Research Article - Biomedical Research (2017) Volume 28, Issue 17
Impact of cone beam computed tomography on measurement of bone density in periapical lesions following endodontic treatment: A clinical study
Adel S Alagl1, Sumit Bedi2*, Khalid Hassan1 and Zainab AL-AQL3
1Department of Preventive Dental Sciences, Division of Periodontics, College of Dentistry, University of Dammam, Dammam, Saudi Arabia
2Department of Preventive Dental Sciences, Division of Paediatric Dentistry, College of Dentistry, University of Dammam, Dammam, Saudi Arabia
3Dental Services, King Abdulaziz Medical City, National Guard Health Affairs, Jeddah, Saudi Arabia
- *Corresponding Author:
- Sumit Bedi
Department of Preventive Dental Sciences
College of Dentistry
University of Dammam, Saudi Arabia
Accepted on August 18, 2017
Abstract
Objective: Differences in bone density before and after regenerative endodontic treatment were examined in teeth with periapical lesions in Hounsfield Units (HUs) by using Cone Beam Computed Tomography (CBCT).
Materials and Methods: 8 patients requiring regenerative endodontic treatment for periapical lesions underwent CBCT scan before and after treatment. Their bone densities were calculated in HUs. The study included 10 teeth with periapical lesions measuring 4-8 mm in diameter. HU measurements were taken from an area of 3.5 mm2 on the CBCT images where the bone density was lowest, before and after treatment. The initial and final measurements were compared statistically by using paired samples statistics at the 5% significance level.
Results: There was a highly significant difference between the initial and final measurements (HU values) before and 1 year after endodontic treatment (P<0.0001).
Conclusion: We conclude that CBCT is a valuable tool for detecting the bone growth in periapical lesions.
Keywords
Bone density, Cone-beam computed tomography, Hounsfield units, Regenerative endodontics, Platelet-rich plasma, Mineral trioxide aggregate.
Introduction
Dental pulp necrosis can occur as a result of caries, trauma, and operative dental procedures and often comprises of a mixed, predominantly gram-negative, anaerobic bacterial flora [1]. These infections generally result in development of an immune response in the periapical region, and microbial infection can cause the development of periapical lesions that cause periapical bone destruction [2]. The clinical management of these young permanent teeth periapical pathology along with arrested root development poses a great challenge for dentists [3,4]. The healing of large periapical lesions might be stimulated by simple nonsurgical treatment by using proper infection control [5]. Earlier calcium hydroxide [4] has been used successfully for such conditions, which intended at formation of an apical hard tissue barricade after multiple visits used to be the treatment of choice for such cases. But with the advent of Mineral Trioxide Aggregate (MTA); a synthetic wall in apical third of root can be formed promptly with a very high success rate as proven by many milestone studies [6-8] and this has reduced the number of required clinical sessions also. But the disadvantage common to both calcium hydroxide and MTA is that they do not allow the continuance of root development resulting in thin dentin walls and hence a feeble root structure [9,10]. The clinical management of such a perplexing clinical challenge is to tickle the regenerative tissues of the pulpdentine complex to complete apical development [3]. Lately, various authors have reported the concept of regenerative endodontics [10-12]. Recent advances in tissue engineering focus upon three key essentials for tissue regeneration: the adult stem cells, the signalling molecules and a 3-dimensional physical scaffold that can sustain cell growth and differentiation. Recently, Platelet-Rich Plasma (PRP) has been mentioned as a source for growth factors and potentially ideal scaffold for regenerative endodontic treatment regimens due to their recognition to maintain vitality of pulp tissues by promoting cell growth and transport of growth factors in a disinfected environment [13-16]. Various case series and in vivo studies have been published in literature concerning platelet-rich plasma for successful regeneration in endodontics [14,15,17,18]. The periapical radiograph is an important instrument for assessing periapical bone pathology [19]. Many studies have indicated that periapical radiography is not a permanent reflection of the presence of a lesion and does not reflect the real size of the lesion or its spatial relationship to anatomical structures [20,21]. Moreover, radiographs are 2- dimensional representations of 3-Dimensional (3D) structures, and certain clinical features might not be reflected in radiographic changes [22].
Recently, new imaging modalities have been developed for use as diagnostic tools in dental radiology. In particular, Cone Beam Computed Tomography (CBCT) provides detailed highresolution images of oral structures and enables the early detection of bone lesions [23,24]. CBCT has been used in many clinical and nonclinical investigations [25]. It has been reported that CBCT can be used to differentiate between cysts and granulomas on the basis of the marked difference in density between the contents of the cyst cavity and granulomatous tissue [26,27]. In the periapical region, the mineral density can be measured in Hounsfield Units (HUs) by using CBCT [28]. This study was conducted to examine changes in bone density in periapical lesions before and after regenerative endodontic treatment by using CBCT.
Materials and Methods
In our study (approved by the University of Dammam Institutional review board; IRB-2014-02-029), 8 healthy patients with young permanent single rooted teeth were selected from patients attending the outpatient dental clinics at College of Dentistry, University of Dammam. None of the teeth had been previously treated for necrosis. A written informed consent was acquired from the patients/guardians. Institutional ethical clearance was obtained for this study.
Inclusion criteria for teeth were as follows:
1. Pulp necrosis, with periapical lesions and an immature apex
2. Probability of restoration of teeth.
3. No pathologic mobility, ankylosis, root fracture, or probing depths>3 mm
4. Patients not allergic to medicaments and antibiotics necessary to complete procedure.
Pulp necrosis was tentatively diagnosed by dental history and clinical examination, which included electric pulp testing and cold testing. Clinical signs and symptoms such as pain, swelling, fistula, and sensitivity to percussion and palpation were also noted. From 8 patients, 10 non-vital young permanent teeth with either apical periodontitis or abscesses showing negative response on pulp testing were included. The samples with periapical lesions were grouped according to the size of the lesion as follows:
Score 1: Periapical radiolucency˃3-5 mm
Score 2: Periapical radiolucency˃6-8 mm
Score 3: Periapical radiolucency˃8 mm
Treatment procedures
Regenerative treatment was performed according to the American Association of Endodontists protocol. Under rubber dam isolation, access opening was made followed by copious, gentle irrigation with 1.5% sodium hypochlorite and the working length was determined by using a size 20 sterile Kfile. Canals were not instrumented but were irrigated copiously with 2.5% NaOCl (20 ml), sterile saline (20 ml), and 0.12% chlorhexidine (10 ml) and dried with sterile paper points. After sealing the pulp chamber with a dentin bonding agent to minimize the risk of staining, 1:1:1 of metronidazole, ciprofloxacin, and minocycline were pulverized and mixed with distilled water to a final concentration of 0.1 mg/ml till a creamy paste was formed which was placed in the canal below the Cemento-Enamel Junction (CEJ) to minimize crown staining, by using a lentulo-spiral in a slow-speed handpiece. Canals were sealed with reinforced with zinc oxide-eugenol cement and patients were recalled after 3 weeks. In cases of any persistent signs and symptoms of infection (i.e. purulent drainage, failure to resolve pain, swelling, fistula, and sensitivity to percussion and palpation), triple antibiotic paste was applied again.
After 3 w, response to initial treatment was assessed and if asymptomatic, antibiotic paste was removed by copious irrigation with 20 ml of 17% EDTA and normal saline followed by drying of canals with sterile paper points and PRP scaffold was created.
Platelet rich plasma was prepared according to Dohan et al [29]. PRP was combined with equal volumes of sterile saline solution containing 10% calcium chloride and sterile bovine thrombin (100 U/ml) to achieve coagulation. The PRP was then injected into the root canal up to the level of CEJ. The final restoration was completed with white MTA, glass ionomer cement, and composite resin during the same visit to achieve coronal seal.
For all the cases, CBCT (I-Cat; Imaging Sciences International, Hatfield, PA) was used to take images of the maxillofacial area at a setting of 120 kVp and 3-7 mA. Images were obtained with an exposure time of 9 s. Each scan was taken over 360º with a voxel size of 0.3 mm. The lesion size, bone density (HU) and root length were measured.
Bone density evaluation from CBCT images
Clinical and radiographic follow-up examinations were done every three months during a 12-month follow-up period by the same Paediatric dentist for all cases. Treated teeth were examined for vitality by using both electric pulp and cold testing, and were recorded as responding positively to vitality testing only if they responded positively to both tests.
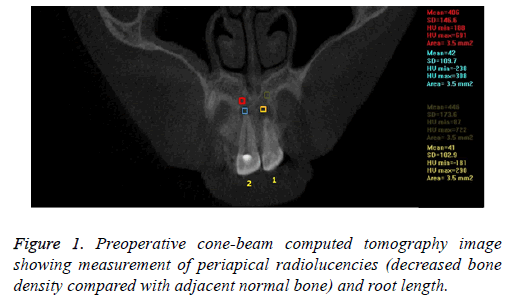
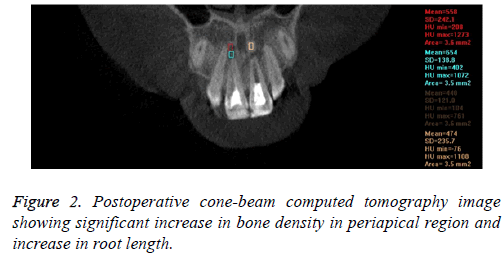
In the pre-treatment CBCT images, the bone density (HU) was recorded from CBCT coronal plane images with a 3.5 mm2 area in periapical lesion areas and normal adjoining bone regions (Figure 1). Post treatment CT scan was done at 3 months and then repeated at least every 3-12 months depending on circumstances. At the end of 12 months, CBCT images were independently evaluated for lesion size, periapical healing and apical closure (Figure 2). The mean of the values of the initial and final measurements for bone density (HU) is shown in Table 1.
| Case | Initial | Follow up |
|---|---|---|
| 1 | 65 | 405 |
| 2 | 135 | 535 |
| 3 | 67 | 311 |
| 4 | 53 | 461 |
| 5 | 161 | 555 |
| 6 | 121 | 612 |
| 7 | 83 | 475 |
| 8 | 142 | 562 |
| 9 | 111 | 595 |
| 10 | 215 | 613 |
Table 1. Measurements of bone density in Hus.
The same pediatric dentist re-examined all the radiographs 1 month after the preliminary examination and intra-examiner validity was assessed by using kappa statistics. Kappa values ranged from 0.8 to 1, signifying good reliability.
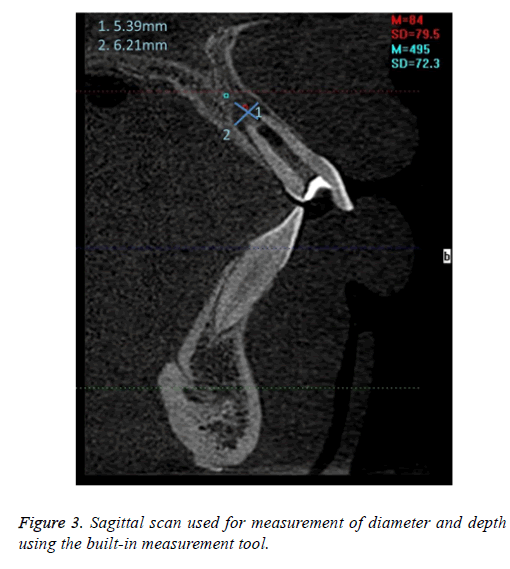
CBCT measurements were obtained with Dolphin 3D imaging (Dolphin Imaging Systems, California) on a computer workstation running under Microsoft Windows XP professional SP-1 (Microsoft Corp, Redmond, WA). Sagittal view CBCT images were evaluated to measure lesion size using the built-in measurement tools (Figure 3).
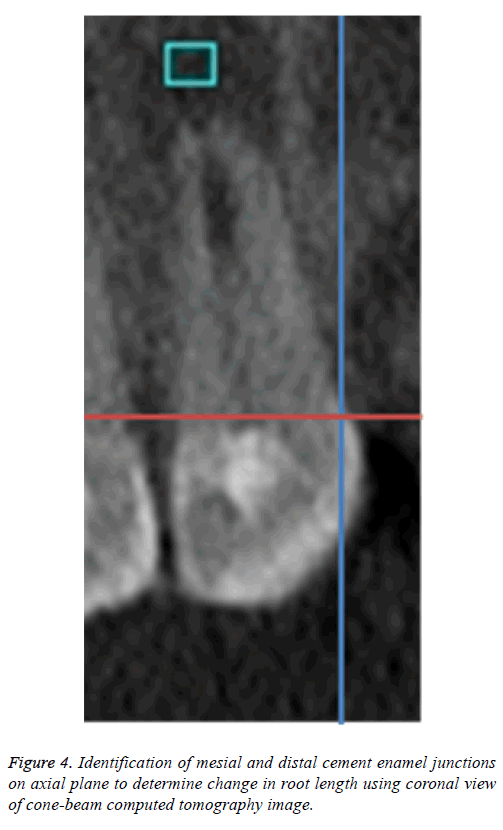
To standardize the root length measurements, the alignment for each sample was attuned using coronal view; the sagittal and axial planes were adjusted to intersect on CEJ (Figure 4). Root length was measured along the long axis of each tooth from the most apical point of the root perpendicular to the line connecting the mesial and distal CEJs as a measure of standardization [30] to determine the change in the length postoperatively.
Statistical analysis
Data was analysed using the Statistical Package for Social Sciences version 13 (SPSS, Chicago, IL). CBCT data were assessed by using Paired t-test with P value<0.001 considered to be statistically significant.
Results
The initial and final measurements were compared statistically, and the results are shown in Table 2. For the bone density assessment of the periapical lesions, the lowest bone density value before endodontic treatment was +53 HUs, and the highest bone density after endodontic treatment was +613 HUs. There was a significant relationship between the values before and 1 year after endodontic treatment (P<0.001).
| n=10 | Mean | Standard deviation | Standard error of the mean | 95% confidence interval of the difference | P value | |
|---|---|---|---|---|---|---|
| Lower | Upper | |||||
| Initial-final | -397.1 | 74.64 | 22.05 | -446.98 | -347.22 | ˂0.0001 |
Table 2. Comparison of bone densities (HUs).
Discussion
Periapical lesions may be detected with conventional radiography only 15 to 30 d after the development of the disease [31]. However, with advancements in technology like CBCT imaging, it is possible to detect periapical lesions as soon as 7 d after it develops. The detection and characterization of these lesions represent an important preoperative factor that may influence the outcome of root canal treatment; thus, early diagnosis is essential [4,5].
Liang et al. [31] reported that the 3D CBCT has been found to be more sensitive than periapical radiographs in detecting periapical lesion treatment outcomes. Bone density values from CBCT scans might not correspond to values obtained with periapical radiographs. This study was conducted to examine whether bone density values can be assessed in teeth with periapical lesions by using CBCT. To standardize the study, we enrolled patients with lesions measuring 4-8 mm in diameter. All of the endodontic treatments and bone density values were performed and measured by the same researcher. A region of 3.5 mm2 was measured by using CBCT. The lowest bone density values were recorded in HUs on scanning the periapical radiolucent area in the buccopalatal direction.
The HU is the numeric unit of an electron density [32] that is determined for each pixel of a conventional CT image. This value of the unit depends on the composition and nature of the tissue imaged and is used to represent the density of the tissue [33]. The Hounsfield index is an accepted, standardized scale to report reconstructed CT values and is a measure of X-ray attenuation that varies according to tissue density [34].
HUs can be used to distinguish tissue types, such as cysts from granulomas, e.g., air=-1000 HUs, water=0 HUs, muscle=+40 HUs, and bone $+400 HUs [21]. The Misch bone density classification is as follows: D1, dense cortical bone; D2, thick dense to porous cortical bone on the crest and coarse trabecular bone within; D3, thin porous cortical bone on the crest and fine trabecular bone within; D4, fine trabecular bone; D5, immature, non-mineralized bone [35].
Moreover, the Misch bone density classification might be evaluated on CT images on the basis of the correlation with a range of HUs; Dl corresponds to values>1250 HUs; D2, 850-1250 HUs; D3, 350-850 HUs; D4, 150-350 HUs; and D5, <150 HUs [24]. In this study, the HU values ranged from 53-613 HUs. Bone density D3 was very common in the maxilla [35]. After treatment, the bone tissue values of the HUs measured in our study corresponded to the classification of Rosenberg et al [33]. One year after treatment, 9 cases were D3 and one was D4 according to the Misch classification. The values increased over the course of the study. These results corroborate with the results of previous studies by Kaya et al. [36] and Venskutonis et al. [37].
To evaluate the healing of periapical lesions, Estrela et al. [24] successfully used CBCT and periapical index scoring to measure radiolucent areas. In the present study, instead of using an index to score bone density, we quantitatively measured bone density. With successful regenerative endodontic treatment, a decrease in the size of the radiolucent area and bone tissue healing with increased root length and apical closure were observed. All these parameters could be evaluated with the 3D CBCT. Diederichs et al. [38] reported the measurement of bone density in HUs by CT. Moreover, conventional radiography should be used cautiously to identify periapical lesions because of the increased probability of an inaccurate diagnosis. In contrast, CBCT in endodontics is very accurate at detecting periapical lesions, even in the earliest stages, and in aiding with differential diagnosis [39].
The high radiation dosage is a major concern with the use of CBCT. The doses from CBCT are significantly lower than from conventional medical CT but higher than those from traditional views used in dentistry [40,41]. The reported exposure from a full-mouth series of analog radiographs (Dspeed) is 150 mSv [42]. In comparison, a roundtrip airplane flight from Paris to Tokyo exposes passengers to an effective dose of 139 mSv [43]. According to the manufacturer’s data, the radiation dose emitted in our study was 32 mSv (I-CAT scan time, 10 s). The radiation dose emitted in this study was similar to that from 3-4 intraoral radiographs. The advances in medical imaging and the use of techniques like filtering will further help us in understanding the details of the bone response in future [44].
Conclusion
Within the limitations of this study, CBCT is an effective method for quantitative measurement of bone density in periapical area of teeth requiring endodontic treatment and provides considerably more information for oral diagnostic purposes than traditional methods.
With 3D CBCT images, a significant decrease in the size of the radiolucent area, closure of root apex and bone tissue healing can be observed following endodontic treatment. Use of the advancements like noise reduction and filtering in the field of medical imaging will reveal more intricate details in future.
Conflict of Interest
The authors declare that there are no known conflicts of interest associated with this publication and there has been no financial support for this work that could have influenced its outcome.
Funding Acknowledgement
This research study was funded by Deanship of Scientific Research, University of Dammam, Grant #2014138.
References
- Sundqvist G. Taxonomy, ecology, and pathogenicity of the root canal flora. Oral Surg Oral Med Oral Pathol 1994; 78: 522-530.
- Sasaki H, Stashenko P. Interrelationship of dental pulp and apical periodontitis. Bender and Seltzers dental pulp. Chicago: Quintessence Publishing 2012; 277-300.
- Abbott P. Apexification with calcium hydroxide-when should the dressing be changed? The case for regular dressing changes. Aust Endod J 1998; 24: 27-32.
- Foreman PC, Barnes IE. Review of calcium hydroxide. Int Endod J 1990; 23: 283-297.
- Saatchi M. Healing of large periapical lesion: a non-surgical endodontic treatment approach. Aust Endod J 2007; 33: 136-140.
- Bakland L, Andreasen J. Will mineral trioxide aggregate replace calcium hydroxide in treating pulpal and periodontal healing complications subsequent to dental trauma? A review. Dent Traumatol 2012; 28: 25-32.
- Damle SG, Bhattal H, Loomba A. Apexification of anterior teeth: a comparative evaluation of mineral trioxide aggregate and calcium hydroxide paste. J Clin Pediatr Dent 2012; 36: 263-268.
- Chala S, Abouqal R, Rida, S. Apexification of immature teeth with calcium hydroxide or mineral trioxide aggregate: systematic review and meta-analysis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2011; 112: 36-42.
- Petrino JA. Revascularization of necrotic pulp of immature teeth with apical periodontitis. Northwest Dent 2007; 86: 33-35.
- Bose R, Nummikoski P, Hargreaves K. A retrospective evaluation of radiographic outcomes in immature teeth with necrotic root canal systems treated with regenerative endodontic procedures. J Endod 2009; 35: 1343-1349.
- Hargreaves K, Diogenes A, Teixeira F. Treatment options: biological basis of regenerative endodontic procedures. J Endod 2013; 39: 30-43.
- Huang G, Sonoyama W, Liu Y, Liu H, Wang S, Shi S. The hidden treasure in apical papilla: the potential role in pulp/dentin regeneration and bioroot engineering. J Endod 2008; 34: 645-651.
- Bezgin T1, Yilmaz AD, Celik BN, Sönmez H. Concentrated platelet-rich plasma used in root canal revascularization: 2 case reports. Int Endod J 2014; 47: 41-49.
- Sachdeva G, Sachdeva L, Goel M, Bala S. Regenerative endodontic treatment of an immature tooth with a necrotic pulp and apical periodontitis using platelet-rich plasma (PRP) and mineral trioxide aggregate (MTA): a case report. Int Endod J 2015; 48: 902-910.
- Torabinejad M, Turman M. Revitalization of tooth with necrotic pulp and open apex by using platelet-rich plasma: a case report. J Endod 2011; 37: 265-268.
- Hiremath H, Gada N, Kini Y, Kulkarni S, Yakub S, Metgud S. Single-step apical barrier placement in immature teeth using mineral trioxide aggregate and management of periapical inflammatory lesion using platelet-rich plasma and hydroxyapatite. J Endod 2008; 34: 1020-1024.
- Narang I, Mittal N, Mishra N. A comparative evaluation of the blood clot, platelet-rich plasma, and platelet-rich fibrin in regeneration of necrotic immature permanent teeth: A clinical study. Contemp Clin Dent 2015; 6: 63-68.
- Jadhav G, Shah N, Logani A. Revascularization with and without platelet-rich plasma in nonvital, immature, anterior teeth: a pilot clinical study. J Endod 2012; 38: 1581-1587.
- Barbat J, Messer HH. Detectability of artificial periapical lesions using direct digital and conventional radiography. J Endod 1998; 24: 837-842.
- van der Stelt PF. Experimentally produced bone lesions. Oral Surg Oral Med Oral Pathol 1985; 59: 306-312.
- Cotti E, Campisi G, Garau V, Puddu G. A new technique for the study of periapical bone lesions: ultrasound real time imaging. Int Endod J 2002; 35: 148-152.
- Molven O, Halse A, Fristad I. Long-term reliability and observer comparisons in the radiographic diagnosis of periapical disease. Int Endod J 2002; 35: 142-147.
- Arai Y, Tammisalo E, Iwai K, Hashimoto K, Shinoda K. Development of a compact computed tomographic apparatus for dental use. Dentomaxillofac Radiol 1999; 28: 245-248.
- Estrela C, Bueno MR, Azevedo BC, Azevedo JR, Pecora JD. A new periapical index based on cone beam computed tomography. J Endod 2008; 34: 1325-1331.
- Patel S, Dawood A, Pitt Ford T, Whaites E. The potential applications of cone beam computed tomography in the management of endodontic problems. Int Endod J 2007; 40: 818-830.
- Aggarwal V, Logani A, Shah N. The evaluation of computed tomography scans and ultrasounds in the differential diagnosis of periapical lesions. J Endod 2008; 34: 1312-1315.
- Simon JHS, Enciso R, Malfaz JM, Rogers R, Bailey-Perry M, Patel A. Differential diagnosis of large periapical lesions using cone-beam computed tomography measurements and biopsy. J Endod 2006; 32: 833-837.
- Sato I, Ando-Kurihara N, Kota K, Iwaku M, Hoshino E. Sterilization of infected root-canal dentine by topical application of a mixture of ciprofloxacin, metronidazole and minocycline in situ. Int Endod J 1996; 29: 118-124.
- Dohan DM, Choukroun J, Diss A, Dohan SL, Dohan AJ, Mouhyi J, Gogly B. Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part I: technological concepts and evolution. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2006; 101: 37-44.
- Sherrard JF, Rossouw PE, Benson BW, Carrillo R, Buschang PH. Accuracy and reliability of tooth and root lengths measured on cone-beam computed tomographs. Am J Orthod Dentofacial Orthop 2010; 137: 100-108.
- Liang YH, Li G, Wesselink PR, Wu MK. Endodontic outcome predictors identified with periapical radiographs and cone-beam computed tomography scans. J Endod 2011; 37: 326-331.
- Hatton J, McCurdy B, Greer PB. Cone beam computerized tomography: the effect of calibration of the Hounsfield unit number to electron density on dose calculation accuracy for adaptive radiation therapy. Phys Med Biol 2009; 54: 329-346.
- Rosenberg PA, Frisbie J, Lee J. Evaluation of pathologists (histopathology) and radiologists (cone beam computed tomography) differentiating radicular cysts from granulomas. J Endod 2010; 36: 423-428.
- Shapurian T, Damoulis PD, Reiser GM, Griffin TJ, Rand WM. Quantitative evaluation of bone density using the Hounsfield index. Int J Oral Maxillofac Implants 2006; 21: 290-297.
- Misch CE. Bone character: second vital implant criterion. Dent Today 1988; 7: 39-40.
- Kaya S, Yavuz I, Uysal I, Akkuay Z. Measuring bone density in healing periapical lesions by using cone beam computed tomography: a clinical investigation. J Endod 2012; 38: 28-31.
- Venskutonis T, Plotino G, Tocci L, Gambarini G, Maminskas J, Juodzbalys G. Periapical and endodontic status scale based on periapical bone lesions and endodontic treatment quality evaluation using cone-beam computed tomography. J Endod 2015; 41: 190-196.
- Diederichs CG, Engelke WG, Richter B, Hermann KP, Oestmann JW. Must radiation dose for CT of the maxilla and mandible be higher than that for conventional panoramic radiography? AJNR Am J Neuroradiol 1996; 17: 1758-1760.
- Estrela C, Bueno MR, Leles CR, Azevedo B, Azevedo JR. Accuracy of cone beam computed tomography, panoramic and periapical radiographic for detection of apical periodontitis. J Endod 2008; 34: 273-279.
- Ludlow JB, Davies-Ludlow LE, Brooks SL, Howerton WB. Dosimetry of 3 CBCT devices for oral and maxillofacial radiology: CB Mercuray, NewTom 3G and i-CAT. Dentomaxillofac Radiol 2006; 35: 219-226.
- Schulze D, Heiland M, Thurmann H, Adam G. Radiation exposure during midfacial imaging using 4- and 16-slice computed tomography, cone beam computed tomography systems and conventional radiography. Dentomaxillofac Radiol 2004; 33: 83-86.
- Frederiksen NL. X rays: what is the risk? Tex Dent J 1995; 112: 68-72.
- Bottollier-Depois JF, Trompier F, Clairand I. Exposure of aircraft crew to cosmic radiation: on-board intercomparison of various dosemeters. Radiat Prot Dosimetry 2004; 110: 411-415.
- Parsian A, Mehdi R, Noradin G. A hybrid neural network-gray wolf optimization algorithm for melanoma detection. Biomed Res 2017; 28: 3408-3411.