Research Article - Biomedical Research (2016) Volume 27, Issue 4
Screening and analysis of anti-A431 cell proliferative fraction of Euphorbia fischeriana Steud. extract
Jiawen Wu1*, Hong Sun2, Jia Huo1, Yuan Wang1 and Jing Li1
1Department of Dermatology, The Second Hospital of Xi’an Jiaotong University, Xi’an 710004, Shaanxi, People’s Republic of PR China
2Department of Neurology, The Second Hospital of Xi’an Jiaotong University, Xi’an, 710004, Shaanxi, People’s Republic of PR China
- *Corresponding Author:
- Jiawen Wu
Department of Dermatology, The Second Hospital of Xi’an Jiaotong University, PR China
Accepted on April 14, 2016
Abstract
To investigate the anti-human epidermal squamous-cell carcinoma A431 cell proliferative fraction of Euphorbia fischeriana Steud. and to determine chamaejasmine B content in the fraction. Anti-A431 cell proliferative activity of various Euphorbia fischeriana Steud. fractions, which were extracted using ethanol extraction and with different solvents, were screened by MTT assay to identify the active fraction. Chamaejasmine B content in the active fraction was determined by HPLC. Ethyl ether fraction showed more pronounced anti-proliferation activity on A431 cells. Ethyl ether fraction was the active fraction, in which chamaejasmine b content was 30.6%. IC50 of ethyl ether fraction of Euphorbia fischeriana Steud. for A43l cells at 48 h was 40 μmol/L. At this dose, A431 cell growth inhibition rate was (53.2 ± 0.31%). Ethyl ether extract is the anti-A431 cell proliferative fraction of Euphorbia fischeriana Steud. HPLC determination of chamaejasmine B content in active fraction is an accurate, reproducible and convenient method. We preliminarily conclude that the higher the chamaejasmine B content in the active fraction, the stronger its anti-proliferative effect on A431 cells.
Keywords
Euphorbia fischeriana Steud, Activity screening, A431 cell, HPLC.
Introduction
Chinese drug Langdu Daji is the dried root of Euphorbia fischeriana Steud. in the genus Euphorbia of the family Euphorbiaceae. According to the records of past dynasties, Euphorbia fischeriana Steud. is bitter, pungent, neutral in nature and toxic, with edema-relieving, phlegm-eliminating, stagnation-dispersing and insecticidal functions. Further research at home and abroad in recent years has shown that Euphorbia fischeriana Steud. contains multiple types of constituents, such as diterpenoids, triterpenoids, sterols and phenolic acids [1-3]. The herb can be used for treating lymph node tuberculosis, bone tuberculosis, skin tuberculosis, psoriasis, neurodermatitis, chronic bronchitis, vaginal trichomoniasis, etc. [4-6]. Euphorbia fischeriana Steud. has been found to have activity against a variety of cancer cells. However, its anti-proliferative action on A431 cells has never been reported. This study discovers the active anti-A431 cell proliferative fraction of Euphorbia fischeriana Steud. and determines the content of chamaejasmine B, its major constituent. In this study, combined natural medicinal chemical and pharmacological means are employed. Anti-A431 cell proliferative fraction of Euphorbia fischeriana Steud. is screened. Meanwhile, HPLC is used to determine chamaejasmine B content in the active fraction. Human squamous-cell carcinoma A431 cells are selected in this study for investigating the growth inhibitory action and mechanisms of Euphorbia fischeriana Steud., thereby providing basic research evidence for the clinical treatment of skin cancer.
Materials and Methods
Instruments, drugs and reagents
Euphorbia fischeriana Steud. medicinal material (collected in autumn at Qianshan area in Anshan, Liaoning and identified by associate professor Wang Ping at the Pharmacy College of Changchun University of Chinese Medicine as Euphorbia fischeriana Steud.); chamaejasmine B reference (National Institute for the Control of Pharmaceutical and Biological Products, batch number 0726-2542109). DMEM medium was purchased from Gibco, USA; FBS was purchased from Yidelai Company in Nanjing; ART was prepared into a 60 ms/ml stock solution with 5% sodium bicarbonate and stored at -20ºC, which was diluted to the desired concentration with medium before use, final concentration of sodium bicarbonate <0.5% (Kingyork Pharmaceutical Company in Tianjin); DDP, trypan blue, MTT, DMSO, PI, RNase A and Deferoxamine (DFOM) (Sigma); AnnexinV-FITC apoptosis detection kit was purchased from Yaomei Biotechnology Co., Ltd. in Dalian. Microplate reader (model 680, BIO-RAD); flow cytometer (BD-FAC-SCalibur, USA).
Preparation of Euphorbia fischeriana Steud. extract
Euphorbia fischeriana Steud. medicinal material (600 g) was crushed, extracted under heating reflux for 2 h with 2 L of 95% ethanol 3 times, and then filtered while hot. Filtrates were combined, concentrated under reduced pressure until the volume was about 100 ml, then added with an appropriate volume of water and let stand to complete the precipitation. After filtration, precipitate was taken, dried under reduced pressure, added with an appropriate amount of acetone and Soxhlet extracted until colourless. Afterwards, acetone extract was concentrated, dried under reduced pressure, then again Soxhlet extracted with ethyl ether until colourless. Ethyl ether extracted solution was concentrated and dried under reduced pressure to give the ethyl ether extract (fraction I). Meanwhile, the insoluble after ethyl ether extraction was used as acetone extract (fraction II).
Experiments on anti-proliferative activity of Euphorbia fischeriana Steud. extract on human epidermal squamous-cell carcinoma A431 cells [7,8]
Cell culturing: Human epidermal squamous-cell carcinoma A431 cells were provided by Tianhui Biological Products Co. Ltd., which were cultured at 37°c, 5% CO2 conditions. Medium was replaced and cells were passaged once every 2-3 days or harvested for subsequent experiments.
Growth inhibitory activities of Euphorbia fischeriana Steud. extracts A and B on A431 cells
Logarithmic phase A431 cells were seeded in 96-well plates at 5.0 × 104/ml and 100 μL/well. After adherence, cells were cultured synchronously in serum-free DMEM medium for 24 h. Each well was added with 100 μL of DMEM medium containing different concentrations of extracts to make the final extract concentrations be 5, 10, 20, 40, 80 and 160 mg/L. Meanwhile, a control cell group was set up. Each concentration group consisted of 8 replicate wells. After culturing for 48 h, 5 mg/ml MT (20 μL/well) was added and culturing was continued for an additional 4 h. Then, supernatant was discarded, DMSO was added at 150 μl/well, and plates were shaken for 10 min. After complete dissolution of formazan crystals, optical density at 570 nm [D (570)] was measured with a micro plate reader. The experiment was repeated three times.
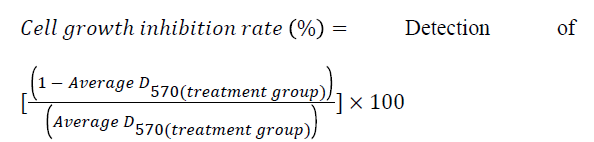
growth inhibitory effect of DDP on A431 cells
Logarithmic phase A431 cells were seeded in 96-well plates. Final concentrations of DDP were set as 2, 4, 6, 8, 10, 20 and 30 μmol/L, respectively. A control cell group was set up and detection was performed after culturing for 48 h. Experiment was repeated three times according to the procedures. "Growth inhibitory activities of Euphorbia fischeriana Steud. extracts A and B on A431 cells".
Detection of effect of DFOM partially blocked Euphorbia fischeriana Steud. extract A on A431 cells
Logarithmic phase A431 cells were seeded in 96-well plates, added with DMEM medium containing DFOM (final concentration 60 μmol/L) at 100 μl/well and preconditioned for 4 h to remove DFOM. Then, extracts with final concentrations of 5, 10, 20, 40, 80 and 160 mg/L were added. Detection was performed after culturing for 48 h and a control cell group was set up. Experiment was repeated three times according to the procedures. "Growth inhibitory activities of Euphorbia fischeriana Steud. extracts A and B on A431 cells".
Optical microscopic observation of morphological changes in A43l cells after treatment with extracts
According to MTT results, IC50 of Euphorbia fischeriana Steud. extract A for A431 cells at 48 h was 40 mg/L, while the safe concentration of DFOM for A431 cells at 4 h was 60 μmol/L. All treatment groups were treated with 40 mg/L extract for 48 h, while all treatment block groups were preconditioned with 60 μmol/L DFOM for 4 h before treating with 40 mg/L extract for 48 h.
Logarithmic phase A431 cells were seeded in 6-well plates. Treatment group, treatment block group and control cell group were set up. Cells in each group were observed for morphological changes by optical microscopy and counted by trypan blue staining. The experiment was repeated three times.
Flow cytometric detection of cell cycle
Logarithmic phase A431 cells were seeded in 100 ml flasks. Treatment group, treatment block group and control cell group were set up. After culturing for 48 h, cells were collected into 1.5 ml EP tubes, washed twice with PBS, fixed in 1 ml of 4 precooled 70% ethanol, stained with PI (final concentration 50 μg/m1) and incubated in the dark for 30 min, followed by detection of cell cycle with a flow cytometer. The experiment was repeated three times.
Flow cytometric detection of apoptosis
Logarithmic phase A431 cells were seeded in 100 ml flasks. Treatment group, treatment block group and control cell group were set up. After culturing for 48 h, cells in each group were collected, washed with PBS, added with 200 μl of reaction binding buffer, 10 μl of PI and 5 μl of AnnexinV-FITC, then incubated in the dark at room temperature for 10 min, followed by detection of apoptosis with a flow cytometer. The experiment was repeated three times.
Statistical processing
ANOVA was performed using SPSS 10.0 statistical software.
Results
Active fraction of Euphorbia fischeriana Steud. Extracts
MTT assay of growth inhibitory effects of Euphorbia fischeriana Steud. extracts on A431 cells
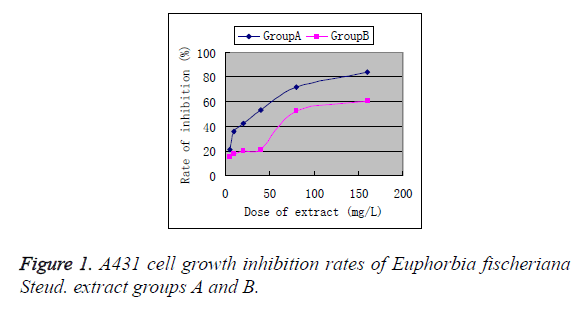
Euphorbia fischeriana Steud. extract groups A and B had final concentrations of 5, 10, 20, 40, 80 and 160 mg/L, respectively. After treating A431 cells for 48 h, MTT results were as follows: IC50 of Euphorbia fischeriana Steud. extract group A for A431 cells was 40 mg/L, while the IC50 of Euphorbia fischeriana Steud. extract group B for A431 cells was 100 mg/L. Within a 5 ~ 40 mg/L low dose range, Euphorbia fischeriana Steud. extract group A had significantly stronger growth inhibitory effect on A431 cells than the normal control cells at the same doses (P<0.01). Furthermore, the A431 cell growth inhibitory effect of Euphorbia fischeriana Steud. extract group A exhibited dose dependence (Figure 1).
Growth inhibitory effects of DFOM partially blocked Euphorbia fischeriana Steud. extract group A on A431 cells
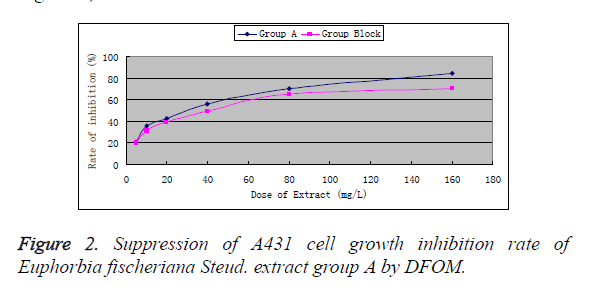
Based on the above results, Euphorbia fischeriana Steud. extract group A was found to have stronger inhibitory effect on A431 cells, so we only studied this group in the following. After preconditioning with DFOM (60 μmol/L) for 4 h and culturing for 48 h, the absorbance was (1.521 ~ 0.209), which showed no significant difference (P>0.05) from that of the drug-free control cell group (1.611 ± 0.239). This indicated that 60 μmoL/L was a safe dose of DFOM. 60 μmol/L DFOM significantly reduced the growth inhibitory effect of Euphorbia fischeriana Steud. extract group A on A431 cells (P<0.01, Figure 2).
Optical microscopic observation of morphological changes in A43l cells after treatment with Euphorbia fischeriana Steud. extract group A
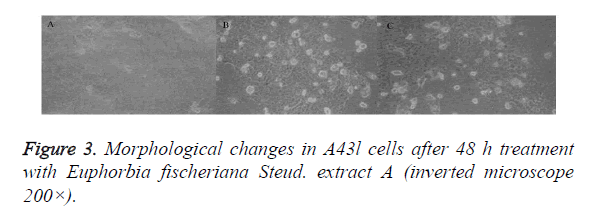
A431 cells in the control group were rounded and full, with good refraction, clear outline and tight intercellular junction. Besides, cell count by trypan blue staining was (2.32 ± 0.12) × 106/ml. In the Euphorbia fischeriana Steud. extract group A, A431 cells became smaller and pseudopodia disappeared. Viable cell count was (1.02 ± 0.09) × 106/ml. Cells were irregularly shaped, with loose intercellular junction, increased secretions and increased number of deaths.
DFOM could partly block the growth inhibitory effect of Euphorbia fischeriana Steud. extract A on A431 cells. In the treatment block group, A431 cells presented insignificant intercellular junction, but were irregular in shape. Viable cell count by trypan blue staining was (1.34 ± 0.12) × 106/ml, which differed significantly from that of the treatment group (P<0.01) (Figure 3).
Flow cytometric detection of A43l cell cycle arrest by Euphorbia fischeriana Steud. extract A
Euphorbia fischeriana Steud. extract A blocked A431 cells in S phase, thus resulting in arrest of A431 cell growth. This finding was consistent with the MTT assay and morphological change results (Table 1).
| Group | G0/G1 | S | G2/M |
|---|---|---|---|
| Control group | 54.21 ± 2.12 | 32.21 ± 2.01 | 13.58 ± 0.63 |
| Treatment group | 29.21 ± 2.90* | 66.54 ± 0.09* | 4.26 ± 1.49 |
Table 1: Flow cytometric detection of effect of Euphorbia fischeriana
Steud. extract A on A431 cell cycle (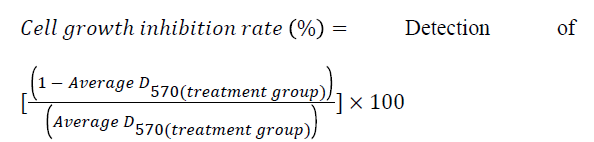 ± s).
± s).
Flow cytometric detection of A43l cell apoptosis induced by Euphorbia fischeriana Steud. extract A
A431 cell apoptosis rate in the treatment group (19.52 ± 2.1) % was significantly higher than the control group [(2.63 ± 2.01) %, P<0.01]. A431 cell apoptosis rate in the treatment block group was (6.98 ± 1.6) %, which was significantly lower than the treatment group (P<0.01), while having no significant difference compared with the control group (P>0.05).
Determination of chamaejasmine B content in Euphorbia fischeriana Steud. extract A [9,10]
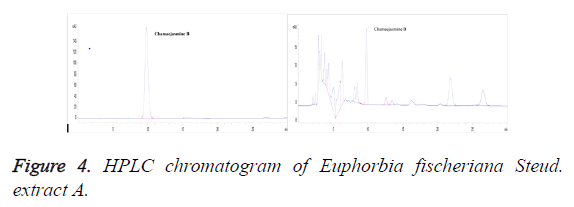
Chromatographic conditions: Column: C18 (250 mm × 4.6 mm, 5 μm; Elite Instrument Co., Ltd. in Dalian); column temperature: 30°c; mobile phase: methanol-water (68:32); flow rate: 1.0 ml/min; detection wavelength: 254 nm; injection volume: 20 μl; number of theoretical plates: should not be less than 2,000 calculated based on chamaejasmine B; resolution from impurity peak: should be greater than 1.5 (Figure 4).
Preparation of reference solution: About 12 mg of chamaejasmine B reference was accurately weighed, placed in a 50 ml volumetric flask, dissolved and diluted to the mark with methanol and shaken uniformly (240 μg/ml). Then, 5 ml of the solution was accurately drawn, placed in a 50 ml volumetric flask, dissolved and diluted to the mark with methanol and shaken uniformly to serve as the reference solution (24 μg/ml).
Preparation of test solution: About 7 mg of Euphorbia fischeriana Steud. extract A powder was accurately weighed, added precisely with 100 ml of methanol, weighed, shaken well and ultrasonicated for 20 min, then removed, let cool and weighed. After replenishing the weight loss with methanol, the solution was filtrated through millipore membrane (0.45 μm) to give the test solution.
Linearity investigation: 2.0, 4.0, 6.0, 8.0 and 10.0 ml of the above reference solution (240 μg/ml) were accurately drawn, placed separately in 50 ml volumetric flasks, dissolved and diluted to the mark with methanol and shaken well to give chamaejasmine B solutions with five different concentrations of 9.6, 19.2, 28.8, 38.4 and 48 μg/ml. Each 20 μl of the above solutions was accurately drawn and injected for determination under the above chromatographic conditions, and then chromatograms were recorded. Regression was performed with peak area (Y) versus concentration (X) to obtain the equation:
Y=83312.13, X=2859.6, r = 0.9998. The result showed good linearity of chamaejasmine B within a 9.6 ~ 48 μg/ml concentration range.
Accuracy test: 20 μl of reference solution (24 μg/ml) was accurately drawn and injected for 5 consecutive times for investigation of accuracy in terms of peak area, RSD=0.54%.
Stability test: The same sample solution was determined in parallel five times at an interval of 1 ~ 2 h for calculation of sample content, RSD=0.78%. The result showed that the sample was stable within 8 h.
Reproducibility test: Five aliquots of Euphorbia fischeriana Steud. extract A were prepared as per the method for preparation of test solution and determined separately. RSD=0.88% in terms of sample content, indicating good reproducibility.
Recovery test: Five aliquots of 5 mg of Euphorbia fischeriana Steud. extract A with known content were accurately weighed, added separately with an appropriate amount of chamaejasmine B reference, prepared as per the method for preparation of reference solution and measured for peak area under the above chromatographic conditions. The results are shown in Table 2.
| Sampling amount (mg) | Sample content (mg) | Amount added (mg) | Measured amount (mg) | Recovery (%) | Average (%) | RSD(%) |
|---|---|---|---|---|---|---|
| 5.01 | 1.53306 | 1.5 | 3.03 | 102.00 | 102.00 | 2.28 |
| 5.07 | 1.55142 | 1.5 | 3.03 | 102.00 | ||
| 5.12 | 1.56672 | 1.5 | 3.04 | 102.67 | ||
| 4.99 | 1.52694 | 1.5 | 3.03 | 102.00 | ||
| 4.32 | 1.62792 | 1.5 | 3.11 | 107.33 |
Table 2. Recovery test results.
Sample content determination: Samples from three different batches were taken, sample and reference solutions were prepared as per the prescribed methods and chamaejasmine B contents were determined separately. The results are shown in Table 3.
| Extract number | Average content (%) | RSD (%) |
|---|---|---|
| 20150506 | 30.6 | 2.12 |
| 20150507 | 26.7 | 1.86 |
| 20150508 | 32.1 | 2.43 |
Table 3. Determination results of chamaejasmine B content in three batches of extracts (n=4).
Discussion
Euphorbia fischeriana Steud. can treat oedema, abdominal distension, phlegm accumulation, scrofula, bone tuberculosis, skin scabies, and malignant sore and vaginal trichomoniasis. In Mongolian medicine, the herb is used for treating "sticky" symptom, anthrax, laryngeal prominence, oedema, carbuncles, scabies, ringworm, impetigo, gout, joint pain and fever. Recent studies have shown that drugs from Euphorbia fischeriana Steud. have cytotoxicity against a variety of tumour cell lines [11-13]. Euphorbia fischeriana Steud.'s effect on skin squamous-cell carcinoma has little been reported so far. Results of the present study show that the anti-proliferative action of Euphorbia fischeriana Steud. extract A on human epidermal squamous-cell carcinoma cell line A431 is achieved mainly by arresting cells in S phase and inducing apoptosis, thereby exerting specific cytotoxicity. Experimental results revealed that compared to DDP, Euphorbia fischeriana Steud. extract A may be more beneficial in the treatment of relevant clinical diseases as an antineoplastic with highly specific cytotoxicity and small side effects.
This study finds that Euphorbia fischeriana Steud. extract A not only can evidently inhibit the proliferation of A431 cells, but can also promote their apoptosis. To investigate this mechanism of action of Euphorbia fischeriana Steud. extract A, we reduce high Fe2+ concentration in A431 cells by DFOM preconditioning, and find decreased A43l cell apoptosis rate caused by Euphorbia fischeriana Steud. extract A, suggesting possible association between its induction of apoptosis and intracellular Fe2+ mediation. This is consistent with the finding that Fe2+ down-regulation reduced apoptosis reported in the literature [14]. Research has shown that cancer cells have exuberant nucleic acid metabolism which require large amounts of Fe2+; highly dense transferrin receptors are present on the surface of most tumour cells, while normal cells have less transferrin receptors; therefore, A431 cells can absorb large amounts of Fe2+ via the surface transferrin receptors [15]. When Euphorbia fischeriana Steud. extract A enters A431 cells and reacts with Fe2+, it can produce free radicals while decreasing the intracellular Fe2+ and reducing DNA synthesis to exert the tumour cell killing and inhibiting effects. Meanwhile, experimental data show that DFOM cannot completely block the apoptotic effect of Euphorbia fischeriana Steud. extract A on A431 cells, suggesting that the extract may induce A431 cell apoptosis through other mechanisms as well.
The present study finds that after treatment with Euphorbia fischeriana Steud. extract A, A43l cells are arrested in S phase, that is, S phase cells accumulate largely, which is probably associated with Akt-NF-κB, NF-κB and (VHL)/(HIF-1α) signalling pathways [16-18]. Whether the increase in S phase cells is related to the anti-proliferative activity of Euphorbia fischeriana Steud. extract A needs experimental verification.
This study establishes a method for determining chamaejasmine B content in Euphorbia fischeriana Steud. extract A. The method can provide the basis for quality control of herb extracts, which is of positive significance to the extraction process of Chinese drugs.
Acknowledgements
This work is supported in part by a grant from the National Natural Science Foundation of China (NSFC, Grant No. 81502749).
References
- Wang M, Wang QH, Wei Q, Lia J, Guo C, Yang BY, Kuang HX. Two new ent-atisanes from the root of Euphorbia fischerianaSteud.Natural Product Research: Formerly Natural Product Letters 2016; 30: 144-149.
- Pei YH, Han B, Jia ZH, Nikaido T, Koike K.Fischeria A. A novel norditerpene lactone from Euphorbia fischeriana.Tetrahedron Letters 1999; 40: 951-952.
- Uto T, Qin GW, Morinag O, Shoyama Y. 17-Hydroxy-jolkinolide B. Aditerpenoid from Euphorbia fischeriana, inhibits inflammatory mediators but activates heme oxygenase-1 expression in lipopolysaccharide-stimulated murine macrophages.International Immunopharmacology 2012; 12: 101-109.
- WangJH, ZhangK, NiuHY, ShuLH, YueDM, LiDY, HeP. Jolkinolide B from Euphorbia fischerianaSteud induces in human leukemic cells apoptosis via JAK2/STAT3 pathways. Int J ClinPharmacolTher 2013; 51: 170-178.
- Sun YX, Liu JC. ChemInform Abstract: Chemical Constituents and Biological Activities of Euphorbia fischerianaSteud. ChemInform 2011; 8: 1205-1214.
- Wang JH, Zhou YJ, BaiX, He P. Jolkinolide B from Euphorbia fischerianaSteud induces apoptosis in human leukemic U937 cells through PI3K/Akt and XIAP pathways. Molecules and Cells 2011; 32: 451-457.
- Kamathama S, Kumarb N, Gudipallic P. Isolation and characterization of gallic acid and methyl gallate from the seed coats of GivotiarottleriformisGriff. and their anti-proliferative effect on human epidermoid carcinoma A431 cells. Toxicology Reports 2015; 2: 520-529.
- Rajendran I, Dhandapani H, Anantanarayanan R, Rajaram R. Apigenin mediated gold nanoparticle synthesis and their anti-cancer effect on human epidermoid carcinoma (A431) cells. RSC Advances 2015; 5: 51055-51066.
- Qiao CF, Han QB, He ZD, Song JZ, Xu HX. HPLC determination of jolkinolide A and jolkinolide B inChinese herb medicine“Lang-du”. Chinese Journal of Pharmaceutical Analysis 2006; 26: 1204-1206.
- Li Y, Liu XL, Cai ZG, Zhang SX. LC-ESI-MS/MS analysis and pharmacokinetics of jolkinolide B, a potential antitumor active component isolated from Euphorbia fischeriana, in rat plasma. Biomedical Chromatography 2014; 28: 193-196.
- Liu YX, Wang MZ, Sun XF. Clinical analysis of 72 epileptic patients treated with alkaline extract of Euphorbia fisheriana. ZhongguoZhong Xi Yi Jie He ZaZhi 1994; 14: 282-284: 261.
- Wang Y, Ma XQ, Yan SS, Shen SS, Zhu HL, Gu Y, Wang HB, Qin GW, Yu Q. 17-Hydroxy-jolkinolide B Inhibits Signal Transducers and Activators of Transcription 3 Signalling by Covalently Cross-Linking Janus Kinases and Induces Apoptosis of Human Cancer Cells. Cancer Research 2009; 69: 7302-7307.
- Yan SS, Li Y, Wang Y, Shen SS, Gu Y, Wang HB, Qin GW, Yu Q. 17-Acetoxyjolkinolide B irreversibly inhibits IκB kinase and induces apoptosis of tumour cells. Molecular Cancer Therapeutics 2008; 7: 1523-1532.
- Warren G, Davoust J, Cockeroft A. Recycling of transferrin receptors in A431 cellsisinhibited duringmitosis. EMBO J 1984; 3: 2217-2225.
- Singh NP, Lai H. Selective toxicity of dihydroartemisinin and holotransferrin toward human breast cells. Life Sci 2001; 70: 49-56.
- Pan XY, Zhang MJ, Zeng XY, Lin J, Li L, Li M, Zhao W. Euphorbia fischeriana extract reactivates latent HIV through nuclear factor-κB pathway. Journal of Southern Medical University 2015; 11: 1614-1618.
- Cui HX, Wang M, Yuan JX, Liu JC. Effect of ethyl gallate on human breast cancer MDA-MB-231 cell invasion and its mechanism. ActaPharmaceuticaSinica 2015; 1: 45-49.
- Yang Y, Liu JC, Cong H, Han CC, Ma LW, Jian BY. Effect of 12-deoxyphorbol-13-palmitate on VEGF expression in human breast cancer MCF-7 cells and relevant mechanisms. Chinese Journal of Experimental Traditional Medical Formulae 2015; 2: 120-124.