statistics analysis was conducted using SPSS 17.0, the paired t-test was applied on paired samples, and t-test was applied on two samples according to completely random design. Comparison of rate between two samples was applied using χ2 test, and the measurement data or level data were conducted using rank sum test. Ρ<0.05 is defined as significant difference.
Results
Clinical parameters and Hp infection
After carbon 13 urea breath tests, 43 of 80 subjects were determined to be Hp-positive, who were then included in the experiment after radical Hp routine treatment., the clinical parameters including gender, gastroscopy diagnosis grade, age, etc. were shown in Table 1. There were no significant differences (Ρ>0.05) in various parameters between CM group and CON group.
| Group | N | Gender | EDG | Age (Y) | ||
|---|---|---|---|---|---|---|
| Male | Femal | A2 | H1 | |||
| CM | 40 | 24 | 16 | 23 | 17 | 58.10 ± 6.58 |
| Hp (+) | 25 | 11 | 14 | 15 | 10 | 60.09 ± 5.67 |
| Hp (-) | 15 | 8 | 7 | 6 | 9 | 57.26 ± 4.32 |
| CON | 40 | 21 | 19 | 18 | 22 | 60.05 ± 7.01 |
| Hp (+) | 18 | 10 | 8 | 12 | 6 | 62.07 ± 6.02 |
| Hp (-) | 22 | 11 | 11 | 13 | 9 | 58.05 ± 5.31 |
| 0.457 | 1.251 | 1.283 | ||||
| P | 0.499 | 0.263 | 0.101 | |||
Table 1. Clinical parameter.
Changes of effective rate and gastric mucosa after MWQD intervention
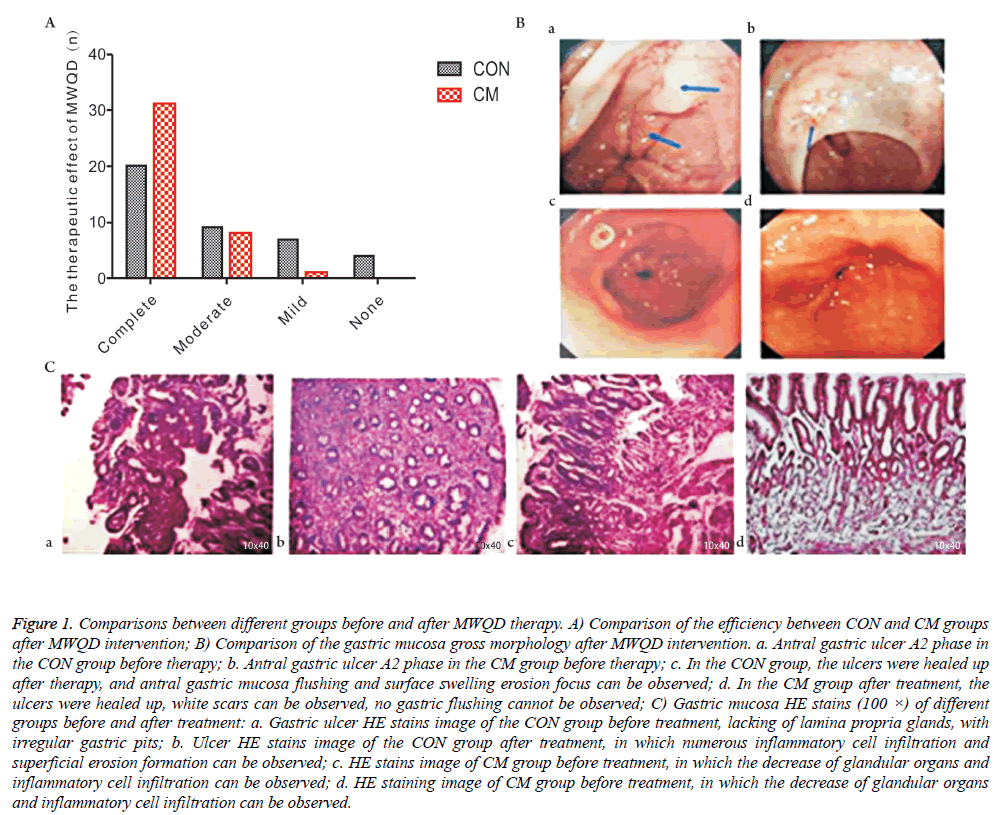
As shown in Figure 1A, based on the mucosa improvement condition under the gastroscope, the gross effective rate of CON group is 90% and the gross effective rate of CM group is 100%. The results suggest that the effective rate of the use of pantoprazole enteric-coated capsules is higher than that of the combined use of MWQD. As shown in Figure 1B, after the addition of MWQD, gastric mucosa inflammation in the CM group was improved compared with the result in the CON group. Though the former ulcers of the subjects in the CON group healed, some of them had erosion focus and swelling red of gastric mucosa after two months continual administration of Aspirin for two months. HE staining results also indicate that, after the additional application of MWQD, the gastric mucosa inflammation of the subjects in the CM group had better improvements that that in the CON group. As shown in Figure 1C, some patients showed surface erosion focus in gastric mucosa.
Figure 1: Comparisons between different groups before and after MWQD therapy. A) Comparison of the efficiency between CON and CM groups after MWQD intervention; B) Comparison of the gastric mucosa gross morphology after MWQD intervention. a. Antral gastric ulcer A2 phase in the CON group before therapy; b. Antral gastric ulcer A2 phase in the CM group before therapy; c. In the CON group, the ulcers were healed up after therapy, and antral gastric mucosa flushing and surface swelling erosion focus can be observed; d. In the CM group after treatment, the ulcers were healed up, white scars can be observed, no gastric flushing cannot be observed; C) Gastric mucosa HE stains (100 ×) of different groups before and after treatment: a. Gastric ulcer HE stains image of the CON group before treatment, lacking of lamina propria glands, with irregular gastric pits; b. Ulcer HE stains image of the CON group after treatment, in which numerous inflammatory cell infiltration and superficial erosion formation can be observed; c. HE stains image of CM group before treatment, in which the decrease of glandular organs and inflammatory cell infiltration can be observed; d. HE staining image of CM group before treatment, in which the decrease of glandular organs and inflammatory cell infiltration can be observed.
Expression of IL-1, TNF-α, PGE2 and COX-2
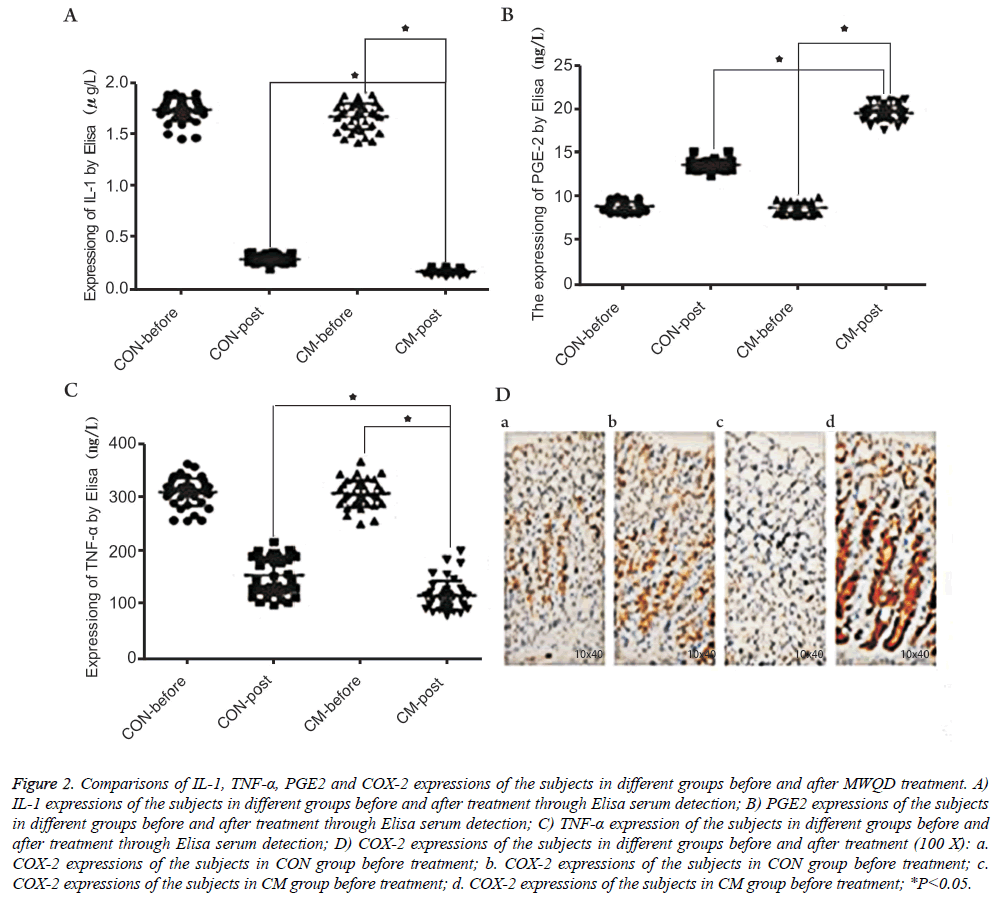
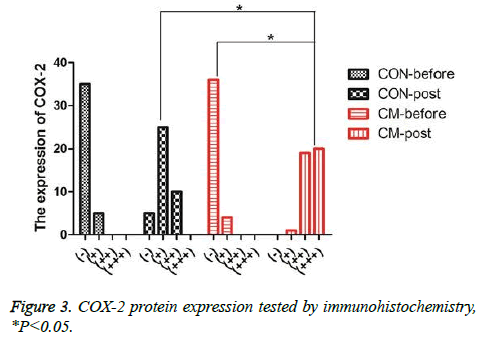
The serums of the patients before and after treatment were selected and then the levels of IL-1, TNF-α and PGE2 were detected using Elisa. The results were shown in Figures 2A-2C. One can observe that, after MWQD application, the levels of IL-1 and TNF-α of the patients in the CM group were significantly reduced (Ρ<0.05), and the values of the patients after two-months reexamination differed in the CM and CON groups. However, PGE2 in CM group was significantly increased (Ρ<0.05) compared with the result in CON group. To further study the gastric mucosa repair function of MWQD, the expression of COX-2 was determined via immunohistochemistry. As shown in Figures 2D and 3, COX-2 was mainly expressed to be strongly-positive in CM group, and to be weekly-positive in CON group. The expressions of COX-2 of two groups showed significant difference (Ρ<0.05).
Figure 2: Comparisons of IL-1, TNF-α, PGE2 and COX-2 expressions of the subjects in different groups before and after MWQD treatment. A) IL-1 expressions of the subjects in different groups before and after treatment through Elisa serum detection; B) PGE2 expressions of the subjects in different groups before and after treatment through Elisa serum detection; C) TNF-α expression of the subjects in different groups before and after treatment through Elisa serum detection; D) COX-2 expressions of the subjects in different groups before and after treatment (100 X): a. COX-2 expressions of the subjects in CON group before treatment; b. COX-2 expressions of the subjects in CON group before treatment; c. COX-2 expressions of the subjects in CM group before treatment; d. COX-2 expressions of the subjects in CM group before treatment; *P<0.05.
Discussion
The results of this study shown that, based on the mucosa improvement conditions under the gastroscope, the gross effective rate of CON group was 90% and the gross effective rate of CM group was 100%. The results suggested that the effective rate under a single use of Pantoprazole Enteric-coated Capsules was lower than that of combined use of MWQD. HE staining results also indicated that, after the additional application of MWQD, the gastric mucosa inflammation of the patients in the CM group had better improvements than that in the CON group. In addition, IL-1, TNF-α, and PGE-2 testing results shown that, after MWQD application, the levels of IL-1 and TNF-α of the subjects in the CM group were significantly reduced, and the values of the subjects after two-months reexamination differed in the CM and CON groups, indicating the MWQD’s functions in lowering inflammatory factor. However, PGE2 in CM group was significantly increased compared with the result in CON group, suggesting that MWQD can improve the healing of gastric ulcer and protects gastric mucosa by highering PGE2 expression. The further study of the gastric mucosa repair function of MWQD was shown that, COX-2 was mainly expressed to be stronglypositive in CM group, and to be weekly-positive in CON group. The expressions of COX-2 of two groups showed significant difference.
Furthermore, the study showed that two indicators of the patients were higher before treatment. The gastric mucosa ulcers were formed under gastroscopy, and a bunch of inflammatory cell infiltration was visible after HE staining. Decrease of inflammatory cytokines is higher (Ρ<0.05) in the CM group than that in the CON group after treatment. Although some patients' ulcers were healed up after endoscopic treatment in CON group, gastric inflammation still existed, even the superficial erosion were formed, while the CM group has no significant inflammatory infiltrates, indicating the anti-inflammatory effects of MWQD. Generally, it is considered that PGE2 can inhibit gastric acid secretion and promote gastric mucosal blood flow restore so that it can improve the self-repair of the damaged gastric mucosa [21]. In this study, average the increase of PGE2 level in the two groups after treatment, the result of CM group is significantly higher than that of CON group (Ρ<0.05), suggesting MWQD can promote the synthesis of PGE2 and thus contribute to gastric ulcer healing and mucosal recovery.
Previous studies have shown that NSAID is mainly caused by COX inhibiting PGE2-mediated synthesis [22,23]. To further study the mechanism of MWQD, COX2 expressions of objects were detected using immunohistochemical methods. Similarly, it is consistent with the PGE2 expression trend. After the addition of MWQD, COX2 expression was higher in CM group than that in CON group (Ρ<0.05). Therefore, MWQD may inhibit the expression of inflammatory cytokines, and thus increase PGE2/COX2 expression to enhance the gastric mucosal protection and self-healing capabilities. The former studies confirmed that inflammatory cytokines IL-1 and TNF-α may promote the synthesis of PGE2/COX2, i.e., the three trends were positively correlated, and play an important role in the formation of oral cancer [18]. In the present study, the inflammatory cytokines was inhibited, and PGE2/COX2 expressions were increased to promote ulcer recovery. It may be related to two-way modulation of MWQD. The specific mechanism is expected to be studied in further research.
Conclusion
In conclusion, this study confirmed that the joint use of aspirin and MWQD can significantly improve the clinical effective rates of NSAIDU, reduce repetitive damages of NSAIDs on the gastric mucosa, reduce inflammation and promote healing of ulcers, which can provide a treatment direction for long-term use of low doses of NSAIDS such as aspirin. However, due to the limitations on the number of clinical cases and the scarcity of clinical specimens, we failed to have in-depth progress in the mechanism, and the molecular mechanisms of MWQD will be the focus in further studies.
Acknowledgements
The authors would like to thank the financial support of science and technology project of Jiangsu Provincial Administration of traditional Chinese Medicine (YB2015053).
References
- Vane JR. Inhibition of prostaglandin synthesis as a mechanism of action for aspirin-like drugs. Nat New Biol 1971; 231: 232-235.
- Wallace JL. Prostaglandins, NSAIDs, and gastric mucosal protection: why doesn't the stomach digest itself? Physiol Rev 2008; 88: 1547-1565.
- Malfertheiner P, Chan FK, McColl KE. Peptic ulcer disease. Lancet 2009; 374: 1449-1461.
- Lanza FL, Chan FK, Quigley EM; Practice Parameters Committee of the American College of Gastroenterology. Guidelines for prevention of NSAID-related ulcer complications. Am J Gastroenterol 2009; 104: 728-738.
- Laine L. Proton pump inhibitor co-therapy with nonsteroidal anti-inflammatory drugs-nice or necessary? Rev Gastroenterol Disord 2004; 4: S33-41.
- Holtmann G, Gschossmann J, Buenger L, Gerken G, Talley NJ. Do changes in visceral sensory function determine the development of dyspepsia during treatment with aspirin? Gastroenterology 2002; 123: 1451-1458.
- Hsu PI, Tsai TJ. Epidemiology of Upper Gastrointestinal Damage Associated with Low-Dose Aspirin. Curr Pharm Des 2015; 21: 5049-5055.
- Lai KC, Lau CS, Lp WY, Wong BC, Hui WM, Hu WH, Wong RW, Lam SK. Effect of treatment of Helicobacter pylori on the prevention of gastroduodenal ulcers in patients receiving long-term NSAIDs: a double-blind, placebo-controlled trial. Aliment Pharmacol Ther 2003; 17: 799-805.
- Hawkey CJ, Tulassay Z, Szczepanski L. Randomised controlled trial of Helicobacter pylori eradication in patients on non-steroidal anti-inflammatory drugs: HELP NSAIDs study. Helicobacter Eradication for Lesion Prevention. Lancet 1998; 352: 1016-1021.
- Kono Y, Okada H, Takenaka R. Does Helicobacter pylori Exacerbate Gastric Mucosal Injury in Users of Nonsteroidal Anti-Inflammatory Drugs? A Multicenter, Retrospective, Case-Control Study. Gut Liver 2016; 10: 69-75.
- Sung JJ, Lau JY, Ching JY, Wu JC, Lee YT. Continuation of low-dose aspirin therapy in peptic ulcer bleeding: a randomized trial. Ann Intern Med 2010; 152: 1-9.
- Hayden M, Pignone M, Phillips C, Mulrow C. Aspirin for the primary prevention of cardiovascular events: a summary of the evidence for the U.S. Preventive Services Task Force. Ann Intern Med 2002; 136: 161-172.
- Rouzer CA, Marnett LJ. Cyclooxygenases: structural and functional insights. J Lipid Res 2009; 50: S29-34.
- Masferrer JL, Zweifel BS, Manning PT, Hauser SD, Leahy KM. Selective inhibition of inducible cyclooxygenase 2 in vivo is antiinflammatory and nonulcerogenic. Proc Natl Acad Sci U S A 1994; 91: 3228-3232.
- Laugenbach R, Loftin C, Lee C, Tiano H. Cyclooxygenase knockout mice: models for elucidating isoform-specific functions. Biochem Pharmacol 1999; 58: 1237-1246.
- Masferrer JL, Zweifel BS, Manning PT, Hauser SD, Leahy KM. Selective inhibition of inducible cyclooxygenase 2 in vivo is antiinflammatory and nonulcerogenic. Proc Natl Acad Sci U S A 1994; 91: 3228-3232.
- Choi SH, Langenbach R, Bosetti F. Genetic deletion or pharmacological inhibition of cyclooxygenase-1 attenuate lipopolysaccharide-induced inflammatory response and brain injury. FASEB J 2008; 22: 1491-1501.
- Chang LY, Wan HC, Lai YL, Chou IC, Chen YT, Hung SL. Areca nut extracts increased the expression of cyclooxygenase-2, prostaglandin E2 and interleukin-1alpha in human immune cells via oxidative stress. Arch Oral Biol 2013; 58: 1523-1531.
- Zhao X, Dou W, He L, Liang S, Tie J, Liu C, Li T, Mo P, Shi Y. MicroRNA-7 functions as an anti-metastatic microRNA in gastric cancer by targeting insulin-like growth factor-1 receptor. Oncogene 2013; 32: 1363-1372.
- Zheng XY. Guidelines for the research of new clinical drugs of Chinesemedicine(trial implementation). Beijing: China Medical Science Press 2002; 233-237.
- Gyires K, Németh J, Zádori ZS. Gastric mucosal protection and central nervous system. Curr Pharm Des 2013; 19: 34-39.
- Whittle BJ. Gastrointestinal effects of nonsteroidal anti-inflammatory drugs. Fundam Clin Pharmacol 2003; 17: 301-313.
- Lamarque D. Pathogenesis of gastroduodenal lesions induced by non-steroidal anti-inflammatory drugs. Gastroenterol Clin Biol 2004; 28: C18-26.