- Biomedical Research (2016) Volume 27, Issue 3
Prevalence and antimicrobial susceptibility pattern of staphylococci and streptococci causing ocular infections from a tertiary eye care hospital, South India.
Haridas Sreekumar1, Palanisamy Manikandan2,3,4, Prithiviraj Aiswariya1, Venkatapathy Narendran2, Perumal Gomathi2, Ravindran Priya1, Kanesan Panneer Selvam5 and Coimbatore Subramanian Shobana6*
1Department of Microbiology, Dr. G. R. Damodaran College of Science, Coimbatore - 641 014, Tamilnadu, India
2Department of Microbiology, Aravind Eye Hospital and Post Graduate Institute of Ophthalmology, Coimbatore - 641 014, Tamilnadu, India
3Department of Medical Laboratory Technology, College of Applied Medical Sciences, Majmaah University, Al- Majmaah, KSA
4Greenlink Analytical and Research Laboratory India Private Ltd., Coimbatore, Tamilnadu, India
5Department of Microbiology, M.R Government Arts College, Mannargudi – 614 001, Tamilnadu, India
6Department of Microbiology, PSG College of Arts & Science, Coimbatore - 641 014, Tamilnadu, India
- *Corresponding Author:
- Coimbatore Subramanian Shobana
Department of Microbiology
PSG College of Arts & Science
India.
Accepted date: February 28, 2016
Abstract
Gram positive cocci viz., streptococci and staphylococci are found to be more predominant among ocular infections. Treatment of bacterial eye infections may engross empirical therapy with topical ophthalmic broad spectrum antibiotic formulations which is a prevailing practice among ophthalmologists. Ocular samples were collected from patients attending a tertiary eye care hospital, Coimbatore, south India from a total of 1802 ocular samples viz., Gram positive cocci (n=329), other bacteria & actinomycetes (n=561) and fungi (n=716), were isolated. Out of 329 Gram positive cocci, a total of 200 isolates viz., Streptococcus pneumoniae (n=76), coagulase negative staphylococci (CoNs) (n=49), Staphylococcus aureus (n=40), S. viridans (n=33) and S. pyogenes (n=2). The results revealed that the patients belonging to the age group between 60-80 years were most affected and male preponderance was observed. The isolates were subjected to antibiogram analysis with 15 different antibiotics according to the Clinical and Laboratory Standards Institute (CLSI) guidelines. Antibiotic viz., ampicillin, ofloxacin, gatifloxacin, levofloxacin, cefotaxime, cefazolin, piperacillin and vancomycin were found to effective against Gram positive cocci ocular infections.
Keywords
Eye, Infections, Staphylococcus, Streptococcus, Antibiotic susceptibility.
Introduction
Eye is a unique and necessary organ that is constantly exposed to the external environment. Infection and inflammation of the ocular regions may lead to blindness if prompt and appropriate therapy is not instituted [1]. Suppurative keratitis or corneal ulcer can cause corneal opacity & perforation and is the second most common cause for monoocular blindness in developing countries after cataract. Depending on the geographical location, the aetiological factor may be fungus or bacterium [2].
Corneal ulcer involves the disruption of the epithelial layer with involvement of the corneal stroma [3]. Bacterial endophthalmitis can occur following the introduction of an infectious agent into the posterior segment of the eye, causing intraocular infection and inflammation that despite appropriate therapeutic intervention frequently results in visual loss. It occurs more frequently following cataract surgery, the most commonly performed type of intraocular surgery. Intraocular lens implantation and penetrating keratoplasty (corneal transplantation) are the common ocular surgical procedures with a greater potential for intraocular bacterial contamination through wound leaks and subsequently lead to high risk of acute postoperative endophthalmitis [4].
The defence mechanisms present in the eye prevents the adverse activity of microorganisms within the ocular surface [5] and the infection due to trauma and other invasive procedures modify the normal flora leading to ocular infections [6]. Gram positive cocci are the most common opportunistic pathogens as a primary cause of bacterial eye infections. Staphylococci are normal inhabitants of skin and mucous membrane and have overcome most of the antimicrobial agents.
The genus Staphylococcus includes many pathogenic species in which Staphylococcus aureus and coagulase negative S. aureus (CoNS) are most important. Streptococci are normally found in respiratory, urinary and gastrointestinal tract, among which S. pneumoniae & S. viridans are common amongst ocular pathogens [7]. Coagulase-negative staphylococci are responsible for about 70% of post-operative endophthalmitis, followed by S. aureus, S. viridans, other Gram positive and Gram negative bacteria [4].
In most of the cases when the pathogen is not yet known, the choice of antimicrobial agents is commonly made empirically. Bharathi et al. [8] stated that an understanding of aetiological agents is vital to institute therapy and prevention of the disease entity and therefore it is necessary to carry out laboratory investigations including direct microscopy and culture. Once the organism has been identified, the effective antimicrobial agent is chosen according to the results of antibiotic susceptibility testing.
The data of in vitro antibiotic susceptibility testing will aid the clinicians to device an appropriate empirical treatment strategies for treating the ocular infections [9]. The purpose of this study was to analyse the prevalence of Gram positive cocci in ocular infections among the patients attending a tertiary eye care hospital, south India and to determine the in vitro susceptibility of these isolates to commonly prescribed antibiotics for ocular infections.
Materials and Methods
Location and duration of study
This study was carried out from September 2012 to December 2013 among the patients attending the Ocular Microbiology Department at Aravind Eye Hospital and Post Graduate Institute of Opthalmology, Coimbatore, Tamilnadu, India.
Collection and processing of ocular samples
The samples were collected from cases suspected of having ocular infection after a thorough examination by an ophthalmologist. Conjunctival swabs from the preoperative screening were collected using a sterile swab under aseptic conditions. The corneal scrapings were collected from patients with clinically suspected keratitis using a sterile Kimura's spatula by an ophthalmologist.
The aqueous tap and vitreous tap specimens from the endophthalmitis cases were obtained by an ophthalmologist through aspiration with the help of sterile tuberculin syringe and 22 gauge needles. A part of the specimen was subjected to direct microscopy after Gram staining and the rest was inoculated in brain heart infusion broth (BHI) & streaked on 5% sheep blood agar and chocolate agar plates and incubated at 37°C for 24-48 h. A culture was considered positive if growth was obtained in at least two media [10,11,12].
Identification and characterization of Gram positive cocci
The isolates were subjected to Gram staining for the presumptive identification of the cocci in chains and clusters. The isolates of staphylococci were analysed for coagulase production and mannitol fermentation. Streptococci were further tested for optochin sensitivity and bile solubility. Speciation of the isolates was carried out using standard biochemical test viz., indole production, methyl red, Voges- Proskauer test, citrate utilisation, oxidase test, catalase production test, glucose fermentation, nitrate reduction and esculin hydrolysis [12,13]. Representative isolates were further reconfirmed with KB004 HiStaph™ identification kit for CoNS and KB005A HiStrep™ identification kit for Streptococcus sp.
Antimicrobial susceptibility testing
Antibiotic susceptibility test was performed on Muller Hinton agar in accordance with the procedure outlined by CLSI [14]. A total of fifteen antibiotics (HiMedia, Mumbai, India) belonging to eight different classes viz., Aminobenzyl penicillin (ampicillin; 2 μg), fluroquinolones (ciprofloxacin; 5 μg, ofloxacin; 5 μg, gatifloxacin; 5 μg, moxifloxacin; 5 μg & levofloxacin; 5 μg), cephalosporins (cefotaxime; 30 μg, cefuroxime; 30 μg & cefazolin; 30 μg), ureidopenicillin (piperacillin; 10 μg), glycopeptides (vancomycin; 30 μg), chloromphenical (10 μg), tetracycline (30 μg) and aminoglycoside (tobramycin; 10 μg & gentamycin; 10 μg), at varying concentrations were used for the analysis.
For inoculum preparation, few colonies of the test isolates were transferred aseptically into sterile BHI broth and incubated at 35°C for 2 to 5 h. The broth culture was adjusted to the turbidity equivalent of 0.5 McFarland standards. Sterile Muller Hinton agar plates (with 5% sheep blood for streptococci) was inoculated, the discs were placed and incubated as per the standard procedures [14]. After incubation, the plates were observed for the zone of bacterostatis. The reading was performed by measuring the diameter of the inhibiting zone around the disc, in agreement with the CLSI criteria for all antibiotics, with one of three resulting grades: resistant, intermediately sensitive, or susceptible. S. pneumoniae (ATCC 49619) and S. aureus (ATCC 29213) were used in every test for quality control. For the confirmation of methicillin resistance among S. aureus, disc diffusion test was performed with cefoxitime (30 μg) and oxacillin (1 μg) discs [14].
Results
From 1802 culture positive ocular samples processed during the 16 months study period, 329, 561 and 716 accounted for Gram positive cocci, other bacteria & actinomycetes and fungi, respectively. Of 329 Gram positive cocci, 116 (35.25%), 112 (34.04%), 40 (12.15%), 36 (10.94), and 25 (7.59%) were from corneal ulcer/keratitis, preoperative screening, endophthalmitis, other infected cases (secondary infections, pus, scleral abscess, intra ocular lens culture, sutures, buccal space infection, etc.) and other cultures (mainly corneal button, eye ball and scleral rim), respectively.
From table 1, it is evident that highest number (n=60) of S. pneumoniae were isolated from corneal ulcer cases. Further, S. pneumoniae was the most frequent (n=133) Gram positive bacteria isolated during the study period. However, CoNS (n=20) was the major causative agent of endophthalmitis and also the second most frequently isolated Gram positive bacteria (n=92) next to S. pneumoniae. Among other infected cases, S. aureus was isolated in higher number (n=13) when compared to other isolates. S. viridans occurred more frequently in corneal ulcer (n=17), followed by preoperative screening (n=12) and endophthalmitis (n=10). Also, 2 isolates of S. pyogenes were obtained from corneal ulcer.
| Gram positive cocci isolates | Type of eye infection | |||||
|---|---|---|---|---|---|---|
| Corneal ulcer | Preoperative screening |
Endophthalmitis | other Infected cases |
Other cultures |
Total (%) | |
| S. pneumoniae | 60 | 57 | 4 | 9 | 3 | 133 (40.42%) |
| CoNS | 15 | 33 | 20 | 8 | 16 | 92 (27.96%) |
| S. aureus | 22 | 10 | 6 | 13 | 5 | 56 (17.02%) |
| S. viridans | 17 | 12 | 10 | 6 | 1 | 46 (13.98%) |
| S. pyogenes | 2 | - | - | - | - | 2 (0.60%) |
| Total (%) | 116 (35.25%) | 112 (34.04%) | 40 (12.15%) | 36 (10.94%) | 25 (7.59%) | 329 (100%) |
Table 1: Types and number of isolates of Gram positive cocci from ocular infections.
Overall, a total of 133, 92, 56, 46 and 2 isolates of S. pneumoniae, CoNS, S. aureus, S.viridans and S. pyogenes, respectively were obtained in the study. From these isolates, a total of 200 strains viz., 76 S. pneumoniae, 49 CoNS, 40 S. aureus, 33 S.viridans and 2 S. pyogenes isolates were subjected for further analysis. Among the selected cases with Gram positive cocci eye infections, male preponderance (n=123) was noted. The age groups of the target patients ranged between 1 month and 80 years and it was found that the individuals between 60 and 80 years was most affected with Gram positive cocci eye infections (Table 2).
| Age group | Male n=123 |
Female n=77 |
Total number of individuals (n=200) |
|---|---|---|---|
| 0-20 | 12 | 6 | 18 (9%) |
| 20-40 | 22 | 9 | 31 (15.5%) |
| 40-60 | 25 | 27 | 52 (26%) |
| 60-80 | 64 | 35 | 99 (49.5%) |
Table 2: Age wise distribution of the target patients with Gram positive cocci eye infections.
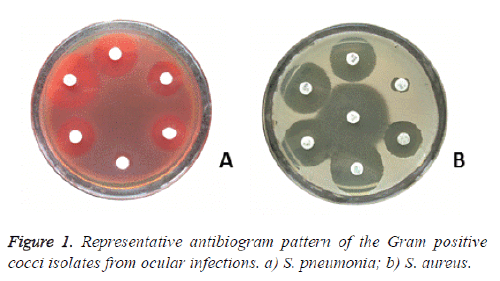
The antibiotic susceptibility analysis (Figure 1 and Table 3) revealed that all the isolates of streptococci were susceptible to aminobenzyl penicillin, ureidopenicillin, fluroquinolones, cephalosporins & glycopeptides, except 1 & 8 isolates of S. pneumoniae were intermediately susceptible to cefotaxime & chloramphenical, respectively.
| Isolates | Streptococci (n=111) | Staphylococci (n=89) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| S. pneumoniae N=76 n (%) | S. viridans N=33 n (%) | S. pyogenes N=2 n (%) | CoNS N=49 n (%) | S. aureus N=40 n (%) | ||||||||||
| Sensitivity pattern | S | I | R | S | I | R | S | R | S | I | R | S | I | R |
| AMP1 (2µg) | 76 (100) | - | - | 33 (100) | - | - | 2 (100) | - | 49 (100) | - | - | 38 (95) | - | 2 (5) |
| CIP2 (5 µg) | 76 (100) | - | - | 33 (100) | - | - | 2 (100) | - | 43 (87.75) | 6 (12.24) | - | (34) 85 | 4 (10) | 2 (5) |
| OFX3 (5 µg) | 76 (100) | - | - | 33 (100) | - | - | 2 (100) | - | 49 (100) | - | - | 38 (95) | - | 2 (5) |
| GAT4 (5 µg) | 76 (100) | - | - | 33 (100) | - | - | 2 (100) | - | 49 (100) | - | - | 38 (95) | - | 2 (5) |
| MXF5 (5 µg) | 76 (100) | - | - | 33 (100) | - | - | 2 (100) | - | 47 (95.92) | 2 (4.08) | - | 37 (92.5) | 1 (2.5) | 2 (5) |
| LVX6 (5 µg) | 76 (100) | - | - | 33 (100) | - | - | 2 (100) | - | 49 (100) | - | - | 38 (95) | - | 2 (5) |
| CTX7 (30 µg) | 75 (98.68) | 1 (1.32) | - | 33 (100) | - | - | 2 (100) | - | 49 (100) | - | - | 38 (95) | - | 2 (5) |
| CXM8 (30 µg) | 76 (100) | - | - | 33 (100) | - | - | 2 (100) | - | 49 (100) | - | - | 38 (95) | - | 2 (5) |
| CFZ9 (30 µg) | 76 (100) | - | - | 33 (100) | - | - | 2 (100) | - | 49 (100) | - | - | 38 (95) | - | 2 (5) |
| PIP10 (10 µg) | 76 (100) | - | - | 33 (100) | - | - | 2 (100) | - | 49 (100) | - | - | 38 (95) | - | 2 (5) |
| VAN11 (30 µg) | 76 (100) | - | - | 33 (100) | - | - | 2 (100) | - | 49 (100) | - | - | 38 (95) | - | 2 (5) |
| CHL12 (10 µg) | 68 (89.47) | 8 (10.53) | - | 33 (100) | - | - | 2 (100) | - | 49 (100) | - | - | 38 (95) | - | 2 (5) |
| TET13 (30 µg) | 62 (81.57) | 10 (13.16) | 4 (5.26) | 7 (21.21) | 4 (12.12) | 22 (66.67) | 1 (50) | 1 (50) | 47 (95.92) | - | 2 (4.08) | 37 (92.5) | 1 (2.5) | 2 (5) |
| TOB14 (10 µg) | 76 (100) | - | - | 27 (81.81) | - | 6 (18.18) | 2 (100) | - | 49 (100) | - | - | 35 (87.5) | - | 5 (12.5) |
| GEN15 (10 µg) | 0 | - | 76 (100) | 0 | - | 33 (100) | 0 | 2 (100) | 47 (95.92) | - | 2 (4.08) | 33 (82.5) | - | 7 (17.5) |
| CONs=Coagulase negative staphylococci; N=number of isolates tested; S=Sensitive; I=Intermediate; R=Resistant; 1Ampicillin; 2Ciprofloxacin; 3Ofloxacin; 4Gatifloxacin; 5Moxifloxacin; 6Levofloxacin; 7Cefotaxime; 8Cefuroxime; 9Cefazolin; 10Piperacillin; 11Vancomycin; 12Chloromphenical; 13Tetracycline; 14Tobramycin; 15Gentamycin | ||||||||||||||
Table 3: Antibiotic sensitivity of Gram positive cocci (n=200) from ocular infections.
Exactly, 10 & 4 isolates of S. pneumoniae, 4 & 22 isolates of S. viridians and 1 isolate of S. pyogenes were intermediately susceptible & resistant, respectively to tetracycline. Except 6 isolates of S. viridians, all the isolates of streptococci were sensitive to tobramycin.
All the streptococcal isolates were completely resistant to gentamycin. Among the 49 isolates of CoNS tested, 6 & 2 isolates were intermediately susceptible to ciprofloxacin & moxifloxacin, respectively 1 isolate each was resistant to tetracycline and gentamycin, respectively.
Of 40 isolates of S. aureus analysed, 4, 1 and 1 isolates were intermediately susceptible to ciprofloxacin, moxifloxacin and tetracycline, respectively.
Precisely, 5 and 7 isolates of S. aureus were resistant to tobramycin and gentamycin, respectively and 2 isolates of S. aureus were resistant to rest of the antibiotics. Upon further analysis of the resistant isolates of S. aureus, a total of 2 isolates were determined to be MRSA.
Discussion
Most of the ocular bacterial infections are due to Gram positive bacteria than Gram negative bacteria [15]. The present study primarily focused on Gram positive cocci causing eye infections. A total of 1802 samples were collected from patients attending tertiary care eye hospital between September 2012 and December 2013. The predominant isolates were S. pneumoniae (40.42%) followed by CoNS (27.96%). Kaliamurthy [15] reported that Staphylococcus spp. (64.5%) followed by Streptococcus spp. (12.3%) as the most common Gram positive cocci isolated from keratitis. Staphylococci being the predominant Gram positive cocci among the eye infections have been reported by many authors [5,7,16,17,18,19]. Although in the current analysis, S. pneumoniae was frequently isolated from corneal ulcer and preoperative screening, CoNS was the most frequently isolated bacteria from endophthalmitis cases, which is in accordance to Kaliamurthy et al. [15], Callegen et al. [20] and Durand [21]. Similar to our findings, Leck et al. [22] reported that in India streptococci accounted for 46.8% of corneal ulcer followed by staphylococci (26.8%). In the present study, of 200 patients, individuals between 60-80 years (49.5% of 200) and 40-60 years (26% of 200) were most affected with Gram positive cocci eye infections and similar results have been observed by Sreenivasan et al. [23], Muluye et al. [24] and Tesfaye et al. [25]. Male preponderance (123 of 200) of Gram positive cocci eye infection which was observed in the present study was also reported by several authors [15,24,26,27]. The development of antibiotic resistance to the commonly used topical antibiotics is an important problem worldwide. Therefore antibiotic susceptibility analysis would reveal the varying resistance pattern among the isolates and will help the clinicians to design an appropriate treatment regimen. In the present study all the streptococci were resistant to gentamycin. Among S. pneumoniae (n=76), 5.26% were resistant to tetracycline and 10.53% & 13.6% were intermediately susceptible to chloramphenical and tetracycline, respectively. Among S. viridians (n=33) 66.67% and 18.8% were resistant to tetracycline and tobramycin, respectively. Exactly, 4% of S. viridians were intermediately susceptible to tetracycline. Of the two isolates of S. pyogenes tested, one was resistant to tetracycline. The resistance to gentamycin and tobramycin in the present study correlates with the reports of Chalita [28]. In the present study, the recommended antibiotics for streptococcal ocular infections were ampicillin, the fluroquinolones tested, cefotaxime, cefazolin, piperacillin and vancomycin. The highest efficacy of fluroquinolones against Gram positive streptococci has been reported by Kaliamurthy et al. [15], Ramesh et al. [17], Reddy et al. [7] and Hofling- Lima et al. [29]. All the antibiotics analysed were effective against CoNS (n=49) isolates. Exactly, two isolates each were resistant to tetracycline and gentamycin, respectively. Also, 6 and 2 isolates of CoNS were intermediately susceptible to ciprofloxacin and moxifloxacin, respectively. Sensitivity of CoNS to most of the antibiotics has also been demonstrated by Shifraew et al. [19] and Bharati et al. [5]. But, Manikandan et al. [30] reported that 11.4% of CoNS were resistant to the entire antibiotic tested. Of 40 isolates of S. aureus analysed, 4, 1 and 1 isolates were intermediately susceptible to ciprofloxacin, moxifloxacin and tetracycline, respectively. Further, 5 and 7 isolates were resistant to the aminoglycosides viz., tobramycin and gentamycin, respectively and similar results were reported by Bharati et al. [5]. Of 40 isolates of S. aureus, exactly, 2 isolates were identified to be MRSA. Rajaduraipandi et al. [31] reported 8 MRSA from a total of 20 S. aureus isolated from conjunctival sample. Kang et al. [32] reported 57.6 % of MRSA from ocular infection in Taiwan. Vola et al. [33] reported 56 MRSA isolates from 566 S. aureus isolates from ocular infections. Nithya et al. [34] reported 33 MRSA of 127 S. aureus from ocular infections in south India.
Antibiotic resistance among ocular pathogens is increasing in parallel with the increase seen over the years in bacteria associated with systemic infections [35]. Antibiotic resistance in S. aureus can be attributed to horizontal gene transfer, primarily in hospitals as the selective pressures for resistance are maximum [36]. The emergence of antibiotic-resistant bacteria can thus be attributed to the use of antibiotics. Reduction in prescribing antibiotic is one of the effective ways to lessen the selection pressure and to prevent overuse and misuse of antibiotics, an up-to-date practical guideline, on the basis of case-by-case scientific data, for proper antibiotic prescribing should be developed [37]. Van Bambeke et al. [38] stated that antibacterial resistance in S. pneumoniae is increasing worldwide, affecting principally beta-lactams and macrolides followed by fluoroquinolone. The complete resistance of pneumococci, viridians streptococci and S. pyogenes to gentamycin was noted in the study. Salles et al. [39] reported that ribosomal mutation can result in aminoglycoside resistance in vitro but most resistance in the clinic is mediated by aminoglycoside modifying enzymes (AMEs).
Nuermberger and Bishai [40] reported that appropriate use of antibiotics and the introduction of the pneumococcal conjugate vaccines appear to offer the most promise for limiting the spread of drug resistance among pneumococci.
It could be concluded that, Gram positive cocci frequently causes ocular infection and the occurrence of two isolates of MRSA and emergence of intermediately susceptible and resistant strains must be given due consideration in this part of the country. In the present study, in vitro susceptibility analysis confirmed that ampicillin, ofloxacin, gatifloxacin, levofloxacin, cefotaxime, cefazolin, piperacillin and vancomycin demonstrated highest efficacy against Gram positive cocci isolates. These antibiotics may be considered as first line drug of choice for treating Gram positive cocci ocular infections as far as this region of study is concerned.
References
- Callegan M, Gregory-Ksander M, Willcox M, Lightman S. Ocular Inflammation and Infection. Int J Inflam 2012; 403520.
- Sherwal BL, Verma AK. Epidemiology of ocular infection due to bacteria and fungus – a prospective study. JK science 2008; 10:127-131.
- Liesegang TJ.Bacterial keratitis. Infect Dis Clin North Am 1992; 6: 815-829.
- Han DP, Wisniewski SR, Wilson LA, Barza M, Vine AK, Doft BH, Kelsey SF. Spectrum and susceptibilities of microbilogic isolates in the endophthalmitis vitrectomy study. Am J Ophthalmol 1996; 122:1-17.
- Bharathi MJ, Ramakrishnan R, Shivakumar C, Meenakshi R, Lionalraj D. Etiology and antibacterial susceptibility pattern of community acquired bacterial ocular infections in a tertiary eye care hospital in south India. Indian J Ophthalmol 2010; 58:497-507.
- Mantelli F, Mauris J, Argueso P. Ocular surface epithelial barrier and other mechanisms of mucosal protection: from allergy to infectious diseases. Curr Opin Allergy Clin Immunol 2013; 13:563-568.
- Reddy AK, Garg P, Alam MR, Gopinathan U, Sharma S, Krishnaiah S. Comparison of in vitro susceptibilities of Gram-positive cocci isolated from ocular infections against the second and fourth generation quinolones at a tertiary eye care centre in South India. Eye 2010; 24:170-174.
- Bharathi MJ, Ramakrishnan R, Vasu S, Meenakshi R, Shivkumar C, Palaniappan R. Epidemiology of bacterial keratitis in a referral centre in south India. Indian J Med Microbiol 2003; 21:239-245.
- Ubani UA. Common bacterial isolates from infected eyes. J Ni Optom Assoc 2009; 15:40-47.
- Sharma S .Diagnosis of infectious diseases of the eye. Eye 2012; 26:177-184
- Srinivasan M, Gonzales CA, George C, Cevallos V, Mascarenhas JM, Asokan B, Wilkins J, Smolin G, Whitcher JP. Epidemiology and aetiological diagnosis of corneal ulceration in Madurai, south India. Br J Ophthalmol 1997; 81: 965-971.
- Whitman WB, Goodfellow M, Kämpfer P, Busse HJ, Trujillo ME, Ludwig WSuzuki Ki. Bergey’s Manual of Systematic Bacteriology.2nd ed 5, parts A and B Springer-Verlag 2012.
- Boyle VJ, Fancher, M.E, Ross RW Jr. Rapid, modified Kirby-Bauer susceptibility test with single, high concentration antimicrobial disks. Antimicrob Agents Chemother 1973; 3: 418–424.
- CLSI. performance standards for antimicrobial disk susceptibility tests; approved standard-eleventh edition. CLSI document M02-A11. Wayne, PA: Clinical and Laboratory Standards Institute; 2012.
- Kaliamurthy J, Kalavathy CM, Parmar P, Nelson Jesudasan CA, Thomas PA. Spectrum of bacterial keratitis at a tertiary eye care centre in India.Biomed Res Int 2013.
- Mshangila B, Paddy M, KajumbulaH,Agaba –Ateenyi C,Kahwa B ,Seni J. External ocular surface bacteril isolates and their antimicrobial susceptibility patterns among pre-operative cataract patients at mulago national hospital in kampala, Uganda. BMC Ophthalmol 2013.
- Ramesh S, Ramakrishnan R, Bharathi MJ, Amuthan M, Viswanathan S. Prevalence of bacterial pathogens causing ocular infections in South India. Indian J PatholMicrobiol 2010; 53:281-286.
- Summaiya AM, Neeta DK, and Sangita BR . Ocular infections: rational approach to antibiotic therapy. Nat J Med Res 2012; 2: 22-24.
- Shiferaw B, Gelaw B, Assefa A, Assefa Y, Addis Z. Bacterial isolates and their antimicrobial susceptibility pattern among patients with external ocular infections at borumeda hospital, northeast Ethiopia. BMC Ophthalmol 2015.
- Callegan MC, Engelbert M, Parke II DW, Jett BD, Gilmore MS. bacterial endophthalmitis: epidemiology, therapeutics, and bacterium-host interactions. ClinMicrobiol Rev 2002;15: 111–124.
- Durand M L. Endophthalmitis. ClinMicrobiol Infect 2013; 19:227–234.
- Leck AK, Thomas PA, Hagan M, Kaliamurthy J, Ackuaku E, John M, Newman MJ, Codjoe FS, Opintan JA, Kalavathy CM, Essuman V, Jesudasan CA, Johnson GJ. Aetiology of suppurative corneal ulcers in Ghana and south India, and epidemiology of fungal keratitis. Br J Ophthalmol 2002; 86:1211-1215.
- Srinivasan M, Gonzales C, George C, Cevallos V, Mascarenhas J, Asokan B, Whitcher J. Epidemiology and aetiological diagnosis of corneal ulceration in Madurai, south India. Br J Ophthalmol 1997; 81: 965–971.
- Muluye D, Wondimeneh Y, Ferede G, Nega T, Adane K, Biadgo B, Tesfa H, Moges F. Types and drug susceptibility patterns of bacterial isolates from eye discharge samples at Gondar University Hospital, Northwest Ethiopia. BMC Res Notes 2014; 7: 292.
- Tesfaye T, Beyene G, Gelaw Y, Bekele S, Saravanan M.Bacterial Profile and antimicrobial susceptibility pattern of external ocular infections in jimma university specialized hospital, Southwest Ethiopia. Ajidm 2013;1:13-20.
- Chuang CC, Hsiao CH, Tan HY, Ma DHK, Lin KKI, Chang CJ, Huang YCStaphylococcus aureus ocular infection: methicillin-resistance, clinical features, and antibiotic susceptibilities. PLoS One 2012; 7.
- Henry CR, Flynn HW, Miller D, Forster RK, Alfonso EC. Infectious keratitis progressing to endophthalmitis: A 15-year-study of microbiology, associated factors, and clinical outcomes. Ophthalmology 2012; 119:2443-2449.
- Chalita MR, Höfling-Lima AL, Paranhos A Jr, Schor P, Belfort R Jr. Shifting trends in in vitro antibiotic susceptibilities for common ocular isolates during a period of 15 years. Am J Ophthalmol 2004; 137:43-51.
- Höfling-Lima AL, Belfort JrR, Moeller CTA, Bruno BC, Sousa LB de, FreitasDde. In vitro antibiotic susceptibilities of ocular bacteria isolates from the cornea and conjunctiva to moxifloxacin, gatifloxacin and other fluoroquinolones. Arg Bras. Ofthlamol 2004; 67:883-886.
- Manikandan P, Bhaskar M, Revathy R, John RK, Narendran K, Narendran V. Speciation of coagulase negative Staphylococcus causing bacterial keratitis. Indian J Ophthalmol 2005; 5:59-60.
- Rajaduraipandi K, Mani KR, Panneerselvam K, Mani M, Bhaskar M, Manikandan P Prevalence and antimicrobial susceptibility pattern of methicillin resistant Staphylococcus aureus: a multicentre study. Indian J Med Microbiol 2006; 24:34-8.
- Kang YC, Hsiao CH, Yeh LK, Ma DHK, Chen PYF, Lin HC, Tan HY, Chen HC, Chen SY, Huang YC. Methicillin-Resistant Staphylococcus aureus Ocular infection in Taiwan: Clinical Features, Genotying, and Antibiotic Susceptibility. Medicine (Baltimore) 2015; 94.
- Vola EM, Moriyama AS, Lisboa R, Vola MM, Hirai FE, Bispo PJM, Hofling –Lima AL. Prevalence and antibiotic susceptibility of methicillin-resistant Staphylococcus aureus in ocular infections. Arq Bras Oftalmol 2013; 76:350-353
- Nithya V, Rathinam S, Lalitha P. Molecular Epidemiology of Methicillin Resistant Staphylococcus aureus (MRSA) Causing Ocular Infections in South India. Invest Ophthalmol Vis Sci 2013; 54: 2162
- Sharma S. Antibiotic resistance in ocular bacterial pathogens. Indian J Med Microbiol 2011; 29: 218-221
- Chambers HF, DeLeo FR. Waves of Resistance: Staphylococcus aureus in the Antibiotic Era. Nat Rev Microbiol 2009;7:629-641.
- Lee C-R, Cho IH, Jeong BC, Lee SH. Strategies to Minimize Antibiotic Resistance. Int J Environ Res Public Health 2013; 10: 4274-4305.
- Van Bambeke F, Reinert RR, Appelbaum PC, Tulkens PM, Peetermans WE. Multidrug-resistant Streptococcus pneumoniae infections: current and future therapeutic options. Drugs. 2007; 67:2355-2382.
- Salles C, Creancier L, Claverys JP, Mejean V. The high level streptomycin resistance gene from Streptococcus pneumoniae is a homologue of the ribosomal protein S12 gene from Escherichia coli. Nucleic Acids Res 1992; 20: 6103.
- Nuermberger EL, Bishai WR. Antibiotic Resistance in Streptococcus pneumoniae: What Does the Future Hold? Clin Infect Dis 2004; 38: S363-S37.