Research Article - Biomedical Research (2017) Volume 28, Issue 3
Prognostic value of neutrophil to lymphocyte ratio in patients with nasopharyngeal carcinoma: A meta-analysis
Bin Yu1,2, Zhonghua Li2, Qingping Zheng2, Zhanxiong Luo2, Jing Li2, Yuanyuan Zhou2, Leili Wang2, Chanjuan He2, Fengrong Shi2 and Rensheng Wang1*1Guangxi Medical University, Nanning, Guangxi 530021, China
2Deptartment of Radiation Oncology, Liuzhou People's Hospital, Liuzhou, Guangxi, China
Accepted on September 9, 2016
Abstract
This study was aimed to investigate the prognostic significance of Neutrophil to Lymphocyte Ratio (NLR) for patients with Nasopharyngeal Carcinoma (NPC) by the analytic approach of meta-analysis. We searched databases of PubMed, Emabse and Web of Science and included 6 studies with 3,469 patients in the meta-analysis. All included studies were from China. The association between NLR and four end-points events, Overall Survival (OS), Progression-Free Survival (PFS), Disease-Specific Survival (DSS) and Distant Metastasis-Free Survival (DMFS) was evaluated by using STATA version 12.0. Elevated NLR was associated with poor OS (HR=1.73, 95% CI: 1.37-2.13, p<0.001), PFS (Hazard Ratio (HR) =1.57, 95% CI: 1.19-2.09, p=0.002), DSS (HR=1.41, 95% CI: 1.19-1.66, p<0.001) and DMFS (HR=1.91 with 95% CI: 1.35-2.71, p<0.001). Furthermore, there was no significant heterogeneity and publication bias for all analyses in our study, indicating the robustness and credibility of this metaanalysis. Our study showed that elevated NLR was a significant biomarker for poor OS, PFS, DSS and DMFS in NPC. NLR was a potential valuable marker for NPC treatment.
Keywords
Meta-analysis, Neutrophil to lymphocyte ratio, Nasopharyngeal carcinoma, Biomarker.
Introduction
Nasopharyngeal Carcinoma (NPC) is a rare cancer type worldwide, especially less seen in Western countries and Africa. However, in South-East Asia and Southern China, NPC is endemic [1]. In 2010, the incidence rate of NPC is 4.33/100,000 for male patients and 1.92/100,000 for female patients in China, respectively [2]. NPC is generally sensitive to radiotherapy and chemotherapy, therefore, radiotherapy alone or in combination with chemotherapy is the recommended standard treatment for first diagnosed NPC, either for non-metastatic or metastatic disease [3,4]. Furthermore, the fast growing of concurrent chemo radiotherapy and targeted therapy provides multiple treatment options for NPC [5]. Faced with various therapy modalities, it is essential for clinicians to select more optimum therapy for individualized patients. Currently, the prognosis for NPC is often based on Tumour-Node-Metastasis (TNM) stage [6], whereas its prognostic efficiency is usually compromised due to unsatisfactory specificity and sensitivity. As a result, novel and low cost prognostic biomarkers are urgently needed.
Current evidence demonstrates that inflammatory cells play a pivotal role in cancer occurrence and progression [7,8]. In tumour microenvironment, inflammatory cells secrete a variety of cytokines to promote cancer development, whereas, innate immune responses could simultaneously up regulate the amount and activity of lymphocytes to attack cancer cells [8]. Neutrophil-to-Lymphocyte Ratio (NLR) is easy to obtain from peripheral blood count and can also reflect the host inflammation responses. Previous have shown that NLR was a significant prognostic marker in various solid tumours, including non-small cell lung cancer [9], hepatocellular carcinoma [10], breast cancer [11], gastric cancer [12] and prostate cancer [13]. Meanwhile, accumulated evidence showed that NLR also possessed potential prognostic value for NPC [14-16], while some other studies [17-19] did not detect the impact of NLR on NPC prognostication. To comprehensively clarify the different results from various studies, here, we employed the analytic method of meta-analysis. We thoroughly search relevant publications and meta-analysed the data to investigate the precise prognostic role of NLR in NPC.
Material and Methods
Literature search
A literature search was conducted on April, 2016 through the databases of PubMed, Embase and ISI Web of Science with the following search items: “Neutrophil to Lymphocyte Ratio”, “NLR”, “Nasopharyngeal Carcinoma”, “NPC”, and “nasopharyngeal neoplasms (Medical Subject Headings (MeSH) Terms)”. Only English publications were included. Reference lists of relevant articles were also manually screened to find additional studies.
Selection criteria
Eligible studies have to meet these criteria: (i) patients were pathologically confirmed NPC; (ii) NLR was obtained using blood based method prior treatment; (iii) data regarding the association between NLR and survival outcomes were presented or could be computed with Parmar’s method [20], survival outcomes included Overall Survival (OS), Progression-Free Survival (PFS), Disease-Specific Survival (DSS) and Distant Metastasis-Free Survival (DMFS); (iv) published in English. Letters, reviews, meeting abstracts, nonhuman studies and publications with insufficient information and sample size less than 50 were excluded. OS was measured as the first date of treatment to the date of death [19]. PFS was defined as the time from treatment to the first observation of local or regional recurrence or distant metastasis [19]. DSS was measured as the first date of treatment to the date of death because of disease. DMFS was defined as the time from treatment to the first observation of distant metastasis.
Data extraction and quality assessment
Two investigators (YB and ZHL) extracted the following information from eligible articles: name of first author, country, year, number of patients, treatment strategies, cut-off value for NLR and survival outcomes. Discrepancies between two investigators were resolved by discussion with the third investigator (RSW). The quality assessment of included studies was independently applied using the Newcastle-Ottawa Scale (NOS) [21]. The total possible score of NOS was 9 stars. Studies with>6 stars were regarded as being of good quality.
Statistical analysis
Statistical analysis was performed using Stata version 12.0 (Stata-Corp, College Station, Texas, USA). Hazard ratios (HRs) and their 95% CIs were used to evaluate the association between NLR and survival outcomes. HRs and 95% CIs were extracted from included studies to calculate pooled HRs. Both fixed effects model and random effects model were carried out and heterogeneity was assessed using the Cochran Q test and I2 statistic. I2>50% or Ph<0.1 indicated significant heterogeneity, then results from random effects model were picked, otherwise fixed effects model was selected. Potential risk of publication bias was evaluated by Begg’s test and visual inspection of funnel plots. Statistical significant level was set at p<0.05.
Results
Basic characteristics of included studies
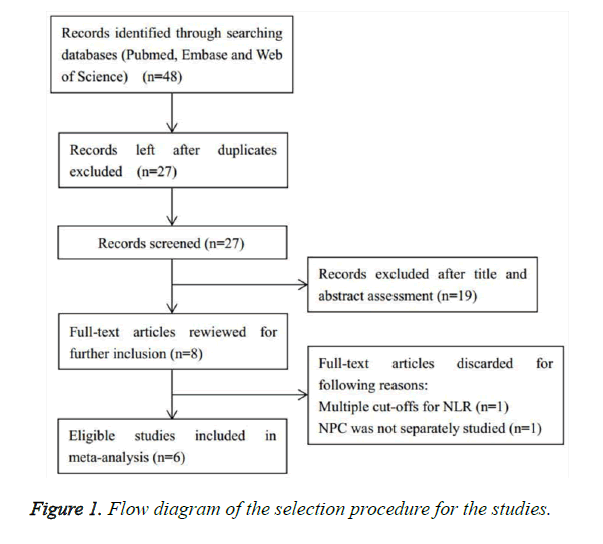
Initial literature search identified 48 records from electronic databases. Then duplicate publications were rejected and titles and abstracts were screened. Eight studies came into full text evaluation whereas one study [22] provided various cut-off values for NLR and another article [23] investigated head and neck cancer rather than NPC alone, so these two studies were excluded [22,23]. Finally, six studies involving 3,469 patients were included for meta-analysis [14-19]. The literature selection process was shown in Figure 1. The articles were published from 2011 to 2015 and were all conducted in China. Sample sizes ranged from 211 to 1,895 and the cut-off values for NLR ranged from 2.5 to 5. The relevant characteristics of included studies were presented in Table 1.
| Author | Year | NOS | Country | Patients | Gender | Age | Research | Follow-up | Histological | Metastasis | Treatment | Cut-off | Survival |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| score | (n) | (M/F) | (median, range) | Time | (month) | Type | At diagnosis | Outcome | |||||
| An | 2011 | 8 | China | 363 | 274/89 | 47 (12-76) | 2001-2002 | 62 (2-92) | NPC | No | Radiotherapy | 3.73 | DSS,DMFS |
| Chang | 2013 | 7 | China | 1,895 | 1455/440 | NA | 2001-2004 | 60 | NPC | No | Radiotherapy | 2.5 | DSS |
| Chen | 2014 | 8 | China | 211 | 181/31 | 46 (14-72) | 2005-2011 | 60 | NPC | Yes | Chemotherapy | 5 | OS,PFS |
| Jin | 2015 | 6 | China | 229 | 196/33 | 45 (18-72) | 2006-2011 | To 2012 | NPC | Yes | Chemotherapy | 3.6 | OS |
| Li | 2015 | 8 | China | 520 | 400/120 | 43 (13-78) | 2001-2009 | 88.4 (4.2-150.6) | NPC | No | Radiotherapy | 2.81 | DMFS |
| Sun | 2015 | 7 | China | 251 | 180/71 | 46 (15-76) | 2008-2011 | 50 (5-84) | NPC | No | Chemotherapy | 2.6 | OS,PFS |
| NPC: Nasopharyngeal Carcinoma; DSS: Disease-Specific Survival; DMFS: Distant Metastasis-Free Survival; RFS: Recurrence-Free Survival; OS: Overall Survival; PFS: Progression-Free Survival; NOS, Newcastle-Ottawa Scale. | |||||||||||||
Table 1. Main information of eligible studies.
Prognostic value of NLR for OS, PFS, DSS and DMFS
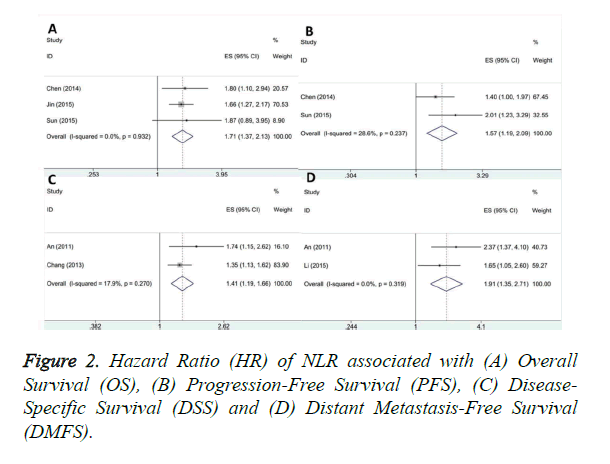
A total of three studies containing 691 patients investigated the relationship between NLR and OS [16,17,19]. The pooled HR was 1.73 and 95% CI: 1.37-2.13, p<0.001, without heterogeneity (I2=0, Ph=0.932) as shown in Table 2 and Figure 2A. For PFS analysis, two studies with 462 subjects provided the results [16,19]. The combined data from these two investigations was: HR=1.57, 95% CI: 1.19-2.09, p=0.002, with non-significant heterogeneity (I2=28.6%, Ph=0.237) as shown in Table 2 and Figure 2B. Pooled HR and 95% CI from two studies including 2,258 patients was HR=1.41, 95% CI: 1.19-1.66, p<0.001, I2=17.9%, Ph=0.27 as shown in Table 2 and Figure 2C, indicating high NLR was correlated with worse DSS [14,15]. Moreover, two articles with 883 patients investigated the prognostic role of NLR on DMFS [14,18]. Our pooled data showed that HR was 1.91 with 95% CI: 1.35-2.71, p<0.001, and there was no heterogeneity (I2=0, Ph=0.319) as shown in Table 2 and Figure 2D.
| Variables | Studies (n) |
Patients (n) |
Fixed-effects model HR (95% CI) p |
Random-effects model HR (95% CI) p |
Heterogeneity I2 (%) Ph |
|||
|---|---|---|---|---|---|---|---|---|
| OS | 3 | 691 | 1.71 (1.37-2.13) | <0.001 | 1.71 (1.37-2.13) | <0.001 | 0 | 0.932 |
| PFS | 2 | 462 | 1.57 (1.19-2.09) | 0.002 | 1.6 (1.14-2.26) | 0.007 | 28.6 | 0.237 |
| DSS | 2 | 2,258 | 1.41 (1.19-1.66) | <0.001 | 1.43 (1.16-1.76) | 0.001 | 17.9 | 0.27 |
| DMFS | 2 | 883 | 1.91 (1.35-2.71) | <0.001 | 1.91 (1.35-2.71) | <0.001 | 0 | 0.319 |
Table 2. Main results of the meta-analysis for NLR in NPC prognosis.
Assessment for publication bias
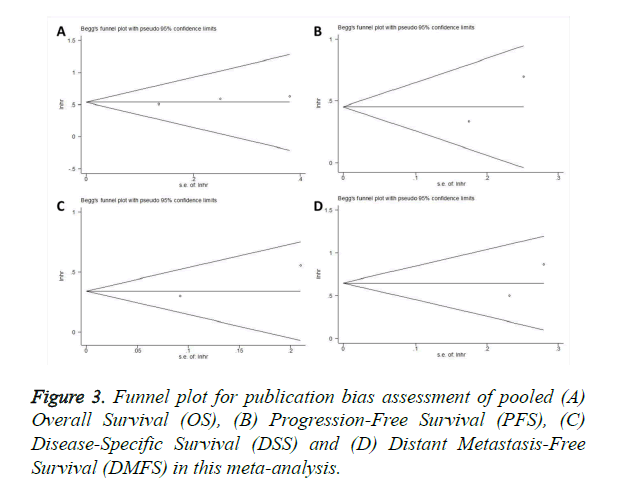
Begg’s test was adopted to assess the publication bias. As shown in Figure 3, there was no significant publication bias for OS, PFS, DSS or DMFS analysis. The p values of Begg’s test for OS, PFS, DSS, and DMFS were 0.296, 1, 1, and 1, respectively. These results demonstrated that this study was meaningful and statistically reliable.
Discussion
NLR is a serum based parameter which can reflect the immune responses status in cancer patients [24]. Previous studies have reported that NLR could be used as an efficient prognostic marker in various cancer forms whereas its prognostic value for NPC was still controversial. In the present study, we used meta-analysis approach and showed that elevated NLR was associated with worse OS, PFS, DSS and DMFS in patients with NPC. We for the first time systematically and comprehensively investigated the correlation between NLR and four end point events for NPC patients in a single study. Furthermore, the results of our analysis were robust and credible. To our knowledge, this is the first meta-analysis exploring the prognostic role of NLR for survival analyses in NPC.
In the last two decades, growing evidence showed that there was a strong linkage between inflammation and cancer [8,25]. By recruiting immunosuppressive cells and activating chemokines, cancer cells induce immune tolerance in tumour microenvironment and result in cancer development and metastasis. On the one hand, neutrophils have been reported to facilitate tumour growth and metastasis through releasing Reactive Oxygen Species (ROS) and Nitric Oxide (NO) [26]. In addition, tumour-associated neutrophils have been shown to secrete inflammatory mediators including IL-1, TNF and IL-6 to promote angiogenesis [27]. On the other hand, lymphocytes play an important role in antitumor activity, elevated Tumour Infiltrating Lymphocytes (TIL) have been reported to correlate with prolonged survival in different cancer types [28,29]. The NLR represents an index of pro-tumour and antitumor activity and it is widely available because it is derived from routine test in daily clinical practice.
The results from our study revealed that elevated NLR was a biomarker for worse survival outcomes, which was in line with results from other cancer types, such as lung cancer [9], hepatocellular carcinoma [10] and breast cancer [11]. We have noted that previous meta-analyses have explored the role of NLR for prognostication for various cancers. Whereas, NPC, as a malignancy with high incidence rate in Southern China, the prognostic value of NLR for which was still conflicting based on results from different reports. In the current study, we first clarify the relationship between high NLR and OS, PFS, DSS and DMFS in NPC. Our study provided rational evidence for potential usage of NLR for prognosis.
There were several limitations need to be addressed. First, the meta-analysis only included primary studies from China, which may introduce selection bias because patients with other ethnicity backgrounds were not enrolled. Second, the sample size was relatively small and limited number of studies was recruited for each survival analysis, which inhibits us to performed subgroup analysis.
In summary, our meta-analysis demonstrated that elevated NLR was associated with poor OS, PFS, DSS and DMFS in patients with NPC. Our results provided evidence to recommend NLR as a significant prognostic marker for NPC. However, due to above limitations, large scale studies with diverse ethnicitical patients are needed to verify our findings.
Acknowledgements
This study was supported by Guangxi appropriate Medical and Health Technology Research and Development project (Grant No. S201415-07) and Guangxi Health Department self-funded research projects (Grant No. Z2014432).
References
- Chang ET, Adami HO. The enigmatic epidemiology of nasopharyngeal carcinoma. Cancer Epidemiol Biomarkers Prev 2006; 15: 1765-1777.
- Wei KR, Zheng RS, Zhang SW, Liang ZH, Ou ZX and Chen WQ. Nasopharyngeal carcinoma incidence and mortality in China in 2010. Chin J Cancer 2014; 33: 381-387.
- Lee AW, Lin JC, Ng WT. Current management of nasopharyngeal cancer. Semin Radiat Oncol 2012; 22: 233-244.
- Ma BB, Hui EP, Chan AT. Systemic approach to improving treatment outcome in nasopharyngeal carcinoma: current and future directions. Cancer Sci 2008; 99: 1311-1318.
- Li XM, Huang WG, Yi H, Cheng AL, Xiao ZQ. Proteomic analysis to identify cytokeratin 18 as a novel biomarker of nasopharyngeal carcinoma. J Cancer Res Clin Oncol 2009; 135: 1763-1775.
- Guigay J. Advances in nasopharyngeal carcinoma. Curr Opin Oncol 2008; 20: 264-269.
- Balkwill F, Mantovani A. Cancer and inflammation: implications for pharmacology and therapeutics. Clin Pharmacol Ther 2010; 87: 401-406.
- Mantovani A, Allavena P, Sica A, Balkwill F. Cancer related inflammation. Nature 2008; 454: 436-444.
- Gu XB, Tian T, Tian XJ, Zhang XJ. Prognostic significance of neutrophil-to-lymphocyte ratio in non-small cell lung cancer: a meta-analysis. Sci Rep 2015; 5: 12493.
- Xiao WK, Chen D, Li SQ, Fu SJ, Peng BG, Liang LJ. Prognostic significance of neutrophil-lymphocyte ratio in hepatocellular carcinoma: a meta-analysis. BMC Cancer 2014; 14: 117.
- Chen J, Deng Q, Pan Y, He B, Ying H. Prognostic value of neutrophil-to-lymphocyte ratio in breast cancer. FEBS Open Bio 2015; 5: 502-507.
- Zhang X, Zhang W, Feng LJ. Prognostic significance of neutrophil lymphocyte ratio in patients with gastric cancer: a meta-analysis. PLoS One 2014; 9: e111906.
- Gu X, Gao X, Li X, Qi X, Ma M. Prognostic significance of neutrophil-to-lymphocyte ratio in prostate cancer: evidence from 16,266 patients. Sci Rep 2016; 6: 22089.
- An X, Ding PR, Wang FH, Jiang WQ, Li YH. Elevated neutrophil to lymphocyte ratio predicts poor prognosis in nasopharyngeal carcinoma. Tumour Biol 2011; 32: 317-324.
- Chang H, Gao J, Xu BQ, Guo SP, Lu RB, Li G, Huang SM, Han F, Liu ZG, Tao YL, Tu ZW, Chen C, Li XH, Xia YF. Haemoglobin, neutrophil to lymphocyte ratio and platelet count improve prognosis prediction of the TNM staging system in nasopharyngeal carcinoma: development and validation in 3237 patients from a single institution. Clin Oncol 2013; 25: 639-646.
- Chen C, Sun P, Dai QS, Weng HW, Li HP, Ye S. The Glasgow prognostic score predicts poor survival in cisplatin-based treated patients with metastatic nasopharyngeal carcinoma. Plos One 2014; 9: e112581.
- Jin Y, Ye X, He C, Zhang B, Zhang Y. Pre-treatment neutrophil-to-lymphocyte ratio as predictor of survival for patients with metastatic nasopharyngeal carcinoma. Head Neck 2015; 37: 69-75.
- Li AC, Xiao WW, Wang L, Shen GZ, Xu AA, Cao YQ, Huang SM, Lin CG, Han F, Deng XW, Zhao C. Risk factors and prediction score model for distant metastasis in nasopharyngeal carcinoma treated with intensity modulated radiotherapy. Tumor Biol 2015; 36: 8349-8357.
- Sun W, Zhang L, Luo M, Hu G, Mei Q, Liu D, Long G, Hu G. Pre-treatment hematologic markers as prognostic factors in patients with nasopharyngeal carcinoma: Neutrophil-lymphocyte ratio and platelet-lymphocyte ratio. Head Neck 2015; 38: E1332-E13340.
- Parmar MK, Torri V, Stewart L. Extracting summary statistics to perform meta-analyses of the published literature for survival endpoints. Stat Med 1998; 17: 2815-2834.
- Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol 2010; 25: 603-605.
- He JR, Shen GP, Ren ZF, Qin H, Cui C, Zhang Y, Zeng YX, Jia WH. Pre-treatment levels of peripheral neutrophils and lymphocytes as independent prognostic factors in patients with nasopharyngeal carcinoma. J Sci Spec Head Neck 2012; 34: 1769-1776.
- Charles KA, Harris BDW, Haddad CR, Clarke SJ, Guminski A, Stevens M, Dodds T, Gill AJ, Back M, Veivers D, Eade T. Systemic inflammation is an independent predictive marker of clinical outcomes in mucosal squamous cell carcinoma of the head and neck in oropharyngeal and non-oropharyngeal patients. Bmc Cancer 2016; 16: 124.
- Templeton AJ, McNamara MG, Eruga B, Vera-Badillo FE, Aneja P. Prognostic role of neutrophil-to-lymphocyte ratio in solid tumours: a systematic review and meta-analysis. J Natl Cancer Inst 2014; 106: 124.
- Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell 2011; 144: 646-674.
- De Larco JE, Wuertz BR, Furcht LT. The potential role of neutrophils in promoting the metastatic phenotype of tumours releasing interleukin-8. Clin Cancer Res 2004; 10: 4895-4900.
- An X, Ding PR, Li YH, Wang FH, Shi YX. Elevated neutrophil to lymphocyte ratio predicts survival in advanced pancreatic cancer. Biomarkers 2010; 15: 516-522.
- Leitch EF, Chakrabarti M, Crozier JE, McKee RF, Anderson JH, Horgan PG, McMillan DC. Comparison of the prognostic value of selected markers of the systemic inflammatory response in patients with colorectal cancer. Br J Cancer 2007; 97: 1266-1270.
- Nieto Y, Nawaz S, Shpall EJ, Bearman SI, Murphy J. Long-term analysis and prospective validation of a prognostic model for patients with high-risk primary breast cancer receiving high dose chemotherapy. Clin Cancer Res 2004; 10: 2609-2617.