Research Article - Biomedical Research (2017) Volume 28, Issue 10
Expression of vascular endothelial growth factor and angiopoietin-1 in postnatal rats with hyperoxia-induced lung injury and its effect on pulmonary development
Ling Wang, Hui Lv*, Wei Zhou, Li Tao and Juan He
Department of Neonate, Guangzhou Women and Children’s Medical Center, Guangzhou Medical University, Guangzhou, PR China
- *Corresponding Author:
- Hui Lv
Department of Neonate
Guangzhou Women and Children’s Medical Center
Guangzhou Medical University, PR China
Accepted date: March 2, 2017
Abstract
It is well-known that Vascular Endothelial Growth Factor (VEGF) and Angiopoietin (Ang)-1 are important regulators of alveolar and pulmonary vascular development. This study investigated the expression of VEGF and Ang-1 in postnatal rats with hyperoxia-induced lung injury and its effect on pulmonary development. Forty-eight 3-day-old Sprague-Dawley rats were randomly divided into 2 groups and exposed to either hyperoxia (≥ 95%) or air. Real-time PCR and Western blotting were used to measure the mRNA and protein expression levels of VEGF and Ang-1 in pulmonary tissues. No significant differences in the expression levels of VEGF and Ang-1 protein or mRNA were noted between the control and hyperoxia groups on days 1 and 3 (P>0.05); however, an obvious significant difference was noted on day 7 (P<0.05). The mRNA expression levels of VEGF and Ang-1 were 0.722 ± 0.372 and 0.828 ± 0.462, respectively, in the control group and 0.239 ± 0.293 and 0.327 ± 0.184, respectively, in the hyperoxia group. The protein expression levels of VEGF and Ang-1 were 0.632 ± 0.289 and 0.573 ± 0.436, respectively, in the control group, 0.358 ± 0.128 and 0.204 ± 0.068, respectively, in the hyperoxia group. The pulmonary tissues of the hyperoxia group manifested dysplastic characteristics with alveolar simplification, a reduction in alveolar numbers, and retardation in microvascular development. Since VEGF and Ang-1 are important regulators of alveolar and pulmonary vascular development that are involved in the pathogenesis of lung injury and development, their expression profile may be helpful for studying the pathogenic mechanism of and treatment for bronchopulmonary dysplasia.
Keywords
Bronchopulmonary dysplasia, Hyperoxia, Vascular endothelial growth factor, Angiopoietin.
Introduction
Bronchopulmonary Dysplasia (BPD) is a chronic lung disease of premature infants and usually induces severe complications that cause pulmonary dysfunction and dysplasia [1,2]. With the development of mechanical ventilation and application of exogenous pulmonary surfactant, the survival rate of premature infants has had an obvious increase; however, the incidence rate of BPD has also increased resulting in a reduction of the survival rate and quality of life [3-5]. In this study, postnatal rats were exposed to hyperoxia for obtaining acute lung injury rat models, which showed that the pathological changes complied with the occurrence and development of BPD in very premature infants. Although rats born at term have structurally immature lungs, they are functionally mature and require no intubation, ventilation, or treatment with synthetic surfactant. The last stage of pulmonary development in rats is also called alveolarization and starts 3 days after birth, which is at approximately the same time as humans with 26 to 28 weeks of gestation. In this stage, the lungs are not well developed and hyperoxia easily causes lung injury. Hyperoxic gas above 95% is the common concentration used to study BPD-like lung injury. Short-term hyperoxia could lead to an acute lung injury resulting in impaired alveolarization with fewer and enlarged alveolar air spaces [6,7]. Studies have demonstrated that Vascular Endothelial Growth Factor (VEGF) is a key regulator of pulmonary vascular growth and maintenance throughout the embryonic, fetal, and postnatal periods [8-10]. Angiopoietin (Ang) is a vascular growth factor that promotes the interaction between endothelial and adventitial cells and aids in vascular maturity [11]. However, the dynamic expression of VEGF and Ang-1 and their role in acute lung injury remain unclear. In this study, a lung injury rat model was developed using hyperoxiaexposed postnatal rats in order to measure the expression levels of VEGF and Ang-1 in pulmonary tissues and evaluating the effects on pulmonary development; thereby, providing further information on BPD.
Materials and Methods
Developing a rat model of acute lung injury by hyperoxia exposure
Four pregnant Sprague-Dawley rats that were purchased from the experimental animal center of Guangzhou Medical University gave birth to 55 new-born rats. Forty-eight 3-dayold Sprague-Dawley rats, of either sex, were randomly divided into a control and hyperoxia group. Each group had 24 rats. Oxygen boxes were set at 3 different pores, including the entering pore, vent pore, and testing pore. Oxygen flow was set to 6 L/min and oxygen concentration to >95%; the levels were monitored using an oxygen concentration determinator in the box. In the hyperoxia group, postnatal rats and their mothers were fed in the oxygen boxes for 7 days. Meanwhile, the controls were placed in standard rat cages. All the maternal rats were fed with standard rat pellets and water, which were added every day. Postnatal rats were nursed and their growth and vitality were observed [12]. This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. The animal use protocol has been reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of Guangzhou Medical University.
Sample collection and preparation
After deep anesthesia with an intraperitoneal injection of 10% chloral hydrate on days 1, 3, and 7 in 8 random rats from each group, the rats were put down by cervical dislocation and their lungs were separated. The entire portions of the left lungs were immediately rinsed in ice-cold PBS and snap-frozen in liquid nitrogen for real-time PCR and Western blotting; the entire portions of the right lungs were immediately fixed in situ by intratracheal instillation of 4% paraformaldehyde for pathological sections. The morphological changes of the lung tissues were examined by light microscopy.
Morphological observation of lung tissues
Five micro sections were stained with Hematoxylin and Eosin (HE) and examined. The morphological changes in structures in the hyperoxia group were observed under the light microscope and compared to that of the control group.
Detection of the VEGF and Ang-1 mRNA expression levels in lung tissues
Real-time PCR was used to determine the mRNA expression levels of VEGF and Ang-1. Total RNA of lung tissues were extracted with Trizol (Invitrogen Company, USA) and TaKaRa reverse transcription kits (TaKaRa Company, Japan) were used to synthesize cDNA for PCR amplification. The primer sequences were as follows: 5’- GAGTATATCTTCAAGCCGTCCTGT-3’ (VEGF sense), 5’- ATCTGCATAGTGACGTTGCTCTC-3’(VEGF anti-sense), with a 118 bp amplified product; 5’- GGATCGAGCTGATGGACTG-3’ (Ang-1 sense), 5’- TCAAGCTGCTCTGTTTGCCT-3’ (Ang-1 anti-sense), with a 135 bp amplified product; 5’- ACGGTCAGGTCATCACTATC-3’ (β-actin sense), 5’- TGCCACAGGATTCCATACC-3’ (β-actin anti-sense), with a 109 bp amplified product. The PCR conditions were as follows: an initial melting step at 95°C for 5 min, followed by 40 cycles of denaturation at 95°C for 5 s, an appropriate annealing temperature of 52°C for 20 s, with a final extension step at 72°C for 5 min. Subsequently the transcripts were checked using a 2% agarose gel for gel electrophoresis and stained with Ethidium Bromide (EB). The results were observed under an ultraviolet lamp. cDNA was synthesized to establish the reaction system and perform real-time PCR. Ct values were used to calculate the relative amount of mRNA expression levels using a standard curve. The ratio of objective gene to internal reference gene represented the relative mRNA expression levels.
Detection of the VEGF and Ang-1 protein expression levels in lung tissues
A Western blotting assay was used to detect the protein expression levels of VEGF and Ang-1 in lung tissues. The total protein of the lung tissues was extracted and the protein expression levels were measured by BCA assay. In brief, the procedure used for the Western blot assay was as follows: SDS-PAGE electrophoresis, half-dry transferring membranes, blocking antibodies, primary antibodies (rabbit anti-rat VEGF antibody (Santa Cruz Biotechnology, USA), rabbit anti-rat Ang-1 antibody (Millipore Company, USA) and rabbit anti-rat beta-actin antibody (Cell Signaling Technology, USA)), secondary antibodies (HRP-labeled goat anti-rabbit IgG antibody (Santa Cruz Biotechnology, USA), HRP-labeled anti-biotinylated antibody (Cell Signaling Technology, USA)), incubation, imaging and pictures were scanned and analysed by Image-ProPlus Version 6.0 software (Immunochemiluminescence kits, Pierce Biotechnology, USA). The ratio of the gray value of objective protein to the value of the internal reference protein represented the relative protein expression levels.
Statistical analysis
Statistical analysis was performed using the SPSS 16.0 software (http://www.spss.com or SPSS Inc.). Measured data was presented as mean ± SD (x? ± s). The results from the control group on day 1 were used as the reference and the internal reference gene data were adjusted. Data were tested for normal distribution. A two-sample t-test and one-way ANOVA were used to compare data between the 2 groups. P<0.05 was considered to be statistically significant.
Results
Postnatal rat model and the morphological changes of the lung tissues
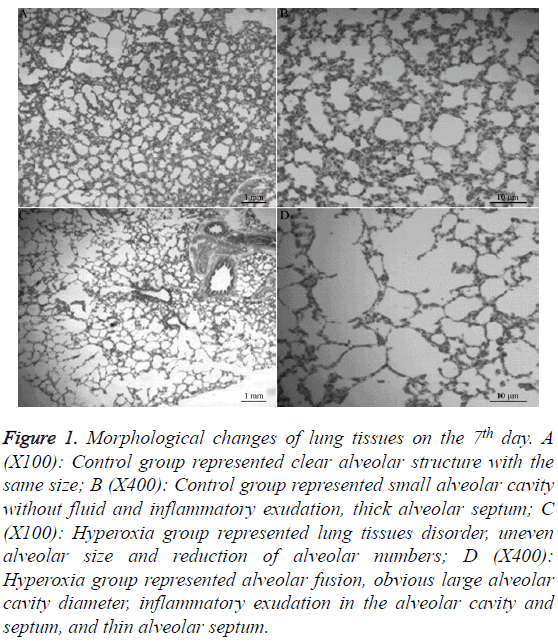
In the control group, postnatal rats grew well with weight gains of 2-3 g every day and a 1.0 cm to 1.5 cm increase per day in length; however, in the hyperoxia group, with the longer time of exposure to hyperoxia, the postnatal rats had thinner bodies, less vitality, blunted responses, and slower increases in weight and length. Two postnatal rats died on day 5 and 6, respectively, in the hyperoxia group; the total mortality rate was 8.33%. In the control group, low power light microscopy (X100) showed no obvious pathological changes with clear alveolar structures that were similar in size, a small alveolar cavity without inflammatory exudates, and a thick alveolar septum. In the hyperoxia group, the lung tissues became disordered with the longer exposure to hyperoxia and the alveolar wall became thinner. Furthermore, the following findings indicated that development and differentiation was impaired in the pulmonary tissues: alveolar simplification, reduction in the number of alveoli, some alveolar fusion, an increase in alveolar volume, reduction in alveolar area, obviously enlarged alveolar cavity diameter, some inflammatory exudates, thin alveolar septum, and an increased number of fibroblast cells and collagen-like substances. High power light microscopy (X400) found that the alveolar wall was entirely without obvious cast-off cells in the control group, but in the hyperoxia group, red cells, microphages and cast-off lung endothelial cells were seen in the alveolus. In addition, the alveolar septum became thin and interstitial cells increased, and the small vessels were expanded and congested (Figure 1).
Figure 1: Morphological changes of lung tissues on the 7th day. A (X100): Control group represented clear alveolar structure with the same size; B (X400): Control group represented small alveolar cavity without fluid and inflammatory exudation, thick alveolar septum; C (X100): Hyperoxia group represented lung tissues disorder, uneven alveolar size and reduction of alveolar numbers; D (X400): Hyperoxia group represented alveolar fusion, obvious large alveolar cavity diameter, inflammatory exudation in the alveolar cavity and septum, and thin alveolar septum.
The VEGF and Ang-1 mRNA expression levels in lung tissues
Compared to the control group, a longer exposure to hyperoxia gradually down-regulated the mRNA expression levels of VEGF and Ang-1 and on day 7, the decrease in mRNA expression levels of VEGF and Ang-1 reached statistical significance at 66.9% and 60.5% (P=0.012<0.05, P=0.013<0.05), respectively. Furthermore, there was no statistically significant differences in the mRNA expression levels of VEGF and Ang-1 on days 1 and 3 between the 2 groups (P>0.05, Table 1).
| Group | Relative level of VEGF mRNA | Relative level of Ang-1mRNA | ||||
|---|---|---|---|---|---|---|
| 1st day | 3rd day | 7th day | 1st day | 3rd day | 7th day | |
| Control group | 1.000 ± 0.366 | 0.931 ± 0.370 | 0.722 ± 0.372 | 1.000 ± 1.108 | 0.907 ± 1.019 | 0.828 ± 0.462 |
| Hyperoxia group | 0.985 ± 0.403* | 0.897 ± 0.512? | 0.239 ± 0.293? | 1.011 ± 1.131? | 0.843 ± 0.388? | 0.327 ± 0.184? |
Table 1: Comparisons of relative levels of VEGF and Ang-1 mRNA (?x ± s).
The VEGF and Ang-1 protein expression levels in lung tissues
Compared with the control group, the protein expression levels of VEGF and Ang-1 were gradually down-regulated with a longer duration of exposure to hyperoxia and on day 7, the protein expression levels of VEGF and Ang-1 had obviously significantly declined to 43.4% and 64.4% (P=0.035, P=0.048), respectively. Moreover, there were no statistically significant differences in the protein expression levels of VEGF and Ang-1 on the day 1 and 3 between the 2 groups (P>0.05, Table 2, Figure 2).
| Group | Relative level of VEGF protein | Relative level of Ang-1 protein | ||||
|---|---|---|---|---|---|---|
| 1st day | 3rd day | 7th day | 1stday | 3rd day | 7th day | |
| Control group | 1.000 ± 0.498 | 0.787 ± 0.544 | 0.632 ± 0.289 | 1.000 ± 0.858 | 0.995 ± 0.871 | 0.573 ± 0.436 |
| Hyperoxia group | 0.960 ± 0.619* | 0.730 ± 0.342? | 0.358 ± 0.128? | 0.755 ± 0.414? | 0.711 ± 0.539? | 0.204 ± 0.068? |
Table 2: Comparisons of relative levels of VEGF and Ang-1 proteins (?x ± s).
Figure 2: Protein levels of VEGF and Ang-1 in lung tissues (Western blotting). Lane 1, 3 and 5 were presented the levels of VEGF, Ang-1 and β-actin on the 1st, 3rd and 7thdays, respectively in control group; Lane 2, 4 and 6 were presented the levels of VEGF, Ang-1 and β-actin on the 1st, 3rd and 7th days, respectively in hyperoxia group.
Discussion
This study investigated the differential expression of VEGF and Ang-1 in postnatal rats with hyperoxia-induced lung injury and their effects on pulmonary development using biochemical and histological data. The decreased expression of VEGF and Ang-1 and morphological changes of lung tissues in the hyperoxia group demonstrated the role of these factors in pulmonary development. Moreover, the morphological changes of pulmonary tissue in the hyperoxia group manifested as dysplastic characteristics with alveolar simplification, a reduction in the number of alveoli, and retardation of microvascular development, which are recognized as histological characteristics of lung injury that influences normal lung development. Three-day-old postnatal rats were exposed to hyperoxic gases at a concentration that was greater than 95% for 1, 3, and 7 days. The longer exposure to hyperoxic gas resulted in the following pathological morphology that was observed: alveolar dysplastic characteristics with thin alveolar walls, alveolar simplification, uneven alveolar sizes, and a reduction in the number of alveoli and capillaries. This is similar to the pathogenesis of BPD, thus indicating that a model was established successfully. The new BPD has different clinical processes and pathological changes in premature infants, which presents with injury to alveolar and vascular development, including simplified alveolar structures, dysmorphic capillary configuration, and variable interstitial cellularity [13,14]. Previous studies mainly focused on the retardation of alveolar development and few researchers studied the injury to pulmonary vascular development. Some studies indicate that alveolarization and abnormal vascular growth are necessary and the pulmonary vascular networks synchronously expanded to form an effective air-blood barrier. Furthermore, in pulmonary development, pulmonary vascular development promotes normal alveolar growth and is involved in the maintenance of the alveolar structures, which indicates that pulmonary vascular development plays a critical role in the development of the lung as a whole [15]. Premature birth and its therapy might block alveolar and vascular development, causing retardation in alveolar and microvascular development, which leads to a reduction of alveoli and capillaries, eventually reducing the blood surface area for gas exchange [16]. This implies that abnormal pulmonary vascular development at an early stage might play a key role in the mechanism of BPD.
The normal progress of alveolarization throughout the development of the lungs is highly dependent on angiogenesis. VEGF, which mainly promotes formation of primitive vascular networks, is a powerful and important regulating factor for pulmonary vascular growth and maintenance during the entire fetal period and in the postnatal period [17-19]. In addition, Ang-1 is part of a family of vascular growth factors that play a role in embryonic and postnatal angiogenesis and promotes the formation of mature vascular networks that have space structures [20,21]. Here, our results showed that the mRNA and protein expression levels of VEGF and Ang-1 were slightly down-regulated, but not significantly on days 1, 3, and 7 in the control group. The alveolarization phase includes bulk alveolarization and continued alveolarization, which focuses on alveolar formation and airspace volume, respectively [22]. However, the mRNA and protein expression levels of VEGF and Ang-1 were significantly lower on day 7 in the hyperoxia group than in the control group, which indicated that the mRNA and protein levels of VEGF and Ang-1 in lung tissue were obviously down-regulated in BPD-like lung injury induced by hyperoxia. Moreover, it might result in the degradation of alveolar structures and lung vascular dysplasia. Therefore, we speculated that VEGF and Ang-1 were important regulators for pulmonary development, but the exact mechanisms require further studies [23], which would be beneficial towards new clinical treatments of BPD. To determine whether we can regulate the expression of VEGF and Ang-1 to improve the alveolar and microvascular development in lung injury, more clinical practice and future research is needed. At this time, we were not able to ultimately improve the pathogenic structures of BPD; however, some researchers have found that the transplantation of mesenchymal stem cells in BPD animal models could alleviate the damage to lung tissue and promote lung development and the regenerative repair of lung tissues.
Conclusion
VEGF and Ang-1 are important regulators of alveolar and lung vascular development, which is involved in the pathogenesis of lung injury and pulmonary development. In addition, it is significant for further research into the pathogenic mechanism of and treatment for Bronchopulmonary Dysplasia (BPD).
Acknowledgements
This study was supported by Medical Science and Technology Program of Guangzhou city (20141A011031).
Conflicts of Interest
All of the authors declare that they have no conflicts of interest regarding this paper.
References
- Jobe AH, Bancalari E. Bronchopulmonary dysplasia. Am J Respir Crit Care Med 2001; 163: 1723-1729.
- Mailaparambil B, Krueger M, Heizmann U, Schlegel K, Heinze J. Genetic and epidemiological risk factors in the development of bronchopulmonary dysplasia. Dis Markers 2010; 29: 1-9.
- Gortner L, Monz D, Mildau C, Shen J, Kasoha M, Laschke MW, Roolfs T, Schmiedl A, Meier C, Tutdibi E. Bronchopulmonary dysplasia in a double-hit mouse model induced by intrauterine hypoxia and postnatal hyperoxia: closer to clinical features? Ann Anat 2013; 195: 351-358.
- Gray PH, Sarkar S, Young J, Rogers YM. Conductive hearing loss in preterm infants with bronchopulmonary dysplasia. J Paediatr Child Health 2001; 37: 278-282.
- Short EJ, Klein NK, Lewis BA, Fulton S, Eisengart S. Cognitive and academic consequences of bronchopulmonary dysplasia and very low birth weight: 8-year-old outcomes. Pediatrics 2003; 112: 359.
- Balasubramaniam V, Mervis CF, Maxey AM, Markham NE, Abman SH. Hyperoxia reduces bone marrow, circulating, and lung endothelial progenitor cells in the developing lung: implications for the pathogenesis of bronchopulmonary dysplasia. Am J Physiol Lung Cell Mol Physiol 2007; 292: 1073-1084.
- Stenger MR, Rose MJ, Joshi MS, Rogers LK, Chicoine LG, Bauer JA, Nelin LD. Inhaled nitric oxide prevents 3-nitrotyrosine formation in the lungs of neonatal mice exposed to >95% oxygen. Lung 2010; 188: 217-227.
- Abman SH. Impaired vascular endothelial growth factor signaling in the pathogenesis of neonatal pulmonary vascular disease. Adv Exp Med Biol 2010; 661: 323-335.
- Kumar VH, Ryan RM. Growth factors in the fetal and neonatal lung. Front Biosci 2004; 9: 464-480.
- Meller S, Bhandari V. VEGF levels in humans and animal models with RDS and BPD: temporal relationships. Exp Lung Res 2012; 38: 192-203.
- De Paepe ME, Greco D, Mao Q. Angiogenesis-related gene expression profiling in ventilated preterm human lungs. Exp Lung Res 2010; 36: 399-410.
- OReilly M, Thebaud B. Animal models of bronchopulmonary dysplasia. The term rat models. Am J Physiol Lung Cell Mol Physiol 2014; 307: 948-958.
- Coalson JJ. Pathology of new bronchopulmonary dysplasia. Semin Neonatol 2003; 8: 73-81.
- Jobe AJ. The new BPD: an arrest of lung development. Pediatr Res 1999; 46: 641-643.
- Abman SH. Bronchopulmonary dysplasia: a vascular hypothesis. Am J Respir Crit Care Med 2001; 164: 1755-1756.
- DAngio CT, Maniscalco WM. The role of vascular growth factors in hyperoxia-induced injury to the developing lung. Front Biosci 2002; 7: 1609-1623.
- Akeson AL, Greenberg JM, Cameron JE, Thompson FY, Brooks SK, Wiginton D, Whitsett JA. Temporal and spatial regulation of VEGF-A controls vascular patterning in the embryonic lung. Dev Biol 2003; 264: 443-455.
- Bhatt AJ, Pryhuber GS, Huyck H, Watkins RH, Metlay LA, Maniscalco WM. Disrupted pulmonary vasculature and decreased vascular endothelial growth factor, Flt-1, and TIE-2 in human infants dying with bronchopulmonary dysplasia. Am J Respir Crit Care Med 2001; 164: 1971-1980.
- Ferrara N, Gerber HP, LeCouter J. The biology of VEGF and its receptors. Nat Med 2003; 9: 669-676.
- Guo Q, Jin J, Yuan JX, Zeifman A, Chen J. VEGF, Bcl-2 and Bad regulated by angiopoietin-1 in oleic acid induced acute lung injury. Biochem Biophys Res Commun 2011; 413: 630-636.
- Jakkula M, Le Cras TD, Gebb S, Hirth KP, Tuder RM. Inhibition of angiogenesis decreases alveolarization in the developing rat lung. Am J Physiol Lung Cell Mol Physiol 2000; 279: 600-607.
- Tschanz SA, Salm LA, Roth-Kleiner M, Barre SF, Burri PH. Rat lungs show a biphasic formation of new alveoli during postnatal development. J Appl Physiol 2014; 117: 89-95.
- Niver D. Bronchopulmonary dysplasia: structural challenges and stem cell treatment potential. Adv Neonatal Care 2014; 14: 1-111.