Research Article - Biomedical Research (2017) Volume 28, Issue 1
Experimental occlusal interference on brain activation during gum chewing
Takero Otsuka1*#, Kenichi Sasaguri2#, Yuta Hayashi1, Kentaro Yamada3, Hiroyuki Muranaka4, Yu Takakura5, Hitoshi Usuniwa6, Nagafumi Doi6, Toshitsugu Kawata11Department of Oral Science, Orthodontic Division, Kanagawa Dental University Graduate School, Yokosuka, Kanagawa, Japan
2Department of Dentistry, Oral and Maxillofacial Surgery, Jichi Medical University, Shimotsuke, Tochigi, Japan
3Department of Oral Science, Brain Functions and Neuroscience Division, Kanagawa Dental University Graduate School, Yokosuka, Kanagawa, Japan
4Department of Radiology, Faculty of Health Science, Tsukuba International University, Tsuchiura, Ibaraki, Japan
5Toride-Kitasoma Health Medical Center, Medical Association Hospital, Toride, Ibaraki, Japan
6Ibaraki Prefectural Medical Center of Psychiatry, Kasama, Ibaraki, Japan
#All authors equally contributed for the work
- *Corresponding Author:
- Takero Otsuka
Department of Oral Science
Orthodontic division
Kanagawa Dental University Graduate School, Japan
Accepted on June 24, 2016
Abstract
This study aimed to investigate brain functions by analysing changes in intensity of brain activation sites as well as the results of sensory evaluation related to occlusal interference. In 11 subjects, an interference crown was made for the left mandibular first molar, and subjects performed a gum-chewing task with crown overlay (200 μm of occlusal interference) and without (control). Sensation was evaluated using a Visual Analog Scale (VAS), and brain activation sites were evaluated using functional Magnetic Resonance Imaging (fMRI). Results were considered statistically significant at p<0.05. Unpleasant sensation during gum chewing was significantly greater in the interference group than the control group (p<0.01). On fMRI group analysis, the somatosensory-motor cortex, supplementary motor area, the insula, and the cerebellum were activated in both groups, thus establishing an experimental model to analyse the relationship between occlusal interference and brain function. In the interference group, activation was significantly higher in the amygdala (p<0.001), anterior cingulate cortex (p<0.001), prefrontal area (p<0.05), and hypothalamus (p<0.05). Our findings show that stimulation from occlusal interference is rapidly relayed to the brain and related to activation of emotions of discomfort.
Keywords
Gum-chewing, Occlusal interference, fMRI, Hypothalamus, Stress response.
Introduction
Some recent reports suggest that occlusal dysfunction may have systemic effects by acting on brain function [1,2]. We previously reported an experimental model of occlusal interference using experimental animals by increasing the occlusal vertical dimension artificially and attaching wires to the occlusal surface of the maxillary molar teeth in rats. Plasma corticosterone and norepinephrine levels in these rats were significantly elevated, thus eliciting chronic stress. As a result, there were changes in long-term potentiation (LTP) in the hippocampus [3]. In addition, in an experimental model of occlusal interference in monkeys by overlaying metal splints on the maxillary molars, urinary cortisol levels rapidly increased, followed 3 weeks later by significant changes in blood cortisol and the lymphocyte count. There was also alveolar bone resorption thought to be due to occlusal trauma, thus demonstrating that occlusal dysfunction led to bruxism associated with emotional stress [4,5].
Meanwhile, with advances in neuroimaging technology such as functional Magnetic Resonance Imaging (fMRI), Magneto Encephalography (MEG), and near-infrared spectroscopy (NIRS) to noninvasively evaluate brain function, the influence of afferent information from the oral cavity on brain function can now also be investigated in humans in the field of dentistry. Our group previously used fMRI during an alternating clenching-rest task when the mandible was forced into a retrusive position using a splint. During clenching with the mandible in a retrusive position compared to when using a control splint, discomfort as assessed with the Visual Analog Scale (VAS) was significantly greater, and activation in the amygdala and Anterior Cingulate Cortex (ACC) was significantly increased. Those findings showed the possibility that with mandibular deviation, afferent information is rapidly sent to the limbic system, thus eliciting emotional stress [1].
The aim of the present study was to investigate the influence of occlusal dysfunction on brain function. A crown of uniform thickness of about 200 μm was made for each subject using a gold electroforming technique. A chewing task was performed during experimental occlusal interference with overlay (interference group) or no overlay (control group) of the crown on the left mandibular first molar, and brain blood flow and function were measured.
Material and Methods
Subjects
The fMRI data were obtained from 11 healthy subjects (five males and six females, mean age 28.7 years) with no history of psychiatric or neurological disease. Each subject gave written informed consent after receiving an explanation of the aims and method of the study, which was approved by the Committee for Research Ethics of Hospital Prefectural Medical Center of Psychiatry, Kasama, Japan and conformed to the STROBE guidelines for reporting observational studies (http:// www.strobe-statement.org).
Preparation of the crown with a gold electroforming technique for the left mandibular first molar tooth to be used for occlusal interference
This study was carried out by modifying the gold electroforming method developed by Rogers [6]. An impression was made of the lower dental arch in each subject, and a cast was prepared with high strength dental stone. While preserving the mesiodistal surface as much as possible, the left mandibular first molar was segmented and drilled, and a working cast was made.
A dedicated electrically conductive silver lacquer was applied to the crown mold (AGC® Switch Conductive silver lacquer; Wieland Dental & Technik GmbH & Co., KG, Pforzheim, Germany). This was immersed in a dedicated gold bath (AGC® Gold Bath; Wieland Dental & Technik GmbH & Co., KG), a brightener (AGC® Brightener additive; Wieland Dental & Technik GmbH & Co., KG) was added, and AGC® Micro (Wieland Dental & Technik GmbH & Co., KG) was used to fabricate the crown used for interference. Next, an acidic bath, glass beads (mean diameter 75 μm, Shofu Inc., Kyoto, Japan), and a pencil-type sandblaster (mean diameter 75 μm, 2 atmospheres, Hi-Blaster III, Shofu Inc.) were used to remove the silver lacquer from the mold. The crown molds were ultrasonically cleaned in distilled water for 10 min [7-9].
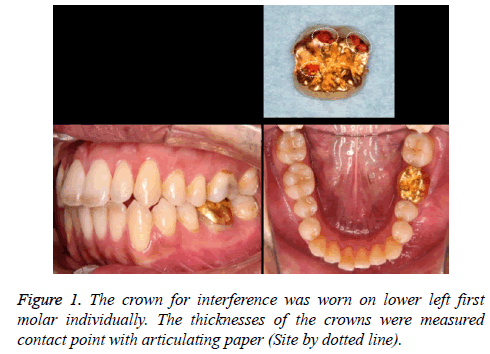
After completion, articulating paper was placed on the crown mold (in the oral cavity), three or four contact points were marked, and the thickness at these sites was measured as shown in Figure 1.
Experimental task
The task paradigm was periods of rhythmic chewing at a rate of approximately 1 Hz as measured with a metronome [10,11]. The gum, which did not have odour or taste components, was specially prepared in the General Laboratory of Lotte Co. Ltd. (Saitama, Japan).
Each subject performed the gum-chewing task with or without the overlay. Each subject performed a series of four cycles of gum chewing, each cycle consisting of 32 s of chewing and 32 s of resting. A recovery/reset period of 20-30 min separated the two chewing series (one with the overlay and one without the overlay as a control) in each subject to eliminate the influence of one task on the other.
VAS questionnaire
Each subject quantified their subjective feeling of discomfort during chewing with a mark on the VAS [12], which ranged from 0 to 10 (0=no discomfort, 10=extreme discomfort). Participants also verbally expressed to an interviewer how they felt during chewing. The paired t-test was used to compare the two VAS scores of each subject, and statistical significance was set at p<0.05.
fMRI protocol
The principle of fMRI is based on the blood oxygenation level dependency (BOLD) effect, which detects signal fluctuations due to changes in the ratio of oxyhemoglobin to deoxyhemoglobin in the blood in relation to brain activity [13]. Functional (T2-weighted) images and anatomical (T1- weighted) images were acquired with a Discovery MR750 3.0- T MRI system (General Electric, Fairfield, CT, USA). The functional images consisted of echo-planar image volumes sensitive to BOLD contrast in the axial orientation (TE=45 ms, TR=4000 ms). The volume included the entire brain with a 64 × 64 matrix and 34 slices (voxel size=3.75 mm × 3.75 mm × 4 mm, slice thickness=3 mm, gap=0.3 mm). Images with 72 volumes were acquired for each experimental condition. Because unstable magnetization routinely degraded the quality of the first eight volumes, the start of the first cycle of chewing was timed to coincide with the beginning of the ninth image volume.
Data analysis
Head motion was corrected using SPM8 (University College London, London, UK). A total of 144 functional images (two runs, 72 image volumes per run) obtained from each subject were normalized to the Montreal Neurological Institute template [14] and spatially smoothed with an 8 mm Gaussian kernel. The data were statistically analysed with the general linear model approach [15] using SPM8. Proportional scaling removed global changes in the BOLD signals. Per cent BOLD signal change for each area of interest was exported to Statcel version 3 (add-in software for Microsoft Excel; OMS Saitama Japan). Results were considered statistically significant at p<0.05.
Results
The crowns used for interference were placed on the left mandibular first molar in each subject, intercuspation was confirmed with articulating paper, and the thickness at each of the three to four contact points with the opposing left maxillary first molar was measured. This confirmed that the fabricated crowns for occlusal interference had nearly uniform thickness of 200 ± 8.636 μm as shown in Figure 1.
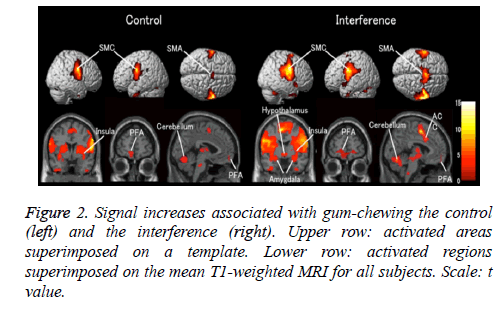
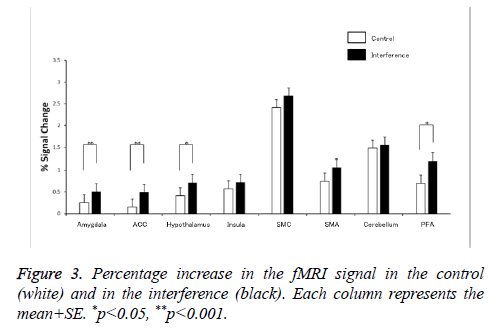
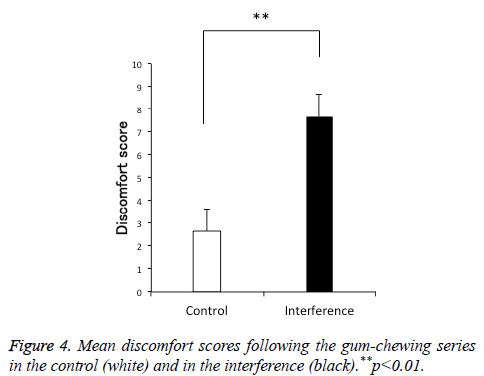
Comparison of the discomfort score using the VAS showed significantly more discomfort in the interference group than in the control group as shown in Figure 2. Group analysis of both chewing tasks (with and without overlay) in the 11 subjects showed activation of the prefrontal area (PFA), somatosensory cortex, supplementary motor area (SMA), insula, amygdala, cerebellum, ACC, and hypothalamus. Activation of the amygdala, ACC, PFA, and hypothalamus was significantly higher in the interference group than in the control group as shown in Figures 3 and 4.
The locations of the most significant foci of activation for these regions are summarized in Table 1, which also shows the anatomical regions with maximal t values in clusters and the coordinates according to the Talairach and Tournoux atlas as shown in Table 1.
| Talairach Coordinates of Max-voxel | ||||||
|---|---|---|---|---|---|---|
| Region of Activation | BA | Maximal t Value | x | y | z | |
| Control | Amygdala | 0.57 | -20 | 2 | -22 | |
| ACC | 0.59 | -2 | 14 | 26 | ||
| Hypothalamus | 1.54 | -2 | -2 | -6 | ||
| Insula | 13 | 6.69 | -34 | 2 | -6 | |
| Sensorimotor cortex | 3.4 | 11.12 | 62 | 0 | 24 | |
| Supplementary motor area | 6 | 5.75 | 6 | -2 | 56 | |
| Cerebellum | 18.38 | 18 | -60 | -20 | ||
| Prefrontal area | 10.46 | 2.42 | -6 | 58 | -12 | |
| 1st molar | Amygdala | 5.93 | -20 | -8 | -14 | |
| ACC | 6.86 | 6 | 10 | 42 | ||
| Hypothalamus | 4.3 | 2 | -2 | -6 | ||
| Insula | 13 | 8.97 | -42 | -4 | 18 | |
| Sensorimotor cortex | 3.4 | 10.86 | 54 | -2 | 28 | |
| Supplementary motor area | 6 | 10.41 | -8 | 0 | 52 | |
| Cerebellum | 14.92 | 20 | -60 | -24 | ||
| Prefrontal area | 10.46 | 4.24 | -10 | 58 | -8 | |
Table 1. Significant Increases in the fMRI Signal during Gumchewing: Anatomical Regions, Brodmann's Area (BA), and Maximal t Values with Co-ordinates as given in the Talairach and Tournoux Atlas.
Discussion
It has recently been shown that rapid input of afferent information from abnormal oral sensation due to mandibular deviation and malocclusion causes activation of brain sites closely related to emotional stress, that this correlates with discomfort as assessed with the VAS, and that this may also affect systemic function [1,2]. Our previous animal study showed that occlusal dysfunction due to experimental occlusal interference increases serum stress hormones and also affects LTP, which is thought to be a parameter of hippocampal function 3. Therefore, in this study, we fabricated a crown for the left mandibular first molar for 200 μm of experimental occlusal interference, and during a gum-chewing task, we used 3.0 Tesla MRI to investigate detailed changes in brain functional responses associated with changes in input of afferent information.
In clinical dental practice, a final prosthesis is usually made with about 100-200 μm of excess height, and this is adjusted chair-side based on the patient's subjective responses. Therefore, we fabricated crowns for experimental occlusal interference with a uniform thickness of about 200 μm using a gold electroforming technique. The gold electroforming technique was originally developed by Rogers [6]. This is now a zincate-based process using a gold solution to deposit pure gold on a master cast [7-9]. This is a convenient fabrication process with high precision and was used to make the crowns for occlusal interference. The measured thickness of these crowns for occlusal interference was 200 ± 8.636 μm. Thus, crowns with nearly uniform thickness could be fabricated.
In a previous study by our group using NIRS, the same vibrotactile stimulus of the first molars compared to other tooth types caused significantly greater activation of the somatosensory cortex [16]. In another study of clenching using splints of a shortened dental arch and complete dental arch, cerebral blood flow was significantly increased for the complete dental arch compared to the shortened dental arch [17]. Therefore, we selected the left mandibular first molar because we considered this advantageous for measurements with brain mapping during experimental occlusal interference.
Our study showed activation of the somatosensory cortex, SMA, insula, and cerebellum in both groups, similar to prior studies of functional brain imaging in humans during gumchewing tasks [10,11,18]. This was thought to establish an experimental model of occlusal interference during gumchewing movement. Chewing movement leads to input of sensory information from the oral cavity to the somatosensory cortex, and this information, via motor association areas such as the premotor cortex and SMA, is thought to activate motor areas that are essential for carrying out these movements [10,11,18]. In addition, the PFA is not simply involved with the generation and control of movement, but is also involved in higher-order behavioural control such as programming, suitable actions, and selection to carry out the target movement [19].
In our study, occlusal interference was thought to increase afferent information input from the oral cavity, thus making normal jaw movement difficult and increasing the functional demands for carrying out chewing movement. As a result, in performing complicated movement related to planning, preparation, and execution of movement in the somatosensory cortex, activation in the SMA, which is thought to function in light of prior memory, also tended to be increased [20]. Furthermore, with increased input of information to the somatosensory cortex and SMA from the oral cavity, activation of the PFA, a site involved in integration and control, was also significantly increased.
The amygdala, an emotional center, distinguishes comfort and discomfort. This type of information is projected to the hypothalamus and periaqueductal gray matter and then is manifested in the body as an emotional response [21,22]. Cerebral association areas, particularly the PFA, are connected with amygdala. In our study, increased information input to the amygdala due to increased activation of the aforementioned PFA was induced by occlusal interference, and afferent information input of discomfort due to occlusal interference resulted in significantly increased activation of the amygdala in the interference group. In addition, the cingulate cortex has reciprocal connections with the amygdala and is thought to play an important role in emotions, cognition, and memory. Especially, the ACC is thought to be involved in learning, eliciting emotions to pain, response selection, and anticipation and avoidance [23]. Activation in both the ACC and amygdala was significantly increased in the occlusion interference group. Therefore, the ACC, like the amygdala, integrates afferent information from the oral cavity and plays an important role in accessing and controlling emotions of discomfort.
The hypothalamus is connected with the amygdala and plays an important role in emotional responses, but brain mapping studies with measurement of hypothalamic activation associated with changes in oral function have not been reported to date. In our previous study, we observed activation near the hypothalamus with individual analysis, but no significant differences with group analysis (Data not shown). Moreover, that study was limited by evaluation with the spatial resolution of a 1.5 Tesla MRI system [1]. Our present study using a 3 Tesla MRI system is the first to show significantly increased activation in the hypothalamus with group analysis in the interference group. The hypothalamus is a regulatory center for the autonomic nervous system, endocrine system, and somatic nervous system, and this controls the body's stress responses [24]. Increased output of information to the hypothalamus caused by increased activation of the amygdala due to occlusal interference is thought to affect the autonomic nervous system, endocrine system, and somatic nervous system and thus greatly influences the expression of emotions. This activation of the amygdala and the hypothalamus as observed in our study suggests that mandibular deviation and occlusal interference (malocclusion) due to inappropriate, irreversible occlusal treatment in the oral cavity may elicit body stress responses associated with emotional responses, may affect the autonomic nervous system and endocrine system, and may lead to adverse systemic effects.
Limitation
One possible limitation of our present study is the small sample size. In addition, we included only healthy volunteers of a young age. There may be changes in occlusion with age, and it is not clear if our results can be generalized beyond young adults with normal occlusion. Larger and more varied samples should be studied to establish the generalizability of our results to patient populations with a variety of occlusions.
Conclusion
Our findings demonstrate not only that oral dysfunction due to occlusal interference (malocclusion) affects oral sensation and chewing movement, but also that the associated discomfort leads to activation of emotional areas, particularly the amygdala and ACC. This further leads to increased activation of the hypothalamus, which is a stress response center. Changes in activation of these emotional areas correlate with subjective assessment, thus suggesting that inappropriate, irreversible occlusal treatment may elicit chronic stress and have systemic effects. Treatment goals in clinical dental practice to date have been based on patients’ subjective responses, but objective treatment parameters have been completely lacking. Therefore, new methods of diagnosis and treatment should urgently be established based on objective findings using neuroimaging technology as a parameter in the future.
Acknowledgements
This study was supported by JSPS KAKENHI Grant Number 24792118. The authors received no financial support and declare no potential conflicts of interest with respect to the authorship and/or publication of this article.
References
- Otsuka T, Watanabe K, Hirano Y. Effects of mandibular deviation on brain activation during clenching: an fMRI preliminary study. Cranio 2009; 27: 88-93.
- Greven M, Otsuka T, Zutz L, Weber B. The amount of TMJ Displacement correlates with brain activity. Cranio 2011; 29: 291-296.
- Kato K, Ono Y, Kubo KY. Occlusal disharmony suppresses long-term potentiation in the rat hippocampal CA1 region. Int J Stomatol Occlusion Med 2010; 3: 71-75.
- Budtz JE. Bruxism and trauma from occlusion. An experimental model in Macaca monkeys. J Clin Periodontol 1980; 7: 149-162.
- BudtzJE. Occlusal dysfunction and stress. An experimental study in macaque monkeys. J Oral Rehabil 1981; 8: 1-9.
- Rogers OW, Armstrong BW. Electroforming a gold matrix for indirect inlays. J Prosthet Dent 1961; 11: 959-966.
- Behrend F. Gold Electroforming System: GES restorations. J Dent Technol 1997; 14(2): 31-37.
- Vence BS. Electroforming technology for galvanoceramic restorations. J Prosthet Dent 1997; 77: 444-449.
- Pettenò D, Schierano G, Bassi F, Bresciano ME, Carossa S. Comparison of marginal fit of 3 different metal-ceramic systems: an in vitro study. Int J Prosthodont 2000; 13: 405-408.
- Onozuka M, Fujita M, Watanabe K. Mapping brain region activity during chewing: A functional magnetic resonance imaging study. J Dent Res 2002; 81: 743-746.
- Onozuka M, Fujita M, Watanabe K. Age-related changes in brain regional activity during chewing: a functional magnetic resonance imaging study. J Dent Res 2003; 82: 657-660.
- Benedetti F, Bernasconi A, Pontiggia A. Depression and neurological disorders. Curr Opin Psychiatry 2006; 19: 14-18.
- Ogawa S, Lee TM, Kay AR, Tank DW. Brain magnetic resonance imaging with contrast dependent on blood oxygenation. Proc Natl Acad Sci U S A 1990; 87: 9868-9672.
- Collins DL, Neelin P, Peters TM, Evans AC. Automatic 3D intersubject registration of MR volumetric data in standardized Talairach space. J Comput Assist Tomogr 1994; 18: 192-205.
- Friston KJ, Holmes AP, Worsley KJ, Poline J-P, Frith CD, Frackowiak RSJ. Statistical parametric maps in functional imaging: A general linear approach. Hum Brain Mapp 1994; 2: 189-210.
- Shimazaki T, Otsuka T, Akimoto S, Kubo KY, Sato S, Sasaguri K. Comparison of Brain Activation via Tooth Stimulation. J Dent Res 2012; 91: 759-763.
- Miyamoto I, Yoshida K, Bessho K. Shortened dental arch and cerebral regional blood volume: an experimental pilot study with optical topography. Cranio 2009; 27: 94-100.
- Takada T, Miyamoto T. A fronto-parietal network for chewing of gum: a study on human subjects with functional magnetic resonance imaging. Neurosci Lett 2004; 360: 137-140.
- Goto K, Hoshi Y, Sata M, Kawahara M, Takahashi M, Murohashi H. Role of the prefrontal cortex in the cognitive control of reaching movements: near-infrared spectroscopy study. J Biomed Opt 2011; 16: 127003.
- Nachev P, Kennard C, Husain M. Functional role of the supplementary and pre-supplementary motor areas. Nat Rev Neurosci 2008; 9: 856-869.
- Buijs RM, Van Eden CG. The integration of stress by the hypothalamus, amygdala and prefrontal cortex: balance between the autonomic nervous system and the neuroendocrine system. Prog Brain Res 2000; 126: 117-132.
- Drabant EM, Kuo JR, Ramel W. Experiential, autonomic, and neural responses during threat anticipation vary as a function of threat intensity and neuroticism. Neuroimage 2011; 55: 401-410.
- Allman JM, Hakeem A, Erwin JM, Nimchinsky E, Hof P. The anterior cingulate cortex. The evolution of an interface between emotion and cognition. Ann N Y Acad Sci 2001; 935: 107-117.
- Austin AW, Wissmann T, von Kanel R. Stress and hemostasis: an update. Semin Thromb Hemost 2013; 39: 902-912.