Research Article - Biomedical Research (2017) Volume 28, Issue 3
Evaluation of the severity of liver cirrhosis using b values based magnetic resonance diffusion weighted imaging of liver lobe
Zhenyu Shu1, Xiangyang Gong1*, Songhua Fang1, Dewang Mao1, Jie Zheng1, Jianguo Xu1, Qiong Song1 and Yueqiao Zhang2
1Department of Radiology, Zhejiang Provincial People's Hospital, Hangzhou 310014, China
2Department of Radiology, Sir Run Run Shaw Hospital, Medical School, Zhejiang University, Hangzhou 310016, China
- *Corresponding Author:
- Xiangyang Gong
Department of Radiology
Zhejiang Provincial People's Hospital, China
Accepted date: August 10, 2016
Abstract
This study aimed to investigate the correlation of magnetic resonance diffusion weighted imaging of liver lobe and Child-Pugh classification in Chinese patients with liver cirrhosis. 72 liver cirrhosis patients and 23 healthy volunteers were enrolled, and patients were divided into class A, B, C according to Child- Pugh classification. All subjects received diffusion weighted imaging with b values set as 0, 400, 600 and 800 s/mm2. The correlation between ADC values of different lobes and Child-Pugh classification was analyzed. ADC values were significantly lower in patients with liver cirrhosis than in healthy controls. Moreover, ADC value was negatively correlated with Child-Pugh classification. At b value of 400 s/mm2 ADC value of left liver lobe was the best indicator to distinguish normal liver from liver cirrhosis and the area under the ROC curve was 0.914. At b value of 600 s/mm2 ADC value of right liver lobe was the best indicator to distinguish class A from class B-C of liver cirrhosis and the area under the ROC curve was 0.834. ADC value of right liver lobe was also the best indicator to distinguish class A-B from class C of liver cirrhosis and the area under the ROC curve was 0.884. These data indicate that ADC value of liver lobe is correlated with liver cirrhosis severity determined by Child-Pugh classification and is a new noninvasive indicator of the severity of liver cirrhosis.
Keywords
Liver cirrhosis, Diffusion-weighted imaging, Apparent diffusion coefficients, ChiId-Pugh staging, Hepatitis B.
Introduction
The incidence of hepatitis B is high in China. Hepatitis B is known to induce severe liver diseases such as liver cirrhosis and liver cancer which are serious threat to people's health. The evaluation of the severity of liver cirrhosis is very important for the selection of appropriate treatment plan and the monitoring of patient response to treatment [1]. Noninvasive Child-Pugh classification is a common method to assess liver function, treatment outcome and prognosis in patients with chronic liver cirrhosis [2]. Recently magnetic resonance Diffusion Weighted Imaging (DWI) has become another noninvasive approach to assess liver cirrhosis by analyzing the changes of water diffusion based on the apparent diffusion coefficient (ADC) [3]. Several studies showed that ADC values of cirrhotic liver were correlated with the results of Child-Pugh classification for the evaluation of the severity of liver cirrhosis [4-7]. However, whether ADC values of cirrhotic liver are correlated with the results of Child-Pugh classification in Chinese patients with liver cirrhosis remains unclear. This study aimed to investigate the correlation of multiple b values based magnetic resonance DWI of liver lobe and Child-Pugh classification in Chinese patients with liver cirrhosis.
Subjects and Methods
Subjects
Total 72 liver cirrhosis patients who visited our hospital between May 2014 and August 2015 were enrolled in this study. All of them had hepatitis B and did not have other liver diseases and serious chronic illness. They included 49 males and 23 females aged 25-60 years (mean 42 years old). Selected patients underwent MRI scans before and biochemical tests of drugs and surgery. Child-Pugh classification was based on ascites, albumin, bilirubin, prothrombin activity and hepatic encephalopathy, and the patients were divided into Child-Pugh class A 22 cases, Child-Pugh class B 26 cases, and Child-Pugh class C 24 cases. In addition, randomly selected 23 healthy volunteers were enrolled who underwent regular physical examination in our hospital, including 15 males and 8 females aged 2l-58 years (mean age 36 years). All volunteers had no history of liver disease, viral hepatitis and liver dysfunction, and no history of long-term drug use and heavy drinking. This study was approved by the Hospital Ethics Committee and all subjects gave informed consent.
Magnetic resonance imaging (MRI)
All subjects underwent MRI using 1.5 T MRI system (Magnetom Avanto, Siemens, Germany) equipped with 12- channel phased-array body coil. Routine MRI procedures included axial two-dimensional spoiled gradient recalled echo T1-weighted imaging (T1WI), fast recovery axial spin echo (FRFSE) and axial fast recovery fast spin-echo T2-weighted imaging (T2WI) with fat suppression. Next, DWI was performed with b values set at 0, 400, 600 and 800 s/mm2. Then contrast agents were injected for conventional contrast-enhanced scanning. The parameters for DWI sequence scanning were: repetition time 2700-3500 msec, echo time 65-80 msec, matrix 96 × 128, field view of 330-380 mm, slice thickness 7 mm, inter-slice gap 2.1 mm, number of excitations 2, and acquisition time 3-4.5 min.
MRI data analysis
All original data were inputted into a workstation (Lconardo Syngo, Siemens Germany) and analyzed according to the consensus of two senior radiologists who were blind to the subject’s clinical data. The liver was divided into four lobes, including the left lateral liver lobe (LLL), the left medial liver lobe (LML), the right liver lobe (RL), and the caudate lobe (CL). Both radiologists calculated ADC values based on defined regions of interest (ROIs), which were approximately 50 mm2 in the left medial liver lobe or the caudate lobe or 200 mm2 in the left lateral or right liver lobe. Three ROIs were selected for each liver lobe and final ADCs were the averaged ADCs of the three ROIs for b values of 0 and 400 sec/mm2, 0 and 600 sec/mm2, and 0 and 800 sec/mm2, respectively.
Statistical analysis
Data were express as mean ± standard deviation (SD) and analyzed by SPSS19.0 statistical software. To test the correlation of liver lobe-based ADCs with Child-Pugh classification, Spearman correlation analysis was performed. Comparisons between two groups were analyzed using Mann- Whitney U tests followed by Bonferroni correction for possible multiple comparisons. The cutoff value of the potentially distinguishing ADC was calculated based on receiver operating characteristic (ROC) analysis of the area under the ROC curve (AUC). P<0.05 was considered as significant difference.
Results
Correlation of ADCs with child-pugh classification of liver cirrhosis
With different b values, ADC values of each classification of liver cirrhosis group were lower than healthy control group (Table 1). In addition, ADC values of each liver lobe with different b values were negatively correlated with Child-Pugh classification (P<0.05, Table 2).
| b value | healthy volunteers (n=23) | patients with liver cirrhosis (n=72) | Child-Pugh classification | |||
|---|---|---|---|---|---|---|
| A (n=22) | B (n=26) | C (n=24) | ||||
| b (0,400) | LLL | 2.03 ± 0.37 | 1.51 ± 0.18 | 1.47 ± 0.17 | 1.53 ± 0.20 | 1.51 ± 0.18 |
| LML | 1.83 ± 0.44 | 1.52 ± 0.19 | 1.52 ± 0.18 | 1.55 ± 0.19 | 1.49 ± 0.21 | |
| RL | 1.65 ± 0.31 | 1.37 ± 0.14 | 1.42 ± 0.14 | 1.37 ± 0.13 | 1.32 ± 0.15 | |
| CL | 1.87 ± 0.38 | 1.43 ± 0.16 | 1.56 ± 0.13 | 1.44 ± 0.14 | 1.33 ± 0.12 | |
| b (0,600) | LLL | 1.80 ± 0.37 | 1.38 ± 0.14 | 1.45 ± 0.14 | 1.42 ± 0.11 | 1.32 ± 0.11 |
| LML | 1.60 ± 0.27 | 1.29 ± 0.13 | 1.37 ± 0.16 | 1.25 ± 0.14 | 1.18 ± 0.09 | |
| RL | 1.62 ± 0.27 | 1.23 ± 0.14 | 1.33 ± 0.18 | 1.24 ± 0.11 | 1.17 ± 0.09 | |
| CL | 1.68 ± 0.31 | 1.28 ± 0.12 | 1.31 ± 0.09 | 1.29 ± 0.12 | 1.21 ± 0.13 | |
| b (0,800) | LLL | 1.61 ± 0.25 | 1.31 ± 0.17 | 1.35 ± 0.14 | 1.31 ± 0.18 | 1.26 ± 0.18 |
| LML | 1.49 ± 0.21 | 1.26 ± 0.14 | 1.27 ± 0.16 | 1.28 ± 0.14 | 1.24 ± 0.12 | |
| RL | 1.42 ± 0.19 | 1.20 ± 0.11 | 1.20 ± 0.12 | 1.23 ± 0.12 | 1.17 ± 0.11 | |
| CL | 1.59 ± 0.25 | 1.32 ± 0.16 | 1.39 ± 0.17 | 1.34 ± 0.16 | 1.26 ± 0.16 | |
| ADC values were expressed as ×10-3mm2/s. LLL: Left Lateral Lobe; LML: Left Liver Lobe; RL: Right Lobe; CL: Caudate Lobe. | ||||||
Table 1: Comparison of ADC values of different groups.
| b value | 400 s/mm2 | 600 s/mm2 | 800 s/mm2 | |
|---|---|---|---|---|
| ADC of LLL | r | -0.45 | -0.42 | -0.48 |
| p | <0.001 | <0.001 | <0.001 | |
| ADC of LML | r | -0.38 | -0.49 | -0.24 |
| p | <0.001 | <0.001 | 0.036 | |
| ADC of RL | r | -0.51 | -0.55 | -0.33 |
| p | <0.001 | <0.001 | 0.002 | |
| ADC of CL | r | -0.61 | -0.57 | -0.51 |
| p | <0.001 | <0.001 | <0.001 | |
| LLL: Left Lateral Lobe; LML: Left Liver Lobe; RL: Right Lobe; CL: Caudate Lobe. | ||||
Table 2: Correlation of ADC values with cirrhosis Child-Pugh classification.
Comparison of ADC values of different liver lobes
ADC values showed significant differences between healthy control group and liver cirrhosis group at different b values. After Bonferroni correction, ADC values of caudate lobe between class A and class B-C as well as between class A-B and class C still showed significant differences when b value was 400 s/mm2. After Bonferroni correction ADC values of left lateral liver lobe and right liver lobe between class A and class B-C as well as between class A-B and class C still showed significant differences, and ADC values of caudate lobe between class A-B and class C still showed significant differences when b value was 600 s/mm2. After Bonferroni correction, ADC values of caudate lobe between class A-B and class C still showed significant differences when b value was 800 s/mm2 (Table 3).
| b value | Normal vs. cirrhosis | Comparison between different Child-Pugh class | ||
|---|---|---|---|---|
| A vs. B-C | A-B vs. C | |||
| b (0,400) | LLL | <0.001* | 0.266 | 0.806 |
| LML | <0.001* | 0.75 | 0.135 | |
| RL | <0.001* | 0.055 | 0.057 | |
| CL | <0.001* | <0.001* | <0.001* | |
| b (0,600) | LLL | <0.001* | 0.003* | <0.001* |
| LML | <0.001* | 0.352 | 0.031 | |
| RL | <0.001* | 0.001* | <0.001* | |
| CL | <0.001* | 0.036 | 0.004* | |
| b (0,800) | LLL | <0.001* | 0.141 | 0.134 |
| LML | <0.001* | 0.859 | 0.373 | |
| RL | <0.001* | 0.99 | 0.066 | |
| CL | <0.001* | 0.054 | 0.012* | |
| LLL: Left Lateral Lobe; LML: Left Liver Lobe; RL: Right Lobe; CL: Caudate Lobe. *P<0.05 indicated significant difference after Bonferroni correction. | ||||
Table 3: ADC values to distinguish different severity of liver cirrhosis.
ROC analysis of the distinguishing ability of ADC values for liver cirrhosis
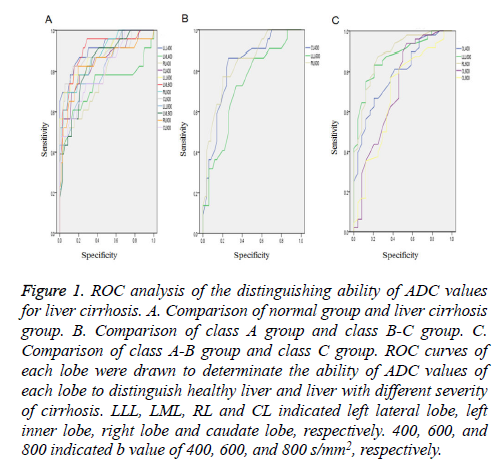
When b value was 400 s/mm2, ADC value of left lateral lobe was the best indicator to distinguish between normal liver and liver cirrhosis, the area under the ROC curve (AUC) was 0.914. With ADC value ≤ 1.68 × 10-3 mm2/s as the standard, the sensitivity was 84.72% and the specificity was 82.61%.
When b value was 600 s/mm2, ADC value of right lobe was the best indicator to distinguish between class A and class B-C and the area under the ROC curve (AUC) was 0.834. With ADC value ≤ 1.32 × 10-3 mm2/s as the standard, the sensitivity was 80% and the specificity was 77.27%. ADC value of right lobe was also the best indicator to distinguish between class AB and class C, and the area under the ROC curve (AUC) was 0.884. With ADC value ≤ 1.21 × 10-3 mm2/s as the standard, the sensitivity was 75% and the specificity was 87.5% (Table 4 and Figures 1A-1C).
Figure 1: ROC analysis of the distinguishing ability of ADC values for liver cirrhosis. A. Comparison of normal group and liver cirrhosis group. B. Comparison of class A group and class B-C group. C. Comparison of class A-B group and class C group. ROC curves of each lobe were drawn to determinate the ability of ADC values of each lobe to distinguish healthy liver and liver with different severity of cirrhosis. LLL, LML, RL and CL indicated left lateral lobe, left inner lobe, right lobe and caudate lobe, respectively. 400, 600, and 800 indicated b value of 400, 600, and 800 s/mm2, respectively.
| b valuelobe | Threshold(×10-3mm2/s) | Staging | AUC | Sensitivity (%) | Specificity (%) | |
|---|---|---|---|---|---|---|
| b (0,400) | LLL | 1.68 | Normal vs. cirrhosis | 0.914 | 84.72 | 82.61 |
| LML | 1.75 | Normal vs. cirrhosis | 0.726 | 86.11 | 60.87 | |
| RL | 1.56 | Normal vs.cirrhosis | 0.788 | 90.28 | 52.17 | |
| CL | 1.55 | Normal vs. cirrhosis | 0.846 | 79.17 | 78.26 | |
| CL | 1.45 | A vs. B-C | 0.82 | 74 | 86.36 | |
| CL | 1.41 | A-B vs. C | 0.788 | 79.17 | 66.67 | |
| b (0,600) | LLL | 1.63 | Normal vs. cirrhosis | 0.877 | 95.83 | 69.57 |
| LML | 1.37 | Normal vs. cirrhosis | 0.902 | 84.72 | 82.61 | |
| RL | 1.46 | Normal vs. cirrhosis | 0.874 | 91.67 | 69.57 | |
| CL | 1.36 | Normal vs. cirrhosis | 0.883 | 86.11 | 82.61 | |
| LLL | 1.37 | A vs. B-C | 0.721 | 66.02 | 72.73 | |
| LLL | 1.32 | A-B vs. C | 0.87 | 79.17 | 83.33 | |
| RL | 1.32 | A vs. B-C | 0.834 | 80 | 77.27 | |
| RL | 1.21 | A-B vs. C | 0.884 | 75 | 87.5 | |
| CL | 1.2 | A-B vs. C | 0.687 | 53.19 | 89.58 | |
| b (0,800) | LLL | 1.66 | Normal vs. cirrhosis | 0.831 | 100 | 52.17 |
| LML | 1.33 | Normal vs. cirrhosis | 0.831 | 70.83 | 82.61 | |
| RL | 1.28 | Normal vs. cirrhosis | 0.832 | 80.56 | 82.61 | |
| CL | 1.42 | Normal vs. cirrhosis | 0.813 | 77.78 | 73.91 | |
| CL | 1.22 | A-B vs. C | 0.692 | 63.83 | 77.08 | |
| LLL: Left Lateral Lobe; LML: Left Liver Lobe; RL: Right Lobe; CL: Caudate Lobe. AUC represented the area under the ROC curve. | ||||||
Table 4: ROC analysis of ADC values to diagnose liver cirrhosis.
Discussion
Early intervention is very important for patients with liver cirrhosis, timely and effective treatment could prevent liver cirrhosis progression and avoid chronic liver failure and end-stage liver disease. Therefore, Child-Pugh classification of patients with liver cirrhosis is necessary to assess the severity of liver cirrhosis [8]. However, Child-Pugh classification has several shortcomings and it is urgent to develop better approaches for the evaluation of liver cirrhosis severity. In this study, we reported that the assessment of liver cirrhosis by magnetic resonance imaging DWI was correlated with that of Child-Pugh classification, suggesting that DWI is a novel non-invasive approach for the diagnosis of liver cirrhosis.
Up to now DWI is the only non-invasive imaging method to detect the free diffusion of water molecules in living tissue [9]. In patients with cirrhosis at different stages of progression, the water content will be different and can be determined by DWI. In this study we chose b value at 400, 600 and 800 s/mm2 to measure ADC values of each liver lobe. We found that ADC values were lower in patients with liver cirrhosis than in healthy controls, consistent with previous report [10]. Interestingly, we found that at any b value ADC values of right lobe was always lower than those in other lobes. The reason may be that during the course of liver cirrhosis the size of right lobe would be reduced. Consequently, the blood flow velocity would be affected and the diffusion of water molecules would be significantly restricted in right lobe compared to other lobes.
Previous study reported that ADC values measured with b value set as 400 s/mm2 were the most accurate for the diagnosis and evaluation of liver cirrhosis [11]. In agreement with previous report, in this study we found that ADC values derived from b value at 400 s/mm2 was the best indicator for the presence of liver cirrhosis, and the AUC reached 0.918. In addition, we found that left lateral lobe was the best region to detect the presence of cirrhosis. During liver cirrhosis, liver cells regenerate to compensate liver function, and liver cell regeneration is the most abundant in left lateral lobe among all lobes. Thus ADC values of left lateral lobe change significantly, and are the best indicator for the presence of cirrhosis [12].
This study has certain limitations. First, we only chose several different b values to measure ADC. ADC values derived from a wider range of b values may be better for the diagnosis and evaluation of liver cirrhosis. Second, we only analyzed the correlation of ADC value of liver lobe with the severity of liver cirrhosis assessed by Child-Pugh classification and not with other indexes of liver cirrhosis severity such as MELD score or fibroscan value [13]. Further studies are needed to analyze the correlation of ADC value of liver lobe with MELD score or fibroscan value.
In summary, ADC value of left liver lobe derived from b value of 400 s/mm2 was the best indicator to judge the presence of liver cirrhosis, and ADC value of right liver lobe derived from b value of 600 s/mm2 was the best indicator to judge the severity of liver cirrhosis. These data indicate that ADC value of liver lobe is a new noninvasive indicator of the severity of liver cirrhosis.
References
- Heintges T, Mohr L, Hensel F. Value of liver biopsy prior to interferon therapy for chronic viral hepatitis. Dig Dis Sci 1998; 43: 1562-1565.
- Pagliaro L. MELD: the end of Child-Pugh classification? J Hepatol 2002; 36: 141-142.
- Guiu B, Cercueil JP. Liver diffusion-weighted MR imaging: the tower of Babel? Eur Radiol 2011; 21: 463-467.
- Hsu FO, Chiou YY, Chen CY, Liu GC, Chu HC, Liu HC. Diffusion-weighted magnetic resonance imaging of the liver in hepatitis B patients with Child-Pugh A cirrhosis. Kaohsiung J Med Sci 2007; 23: 442-446.
- Luciani A, Vignaud A, Cavet M, Nhieu JT, Mallat A, Ruel L. Liver cirrhosis: intravoxel incoherent motion MR imaging-pilot study. Radiology 2008; 249: 891-899.
- Kim I, Jang YJ, Ryeom H, Lee SM, Lee HJ, Kim GC. Variation in hepatic segmental volume distribution according to different causes of liver cirrhosis: CT volumetric evaluation. J Comput Assist Tomogr 2012; 36: 220-225.
- Yoon JH, Lee JM, Baek JH, Shin CI, Kiefer B, Han JK. Evaluation of hepatic fibrosis using intravoxel incoherent motion in diffusion-weighted liver MRI. J Comput Assist Tomogr. 2014; 38: 110-116.
- Tsochatzis EA, Bosch J, Burroughs AK. Liver cirrhosis. Lancet 2014; 383: 1749-1761.
- Murtz P, Krautmacher C, Traber F. Diffusion-weighted whole body MR imaging with background body signal suppression: a feasibility study at 3.0 Tesla. Eur Radiol 2007; 17: 3031-3037.
- Taouli B, Tolia AJ, Losada M. Diffusion-weighted MRI for quantification of liver fibrosis: preliminary experience. AJR 2007; 189: 799-806.
- Girometti R, Furlan A, Esposito G. Relevance of b-values in evaluating liver fibrosis: a study in healthy and cirrhotic subjects using two single-shot spin-echo echo-planar diffusion-weighted sequences. MagnReson Imaging 2008; 28: 411-419.
- Ikegami T, Shimada M, Imura S. The changes of the medial right lobe, transplanted with left lobe liver graft from living donors. Transplantation 2009; 87: 698-703.
- Barone M, Viggiani MT, Amoruso A, Schiraldi S, Zito A, Devito F, Cortese F, Gesualdo M, Brunetti N, Di Leo A, Scicchitano P, Ciccone MM. Endothelial dysfunction correlates with liver fibrosis in chronic HCV infection. Gastroenterol Res Pract 2015; 2015: 682174.