- Biomedical Research (2008) Volume 19, Issue 2
Psychiatric morbidity and survival in newly diagnosed treatment-naive cancer patients - A Study from Malaysia
Suthahar Ariaratnam1*, Ambigga Devi1, Gurpreet Kaur2, Dhachayani Sinniah3, Adlina Suleiman1, Maniam Thambu3, Fuad Ismail3
1Faculty of Medicine, Universiti Teknologi MARA, 40450 Shah Alam, Selangor, Malaysia
2Institute for Public Health, Ministry of Health, Malaysia,
3Faculty of Medicine, Universiti Kebangsaan Malaysia, 56000 Kuala Lumpur
- *Corresponding Author:
- Suthahar Ariaratnam
Faculty of Medicine
Universiti Teknologi MARA
40450 Shah Alam, Selangor
Malaysia
Tel: +603-55442848
Fax: +603-55442831
E-mail: dr.suthahar_a@yahoo.com
Accepted date: April 27 2008
Abstract
This paper was prompted by the necessity to investigate the relationship between psychiatric morbidity and survival of cancer patients. Eighty newly diagnosed treatment-naive patients with cancer were prospectively studied using self-administered questionnaires. Subsequent follow-ups were carried out at 0.5 year and between 1 to 2 years. The prediction of survival time was determined using Cox model. Depression (p = 0.001), stage 4 cancer disease (p = 0.016), neurological (p = 0.032), gastrointestinal tract (p = 0.04), head & neck (p = 0.011), gynecological (p = 0.005) and bone & soft tissue (p = 0.030) malignancies were independent and statistically significant prognostic factor of survival. Depressed patients have almost four fold greater risk of dying than non-depressed patients. Patients with stage 4 cancer illness have five fold greater risk of dying than patients with stage 1 disease. Patients with neurological, gynecological, head and neck, bone & soft tissue and gastro intestinal tract malignancies were found to have approximately thirty-six, twenty-five, twenty-two, sixteen and seven fold greater risk of dy-ing, respectively when compared to subjects with genitourinary cancers. Depression has significant impact on survival of patients with cancer.
Key words
Cancer Patients, Psychiatric Morbidity, Treatment-naïve, survival
Introduction
Psychosocial issues influence disease development and outcome which in turn could affect survival of cancer patients. This has been a subject of much debate. Nevertheless, there have been marked controversies among people who have discussed it. Some have reported correlations between different psychosocial variables [1-4]. On the other hand, others could not confirm such correlations [5,6]. Lewis et al [7] have affirmed that these inconsistency stems from poor methodology. The poor methodology includes (a) inadequately control for a variety of disease-related factors and therapeutic influence, (b) psychosocial variables were assessed after the cessation of treatment, (c) difference in estimating coping strategies, (d) multiple variables were assessed retrospectively, (e) marked variable outcome indicators, (f) seldom use of specific statistical analysis for survival (e.g. Kaplan-Meier plots and Cox method), (g) missing psychosocial data during follow-up, (h) inadequate consideration of differences in tumor staging and (i) subjective or non-standardized methods of evaluation. In addition, Irwin [8] in his recent article reiterated that the question of impact of psychosocial variables on the progression and outcome of cancer is still under discussion and entails more research. Therefore, we were provoked to study a heterogeneous group of cancer patients in which the psychosocial data were assessed on 3 instances and subsequently linked to disease outcome.
Methodology
After the study was approved by the Department of Psychiatry technical and ethics committee,Hospital Universiti Kebangsaan Malaysia (HUKM), permission was obtained from the Head of Department of Oncology &Radiotherapy, HUKM where the sample was eventually gathered.
The sample comprised patients newly diagnosed of cancer who were being referred to the Department of Oncology & Radiotherapy, HUKM, Kuala Lumpur, Malaysia to undergo treatment over a period of 10 months. Subseque ntly follow-up assessments were carried out at 0.5 year and between 1 to 2 years. Eligibility criteria included age 18 years or older, awareness of the diagnosis of cancer within 3 months of assessment and treatment-naive patients. Exclusion criteria included patients with cancer of the oral cavity and adrenal glands since their treatment regimes were not standardized, organic brain syndromes, debilitating illness, previous psychiatric diagnosis, mental retardation and benign conditions.
At the baseline evaluation, subjects were interviewed using questionnaires that included sociodemographic variable (i.e. age, gender, race/ethnicity, religion, marital status, occupation, and education level) and medical variable (i..e. type of cancer and staging). Furthermore, three more questionnaires were used. These being the Hospital Anxiety and Depression Scale (HADS) developed by Zigmond and Snaith [9]. Medical Outcomes Study Short-Form General Health Survey (MOS-SF) developed by Stewart et al [10] and Coping Inventory for Stressful Situations (CISS) developed by Endler & Parker [11]. These 3 instruments were re-administered during the follow-up assessments.
The questionnaires were translated into Malay. Several revisions and back-translations were done to ensure that the translation was a fair one. The HADS has been validated and used previously in other studies [12]. However, to the authors’ knowledge presently the translated version of MOS-SF and CISS has not been used in the local context.
All data were analysed using the Statistical Package for Social Sciences (SPSS) version 10 computer progr am [13].
From distribution questionnaires at baseline to the patient’s death represented the length of survival. For univariate comparison, unadjusted time-to-event analyses were carried out using the Kaplan-Meier estimates [14] and the non-parametric log-rank test [15]. Initially the individual effects of the psychosocial and medical variables were tested. To determine the most significant independent prognostic factors, additional multivariate was done using the Cox model [16]. Psychosocial and medical variables that deemed significant in the univariate tests (such as mental health, gender and stage of the disease) and other variables that proved to affect survival (such as age, occupational status, education level, type of cancer, anxiety, depression total QOL and coping styles) were concurrently entered into the Cox model.
Results
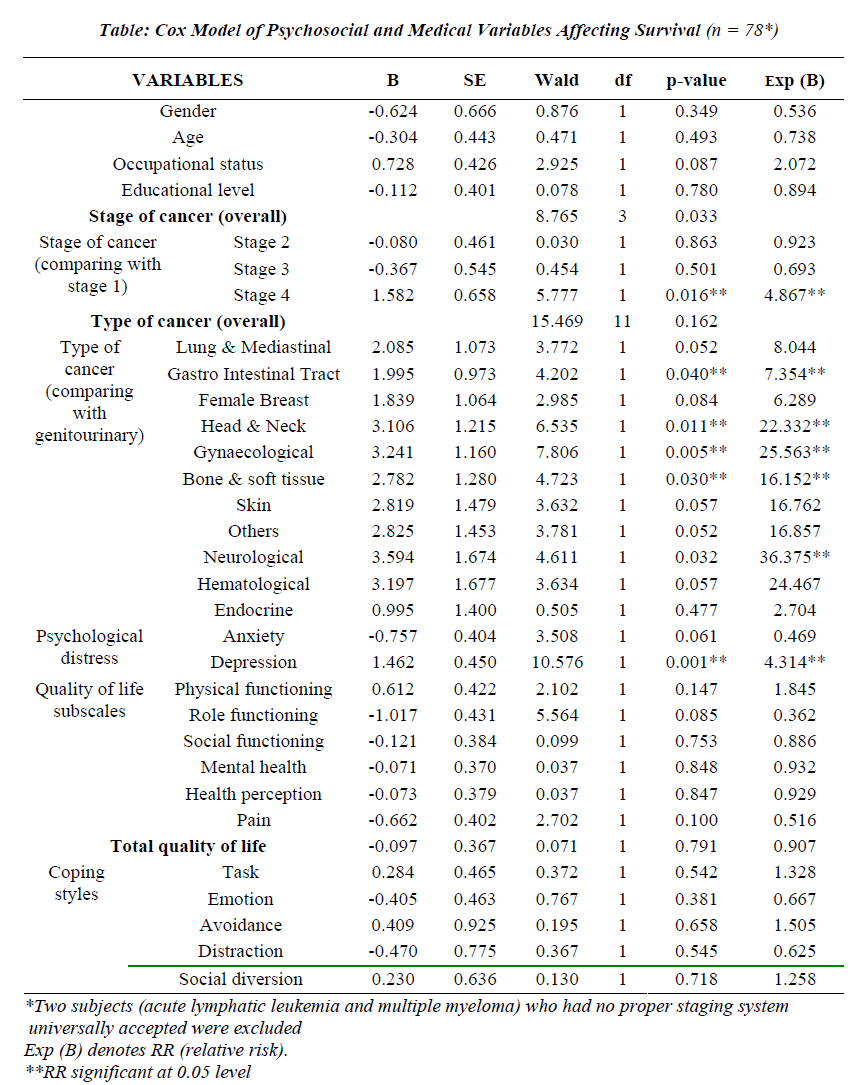
After the exclusion criteria a final cohort of 80 subjects was recruited and later followed-up. Table 1 represents the Cox model. It depicts that stage 4 cancer disease, gastro-intestinal tract, head & neck, neurological, gynaecological, bone and soft tissue malignancies as well as expression were independent and statistically significant prognostic factor of survival during the study period.
Based on relative risk (RR) it was revealed that patients with depression have at least 4- fold greater risk of dying than those without it. It was also noted that stage 4 cancer patients have almost 5 times greater risk of dying than than stage 1 patients. Furthermore, compared to patients with genitourinary cancers, those with neurological, gynaecological, head and neck, bone and soft tissue as well as gastrointestinal tract malignancies were found to have about 36, 26, 22, 16 and 7- fold greater risk of fatality, respectively.
Discussion
Several variables of independent and significant prognostic importance of survival have emerged in the multivariate model namely depression (p = 0.001), stage 4 cancer disease (p = 0.0016), gastrointestinal tract (p = 0.040), head & neck (p = 0.011), neurological (p = 0.032), gynecological (p = 0.005), bone and soft tissue malignancies (p = 0.030). Even though there were no significant differences seen in the cohort based on univariate analysis at baseline and 0.5 years later the Cox model seemed to have taken into account multiple covariates concomitantly. There is no doubt that many confounders were operating which were masking associations and hence produced a spurious relationship in the univariate analysis.
Almost consistent with previous researcher [4] who also examined prospectively the relationship of depression and survival after haematopoietic stem-cell transplantation in 193 patients between 6 and 12 months after similarly controlling for covariates by the Cox model and found that depressed individuals had a three-fold greater risk of dying as compared to non-depressed patients based on relative risk (in our study the RR was 4). However, there was no standardized measure of depression in that study although it was defined as being present in any person who reported being bothered by depression and who had four or more of the following symptoms (anxiety, difficulty concentrating, feelings of isolation, fatigue or loss of memory).
Similarly, Steel et al [3] who had examined a homogenous population of 101 hepatobiliary carcinoma subjects and deduced that depression and reduced survival were found to be associated with diminished natural killer cell numbers. Nonetheless, this study although had used a standardized questionnaire on depression, failed to focus on coping strategies and Quality of life issues which have impact on survival.
Derogatis et al [1] assessed 35 women with metastatic breast cancer and showed that long-term survivors (subjects who lived for 1 year or more) reported more symptoms such as depression, guilt and anxiety while those who died less than 1 year (short-term survivors) were less symptomatic. The short-term survivors demonstrated significant lower levels of hostility and higher level of positive mood. It should be noted that their patients, suffered from predominantly more advanced disease in addition to having a much smaller sample size (n = 35) as opposed to our study (n = 80). In addition, it is recognized that women are generally at higher risk of developing depression than man [17]. Thus, to begin with their subject selection was already biased since it included only females and hence would spuriously report higher level of depression. Therefore, it is only prudent to interpret this study with caution.
Conclusion
From this study we intend to suggest that depression significantly impairs the survival rate in cancer patients.
References
- Derogatis LR, Abeloffm MD, Melisaratos N. Psychological coping mechanisms and survival time in metastatic breast cancer. JAMA 1979; 242: 1504-1508.
- Brown KW, Levy AR, Rosberger Z, et al. Psychological Distress and Cancer Survival: A FollowUp 10 Years after Diagnosis. Psychosom Med 2003; 65: 636-643.
- Steel JL, Geller DA, Gamblin TC, et al. Depression, immunity and survival in patients with hepatobiliary carcinoma. J Clin Oncol 2007; 25: 2397-2405.
- Loberiza FR Jr, Rizzo JD, Bredeson CN, et al. Association of depressive syndrome and early deaths among patients after stemcell transplantation for malignant diseases. J Clin Oncol 2002; 20: 2118-2126.
- Petticrew M, Bell R, Hunter D. Influence of psychological coping on survival and recurrence in people with cancer: systematic review. BMJ 2002; 325: 1066.
- Tross S, Herndon J 2nd, Korzum K, et al. Psychological symptoms and diseasefree and overall survival in women with stage 2 breast cancer. J Natl Cancer Inst 1996; 88: 661-667.
- Lewis C, O'Brien R, Barraclough J. The psychoimmunology of cancer. (2nd ed.). Oxford University Press: Oxford , 2002
- Irwin MR: Depression and risk of Cancer progression. An Elusive link. J Clin Oncol 2007; 25: 2343-2344.
- Zigmond AS, Snaith RP. The Hospital Anxiety and Depression Scale. Acta Psychiatrica Scandinavica 1983; 67: 361-370.
- Stewart AL, Hays RD, Ware JE. Communication: The MOS shortform general health survey: Reliability and validity in a patient population. Medical Care 1988; 26: 724-735.
- Endler NS, Parker JDA. Coping Inventory for Stressful Situations (CISS) Manual (2nd ed.). Multihealth Systems: Toronto, 1999.
- Hatta SM, Hamid AR, Jaafar R, et al. Depressive symptoms among women after abortion. Malaysian J of Psychiatry 1997; 5: 27-33.
- SPSS data entry builder 3.0 user’s guide. SPSS Data Entry. USA: Chicago, 2001.
- Kaplan EL, Meier F. Nonparametric estimation from incomplete observations. J Am Stat Assoc 1958; 58: 457-481.
- Mantel N. Evaluation of survival data and two new rank order statistics arising in its consideration. Cancer Chemother Rep 1996; 50:163-170.
- Cox DR. Regression models and life tables. J R Stat Soc 1972; 34: 187-200.
- Goodwin G. Mood Disorder. In Companion to Psychiatric Studies, Johnstone EC, Owens DGC, Lawrie SM, Sharpe M, Freeman CPL (eds). Churchill Livingstone: Edinburgh, 2004; 421-449.