- Biomedical Research (2015) Volume 26, Issue 1
Potential toxic effects of Zirconia Oxide nanoparticles on liver and kidney factors.
Zahra Arefian1, Fariba Pishbin1, Masoud Negahdary2 and Marziyeh Ajdary3*1Department of Biology, Payame Noor University, IR. of Iran
2Tehran Heart Center, Tehran University of Medical Sciences, Tehran, Iran
3Young Researchers and Elite Club, Khorasgan Branch, Islamic Azad University, Isfahan, Iran.
- *Corresponding Author:
- Marziyeh Ajdar
Young Researchers and Elite Club
Khorasgan Branch
Islamic Azad University
Isfahan, Iran
Accepted date: October 07 2014
Abstract
Nanoparticles In addition to the positive aspects of tending to its toxicity in the environment is unavoidable. This research studies the effects of ZrO2 NPs on the liver and kidney tissues as well as the activities in liver and kidney enzymes in the male rats. This study is done on 40 Wistar rat race, in 4 groups which includes on control and 3 experimental groups that are monitored daily, for the control group we give a Saline Solution and 3 other with 1ml/day nanoparticle by differ-ent doses (50,25,100 ppm) intra-peritoneally. After a 1 week period, samples were unconscious, blood sample were collected from the heart and the ALT, AST, ALP and creatinine were meas-ured. Post-treatment tissue level of malondialdehyde as well as the activities of catalase, glutathione peroxidase and superoxide dismutase were measured in the liver. The statistical raw data was analyzed by SPSS statistical software. The significant difference (p<0.05) in the levels of foregoing factors was obtained by the application of the maximum density of ZrO2NPs (100ppm) in comparison with the control groups. The rats when exposed to a high dosage of nanoparticles reported a significant increase in MDA concentration level while significant decreases were ob-served in GPX , CAT and SOD activities (P<0.001). In the rats which were exposed to high dos-age of nanoparticles, the liver enzyme concentration was significantly increased (p<0.05). The ob-tained results revealed the significant role of ZrO2 as an increasing ROS generation agent and the ROS have induced the development of free radicals.
Keywords
antioxidant, enzyme, kidney, liver, zirconia nanoparticles.
Introduction
Nanoparticles are particles that have a mean diameter and dimensions of about 10-9m. These particles due to their small size have special physical, chemical, mechanical, electric and magnetic properties; for example, they freely enter the cell and can interfere in its natural process. Zir-conium-oxide nanoparticles have become the most com-monly used nanoparticles in different industries such as kitchen appliances and are used every day. And this has increased the necessity for the studies on their safety to use these materials. Oxide nanoparticles, due to their physicochemical properties, are extensively used as drug carriers in treatment of cancer cells in live environments. Also, these nanoparticles have many biomedical applica-tions such as tissue regeneration, safety evaluation, detox-ification of biological fluids, thermotherapy of cancer cells and etc. Oxide nanoparticles result in increase of inflammatory responses in rats treated with these nano-particles. Studies have indicated that these nanoparticles can stop cell cycle at G1 phase. Due to their shape and size, nanoparticles can pass through physiological barriers and leave adverse effects. Our knowledge regarding the toxicity of nanoparticles is very limited. The assumption in this study is that long-terms contact with zirconium oxide nanoparticles results in disturbance in hepatic and renal systems. Aspartate aminotransferase (AST) exists in mitochondrial and cytosolic iso-zymes and is found in liver, muscle, brain and pancreas. Alanine aminotransfer-ase is a systolic enzyme which is specific to liver. Alka-line phosphatase (ALP) is an enzyme which exists in many tissues and is released from liver and bones in high concentrations and the obstruction of biliary tract results in its serum increase. Liver cell damage results in the re-lease of these enzymes into the blood flow. Creatinine is a characteristic enzyme of kidney which enters into the blood flow by kidney damage. The main mechanism of nanoparticles function is not known yet but difference in their vivo and in vitro studies suggest that they can pro-duce reactive oxygen species (ROS) and therefore they can have a role on intracellular calcium concentration, activation of transcription factors and creating change in cytokines. Oxidative stress can be considered as a re-sponse to cellular damage, Oxidative stress occurred due to nanoparticles can have several reasons, ROS can be created directly when both the oxidants and free radicals are present on the particle surface; through entering mito-chondria, as different studies have shown that very small nanoparticles can enter the mitochondria and create phys-ical damages that result in oxidative stress; activation of inflammatory cells such as macrophages and neutrophils of alveoli which are involved in the process of phagocyto-sis of nanoparticles. This can result in the production of reactive oxygen and nitrogen species; metal nanoparticles (iron, copper, chromium, vanadium, zirconium) can cause the production of ROS; DNA damage by ROS production as the result of the nanoparticles present can cause serious and inheritable damages to DNA; cellular oxidative stress is characterized by increase in ROS level, decrease in GSH expression and increase of lipid peroxidation. Dif-ferent mechanisms are proposed for justification of the damaging actions of nanoparticles and the elevation of the ROS level is more important in this regard. Superoxide, hydrogen peroxide and other oxygen radicals can damage DNA, proteins and cell lipids directly. Studies indicated that the cells that are exposed to nanoparticles show a reduction in viability. The cell cycle of PC12 cells is stopped in phase G2/M and induces apoptosis in a dose-dependent process, here mitochondrion is a special orga-nelle affected and the mitochondrial membrane potential is reduced. In addition, it reduces the lipid peroxide pro-duction level and the superoxide dismutase (SOD) acti-vation level. Maybe all of the changes can be attributed to the increase in the intracellular ROS level which low-ers the level of glutathione peroxide, catalase and gluta-thione. Recently, Gao et al concluded that magnetic na-noparticles have intrinsic peroxidase property and can catalyze H2O2. The aim of the present study is explor-ing the toxic effects of zirconium oxide nanoparticles on liver and kidney tissues and to provide necessary precau-tions to industry employees.
Materials and Methods
Materials
ZrOCl2·8H2O, urea, CH3OH, sulphuric acid solution-were used to prepare nanoparticles. Saline, ketamine, Rat, Hematoxylin eosin, and laboratory kit (PARS AZMON. Co of IRAN) were used.
Equipment
XRD, SEM (ZIESS EM 902A), TEM (JEM-200CX), UV-visible, optical microscope (OLYMPUS CX 21 FS1) and spectrophotometer (JENWAY. England) was used.
Preparation of ZrO2 nanoparticles
The ZrO2 nanoparticles were prepared according to the literature. Initially, 2.58 g ZrOCl2·8H2O and 4.80 g urea were dissolved in 20.0 mL CH3OH under stirring to form a colorless solution. The solution was transferred to a 20-mL Teflon-lined stainless steel autoclave, which was heated to 200 °C and maintained at that temperature for 20 h. The obtained white product was post-treated with sulphuric acid solution (0.167 mmol) and then calcined at 645 °C. The final product was analyzed by XRD, UV-visible, SEM (ZIESS EM 902A) and TEM (JEM-200CX).
Study Design
Some male wistar rats aging 2 months and weighting 250±13g were purchased from Isfahan University of Iran and kept at an ambient temperature of 25±2◦C in 12h light and 12h dark cycle. The animals were quarantined for a period of two weeks before the commencement of the experiment. They were placed in a poor ventilated com-partment in which the animals’ cages were kept. They were freely supplied water and food. This study was car-ried out according to the guidelines approved by Institu-tional Animal Ethical Clearance (IAEC).
Experimental design
The 40 rats were divided into four groups (n=10 rats per group) as follows:
Group Ι- normal controlled rats receiving intraperitoneal physiological Saline.
Group ΙΙ- rats treated Intraperitoneal with 1ml of ZrO2NPs, 25ppm concentration.
Group ΙΙΙ- rats treated Intraperitoneal with 1ml of ZrO2NPs, 50ppm concentration.
Group ΙV- rats treated Intraperitoneal with 1ml of ZrO2NPs, 100ppm concentration.
Blood sample collection
The rat did not show any symptoms of toxicity such as change in fur color, weight loss and any other symptom relevant to the morphology and behavior. Rats were anaesthetized by the Ketamine Chloride which was ad-ministered intraperitoneally. About the 8ml of the ani-mals’ blood were collected by cardiac puncture into Lith-ium Heparin bottles. The blood sample was centrifuged at 3000 rpm for 15 minutes in order to measure the concen-tration of ALP, AST, ALT and the creatinine factors then they were submitted to the Spectrophotometer and bio-chemical kit (Pars Azmon). The spectrophotometer is an instrument which is used to measure the properties of light over a specific portion of the electromagnetic spec-trum typically used in spectroscopic analysis to identify the materials. The measured variable is mostly the light intensity but could also for example, be the polarization state. The independent variable is usually the wavelength of the light or a unit directly proportional to the photon energy, such as wave number or electron volts, which has a reciprocal relationship to wavelength. A spectrometer is used in spectroscopy for producing spectral lines and measuring their wavelengths and intensities.
Measurement of Malondialdehyde Levels in Liver
Once finished, the animals were anesthetized with keta-mine. Liver was dissected from the body, rinsed with cold saline solution weighted after dryness promptly, homoge-nized (10%) subsequently and centrifuged separately with tris buffer for 2 minutes with the homogenizer device at 5000rpm speed. In order to prevent the elimination of enzymes and proteins, all the above steps were conducted at 4° c centrifugation, the clear supernatant solution was removed and the bottom sediment was discarded and the clear supernatant solution was used for measurement. Measuring the levels of Malondialdehyde was based on the method of thiobarbituric acid (TBA) reaction and car-ried out at the boiling point temperature. In this experi-ment, the MDA or Malondialdehyde-like materials react-ed with Thiobarbituric acid and provided a pink color at the maximum absorbance which was at 532 nm. The re-action was carried out at pH=2-3° and temperate of 90° c for 15 minutes. After cooling the sample, the absorbance level was observed. To this end, 150 μl of the centrifuged sample were taken and the amount of 1.5cc of Trichloroa-cetic acid and 1.5 cc of TBARS were added and then we put all the samples and standard tubes with different dilu-tions into Binary hot water for 80 min to facilitate the reaction. The solution was centrifuged at 3000 rpm for 10 min and the absorbance was read in a spectrophotometer at a λ =532 nm. Standard curves were prepared based on the dilution of tetraetoxypropane and the obtained absorb-ance peaks of samples were adapted on the standard curves.
Measurement of Superoxide Dismutase Enzyme Activity
The SOD activity was measured according to the inhibit-ing reductions of Nitroblueterazolium by Exanthin-Xanthin oxidase system as the producer of superoxide. In this experiment, the solutions consisted of Xanthine, Xan-thine Oxidase were applied in the Potassium Phosphate Buffer and Nitroblueterazolium. The absorbance of each sample was read every 30 second for 5 min. To gain the level of inhibition by SOD enzyme, the obtained data was concluded from the corresponding formula on the basis of set commercial kit. The activity of enzyme was measured by the level of inhibition adapted on the standard curve in terms of u/mg protein.
Measurement of Catalase Enzyme Activity
The activity of catalase was assayed according to the method of Abie (20). Briefly 0.01 mml/mml of the Etha-nol was added to a definite mass of tissue homogenate and was incubated in ice for half an hour. And Triton X-100, 10% was added to solution with the final concentra-tion of 1%. This solution was used to measure the activity of enzyme. The enzyme reaction was initiated by the ad-dition of 30mM H2O2 to extract sample tissue homoge-nate in Buffer Phosphate Sodium 50mM with pH=70. Then, the absorbance was measured at 40nm within 3 minutes and the enzyme activity was measured in terms of u/mg protein.
Measurement of Glutathione Peroxidase Enzyme Activity
The activity of GPX was assayed according to the Rotac and et al. method. Glutathione Peroxidase oxidized Gluta-thione in tissue, homogenates that results in the retrieve of hydrogen peroxide to water simultaneously. This reaction was stopped after 10 minutes by Tri-chloroacetic acid and remaining glutathione was reactivated by DNTB solution and it led to the formation of colored compounds 420nm, that can be measured with a spectrophotometer. Reagent mixture consists of 0.2ml of ethylene diamine tetra ace-tate (EDTA) 0.8mM, 0.1ml of Sodium oxide 10mM, 0.1ml of Hydrogen-peroxide 2.5mM, 0.2ml homogenate. The mixture was incubated at 37° c for 10 min. The reac-tion was stopped by adding 0.5ml of 10% Tri-chloro-acetic acid and the tubes were centrifuged for 15 min in 2000Rpm. 3ml of Disodium hydrogen 0.8 mM, and 0.1ml DTNB of 0.4% were added to supernatant solution and the resulting color was measured at 420nm accordingly. Glutathione Peroxidase activity was expressed as μ moles of Glutathione oxidized/min/mg protein.
Histological studies of the experimental animals
The vital organs (i.e., liver and kidney) were dissected separately from the rats in all groups. They were fixed with a 10% of formalin neutral buffer solution. The ob-tained sections were stained by Hematoxylin and Eosin procedure and examined under the light microscope. Their photomicrographs were obtained.
Statistical Analysis
Obtained data were stored by use of SPSS software and then delivered to EXCELL program, and edited and then data was excluded from ANOVA table from SPSS pro-grams. The results were offered by average and standard deviation. According to normal distribution of data for comparison of enzyme results in each group before and after investigation we use ANOVA test by repeated measurement and for comparison of groups in each peri-odic time we used ANOVA test and Dunnett test and we consider less significant surface from 0.05.
Results
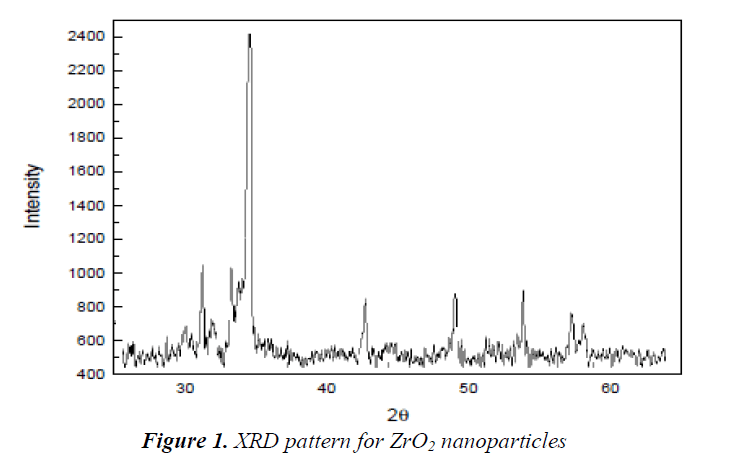
The ZrO2 nanoparticles Diffracted by X-Ray
The XRD pattern Fig. 1 for ZrO2 nanoparticles, the ab-sorption of diffraction peaks are occurred at 2θ values. The peaks that are prominent have been employed for the estimation of sample grain size, through Scherrer equa-tion D = Kλ/ (β cos θ) where K is constant (0.9), λ is the wavelength (λ = 1.5418 A°) (Cu Kα), β is the full width at the half-maximum of the line and θ is the angle of dif-fraction. The estimated grain size employing the relative intensity peak for ZrO2 nanoparticles was found to be 20nm and the addition in sharpness of XRD peaks showed that particles have crystalline nature. All the vari-ous peaks in figure 1 are related to ZrO2 nanoparticles and linked to Joint Committee for Powder Diffraction Studies.
ZrO2 Nanoparticles Electron Microscopic study
Surface attributes are important elements and it is a known fact that the characteristics of a high number of materials and many devices’ performance to a great extent are depended on their surface attributes. In figure 2 (a & b) regular pictures of ZrO2 Nanoparticles can be observed. The two sections of the picture were taken at 150 kV by TEM microscopy. Part (a) of Figure 2 was magnified by 150 KX; however, figure 2, part (b) was magnified by 80 KX. The mean diameter of the synthesized ZrO2 nanoparticles is around 20nm, and includes a very narrow particle distribution.
Serum Analysis
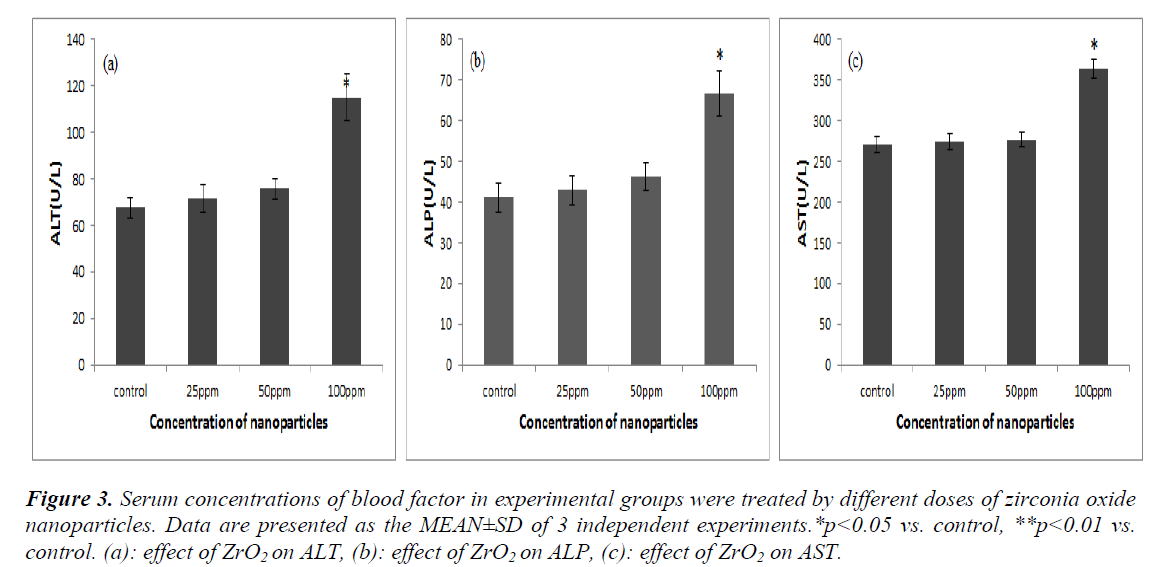
The enzymes such as ALT, AST, and ALP are responsible for the well-functioning of the liver and any damage induced to the liver rising from the use of NPs’ conditions may lead to the appearance of these enzymes within the blood stream. Thus, the effects of ZrO2NPs on the level of different metabolic en-zymes result in the optimum function of the liver. The serum was analyzed and the significant effects of NPs upon the liver damage are shown in (figure3 (a, b, c)). The ALT, ALP, and AST enzymes showed some sig-nificant elevated levels in the treated group in compari-son with the control group (p<0.05).
Figure 3: Serum concentrations of blood factor in experimental groups were treated by different doses of zirconia oxide nanoparticles. Data are presented as the MEAN±SD of 3 independent experiments.*p<0.05 vs. control, **p<0.01 vs. control. (a): effect of ZrO2 on ALT, (b): effect of ZrO2 on ALP, (c): effect of ZrO2 on AST.
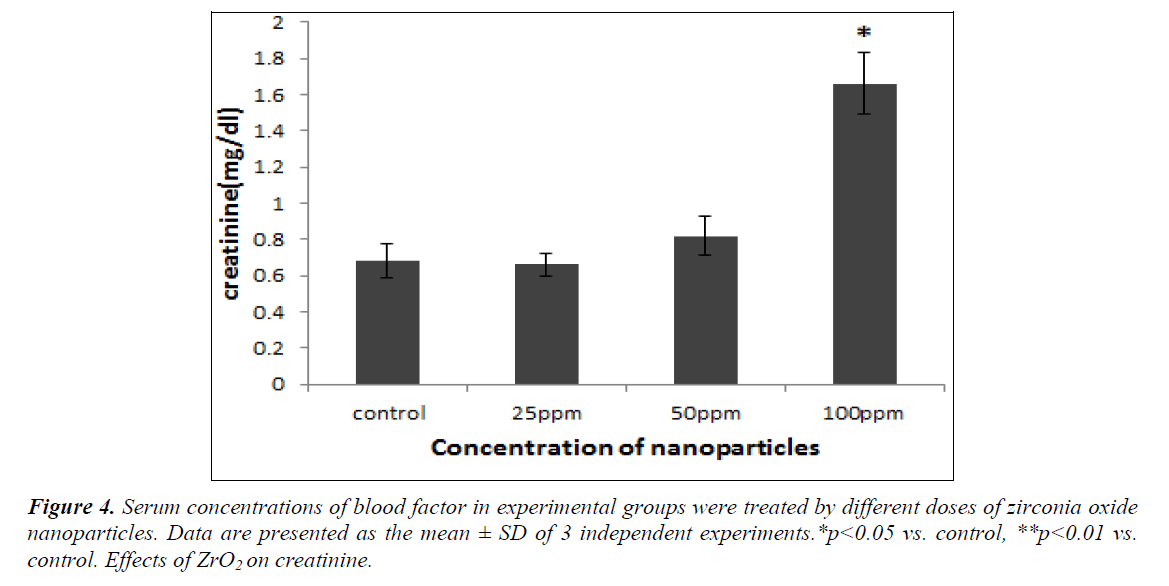
The effects of NPs on kidney damage are shown in the figure4. The level of symptomatic creatinine developed by the renal function has also shown a significant in-crease in comparison with the control group (p<0.05) that shown in the figure 4.
Effects of ZrO2 NPs on the antioxidant system
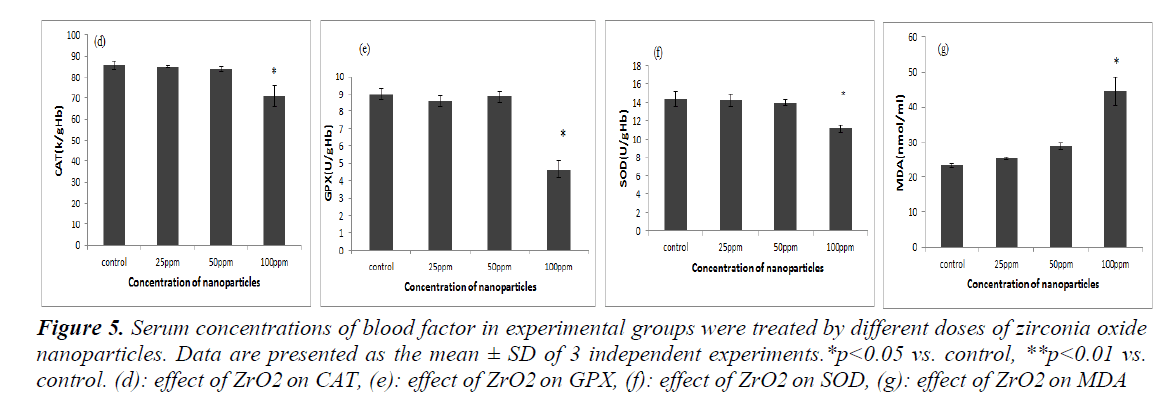
The Catalase, Glutathione Peroxidase and Superoxide Dismutase which are considered as the primary antioxi-dants are responsible for the direct elimination of generat-ed ROS. A significant decrease in the level of enzymes received by the treatment groups respectively (see figure5 (d, e, f), showed a significant difference with the control group (p<0.05). Oxidative stress are generated due to im-balance state of the antioxidant enzymes and induces the generation of free radicals and free radicals cause the generation of apoptosis in turn. In the MDA, a significant increase in the level of enzymes in the treatment groups respectively showed a significant difference with the con-trol group (p<0.05) (see figure 5 (g).
Figure 5: Serum concentrations of blood factor in experimental groups were treated by different doses of zirconia oxide nanoparticles. Data are presented as the mean ± SD of 3 independent experiments.*p<0.05 vs. control, **p<0.01 vs. control. (d): effect of ZrO2 on CAT, (e): effect of ZrO2 on GPX, (f): effect of ZrO2 on SOD, (g): effect of ZrO2 on MDA
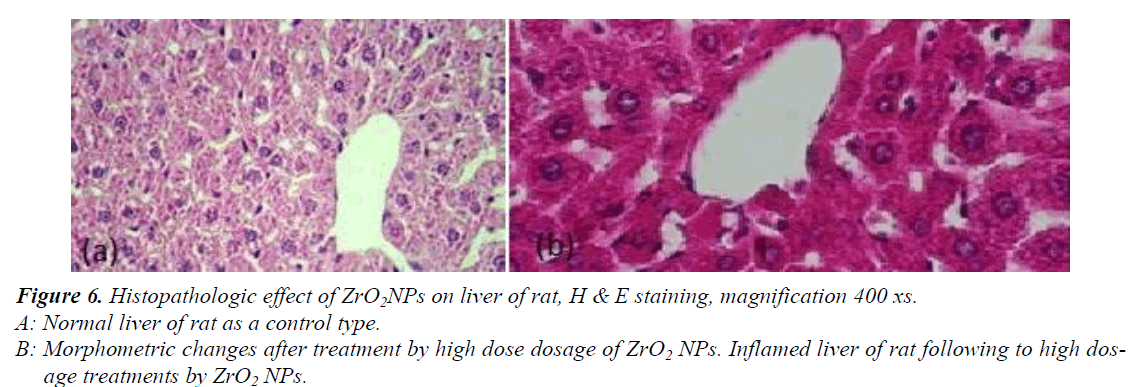
Histo-pathological Studies
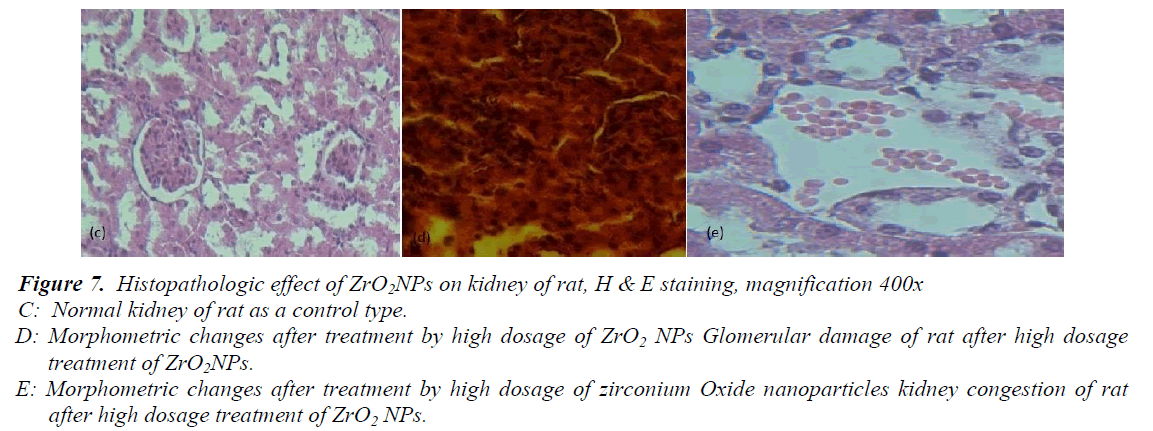
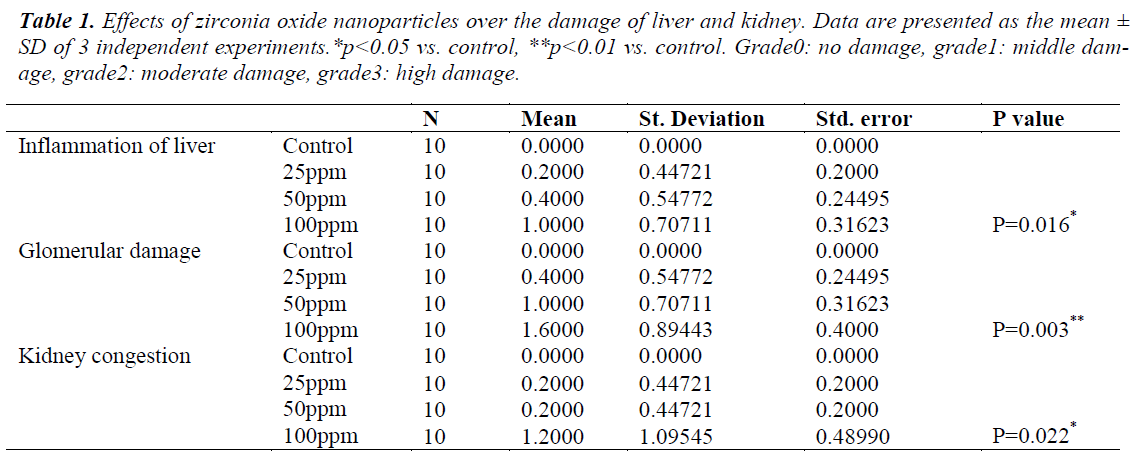
Histological analysis of liver and kidney tissues was carried out in order to examine the potency of ZrO2NPs to the tissue damage. The liver of the control rats showed a normal hepatic architecture of portal triad and central vein (figure6 (a). The rats treated with ZrO2NPs showed a significant difference in inflammation of the liver (table 1) attributed to the fourth group that were received NPs with 100ppm concentration (figure6 (b). The controlled kidney sections showed normal renal cortex and glomerular tufts (figure7 (c). The rats treated with ZrO2NPs showed a significant difference (table 1) in kidney congestion and in destruction of the glomerular capsule space attributed to the fourth group that were received NPs with 100ppm concentration (figure7 (d, e).
Figure 7: Histopathologic effect of ZrO2NPs on kidney of rat, H & E staining, magnification 400x
C: Normal kidney of rat as a control type.
D: Morphometric changes after treatment by high dosage of ZrO2 NPs Glomerular damage of rat after high dosage treatment of ZrO2NPs.
E: Morphometric changes after treatment by high dosage of zirconium Oxide nanoparticles kidney congestion of rat after high dosage treatment of ZrO2 NPs.
Discussion
The main finding of this study was the significant in-crease (p<0.05) in ALP, ALT, AST levels by the admin-istration of 100ppm ZrO2NPs as compared to the normal group. An initial step in detecting the liver damage is to run a simple blood test to determine the presence of cer-tain liver enzymes in blood. Under normal circumstances, these enzymes reside within the cells of the liver. But when the liver is damaged, these enzymes are spilled into the blood stream. The ALT, AST and ALP are available in liver cells in normal conditions; however, in case of damaging to the cell, they transmit into the serum. The absorbed Nano silver bound to the plasma proteins results in its destruction in certain organs such as liver, or causes damage to the mitochondria and declines the Glutathione level. Other results of this study provided a significant increase (p<0.05) in creatinine level with 100 ppm ZrO2NPs concentration as compared to the normal group. An initial step in detecting the kidney damage is running a simple blood test to determine the presence of certain kidney enzymes in the blood. The level of symptomatic creatinine attributed to the renal function was also in-creased significantly in the treated groups in comparison with the Control group. In the analysis associated with the molecular mechanisms of the Anti-oxidative effects of ZrO2NPs in high concentrations showed that they induce the oxidative stress in rat and the effects of ZrO2NPs on GPX and the CAT levels in treated rats were investigated simultaneously. As oxidative stress is due to the increase in the formation of free radicals, it results in damage to the chemical elements of the cell such as protein and lipid structures which in turn results in release of some en-zymes into the blood and in this regard, The levels of some tissue enzymes related to liver such as Aspartate Aminotransferase and Alanine Aminotransferase are in-creased in the blood. Oxidative stress in humans is the result of imbalance in the situation of some antioxidants. In most cases, it finally results in oxidative damage. It seems that reactive oxygen species are produced in every tissue through different mechanisms. Nanotechnology researchers have become familiar with extensive aspects of nanoparticle applications that may have a big role in medicine, prevention and treatment of diseases and pro-duction of drugs. Dewey et al (2009) showed that zinc nanoparticles are able to protect the integrated structure of cell membrane against oxidative damage of free radicals, increase the level of antioxidant enzymes and reduce Malondialdehyde level; while in this study gold nanopar-ticles have resulted in the increase of free radicals produc-tion. Using gold nanoparticles equal to 2.5 mg/kg in in-traperitoneal injection in rats, Selvaraj Barath ManiKanth et al (2010) concluded that gold nanoparticles result in reduction of catalase and glutathione peroxide in healthy and diabetic rats. Many studies in laboratory environment have shown the toxicity of zinc oxide. For example, in 2009 Cory Hanley proved that zinc oxide nanoparticles in culture medium result in the production of reactive oxy-gen species (ROS) and then result in oxidative damage, cell inflammation and death and zinc oxide decomposi-tion in cell culture medium. Also, intracellular ROS has a significant relationship with survival and the level of lac-tate dehydrogenase. In terms of increase in the level of lactate Dehydrogenase and cell death, the results of the present study are compatible with the results of the study by Cory Hanley. The study of zinc oxide antibacterial effects and the measurement of zinc spin resonance in 2010 by Venubabu Thati indicated that aqueous suspen-sions of zinc oxide small nanoparticles results in the pro-duction and increase in the activity of special oxygen and significant increase of oxidative stress. In terms of cell damage, the observations of the present study are compat-ible with the study of Venubabu Thati. The use of magne-sium oxide nanoparticles in Wistar rats showed that mag-nesium oxide nanoparticles result in oxidative stress in rats by reducing the antioxidant capacity and result in sub-acute toxicity in them by reducing superoxide dis-mutase and catalase and the observations of this study are compatible with those results. The use of silver nanoparti-cles at 100 and 200 ppm doses results in the increase of catalase enzyme and with the increase in the level of the used dose, the amount of this enzyme increases too and it was concluded that silver nanoparticles result in oxidative stress and inducing antioxidant enzymes. The use of cop-per nanoparticles at the dose of less than 50 nm resulted in the reduction of superoxide dismutase and catalase and this damage was increased gradually in one day and one week after intra-pulmonary injection. In a study by Liu et al, the researchers showed that the use of copper oxide nanoparticles results in the increase of superoxide dis-mutase and catalase secretion. Oxidative stress is in-creased with the toxicity of nanoparticles and the increase in the production of ROS and oxidative stress can be one of the sign of the toxicity of nanoparticles.
Conclusion
The obtained results reveal the significant role of ZrO2 as an increasing ROS generation agent and the ROS induces the generation of free radicals in turn. Those free radicals cause damage to different tissues. Following, to damage to the liver, its enzymes may leak into the blood stream excessively.
References
- Chen D, Xi T, Bai J. Biological effects induced by nanosilver particles: in vivo study. Biomedical Materials. 2007; 2(3): S126.
- Hsin Y-H, Chen C-F, Huang S, Shih T-S, Lai P-S, Chueh PJ. The apoptotic effect of nanosilver is mediated by a ROS-and JNK-dependent mechanism involving the mitochondrial pathway in NIH3T3 cells. Toxicology letters. 2008;179(3):130-139.
- Oberdörster G, Stone V, Donaldson K. Toxicology of nanoparticles: a historical perspective. Nanotoxicology. 2007; 1(1): 2-25.
- Prijic S, Scancar J, Cemazar M, Bregar VB, Znidarsic A, Sersa G. Increased cellular uptake of biocompatible superparamagnetic iron oxide nanoparticles into malignant cells by an external magnetic field. The Journal of membrane biology. 2010; 236 (1): 167-179.
- Mirković B, Turnšek TL, Kos J. Nanotechnology in the treatment of cancer. Zdravniški Vestnik. 2010; 79(2). Park E-J, Kim H, Kim Y, Yi J, Choi K, Park K. Inflammatory responses may be induced by a single intratracheal instillation of iron nanoparticles in mice. Toxicology. 2010; 275(1): 65-71.
- Stone V, Johnston H, Clift MJ. Air pollution, ultrafine and nanoparticle toxicology: cellular and molecular interactions. IEEE transactions on nanobioscience. 2007; 6(4): 331.
- Anwar MF, Yadav D, Rastogi S, Arora I, Khar RK, Chander J, et al. Modulation of liver and kidney toxicity by herb Withania somnifera for silver nanoparticles: a novel approach for harmonizing between safety and use of nanoparticles. Protoplasma. 2014.
- Buzea C, Pacheco II, Robbie K. Nanomaterials and nanoparticles: sources and toxicity. Biointerphases. 2007; 2(4): MR17-MR71.
- Sikka SC, Rajasekaran M, Hellstrom WJ. Role of oxidative stress and antioxidants in male infertility. Journal of andrology. 1995; 16(6): 464-468.
- Uusitalo LM, Hempel N. Recent advances in intracellular and in vivo ROS sensing: Focus on nanoparticle and nanotube applications. International journal of molecular sciences. 2012; 13(9): 10660-10679.
- Singh N, Manshian B, Jenkins GJ, Griffiths SM, Williams PM, Maffeis TG, et al. NanoGenotoxicology: the DNA damaging potential of engineered nanomaterials. Biomaterials. 2009; 30(23): 3891-3914.
- Li JJe, Muralikrishnan S, Ng C-T, Yung L-YL, Bay B-H. Nanoparticle-induced pulmonary toxicity. Experi- mental biology and medicine. 2010; 235(9): 1025-1033.
- Kim JA, Lee N, Kim BH, Rhee WJ, Yoon S, Hyeon T, et al. Enhancement of neurite outgrowth in PC12 cells by iron oxide nanoparticles. Biomaterials. 2011; 32 (11): 2871-2877.
- Aitken RJ. Free radicals, lipid peroxidation and sperm function. Reproduction, fertility, and development. 1995; 7(4): 659-668.
- Wang J, Sun P, Bao Y, Liu J, An L. Cytotoxicity of single-walled carbon nanotubes on PC12 cells. Toxicology in vitro: An international journal published in association with BIBRA. 2011; 25(1): 242-250.
- Gao L, Zhuang J, Nie L, Zhang J, Zhang Y, Gu N, et al. Intrinsic peroxidase-like activity of ferromagnetic nanoparticles. Nature nanotechnology. 2007; 2(9):577-583.
- Dave SR, Gao X. Monodisperse magnetic nanoparticles for biodetection, imaging, and drug delivery: a versatile and evolving technology. Wiley Interdisciplinary Reviews: Nanomedicine and Nanobiotechnology. 2009; 1(6): 583-609.
- Habig W, Jakoby W. Glutathione S-transferases (rat and human). Methods in enzymology. 1980;77:218-31.
- Aebi H. Catalase in vitro. Methods Enzymol. 1984; 105: 121-126.
- Rotruck J, Pope A, Ganther H, Swanson A, Hafeman DG, Hoekstra W. Selenium: biochemical role as a component of glutathione peroxidase. Science. 1973; 179(4073): 588-590.
- Vahid Yousefi Babadi LN, Azadeh Najafi, Hosein Gholami, Mohammad Ebrahim Beigi Zarji, Jalal Golzadeh, , et al. Evaluation of iron oxide nanoparticles effects on tissue and enzymes of liver in rats. J Pharm Biomed Sci. 2012; 23(23): 1-5.
- Loghman A, Iraj SH, Naghi DA, Pejman M. Histopathologic and apoptotic effect of nanosilver in liver of broiler chickens. African Journal of Biotechnology. 2014; 11(22): 6207-6011.
- Chang ALS, Khosravi V, Egbert B. A case of argyria after colloidal silver ingestion. Journal of cutaneous pathology. 2006; 33(12): 809-811.
- Dawei A, Zhisheng W, Anguo Z. Protective Effects of Nano-ZnO on the Primary Culture Mice Intestinal Epithelial Cells in in vitro Against Oxidative Injury. Journal of Animal and Veterinary Advances. 2009; 8(10): 1964-1967.
- BarathManiKanth S, Kalishwaralal K, Sriram M, Pandian SRK, Youn H-s, Eom S, et al. Research Anti- oxidant effect of gold nanoparticles restrains hyperglycemic conditions in diabetic mice. 2010.
- Hanley C, Thurber A, Hanna C, Punnoose A, Zhang J, Wingett DG. The Influences of Cell Type and ZnO Nanoparticle Size on Immune Cell Cytotoxicity and Cytokine Induction. Nanoscale research letters. 2009; 4(12): 1409-1420.
- Thati V, Roy AS, Ambika Prasad M, Shivannavar C, Gaddad S. Nanostructured zinc oxide enhances the activity of antibiotics against Staphylococcus aureus. J Biosci Tech. 2010; 1(2): 64-69.
- Kiranmai G, Reddy AR. Antioxidant status in MgO nanoparticle-exposed rats. Toxicol Ind Health. 2013 Nov; 29(10): 897-903.
- Yeo M, Kang M. Effects of nanometer sized silver materials on biological toxicity during zebrafish embryogenesis. Bulletin-Korean Chemical Society. 2008; 29(6): 1179.
- Reddy ARN, Colony RK, andhra Pradesh K, Kumar CP. Pulmonary Toxicity of Copper Oxide (CuO) Nanoparticles in Rats. J Med Sci. 2013; 13(7): 571-7.32.
- Liu Z, Liu S, Ren G, Zhang T, Yang Z. Nano-CuO inhibited voltage-gated sodium current of hippocampal CA1 neurons via reactive oxygen species but independent from G-proteins pathway. Journal of Applied Toxicology. 2011; 31(5): 439-445.