- Biomedical Research (2012) Volume 23, Issue 2
Expression of mesenchymal stem cell markers of human adipose tissue surrounding the vas deferens
Habip Akbulut1, Gökhan Cüce2*, Tahsin Murad Aktan2, Selçuk Duman21Unıversıty of Bezmı Alem, Hospıtal of Medıcal Faculty, Department of Urology, Istanbul, Turkey
2Unıversıty of Konya, Faculty of Medıcıne, Deparment of Hıstology and Embryology, Konya, Turkey
- *Corresponding Author:
- Gökhan Cüce
Unıversıty of Konya
Faculty of Meram Medıcıne
Deparment of Hıstology and Embryology Konya, Turkey
Accepted date: January 12 2012
Abstract
Adipose tissue is a source for regenerative medicine applications contains mesenchymal stem cells (MSCs). One of the unique features of MSCs is their trans-differentiation property. Identification of MSCs is accepted by their CD105, CD90 and CD73 markers expression. In lipo aspirate from buttocks and abdominal adipose tissue, these markers were previously shown. In adipose tissue surrounding the vas deferens, we investigated CD105, CD73 and CD90 markers expression. Adipose tissue surrounding the vas deferens was obtained from eight patients who attended to Urology department. Paraffin sections were prepared from adipose tissue after routine histological process. Haematoxylineosin, Toluidine blue and CD105, CD73, CD90 immuno-stainings were performed to the paraffin sections. The strongest expression belonged to CD105, followed by CD90 and finally CD73 which was weakly expressed. Stromal cells derived from the adipose tissue surrounding the vas deferens with similar properties like liposuctioned adipose tissue based on the expression of mesenchymal stem cells markers were reviewed.
Keywords
vas deferens, adipose tissue, mesenchymal stem cells, expression
Introduction
While visceral and subcutaneous fatty tissues are the major locations of adipose tissue in human body [1], there is large deposit of adipose tissue in the entire human body [2].
White adipose tissue has different metabolic properties and functions as a result of the localization area [3,4]. The localization area and the type of adipose tissue has been said to affect the morphology and physiology of adipose tissue [5]. Gene expression profiles of adipose tissue vary depending on its distribution in the body [1,3]. It is possible that the cellular composition may differ according to anatomical localization.
Adipocytes are the most abundant cells in adipose tissue; adipose tissue contains many cell groups such as blood cells, endothelial cells, pre-adipocytes, the fibroblasts and the stromal vascular cells which include smooth muscle cells (2).
The minimum acceptablity of cells to be mesenchymal stem cells (MSCs) is depended on their CD105, CD90 and CD73 markers expression [6,7].
In common surgical lipo-aspirate, samples were previously shown with cells carrying these markers and some authors defined MSCs as adipose derived stem cells (ADSc) [8].
Adipose derived stem cells (ADSc) have been the most preferred source of stem cells since their characterization and isolation over the last 10 years [9]. Adipose derived stem cells can easily be reached and isolated than bone marrow mesenchymal cells [10]. ADSc showed a very high proliferation performance when cultured [9]. MSCs have unique ability of trans-differentiation [7,11,12] and ADSc exhibited the same differentiation capacity compared to bone marrow stem cells, bone, cartilage, adipocytes and skeletal muscle that originated from mesoderm and that’s why ADSc began to be widely used by clinicians in tissue engineering [13]. ADSc isolated from different regions of the body have the capacity to differentiate different myogenic cells in vitro [14].
The paucity of data or any documentation (baseland pubmed search) about MSCs content of the adipose tissue of human vas deferens, this study aimed at searching these cells in this tissue for the expressions of CD105, CD73 and CD90 markers.
Materials and Methods
Adipose tissue surrounding the vas deferens was obtained from each of the eight patients who attended our urology clinic between january and march 2011. The patients were slated for hydrocelectomy and epididymal cyst operations and all the surgical procedures were performed at the Urology department of Bezm-i Alem Medical Faculty. Collected tissues were fixed in 10% formaldehyde and reached Konya within 3 days. Paraffin sections were prepared from the collected samples after routine histological processes at Meram Medical Facuty. The study protocols were reviewed and approved by the Ethics committee of Meram Medicine Faculty. The patients were informed and consent forms were obtained and signed by each of the participants. Toluidine blue histochemistry staining, Haematoxylineosin and CD105, CD73, CD90 immunostainings were performed on the paraffin sections. In this study, CD105 (RB-9291-R7:Neomarker), CD73 (S2054: Epitomics) and CD90 (2694-1:Epitomics) were used. CD73 and CD90 were diluted at one-hundredth (1/100) ratio while CD105 was a concentrated primer antibody.
AEC and substrate were used as chromogen. In the evaluation criteria, four groups were formed according to the prevalence of staining [15].
0: No staining
+1: Insignificant staining
+2: Moderate staining
+3: Strong staining
Results
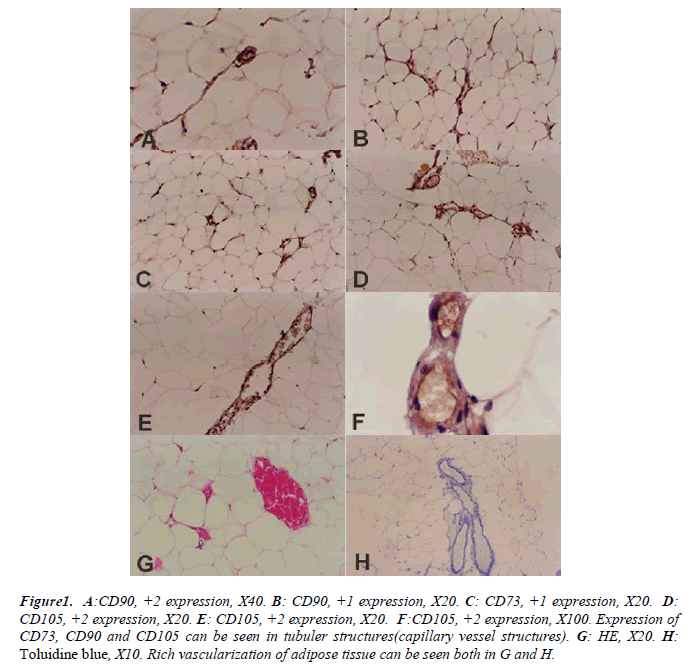
Expression of these markers can be seen in tubular structures (capillaries and vessels) of adipose tissue of vas deferens. Plus one (+1) and (+2) expression of CD90 are shown in Figures B and A respectively CD73 expressions were evaluated +1 in all sections as in Figure C. The +2 expression of CD105 are shown in Figures D, E and F respectively.
Figure 1: A:CD90, +2 expression, X40. B: CD90, +1 expression, X20. C: CD73, +1 expression, X20. D:
CD105, +2 expression, X20. E: CD105, +2 expression, X20. F:CD105, +2 expression, X100. Expression of
CD73, CD90 and CD105 can be seen in tubuler structures(capillary vessel structures). G: HE, X20. H:
Toluidine blue, X10. Rich vascularization of adipose tissue can be seen both in G and H.
A well vascularized adipose tissue can be seen in all the shown sections. Adipocyte cell clusters showed the classical histological ‘honeycomb’ appearance (Figures G and H).
Expression percentages of markers are listed in Tables 1, 2 and 3. The highest expression percentage belonged to CD105 (Table 3), expression percentage of CD90 (Table 2) was moderate and expression percentage of CD73 (Table 1) was lower when compared to CD105 and CD90 in all the sections.
Discussion
In our study, for the first time, we found the cells of adipose tissue surrounding the vas deferens containing markers of MSCs.
There is no unique marker that can identify and separate MSCs from other cell types exactly [16]. Therefore a combination of markers as used in this study could be more reliable.
Fatty acid uptake, lipolysis, energy metabolism control, adipokine and hormone secretion/ expression and many other features vary among adipose localization [17,18,19].
It was claimed adipokines affect the function of surrounding tissues [17]. There might be a double sided reversible effect between adipose and surrounding tissues.
Endothelial cells, syncytiotrophoblasts, connective tissue stromal cells and macrophages express endoglin (CD105), the TGF-b receptor III. Endoglin gene mutation leads to an autosomal dominant disorder called hereditary hemorrhagic telangiectasia. Symptoms of this disorder are wide spread vascular failure and hemorrhage. It can be understood that endoglin has important features in vascular development or endothelial cell interactions [20].
The CD105 expression is not observed in endothelial cells of normal tissue but a powerful expression is observed in endothelial cells of tumor blood vessels [21].
In contrast, CD105 expression is strongly expressed in normal vessels in this study. It has been shown that when MSCs derived from umbilical cord blood begin to differentiate, CD105 expression decreases by a reciprocal effect [22] but increased expressions of CD105 may be before the differentiation of MSCs in adipose tissue in our study.
The results of our work showed that the stromal cells of adipose tissue surrounding the vas deferens have similar properties like liposuctioned adipose tissue according to the expression of mesenchymal stem cells basic markers reported in literature. Here we demonstrated a new finding which has a place in human histology, and till today, the adipose tissue surrounding the vas deferens has never been evaluated as a possible source for MSCs. This study has two strength: firstly,we open a new area for researchers, secondly, the findings add knowledge to scientists in general and to the the urologist in particular, so as to obtain MSCs whenever there is a plan to harvest MSCs for treatment, by this way the need to disturb abdominal region can be eliminated.
References
- Kajimoto K, Hossen MN, Hida K, Ohga N, Akita H, Hyodo M, Hida Y, Harashima H. Isolation and cul- ture of microvascular endothelial cells from murine in- guinal and epididymal adipose tissues. J Immunol Methods. 2010; 357(1-2): 43-50.
- Gomillion CT, Burg KJ. Stem cells and adipose tissue engineering. Biomaterials 2006; 27 (36): 6052-6063
- Casteilla L, Dani C. Adipose tissue-derived cells: from physiology to regenerative medicine. Diabetes Metab. 2006; 32 (5 Pt 1): 393-401.
- Fiseković S. Obesity biological and psychological aspect. Bosn J Basic Med Sci 2005; 5 (4): 30-4.
- Poulos SP, Hausman DB, Hausman GJ. The develop- ment and endocrine functions of adipose tissue. Mol Cell Endocrinol. 2010; 323 (1): 20-34.
- Vallée M, Côté JF, Fradette J. Adipose-tissue engineer- ing: taking advantage of the properties of human adi- pose-derived stem/stromal cells. Pathol Biol. 2009; 57: 309-317.
- Odabaş S, Sayar F, Güven G, Yanikkaya-Demirel G, Pişkin E. Separation of mesenchymal stem cells with magnetic nanosorbents carrying CD105 and CD73 antibodies in flow-through and batch systems. J Chro- matogr B Analyt Technol Biomed Life Sci. 2008; 861: 74-80.
- de Mattos Carvalho A, Alves AL, Golim MA, Moroz A, Hussni CA, de Oliveira PG, Deffune E. Isolation and immunophenotypic characterization of mesenchy- mal stem cells derived from equine species adipose tis- sue. Vet Immunol Immunopathol. 2009; 132: 303-306.
- Philips BJ, Marra KG, Rubin JP. Adipose stem cell- based soft tissue regeneration. Expert Opin Biol Ther. 2012 ;12(2):155-63.
- Choi YS, Vincent LG, Lee AR, Dobke MK, Engler AJ. Mechanical derivation of functional myotubes from adipose-derived stem cells. Doi:10.1016/j.Biomaterials. 2011.12.004.
- Ulicna M, Danisovic L, Danihel L, Vojtassak J. Diabe- tes--adult stem cells as an future alternative therapy? Bratisl Lek Listy. 2009; 110: 773-776.
- Ulicna M, Danisovic L, Vojtassak J.Does cell therapy and tissue engineering represent a promising treatment of diabetic foot ulcers? Bratisl Lek Listy. 2010; 111: 138-143.
- Schäffler A, Büchler C. Concise review: adipose tissue- derived stromal cells basic and clinical implications for novel cell-based therapies. Stem Cells. 2007 Apr; 25 (4): 818-827.
- Yuan Q, Zeng X, Chen L, Peng E, Ye Z. [Comparison o[[f myogenic differentiation ability of adipose-derived stem cells from different sites in rabbit]. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2010; 24: 1228-1232.
- Cuce G, Kalkan SS, Esen HH. Evaluation of TGF beta1 expression and comparison the thickness of different aorta layers in experimental diabetes. Bratisl Lek Listy. 2011;112(6): 318-322.
- Augello A, Kurth TB, De Bari C. Mesenchymal stem cells: a perspective from in vitro cultures to in vivo mi- gration and niches. Eur Cell Mater. 2010; 1:20: 121-33.
- Caesar R, Manieri M, Kelder T, Boekschoten M, Evelo C, Müller M, Kooistra T, Cinti S, Kleemann R, Drevon CA. A combined transcriptomics and lipidomics analy- sis of subcutaneous, epididymal and mesenteric adipose tissue reveals marked functional differences. PLoS One. 2010; 5: e11525.
- Dragusha G, Elezi A, Dragusha S, Gorani D, Begolli L. Treatment benefits on metabolic syndrome with diet and physical activity. Bosn J Basic Med Sci. 2010; 10: 169-176.
- Ginter E, Simko V. Diabetes type 2 pandemic in 21st century. Bratisl Lek Listy. 2010; 111: 134-137.
- 20.Barry FP, Boynton RE, Haynesworth S, Murphy JM, Zaia J. The monoclonal antibody SH-2, raised against human mesenchymal stem cells, recognizes an epitope on endoglin (CD105). Biochem Biophys Res Commun. 1999; 265: 134-139.
- Dales JP, Garcia S, Carpentier S, Andrac L, Ramuz O, Lavaut MN, Allasia C, Bonnier P, Charpin C. Long- Term Prognostic Significance of Neoangiogenesis in Breast Carcinomas: Comparison of Tie-2/Tek, CD105, and CD31 Immunocytochemical Expression. Hum Pa- thol. 2004; 35: 176-183.
- Jin HJ, Park SK, Oh W, Yang YS, Kim SW, Choi SJ. Down-regulation of CD105 is associated with multi- lineage differentiation in human umbilical cord blood- derived mesenchymal stem cells. Biochem Biophys Res Commun. 2009; 381: 676-681.