Research Article - Biomedical Research (2017) Volume 28, Issue 13
HIF-1α, Ki-67, CA153 and CEA levels in breast papillary carcinoma and as potential marker of Western medicine combined with traditional Chinese medicine therapy evaluation
Shu-Guang Yang1#, Fu-Hao Zhu2# and Gang-Ping Wang3,4*
1Department of Traditional Chinese Medicine, Rizhao People's Hospital, PR China
2Department of Pharmacy, Rizhao People's Hospital, PR China
3Department of Pathology, Rizhao People's Hospital, PR China
4Department of Clinical and Research Laboratory, Rizhao People's Hospital, PR China
#These authors contributed equally to this work and they are the co first author.
Accepted on May, 25, 2017
Abstract
Traditional Chinese Medicine (TCM), as an important component of complementary and alternative medicine, has become well known for its significant role in preventing and treating breast lesion with nipple discharge (ND). In the present study, the ND and serum biomarkers level of cancer antigen 153 (CA153) and carcinoembryonic antigen (CEA) were measured and compared with the express of Hypoxia inducible factor-1α (HIF-1α) and nuclear proliferation index Ki-67 in tissue in 93 breast intraductal papillary leision patients with ND, of which, 29 patients diagnosed with intraductal papillary carcinoma received Western medicine EC-P chemotherapy program combined with Runiu recipe TCM treatment. We investigated the significance of TCM as an adjunctive therapy and analyzed the relationship with the levels of biomarkers and clinical outcomes in papillary carcinoma. The results shown that the expression of HIF-1α and Ki-67 in breast papillary carcinomas were higher than those in benign lesions and positivity correlated significantly with ND levels of CA153 and CEA in papillary carcinomas (p<0.05, respectively). TCM combined with Western medicine can relieve the clinical Chinese medical symptoms of papillary carcinoma patients, and decrease CA153 and CEA levels (p<0.05, respectively). This study suggests that HIF-1α, Ki-67, CA153 and CA125 may be considered as potential marker of Western medicine EC-P chemotherapy program combined with TCM therapy evaluation for breast lesions with ND.
Keywords
Traditional Chinese medicine, Breast papillary carcinoma, Biomarker, Nipple discharge, Therapeutic effects.
Introduction
With development of medicine and update of knowledge, breast cancer including intraductal papillary carcinoma therapy has come into a diversified comprehensive treatment stage. In the world including Western countries, Traditional Chinese Medicine (TCM), as an important component of complementary and alternative medicine, has become well known for its significant role in preventing and treating breast lesion with nipple discharge (ND). As the breast mass and breast pain, ND is a relatively common breast complaint accounting for up to 5% of which women seek medical advice. Majority of ND comes forth spontaneously and has a pathological outcome. The most common cause of ND is intraductal papillary lesions, besides intraductal papilloma and intraductal papillary carcinoma. ND also can be the presenting symptom for other type of cancer. In China, breast lesions are different from Western countries, and more traditional herbal medicines are in using. There are no effective molecule biomarkers to evaluate therapeutic effects, especially markers of ND in breast cancer patients, which are rarely studied systemically.
Carcinoembryonic antigen (CEA) was identified as the first human cancer-associated antigen in colon carcinoma in 1965 [1] , and high levels of CEA have been found in ND from breast cancer patients [2], CEA is useful for diagnosis of recurrence and prognosis of breast cancer [3,4]. Cancer antigen 153 (CA153) was mainly used in the management of prognosis, metastasis and recurrence in breast cancer patients [5]. CEA and CA153 in serum were of great value in providing clinical details for the management of recurrence and metastasis for breast cancer. It was verified that high levels of serum CEA and CA153 were associated with a poor prognosis [6]. Preoperative levels of CEA and CA153 in serum were well known as a significant effect on prognosis of breast cancer [7]. However, serum CEA and CA153 levels showed no clinical benefit on diagnosis in patients with early or localised breast cancer [8]. There were some limitations of serum tumor markers in the diagnosis of breast cancer, such as relative limited sensitivity and specificity [8,9].
Breast ND are a promising source of biomarkers for the diagnosis of early-stage breast cancer [10,11]. The human mammary gland comprises discrete ductalalveolar systems in which the breast epithelium exfoliates cells and secretes fluids into the luminal compartment of the gland [10,11]. ND locates in or originates from mammary ducts and generally harbors a benign or malignant breast tumor. ND present in the ducts of nonlactaing women contains concentrated secreted proteins from the breast ductal epithelium [10,11]. Because of its unique cellular and biochemical component that reflect the true microenvironment of the alveolar-ductal system, ND have been recognized for several decades as a potential gold mine of biomarkers for the early diagnosis of breast cancer [12]. In addition, biochemical compounds of physiopathological interest in breast ductal secretions are found at higher concentrations than in the matched serum sample [12].
To investigate whether there are tumor markers in breast cancer with ND can serve as useful diagnostic and treatment biomarkers for breast cancer, for the present study, we examined Hypoxia inducible factor-1α (HIF-1α), nuclear proliferation index Ki-67, CA153 and CEA levels related with breast cancer and analysis their expressions both in serum and ND samples obtained from patients with benign and malignant breast intraductal papillary lesions, to study the relationship of biomarker levels and clinicopathological parameters and clinical outcomes in breast papillary carcinoma.
Materials and Methods
Diagnostic criteria
The pathological categorization was determined according to the current WHO classification system of breast tumor [13]. The pathological diagnosis was verified by histological methods and the results of the immunohistochemistry staining assay independently by two pathologists blinded to the subject’s clinical history. The pathological reading was determined for each biopsy slide with an overall pathological diagnosis determined for each subject. Papilloma with atypical ductal hyperplasia and ductal carcinoma in situ were characterized by the presence of a focal population of monotonous cells with the cytological and architectural features of low-grade ductal neoplasia. Extent and proportion criteria have been used to differentiate papilloma with atypical ductal hyperplasia from ductal carcinoma in situ within a papilloma. The extent cut-off according to some authorities was 3 mm; an intraductal papilloma with atypical ductal hyperplas was diagnosed when the atypical epithelial population was less than 3 mm, while ductal carcinoma in situ within a papilloma was diagnosed when this atypical population was equal or more than 3 mm.
Patients and treatment
By the prospective, randomized and controlled methods, 93 cases breast papillary lesions patients after surgical resection were treated with TCM. There were 70 cases of breast papillary lesions patients with ND in Rizhao People's Hospital (21 patients intraductal papillary carcinoma and 49 patients intraductal papilloma) and 23 cases in Rizhao Traditional Chinese Medicine Hospital (8 patients papillary carcinoma and 15 patients papilloma) from January 2004 to June 2012. Among them there were 29 cases intraductal papillary carcinoma and 64 cases papilloma. 29 patients diagnosed with breast intraductal papillary carcinoma received Western medicine therapy and combined with Runiu recipe TCM treatment. Their age ranged from 20 to 72 years, mean 51.6 year. The effectiveness of TCM of Runiu composition combined with Western medicine on papillary carcinoma after surgical resection was evaluated. The scores of quality of life (QOL) and Chinese medical syndrome were observed to evaluate the clinical effect. The serum and ND biomarkers levels of CA153 and CEA in mammary patients were detected and observed.
Western medicine neoadjuvant chemotherapy
29 patients with breast intraductal papillary carcinoma were treated with Western medicine EC-P chemotherapy program combined Runiu recipe TCM treatment. Western medicine neoadjuvant chemotherapy using EC*4 to P*4 chemotherapy program (EC for 4 cycles followed by paclitaxel chemotherapy for 4 cycles). Four cycles of Epirubicin 90 mg/m2 plus cyclophosphamide 600 mg/m2 q3w followed by four cycles of Paclitaxel 175 mg/m2 q3w (EC-P). Surgery was carried out within 2 weeks after last chemotherapy.
Runiu recipe recipe composition and oral usage
Recipe composition: Runiu recipe composition composed of Angelica, Radices paeoniae alba, radix bupleuri, Tuckahoe, Fried Atractylodes, Immature Tangerine Peel (Pericarpium Citri Reticulatae Viride), Rhizoma Cyperi, Aromatic Turmeric Root-tuber (Radix Curcumae), Tree Peony Bark, Fructus Gardeniae, Citron Fruit, Coix Seed, Morinda officinalis, cornua cervi degelatinatum and Licorice. In TCM theory, breast papillary carcinoma is due to depression of liver-qi, which is made by melancholia and angry , then depression of liver-QI transmits to heat to injury to blood vessels. Spleen is injured by thinking, failing to control blood to overflow from the breast orifice. Hyperactivity of liver fire boils body fluid to be sputum, which stasises with ouerflowing blood in breast to a lump. Radix bupleuri, immature tangerine peel, rhizoma cyperi and Fruit of Citron Fruit, disper stagnated liver qi and dredge channels; Aromatic Turmeric Root-tuber promotes blood circulation for removing blood stasis and relieve qi stagnation; radices, paeoniae alba and angelica nourish liver by nourishing blood; Tuckahoe, Fried Atractylodes and Coix Seed nourish qi to invigorate spleen for eliminating dampness; Tree Peony Bark can clear heat and cool blood, promote blood circulation for removing blood stasis; Morinda officinalis and cornua cervi degelatinatum reinforce kidney to warm yang and coordinate Chong and Conception Vessels. Treatment based on TCM theory differentiation, also called “Bian Zheng Shi Zhi”, is the comprehensive analysis of clinical information that is used to guide the choice of treatment with TCM Runiu recipe composition formulae.
Oral usage: The Patient’s accorded the usage: one dose daily, conventional water decoction, taking juice 400ml, drinking after breakfast and dinner warmly. A course of treatment was one month, taking continuously for 3 months, withdrawal during period.
Measurement of sample biomarkers
All samples were collected before any treatment initiated within 2 days after hospitalization. Nipple was cleansed first with alcohol swabs to remove cellular debris. Droplets of ND were collected in an Eppendorf tube. The tube was then stored in dedicated refrigerator at 4°C. Samples were transported to the laboratory department within 8 hours after collection. For biomarkers analysis, 3 ml of heparinized blood and at least of 0.5 ml of ND were drawn from each individual. The biomarker Concentrations of CA153 and CEA were quantitatively measured using electro chemiluminescence method (Roche E-601, Germany) according to manufacturer’s protocol in the clinical laboratory at local Hospital. The cut-off values for CA153 and CEA in serum were 25.00 U/mL and 3.40 ng/mL respectively, and those in ND were 35.00 U/mL and 9.8 ng/mL, respectively. Commercial reference control sera were used for quality control and calibration.
Immunohistochemistry
Breast tissue samples were fixed in 10% neutral buffered formalin and embedded in paraffin at 4°C for 24 h. Tissue sections at 5 μm thickness were deparaffinized and rehydrated using standard procedures. The specimens were examined under a binocular-dissecting microscope. Immunoreactions were processed using the Ultra SensitiveTM S-P Kit (Maixin- Bio, Fuzhou, Fujian, China) according to the manufacturer's instructions to detect differences in tumor tissue estrogen receptor (ER), progesterone receptor (PR), Ki-67, human epidermal growth factor receptor 2 (HER2/neu) and HIF-1α expression, and signals were visualized using the DAB substrate, which stains the target protein yellow. The pathological specimens were reviewed independently by two pathologists and the pathologists were blinded to the subject's clinical history, and the results of the immunohistochemistry staining assay. The immunoreactivity of positivity for HIF-1α was expressed as the percentage of cancer cells showing nuclear reactivity. For HIF-1α, cells were classified according to the positive rate and color intensity as follows: negative, number of positive cells <25%; positive, brown particles, number of positive cells ≥ 25%. For ER, PR, and Ki-67 expression, the percentage of cancer cells showing nuclear reactivity was recorded after inspection of all of optical fields at 200X, and the mean value was used to score each case. Tumors showing ER or PR expression in more than 10% of cancer cells were considered positive. For the Ki-67 proliferation index, patients were divided into two groups: those with a value between 1% and 14% and those with a value >14%. In addition, regarding the cell membrane reactivity of HER2/neu, oncoprotein expression was evaluated following a similar approach, and the mean value was used to score each case. Tumors expressing HER2/neu in more than 10% of the cancer cells medium to strong intensity expression of complete cell membrane, not only part of the cell membrane, were considered as positive.
Clinicopathological analyses
For analyses, we considered the age at diagnosis, tumor size, status of ER, PR, HER2/neu, HIF-1α expression, and the Ki-67 index in relation to the CA153 and CEA levels in serum and ND. Patients assessed for the expression of ER, PR, HER2/neu and HIF-1α were grouped as positive or negative. Regarding the Ki-67 proliferation index, patients were divided into two groups: those with a Ki-67 proliferation index between 1% and 14% and those with proliferation index >14%.
Statistical analysis
SPSS 17.0 statistical software was used to analyze the data. Enumeration data with chi-squared test (χ2 test) or Fisher’s exact tests, as appropriate. The biomarker concentrations of CA153 and CEA were expressed as the mean and standard deviation (SD). The nonparametric Mann-Whitney U-test was used to determine differences between two groups. The relationship of this dichotomous variable to other clinicopathological correlates was established using χ2 or Fisher’s exact tests, as appropriate. All p values were twosided, p<0.05 was considered statistically significant.
Results
Comparison of serum and ND biomarker levels between papilloma and papillary carcinoma subgroups
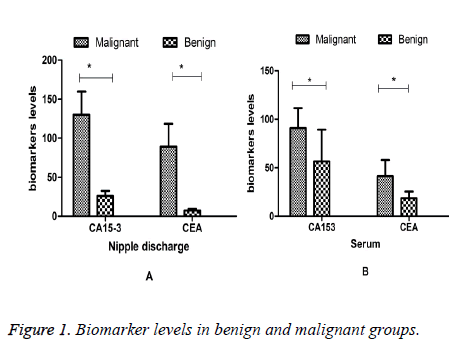
The clinical biological information of a series of 93 cases of breast papillary lesions with ND was summarized in Table 1. Duration of the disease of 57 cases was 2 months to 3 years and 36 cases were less than 2 months. There were significant differences between different discharge color subgroups (p<0.01). The CA153 and CEA levels of ND and serum in different groups were shown in Figure 1. The CA153 and CEA levels of ND (Figure 1a) and serum (Figure 1b) in the malignant group were significantly higher than those in benign group (p<0.05). The CA153 and CEA levels in ND were significantly higher than those in serum (p<0.05) both in intraductal papilloma subgroup and those in papillary carcinoma subgroup.
| Biological parameters | Benign | Malignant |
|---|---|---|
| Number of cases | 64 | 29 |
| Location | ||
| Left | 36 | 18 |
| Right | 28 | 11 |
| Duct amount | ||
| Solitary | 17 | 19 |
| Multiple | 47 | 10 |
| Discharge color | ||
| Bloody | 23 | 21 |
| Nonbloody | 41 | 8 |
| Tumor size | ||
| <2cm | 37 | 18 |
| >2cm | 27 | 11 |
| Type | ||
| Center | 27 | 12 |
| Peripheral | 37 | 17 |
Table 1. The Clinical information of nipple discharge.
Papilloma group included intraductal papilloma (n=36), papilloma with atypical ductal hyperplas (n=11), papilloma with duct expansion (n=11) and papilloma with inflammation (n=6). The ND biomarker levels of CA153 (26.13 ± 6.54 U/ml) and CEA (7.46 ± 1.75 ng/ml) were significantly higher than those in serum (15.61 ± 3.18 U/ml, 2.40 ± 0.72 ng/ml, respectively) in intraductal papilloma subgroups (p<0.05). However, there were no significant difference of ND or serum biomarker levels in papilloma groups (p>0.05). Papillary carcinoma group included papilloma with ductal carcinoma in situ (n=9), papillary carcinoma (n=16) and Encapsulated papillary carcinoma (n=4). There were no difference of ND or serum biomarker levels in papillary carcinoma groups (p>0.05).
ND biomarker levels in HIF-1α and Ki-67 positive and negative expression subgroups of papillary carcinoma
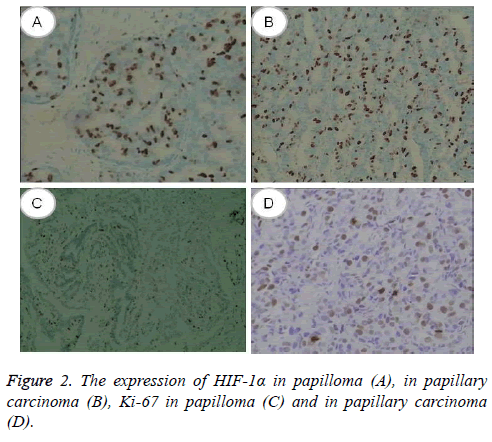
The expression of HIF-1α and Ki-67 was located in cell nuclear, shown in Figure 2. The results shown that the expression of HIF-1α and Ki-67 in breast papillary carcinomas were higher than those in benign lesions (p<0.05, respectively). The ND levels of CA153 and CEA in HIF-1α and Ki-67 positive and negative expression in papillary carcinoma group were shown in Table 2. The levels of CA153 and CEA in HIF-1α and Ki-67 positive subgroups were significantly higher than those in negative patients in papillary carcinoma (p<0.05, respectively), however, the levels of CA153 and CEA in ER/PR positive subgroups were significantly lower than those in negative patients (p<0.05, respectively). The ND levels of CA153 and CEA relation to other different clinical pathological factors were also shown in Table 2. There were no correlation with the age at diagnosis, tumor size, discharge tube hole, location and amount (p>0.05).
| Biological parameters | n | CA l53 U/mL | CEA ng/ml |
|---|---|---|---|
| HIF-1α | |||
| Positive | 20 | 138.61 ± 30.32 | 126.42 ± 34.18 |
| Negative | 9 | 97.24 ± 23.16 | 60.42 ± 15.18 |
| Ki-67 | |||
| >14% | 7 | 134.24 ± 31.13 | 130.07 ± 41.65 |
| ≤ 14% | 22 | 119.67 ± 23.45 | 84.33 ± 31.57 |
| Age at diagnosis | |||
| ≥ 50 years | 19 | 131.01 ± 30.54 | 90.44 ± 29.06 |
| <50 years | 10 | 129.72 ± 29.66 | 88.67 ± 28.38 |
| Discharge tube hole | |||
| solitary | 19 | 136.61 ± 33.39 | 131.42 ± 31.88 |
| multiple | 10 | 115.89 ± 28.25 | 89.99 ± 22.39 |
| Location | |||
| left | 18 | 130.28 ± 29.69 | 90.24 ± 29.33 |
| right | 11 | 130.01 ± 29.77 | 89.01 ± 28.27 |
| Tumour size | |||
| <2 cm | 10 | 129.26 ± 29.93 | 88.95 ± 28.26 |
| >2 cm | 11 | 131.23 ± 31.37 | 91.13 ± 29.84 |
| Tumour amount | |||
| Multi focal | 17 | 131.74 ± 31.64 | 90.91 ± 29.11 |
| Single focal | 12 | 129.51 ± 29.36 | 88.62 ± 28.47 |
| ER status | |||
| Positive | 15 | 126.31 ± 26.74 | 89.79 ± 29.81 |
| Negative | 14 | 180.56 ± 47.12 | 127.25 ± 43.89 |
| PR status | |||
| Positive | 14 | 109.11 ± 24.92 | 70.34 ± 15.23 |
| Negative | 15 | 185.64 ± 49.71 | 130.79 ± 40.23 |
Table 2. ND levels correlation with biological parameters.
Comparison of serum biomarker levels before and after Western medicine combined with TCM therapy
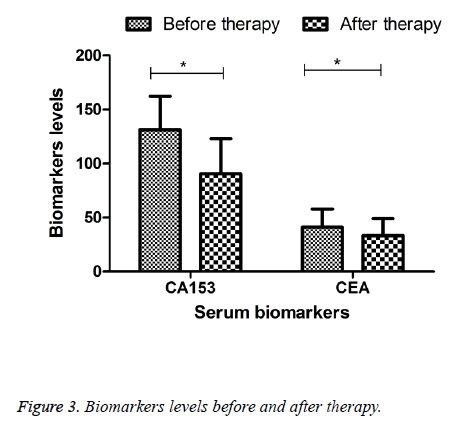
The serum levels of CA153 and CEA were decreased in the patients after Western medicine and combined with TCM therapy compared with the biomarker levels before therapy in papillary carcinoma (p<0.05), shown in Figure 3. TCM composition could decrease the CA153 and CEA tumor biomarker levels after therapy. The improvement of symptoms such as lassitude and fatigue, tidal fever, diet discomfort, stool disorder, soreness and weakness of the lions and knees was superior to that than before, the differences being significant (p<0.05).
Discussion
As the breast mass and breast pain, nipple discharge is a relatively common breast complaint among women [5]. It is classified as normal or abnormal depending on features such as laterality, cycle variation, quantity, color or presentation. Majority of ND comes forth spontaneously and has a pathological outcome. A large portion of patients with pathological ND contain one or several symptoms including unilateral, serous and bloody discharge [14]. The evaluation and management of ND can be undertaken with minimal difficulty by performing a careful history and examination and following a logical thought process linking the type of discharge with the suitable mode of treatment. However, nipple discharge could be a sign of serious abnormality within the breast. Multiple papillomatosis has quite distinct characteristics and decision making can be somewhat challenging. The most common cause of ND is intraductal papillary lesions, besides intraductal papilloma, papilloma with duct expansion inflammation, intraductal cancer, and so on. ND also can be the presenting symptom for other type of cancer.
Intraductal papillary lesions and its association with ND are well known. Breast papillary lesions represent a range of lesions varied morphological, radiological, and pathological features. Such lesions are characterized by formation of epithelial fronds that have both the luminal epithelial and the outer myoepithelial cell layers, supported by a fibrovascular stroma [13]. They are broadly divided into two groups: central and peripheral [13]. Most papillomas are central. Patients present over a wide age range, but most cases occur between age 30 and 50 years [13]. In this study, their age ranged from 20 to 72 years, mean 51.6 years. Central papillomas most frequently present with unilateral sanguineous, or serosanguineous nipple discharge. Presentation as a palpable mass is less common. Mammographic abnormalities include a circumscribed retro-areolar mass of benign appearance, a solitary retro-areolar dilated duct and, rarely, micro calcifications. Small central papillomas may be mammographically occult. Ultrasonography may show a welldefined smooth-walled, solid, hypoechoic nodule or a lobulated, smooth-walled, cystic lesion with solid components. Galactography usually shows an intraluminal filling defect or duct dilatation. Magnetic resonance imaging may be useful in evaluating nipple discharge. Small papillomas often appear as enhancing masses with smooth margins, while larger lesions can have irregular margins. Central papillomas originate in the large ducts, usually of the subareolar region without involving the terminal-duct lobular unit, whereas peripheral papillomas always have their roots in the terminal-duct lobular unit, even if they extend into the ducts. Bloody ND can be the presenting symptom of papillary carcinoma, whereas the more peripherally located lesions may present as a mass. Our present study investigated that there were significant differences in discharge color and type malignant group. Serum biomarkers such as CA153 and CEA can be used as diagnostic and prognostic factors and can also provide valuable information during follow-up. However, serum protein biomarkers show limited diagnostic sensitivity and specificity in stand-alone assays because their levels reflect tumor burden [4]. The clinical in our study groups manifestations that CA153 and CEA in ND can serve as novel biomarkers in the diagnosis and traditional Chinese medicine therapeutic effects evaluation of breast papillary cancer.
The pathogenesis of breast cancer is not fully clear. Many studies have found that breast cancer is mainly a multifactorial disease, occurring as a result of the combined effects of factors. Studies have shown that the proliferation and metastasis of breast cancer cells is a very complex, multistep process, affected by various factors including the common role of environmental, genetic and so on [15]. It is critical for breast cancer management to identify those biomarkers that would provide, at the time of diagnosis, a reliable prognostic assessment and prediction of treatment response [16]. Several studies have shown that certain biological indicators by detecting abnormal expression of HER2/neu, ER, PR etc., can guide clinical diagnosis and treatment activities prognosis. HIF-1a is implicated in carcinogenesis and tumour progression in many human cancers [17,18], including breast cancer. Hypoxia, is a common feature of various cancers [18]. Overexpression of HIF-1a is a frequent feature of malignant disease and is commonly associated with poor prognosis and resistance to conventional chemotherapy [18]. In this study, we have detected expressions of HIF-1α in breast papillary lesions, and analyze the relationship between the expressions and clinicopathological features in breast papillary lesions. Our study shows that the CA153 and CEA levels in HIF-1α positive patients were significantly higher than those in negative patients (respectively, P<0.05). Ki-67, as an easily assessed and reproducible proliferation factor, may be complement to papillary carcinoma as a prognostic tool for selection of Western medicine therapy and combined with Runiu recipe TCM treatmen. In the same way, proliferation study has acquired great value with the diagnosis of breast tumors benign or malignan. In this study, we found a statistically significant association between ki-67 expression and the ND levels of CA153 and CEA in papillary carcinoma. There was also more proliferation measured by immunohistochemical expression of Ki-67 in papillary carcinoma than in papilloma. About high Ki-67, we know that is a factor of poor prognosis in breast cancer, that in patients with breast cancer without axillary lymph node it is an independent prognostic factor in the 87% of the patients who had not received adjuvant medical treatment [19].
Traditional Chinese Medicine (TCM), as an important component of complementary and alternative medicine, has become well known for its significant role in preventing and treating breast lesion with ND. TCM is effective in treating breast papillary lesions and breast cancer, but its theories appear hysteretic in clinical practice, teaching and research of TCM in the treatment of breast papillary cancer [20,21]. Etiology and Pathogenesis of breast papillary carcinoma is due to depression of liver-qi, which is made by melancholia and angry, then depression of liver-QI transmits to heat to injury to blood vessels. Spleen is injured by thinking, failing to control blood to overflow from the breast orifice. Hyperactivity of liver fire boils body fluid to be sputum, which stasises with ouerflowing blood in breast to a lump. Radix bupleuri, immature tangerine peel, rhizoma cyperi and Fruit of Citron Fruit, disper stagnated liver qi and dredge channels; Aromatic Turmeric Root-tuber promotes blood circulation for removing blood stasis and relieve qi stagnation; radices, paeoniae alba and angelica nourish liver by nourishing blood; Tuckahoe, Fried Atractylodes and Coix Seed nourish qi to invigorate spleen for eliminating dampness; Tree Peony Bark can clear heat and cool bood, promote blood circulation for removing blood stasis; Morinda officinalis and cornua cervi degelatinatum reinforce kidney to warm yang and coordinate Chong and Conception Vessels. Treatment based on TCM theory differentiation, also called “Bian Zheng Shi Zhi”, is the comprehensive analysis of clinical information that is used to guide the choice of treatment with TCM Runiu recipe composition formulae. It is the main approach in the clinical practice to increase the effectiveness and safety of TCM treatment in the clinical practice. It is critical for breast cancer management to identify those biomarkers that would provide those factors potentially curable given the availability of early diagnosis and effective therapeutic protocols, at the time of diagnosis, a reliable prognostic assessment and prediction of treatment response. In our study, the total scores of QOL in both groups were superior to those before treatment. Before and after therapy, the Chinese medical syndrome had changed significantly. The obviously improvement of symptoms such as lassitude and fatigue, tidal fever, diet discomfort, stool disorder, soreness and weakness of the lions and knees was superior to that than before, the differences being significant (P<0.05). The CA153 and CEA levels were decreased in the patients after therapy compared with the biomarker levels before therapy. TCM Ruyan recipe composition could decrease the CA153 and CEA tumor biomarker levels.
Conclusion
This finding suggests that nipple discharge and serum CA153 and CEA biomarker levels in papillary carcinoma were significantly higher than those in papilloma, and had a positive correlation with the HIF-1α and Ki-67 expression. The CA153 and CEA levels in nipple discharge were significantly higher than those in serum. The HIF-1α may play a key role during oncogenesis in intraductal papillary lesions. The nipple discharge biomarker levels detection has important value in the diagnosis of intraductal papillary carcinoma. TCM combined with Western medicine can relieve the clinical Chinese medical symptoms of breast intraductal papillary carcinoma patients, and decrease the CA153 and CEA levels. The findings of the present study suggest that tumor marker detection of nipple discharge may be as a new diagnostic method for the diagnosis of the breast intraductal papillary lesions.
Acknowledgements
This work was financially supported by Project of Application Technology Research and Development Project Foundation in Rizhao (2014SZSH02), Medicine and Health Care Science and Technology Development Plan Project Foundation of Shandong Province (2014WS0282), Medical Staff' Science and Technology Innovation Projects of Shandong Province Medical Union Committee (201515) as well as Scientific Research Key Projects of Jining Medical University (JY2013KJ051). The authors would like to thank Prof. Hong-yuan Wang, the pathology department director of Rizhao Traditional Chinese Medicine Hospital, for his valuable cases information.
References
- Gold P, Freedman SO. Specific carcinoembryonic antigens of the human digestive system. J Exp Med 1965; 122: 467-481.
- Kahana L, Bartal AH, Yechieli H, Guttman I, Sheinfeld M. Carcinoembryonic-like substance in breast fluid discharge in benign and malignant breast disease and in milk of lactating women. Isr J Med Sci 1981; 17: 1035-1040.
- Stieber P, Nagel D, Blankenburg I, Heinemann V, Untch M, Bauerfeind I, Di Gioia D. Diagnostic efficacy of CA 15-3 and CEA in the early detection of metastatic breast cancer-A retrospective analysis of kinetics on 743 breast cancer patients. Clinica chimica acta Int J Clin Chem 2015; 448: 228-231.
- Wang G, Qin Y, Zhang J, Zhao J, Liang Y, Zhang Z, Qin M, Sun Y. Nipple discharge of CA15-3, CA125, CEA and TSGF as a new biomarker panel for breast cancer. Int J Mol Sci 2014; 15: 9546-9565.
- Sturgeon CM, Duffy MJ, Stenman UH, Lilja H, Brünner N, Chan DW, Babaian R, Bast RC Jr, Dowell B, Esteva FJ, Haglund C, Harbeck N, Hayes DF, Holten-Andersen M, Klee GG, Lamerz R, Looijenga LH, Molina R, Nielsen HJ, Rittenhouse H, Semjonow A, Shih IeM, Sibley P, Sölétormos G, Stephan C, Sokoll L, Hoffman BR, Diamandis EP; National Academy of Clinical Biochemistry. National Academy of Clinical Biochemistry laboratory medicine practice guidelines for use of tumor markers in testicular, prostate, colorectal, breast, and ovarian cancers. Clin Chem 2008; 54: e11-79.
- Harris L, Fritsche H, Mennel R, Norton L, Ravdin P, Taube S. American Society of Clinical Oncology 2007 update of recommendations for the use of tumor markers in breast cancer. J Clin Oncol 2007; 25: 5287-5312.
- Wu SG, He ZY, Zhou J, Sun JY, Li FY, Lin Q, Guo L, Lin HX. Serum levels of CEA and CA15-3 in different molecular subtypes and prognostic value in Chinese breast cancer. Breast 2014; 23: 88-93.
- Barak V, Goike H, Panaretakis KW, Einarsson R. Clinical utility of cytokeratins as tumor markers. Clin Biochem 2004; 37: 529-540.
- Duffy MJ, Evoy D, McDermott EW. CA 15-3: uses and limitation as a biomarker for breast cancer. Clin Chim Acta 2010; 411: 1869-1874.
- Deutscher SL, Dickerson M, Gui G, Newton J, Holm JE, Vogeltanz-Holm N. Carbohydrate antigens in nipple aspirate fluid predict the presence of atypia and cancer in women requiring diagnostic breast biopsy. BMC Cancer 2010; 10: 519.
- Sauter ER, Wagner-Mann C, Ehya H, Klein-Szanto A. Biologic markers of breast cancer in nipple aspirate fluid and nipple discharge are associated with clinical findings. Cancer Detection Prevention 2007; 31: 50-58.
- Mannello F, Tonti GA, Canestrari F. Nutrients and nipple aspirate fluid composition: the breast microenvironment regulates protein expression and cancer aetiology. Genes Nutr 2008; 3: 77-85.
- Lakhani SR, Ellis IO, Schnitt SJ, Tan PH, Vijver MJVD. WHO classification of tumours of the breast. 4th Ed, IARC Press, France, 2012.
- Furuya C, Kawano H, Yamanouchi T, Oga A, Ueda J, Takahashi M. Combined evaluation of CK5/6, ER, p63, and MUC3 for distinguishing breast intraductal papilloma from ductal carcinoma in situ. Pathol Int 2012; 62: 381-390.
- Sueoka-Aragane N, Sato A, Ide M, Nakamura H, Sotomaru Y. Development of lymphoproliferative diseases by hypoxia inducible factor-1alpha is associated with prolonged lymphocyte survival. PLoS One 2013; 8: e57833.
- Diers AR, Vayalil PK, Oliva CR, Griguer CE, Darley-Usmar V, Hurst DR, Welch DR, Landar A. Mitochondrial bioenergetics of metastatic breast cancer cells in response to dynamic changes in oxygen tension: effects of HIF-1a. PLoS One 2013; 8: e68348.
- Dewi FR, Fatchiyah F. Methylation impact analysis of erythropoietin (EPO) Gene to hypoxia inducible factor-1α (HIF-1α) activity. Bioinformation 2013; 9: 782-787.
- Semenza GL. Defining the role of hypoxia-inducible factor 1 in cancer biology and therapeutics. Oncogene 2010; 29: 625-634.
- Gonzalez-Sistal A, Baltasar-Sánchez A, Menéndez P, Arias JI, Ruibal Á. Breastfeeding and Immunohistochemical Expression of ki-67, p53 and BCL2 in Infiltrating Lobular Breast Carcinoma. PLoS One 2016; 11: e0151093.
- Qi F, Zhao L, Zhou A, Zhang B, Li A, Wang Z, Han J. The advantages of using traditional Chinese medicine as an adjunctive therapy in the whole course of cancer treatment instead of only terminal stage of cancer. BioScience Trends 2015; 9: 16-34.
- Wu CT, Lai JN, Tsai YT. The prescription pattern of chinese herbal products that contain dang–qui and risk of endometrial cancer among tamoxifen–treated female breast cancer survivors in Taiwan: A population–based study. PLoS One 2014; 9: e113887.