Research Article - Biomedical Research (2017) Volume 28, Issue 1
Open microwave ablation in the treatment of spinal metastasis: preliminary experience
Xing Wei, Bingyao Chen*, Li Nan, Yaming Shi, WenWen Wu, Yunqing Ma and Shuxun HouDepartment of Orthopaedic Surgery, the First Affiliated Hospital of PLA General Hospital, Beijing 100048, PR China
- *Corresponding Author:
- Bingyao Chen
Department of Orthopaedic Surgery
The First Affiliated Hospital of PLA General Hospital
PR China
Accepted on June 02, 2016
Abstract
Purpose: This study aims to report the preliminary clinical outcomes of a new procedure using open microwave ablation for the treatment of spinal metastases.
Materials and Methods: Fourteen patients with spinal metastases were treated by open microwave ablation between January 2006 and December 2008. Effectiveness was evaluated using Visual Analog Scale (VAS) and Frankel Classification of Neurological Deficit during the one-month postoperative follow-up.
Results: These procedures were technically successful in all bone lesions. Mean VAS scores during the one-month postoperative follow-up significantly improved compared to preoperative scores (2.3 ± 1.2 vs. 5.1 ± 1.5, P<0.05). Among the six patients who presented with preoperative neurological dysfunction, five patients recovered after surgery; while the remaining patients did not obtain any recovery. Among the eight patients without preoperative neurological dysfunction, one patient complained of loss of total sensory and motor function in the unilateral lower extremity after surgery. However, this was almost restored to normal after three weeks. Local recurrence was observed in three patients. Delayed wound healing occurred in one patient, but eventually healed after a secondary surgery. No wound infection or failure of fixation was observed in any patient. Six patients died after the operation, in which four cases died of primary diseases, and two cases died of cerebral bleeding and pneumonia, respectively.
Conclusions: Open microwave-induced in situ ablation is potentially a promising technique for the treatment of spinal metastases. Further studies with a larger number of samples and longer-term followups are warranted.
Keywords
Microwave, Ablation, Spine, Metastasis, Tumor.
Introduction
Despite the increasing incidence of malignant tumors, there has been an associated improvement in treatments for tumors; resulting in the gradual extension of a patient’s life span [1]. However, the incidence of bone metastases has also increased, and the most common site of bone metastases is in the spine. Conventional therapeutic approaches mainly include analgesic agents, chemotherapy, hormonal therapy, and radiation therapy [2]. However, these approaches are unable to provide stability to an instable spinal column and relieve metastatic epidural spinal cord compression. In these cases, surgery is the optimal surgical selection for the resolution of intractable pain, neurological compromise, and overt or impending spinal instability in patients with spinal metastases. Nevertheless, simple decompression may cause the further destruction of spinal stability, and it cannot prevent the further progress of tumors [1,3,4]. With the continuous advancement and improvement of designs and materials for instruments, spinal surgery has becoming increasingly simple and reliable; and a more aggressive surgical treatment for spinal metastatic tumors has been considered to improve quality of life of a patient.
Furthermore, with the advancement of spondylectomy, the management of solitary spinal metastasis with a wellcontrolled primary lesion using total en bloc spondylectomy has been recommended, in order to obtain better local control [5-9].
Improvements in surgical techniques to the entire vertebral column have made it technically feasible to resect tumors that involve the spine at all levels. The development of spinal instrumentation fixation has allowed surgeons to reconstruct entire vertebral segments after surgery. However, surgical difficulty and profound wounds of en bloc spondylectomy, long operation time, and high blood loss has led to increased risk of death; and this is caused by severe bleeding due to injury to major vessels during perioperative periods [10]. Other severe complications such as paralysis, myocardial infarctions, renal transitory failures, pseudarthrosis, wound necrosis implant failure, and deep infection may also occur [10-12]. For extracompartmental tumors, only marginal or intralesional resection could be performed in patients. Bandiera et al. reported that among the 143 cases received en bloc spondylectomy, wide resection was conducted in 88 cases, marginal resection was conducted in 27 cases, and intralesional resection was conducted in 19 cases with respect to margins [10]. Mini-invasive techniques have been developed for the treatment of spinal metastasis such as radiofrequency ablation, laser ablation, and vertebroplasty [13-17]. Although these procedures could typically reduce pain and improve the quality of life patients, these did not prevent tumor invasion.
In our hospital, we have developed a new technique (namely, open microwave ablation) for the treatment of spinal metastases, in order to reduce surgical trauma and obtain satisfactory local control, adequate spinal decompression and good stability. With the successful application of the in situ ablation of malignant tumors for limb salvage in the extremities, this technique was tentatively applied to spinal metastasis. A relative standard procedure was established, which involved total or partial laminectomy with microwave induced hyperthermia ablation. In this study, we report the preliminary results of a consecutive series of 14 patients with spinal metastases treated with this technique.
Material and Methods
Patients
This study was registered and approved by the local ethics committee on January 2006. This committee approved the screening, treatment and follow-up of these patients. Furthermore, all subjects provided a signed written informed consent. All works were undertaken following the provisions of the Declaration of Helsinki.
Between January 2006 and December 2008, 14 consecutive patients with spinal metastases treated with open microwave-induced hyperthermia ablation at our hospital were included into this study. None of these patients received previous invasive treatment. Inclusion criteria were as follows: (1) patients with Tomita scores ≤ 8; (2) patients who presented with progressive aggravation of nerve dysfunction during the course of radiotherapy; (3) patients who suffered from unbearable high-intensity pain non-responsive to conservative treatment; (4) patients with tumors insensitive to radiotherapy; (5) patients with obvious spinal instability or pathologic vertebral body fracture; (6) preoperative spinal cord injury Frankel grade A or B. Exclusion criteria were as follows: (1) patients with Takuhashi scores >8; (2) patients with no obvious spinal instability; (3) patients who are sensitive to radiotherapy; (4) patients who had complete spinal cord injury or other metastasis; (5) patients who were unable to tolerate surgery due to severe medical complications; (6) patients who were unable to stay in the prone position for a long time; (7) patients who suffered from severe radiation injury or active infection at the incision site after radiotherapy. Clinical characteristics of all patients are summarized in Table 1. Among the patients, nine were male and five were female; and mean age of these patients was 58 ± 6 years (range, 44-73 years). The primary tumors were multiple myeloma (five cases), non-Hodgkin’s lymphoma (NHL, two cases), lung carcinoma (three cases), breast carcinoma (two cases), hepatocellular carcinoma (one case), and renal cell carcinoma (one case). Lesions were located in the thoracic spine (nine cases), lumbar spine (four cases), and thoracolumbar spine (one case). Eleven patients had the involvement of a single segment, while the remaining patients had the involvement of two adjacent segments. According to the Tomita surgical classification of vertebral tumors [18], anatomic sites of the tumors were intracompartmental in three cases and extracompartmental in 11 cases. The Tomita score ranged from 3-6, with a mean of 4.2 ± 1.0.
| Patient | Sex | Age (years) | Diagnosis | Levels | Tomita type | Tomita prognosis score | Operation time (min) | Intraoperative blood loss (mL) | Frankel grade preoperative | Frankel grade postoperative | Local recurrence | Status | Follow-up (months) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | F | 68 | Multiple myelomas | T8 | IV | 3 | 180 | 2200 | D | E | No | Death* | 21 |
| 2 | M | 68 | Multiple myelomas | T7-8 | VI | 4 | 170 | 1600 | E | E | Yes | Alive | 39 |
| 3 | F | 56 | Breast carcinoma | T5 | IV | 3 | 165 | 1200 | - | - | No | Alive | 50 |
| 4 | M | 73 | NHL | T8-10 | VII | 6 | 195 | 500 | A | A | No | Death∆ | 27 |
| 5 | M | 44 | Multiple myelomas | T9 | I | 4 | 125 | 400 | E | E | No | Alive | 33 |
| 6 | F | 57 | NHL | T11 | V | 5 | 215 | 2500 | C | D | Uncertain | DOD | 6 |
| 7 | M | 51 | Liver cell carcinoma | L1 | IV | 5 | 135 | 600 | E | E | No | DOD | 5 |
| 8 | M | 64 | Renal cell carcinoma | T10 | I | 3 | 150 | 600 | E | E | No | Alive | 36 |
| 9 | M | 59 | Lung carcinoma | L3 | IV | 5 | 140 | 800 | - | - | Yes | DOD | 7 |
| 10 | M | 60 | Lung carcinoma | T4 | II | 5 | 155 | 700 | E | D | No | DOD | 6 |
| 11 | M | 51 | Multiple myelomas | T10 | IV | 3 | 130 | 350 | B | D | No | Alive | 27 |
| 12 | F | 49 | Multiple myelomas | L4 | VI | 3 | 145 | 600 | - | - | No | Alive | 26 |
| 13 | F | 58 | Breast carcinoma | L3 | IV | 4 | 120 | 400 | - | - | Yes | Alive | 20 |
| 14 | M | 54 | Lung carcinoma | T11-12 | VII | 6 | 185 | 500 | E | E | No | Alive | 19 |
| NHL: Non-Hodgkin’s Lymphoma; DOD: Death of Disease. *Death of brain bleeding; ∆ Death of lung infection. | |||||||||||||
Table 1. Data of 14 patients who received microwave-induced ablation.
Microwave ablation device
The microwave ablation system was provided by Qiya Medical Instrument Co. (Nanjing, China). Each generator was capable of producing 70 W at 2.45 GHz. Each antenna was connected to a generator by a coaxial cable. The antenna was 3 mm in diameter and 15 cm in length (Figures 1a and 1b). Two antennas were used for a single vertebrae tumor.
Surgical procedure
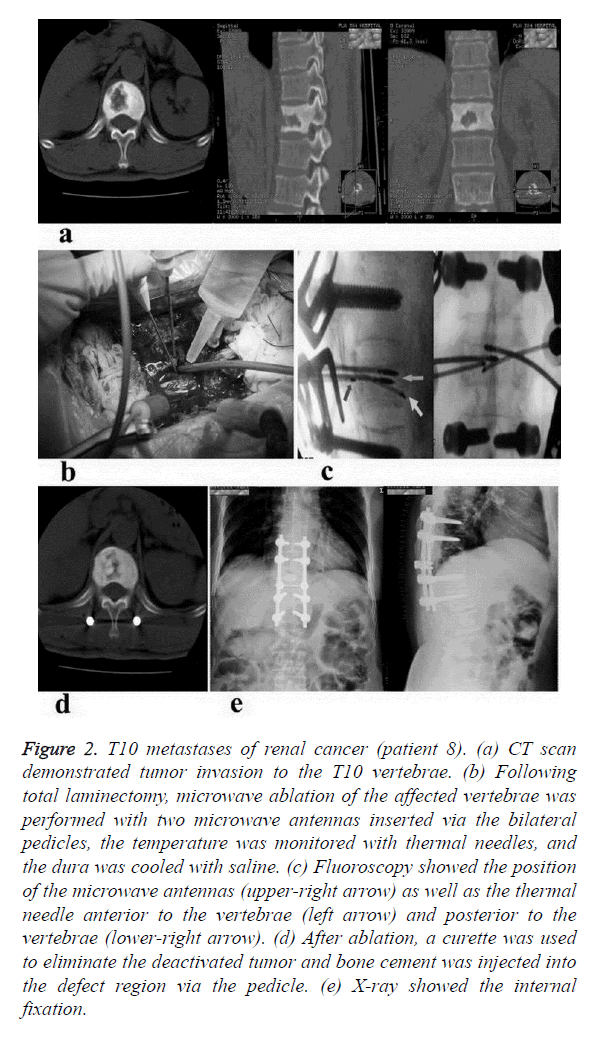
Prior to surgery, computed tomography (CT) scans were performed to determine the location and metastasis of the tumor (Figure 2a). Patients were placed in a prone position under general anesthesia. After a dorsomedial skin incision, paraspinal muscles were detached from the spinous processes, the laminae and the facet joints. Bilateral lamina fenestration or total laminectomy was then performed. The pedicles were enwrapped with cisplatin gauze, and two thermal needles were placed posterior to the vertebrae (to monitor the temperature of the anterior region of the canal) and anterior to the vertebrae (to monitor the temperature of the pleuroperitoneum). Two microwave antennas were inserted via bilateral pedicles (Figures 2b and 2c). Under the guidance of fluoroscopy, the angles and depths of the antennas were adjusted according to the location and range of the tumor; and these were changed one to two times when the tumor had an oversized diameter.
Figure 2. T10 metastases of renal cancer (patient 8). (a) CT scan demonstrated tumor invasion to the T10 vertebrae. (b) Following total laminectomy, microwave ablation of the affected vertebrae was performed with two microwave antennas inserted via the bilateral pedicles, the temperature was monitored with thermal needles, and the dura was cooled with saline. (c) Fluoroscopy showed the position of the microwave antennas (upper-right arrow) as well as the thermal needle anterior to the vertebrae (left arrow) and posterior to the vertebrae (lower-right arrow). (d) After ablation, a curette was used to eliminate the deactivated tumor and bone cement was injected into the defect region via the pedicle. (e) X-ray showed the internal fixation.
Ablation continued for 15-30 minutes with the central tumor temperature reaching 60-100°C. The temperature of the monitoring sites was controlled below 40°C. The dura was cooled from the outside with saline during ablation. Somatosensory evoked potential P40 was used to monitor the function of the nerve and spinal cord. After ablation, the curette was used to eliminate deactivated tissues via the pedicle. Combined vertebroplasty or bone grafts of the affected vertebra (Figure 2d) and internal fixation with the posterior instrumentation at one or two levels below and above the affected vertebra (Figure 2e) were performed to reconstruct of the spine. Fixation length was determined individually to achieve local stabilization. Postoperative management was characterized by wound drainage for 2-3 days. Patients were mobilized after one week, and were instructed to wear a thoracolumbar orthosis for 2-3 months.
Adjuvant treatment
All patients who suffered from myelomas received standard chemotherapy of CHOP, postoperatively. For patients with NHL, case four received four courses of chemotherapy of M2, and was discharged from the hospital because the patient refused further treatment. Furthermore, case six received four courses of chemotherapy in another hospital with unknown medications. In patients who suffered from spinal metastases derived from parenchyma organs, all patients, except for case seven, received chemotherapy and immunomodulatory therapy after the operation; while case seven refused any further treatment and was discharged.
Outcome evaluation and follow-up
Patients were followed-up preoperatively at one, three and six months, 12 months after the procedures, and every six months, thereafter. Pain was scored according to visual analog scale (VAS; range, 0-10) [19]. A higher VAS score equates to a higher level of pain. Neurological status was evaluated according to the Modified Frankel Classification of Neurological Deficit [20]. Pain and neurological scores were recorded after one month, postoperatively. Plain radiographs, CT, magnetic resonance imaging (MRI), or a combination of these modalities were used to detect local recurrence, and evaluate spinal stability in all patients.
Statistics analysis
All analyses were performed using SPSS 13.0 software (SPSS, Chicago, IL, USA). Pre- and post-operative VAS scores were analyzed using paired t-test. P<0.05 was considered statistically significant.
Results
Perioperative outcomes
Technical success was defined as the ability to successfully place the microwave antenna shaft in the center of the lesion and perform compete microwave tumor ablation. These procedures were technically successful in all bone lesions. Mean operative time was 158 ± 23 minutes, which ranged within 120-215 minutes. Mean blood loss during the operation was 925 ± 543 ml, which ranged within 350-2,200 ml.
Prognosis
Six patients died after the operation, among which four cases died of primary diseases including two cases of lung carcinomas, one case of NHL, and one case of hepatocellular carcinoma; while the remaining two patients died of cerebral bleeding and pneumonia, resulting from primary multiple myeloma and NHL, respectively. The life span of the patients that died ranged from 5-27 months after surgery, with an average of 12 ± 8 months. Follow-up time for the eight surviving patients ranged from 19-50 months.
Pain control
Before surgery, all patients complained of back or lower back pain, while six patients also reported combined unilateral or bilateral lower extremity pain. Furthermore, preoperative mean VAS score was 5.1 ± 1.5 (range, 3-9); while follow-up postoperative mean VAS score after one month was 2.3 ± 1.2 (P<0.05, Table 2). Pain relief was achieved in all but one patient.
| Patient | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | Average | |
| VAS score | Pre-operation | 5 | 3 | 4 | 6 | 3 | 9 | 6 | 4 | 5 | 8 | 3 | 5 | 7 | 4 | 5.1 ± 1.5* |
| Post-operation | 2 | 2 | 1 | 3 | 1 | 3 | 5 | 1 | 6 | 3 | 1 | 1 | 2 | 1 | 2.3 ± 1.2 | |
| *P<0.001. | ||||||||||||||||
Table 2. VAS pain score of the 14 patients.
Neurological function
Before surgery, six patients presented with neurological dysfunction, while the other eight patients did not show any neurological dysfunction. In patients presenting with preoperative neurological dysfunction, five patients recovered after surgery, and the remaining patients did not obtain any recovery due to nerve injury caused by the operation. Among patients had no such neurological dysfunction before surgery, seven patients did not exhibit any neurological symptom after surgery, while the remaining patients complained of loss of total sensory and motor function in the unilateral lower extremity at 24 hours after the operation, which were almost restored to normal after three weeks.
Local control
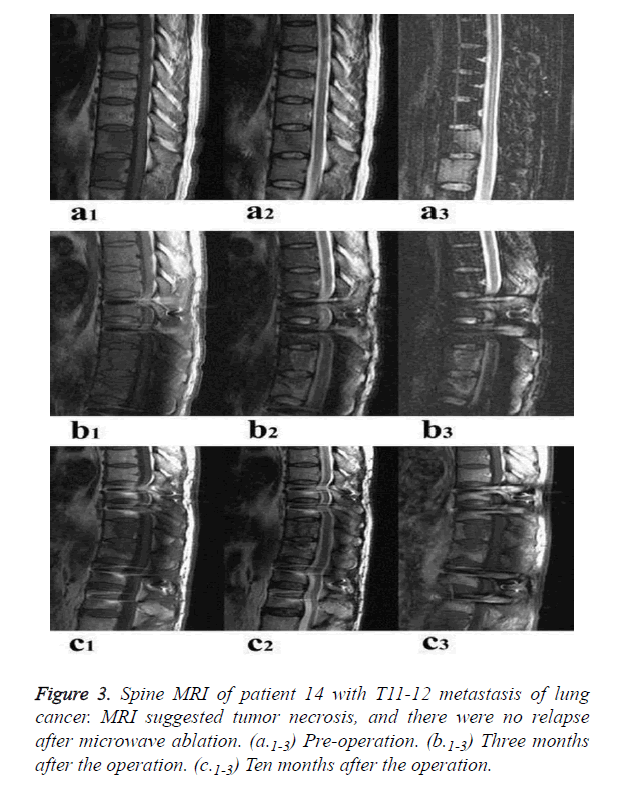
No peri-procedural death was observed. During the follow up period, tumor recurrence was noted in three patients at three, seven and fifteen months after the operation, respectively. No evidence of recurrence was noted in the remaining ten patients. Case six died of multiple metastases; hence, recurrence could not be confirmed due to lack of imaging diagnosis. Figures 3a-3c shows the spinal MRI image of a patient from the operation to 10 month after the operation.
Complications
Delayed wound healing caused by fat liquefaction occurred in one patient (case eight), and was eventually healed after a secondary surgery. No wound infection or failure of fixation was observed in any patient.
Discussion
Microwave ablation has been recognized as an effective therapy to treat tumors such as liver and renal tumors [21-23]. In recent years, there has been growing interest in microwave ablation for tumors in the lung, breast and bones [24-26]. The potential benefits of microwave technology include consistently higher intratumoral temperatures, larger tumor ablation volumes, faster ablation times, ability to use multiple applicators, improved convection profile, optimal heating of cystic masses, and less procedural pain. The use of an inserted microwave antenna can cause a coagulated necrosis zone with a diameter of more than 3 cm in the tumor, while a larger tumor coagulated necrosis can be achieved with the use of multiple microwave antennas.
Spinal metastases are a significant source of morbidity in patients with systemic cancer. To our knowledge, there have been few reports on the use of microwave ablation of metastatic spinal lesions [26-28]. Recently, we have developed a relatively fixed surgical procedure using this technology for the treatment of metastatic spinal tumors. In the present study, we found that microwave-induced ablation was similar to common decompression and internal fixation in terms of surgical trauma, average operative time, and bleeding; and this was superior to that of en bloc spondylectomy. This less invasive procedure is relatively simple and causes no serious complications. The recovery of neurological dysfunction and pain release were achieved in all patients except for one case of preoperative spinal cord injury. Early postoperative functional exercise, even walking, could be performed due to the largely retained integrity of the spine and reliable fixation; leading to an obvious improvement in patients’ quality of life. Local recurrences were found in three of 14 patients, with an acceptable local control rate.
The key to achieve complete tumor ablation is the insertion of bilateral microwave antennas. In this study, microwave antennas were placed under the guidance of fluoroscopy, which depended on the surgeon’s understanding of the imaging data and pedicle screw insertion technique. Our future direction was to design and manufacture an individualized digital navigation template by reverse engineering and 3D printing technique, in order to guide the positioning and insertion of microwave antennas during surgery. For small lesions, single-point ablation can reach the treatment targets. However, for lesions involving whole vertebral bodies or with concomitant paraspinal neoplasms, the angles and depth of these antennas should be adjusted to achieve multi-point ablation.
In clinical practice, spinal cord and nerve root protection were vitally important. In this study, we adopted a series of measurements to reduce the risk of an absolute minimum by adequate spinal canal decompression, exposure of the dura sac and nerve root, continuous intraspinal saline lavage during ablation, real-time monitoring of intraspinal temperature, and separation of the dural and posterior wall of the vertebrae, as much as possible. The distance between the microwave antennas and posterior wall of the vertebrae and pedicle root portion should be no less than 2 cm. This study has several limitations. This study had a small sample size. In addition, this study lacks of a control group, since no other thermal ablation techniques (e.g. RF ablation) were examined. Furthermore, this study was a retrospective study; thus, a prospective study is warranted.
Conclusion
The combination of laminectomy, decompression, internal fixation and microwave-induced ablation is a safety and effective method for the treatment of spinal metastatic carcinoma in terms of pain relief, nerve function improvement, and the local control of short-term tumor recurrence. Future studies with larger numbers of cases and longer-term follow-ups are required.
References
- Bartels RH, van der Linden YM, van der Graaf WT. Spinal extradural metastasis: review of current treatment options. CA Cancer J Clin 2008; 58: 245-259.
- Smith HS. Painful osseous metastases. Pain Physician 2011; 14: E373-403.
- Swift PS. Radiation for spinal metastatic tumors. Orthop Clin North Am 2009; 40: 133-144.
- Sciubba DM, Nguyen T, Gokaslan ZL. Solitary vertebral metastasis. Orthop Clin North Am 2009; 40: 145-154.
- Tomita K, Kawahara N, Murakami H, Demura S. Total en bloc spondylectomy for spinal tumors: improvement of the technique and its associated basic background. J Orthop Sci 2006; 11: 3-12.
- Melcher I, Disch AC, Khodadadyan-Klostermann C, Tohtz S, Smolny M, Stockle U. Primary malignant bone tumors and solitary metastases of the thoracolumbar spine: results by management with total en bloc spondylectomy. Eur Spine J 2007; 16: 1193-1202.
- Aryan HE, Acosta FL, Ames CP. Two-level total en bloc lumbar spondylectomy with dural resection for metastatic renal cell carcinoma. J Clin Neurosci 2008; 15: 70-72.
- Liljenqvist U, Lerner T, Halm H, Buerger H, Gosheger G, Winkelmann W. En bloc spondylectomy in malignant tumors of the spine. Eur Spine J 2008; 17: 600-609.
- Rao G, Suki D, Chakrabarti I, Feiz-Erfan I, Mody MG, McCutcheon IE. Surgical management of primary and metastatic sarcoma of the mobile spine. J Neurosurg Spine 2008; 9: 120-128.
- Bandiera S, Boriani S, Donthineni R, Amendola L, Cappuccio M, Gasbarrini A. Complications of en bloc resections in the spine. Orthop Clin North Am 2009; 40: 125-131.
- Boriani S, Biagini R, De Iure F, Bertoni F, Malaguti MC, Di Fiore M. En bloc resections of bone tumors of the thoracolumbar spine. A preliminary report on 29 patients. Spine (Phila Pa 1976) 1996; 21: 1927-1931.
- Krepler P, Windhager R, Bretschneider W, Toma CD, Kotz R. Total vertebrectomy for primary malignant tumours of the spine. J Bone Joint Surg Br 2002; 84: 712-715.
- Gronemeyer DH, Schirp S, Gevargez A. Image-guided radiofrequency ablation of spinal tumors: preliminary experience with an expandable array electrode. Cancer J 2002; 8: 33-39.
- Halpin RJ, Bendok BR, Liu JC. Minimally invasive treatments for spinal metastases: vertebroplasty, kyphoplasty, and radiofrequency ablation. J Support Oncol 2004; 2: 339-351.
- Ahn H, Mousavi P, Chin L, Roth S, Finkelstein J, Vitken A. The effect of pre-vertebroplasty tumor ablation using laser-induced thermotherapy on biomechanical stability and cement fill in the metastatic spine. Eur Spine J 2007; 16: 1171-1178.
- van der Linden E, Kroft LJ, Dijkstra PD. Treatment of vertebral tumor with posterior wall defect using image-guided radiofrequency ablation combined with vertebroplasty: preliminary results in 12 patients. J Vasc Interv Radiol 2007; 18: 741-747.
- Gevargez A, Groenemeyer DH. Image-guided radiofrequency ablation (RFA) of spinal tumors. Eur J Radiol 2008; 65: 246-252.
- Tomita K, Kawahara N, Baba H, Tsuchiya H, Fujita T, Toribatake Y. Total en bloc spondylectomy. A new surgical technique for primary malignant vertebral tumors. Spine (Phila Pa 1976) 1997; 22: 324-333.
- Wei Z, Zhang K, Ye X, Yang X, Zheng A, Huang G. Computed tomography-guided percutaneous microwave ablation combined with osteoplasty for palliative treatment of painful extraspinal bone metastases from lung cancer. Skeletal Radiol 2015; 44: 1485-1490.
- Frankel HL, Hancock DO, Hyslop G, Melzak J, Michaelis LS, Ungar GH. The value of postural reduction in the initial management of closed injuries of the spine with paraplegia and tetraplegia. I. Paraplegia 1969; 7: 179-192.
- Poggi G, Montagna B, P DIC, Riva G, Bernardo G, Mazzucco M. Microwave ablation of hepatocellular carcinoma using a new percutaneous device: preliminary results. Anticancer Res 2013; 33: 1221-1227.
- Yu J, Liang P, Yu XL, Cheng ZG, Han ZY, Mu MJ. Local tumour progression after ultrasound-guided microwave ablation of liver malignancies: risk factors analysis of 2529 tumours. Eur Radiol 2015; 25: 1119-1126.
- Cristescu M, Abel EJ, Wells S, Ziemlewicz TJ, Hedican SP, Lubner MG. Percutaneous Microwave Ablation of Renal Angiomyolipomas. Cardiovasc Intervent Radiol 2016; 39: 433-440.
- Sun YH, Song PY, Guo Y, Sheng LJ. Computed tomography-guided percutaneous microwave ablation therapy for lung cancer. Genet Mol Res 2015; 14: 4858-4864.
- Zhou W, Jiang Y, Chen L, Ling L, Liang M, Pan H. Image and pathological changes after microwave ablation of breast cancer: a pilot study. Eur J Radiol 2014; 83: 1771-1777.
- Pusceddu C, Sotgia B, Fele RM, Melis L. Treatment of bone metastases with microwave thermal ablation. J Vasc Interv Radiol 2013; 24: 229-233.
- Kastler A, Alnassan H, Pereira PL, Alemann G, Barbe DA, Aubry S. Analgesic effects of microwave ablation of bone and soft tissue tumors under local anesthesia. Pain Med 2013; 14: 1873-1881.
- Kastler A, Alnassan H, Aubry S, Kastler B. Microwave thermal ablation of spinal metastatic bone tumors. J Vasc Interv Radiol 2014; 25: 1470-1475.