Research Article - Biomedical Research (2017) Volume 28, Issue 2
Evaluation of bactericidal activity of thymol-eugenol solution as a potential disinfectant agent
Ling Zhao1 and Xue-an Hao2*1Sterilization and Supply Center, First People’s Hospital of Jining City, Jining, Shandong 272000, PR China
2Disinfection and Hospital Infection Control, Jining Municipal Center for Disease Control and Prevention, Jining, Shandong 272011, PR China
- *Corresponding Author:
- Xue-an Hao
Disinfection and hospital infection control
Jining Municipal Center for Disease Control and Prevention, PR China
Accepted date: June 30, 2016
Abstract
Aim: The emergence of nosocomial infections, which are mainly transmitted by contaminated instruments, there is an urgent need of a novel disinfecting agent, which can able to act by the novel method with less residual concentration. Evaluation of antibacterial activity of thymol-eugenol mixture for potential application as disinfectant agent.
Methods: The reference strains of Escherichia coli, Staphylococcus aureus and Pseudomonas aeruginosa were used for the determination of bactericidal activity for possible disinfectant activity. Docking study with enoyl ACP reductase (FabI) of E. coli was also performed to elucidate the possible mechanism of bactericidal action.
Results: Thymol-eugenol mixture inhibited the growth of the bacterial organism on increasing the concentration and the activity becomes more enhanced on by further increasing the residence time. Furthermore, both thymol and eugenol were found as efficient ligand engaging the active site of enoyl ACP reductase (FabI) of E. coli with estimated free energy of binding of -5.19 kcal/mol and -4.73 kcal/ mol. The estimated inhibition constant of thymol and eugenol was also found in agreement with the excellent bioactivity, 156.07 uM and 343.24 uM, respectively.
Conclusions: The present study corroborated that, thymol-eugenol mixture efficiently inhibits the growth of bacterial organism which easily survive on the medical instruments and can act as potential disinfecting agent.
Keywords
Thymol-Eugenol, Bactericidal activity, Medical instruments.
Introduction
In present scenario, infections caused by bacterial organism created a major risk for the survival of the humans [1]. Once the person got infected with any of the infection, he has to be shifted to hospital for better treatment and management of the disease. After admittance to the hospital, the person has become more vulnerable to Hospital Acquired Infection (HAI) apart from the parent infection [2,3]. Consequently, these HAI are deemed as a most common cause of morbidity and mortality of the admitted individuals and altogether increases the associated health care cost [4]. The post-surgical infections due to the bacterial contamination of the site or instruments pose a significant risk to the condition of the patients [5]. It accounts for over 20% of all healthcare-associated infections in surgical patients and responsible for more than one-third of postoperative deaths worldwide [6].
The surgical procedure involves different types of instruments which need to be sterilized before use. Therefore, it is very imperative to disinfect these instruments for the prevention of HAI [7]. The chemicals used as disinfecting agent have been associated with numerous disadvantages, such as, the residual concentrations of the chemicals and no ideal cleaning product exists for processing all types of instruments because each cleaning product has its specific indications for usage and restrictions [8,9]. Thus, the current research focussed on the search of a novel disinfecting agent which is devoid of any of these problems. Moreover, some instruments are susceptible to heat, which need a special agent for effective disinfection e.g. flexible fibre optic endoscopes.
Plant or plant based products are the chief source of the medicinal agent, exhibiting a diverse array of biological properties [10]. Approximately one-third of the top-selling drugs in the world are natural products or derivatives thereof, because of inherited unmatched chemical diversity and practically devoid of any possible side effects [11]. Thymol is well-known for its disinfectant activity because of its potent antibacterial action [12,13]. On the other hand, Eugenol is another plant based product exhibiting potent antibacterial activity [14,15]. Thus, the present study was aimed for the evaluation of antibacterial activity of thymol-eugenol mixture for potential application as disinfectant agent.
Material and Methods
Chemicals
The chemicals have been procured from Sigma-Aldrich (USA) of analytical grade.
Microorganisms
For analyzing the disinfectant activity, the reference strains of Escherichia coli, Staphylococcus aureus and Pseudomonas aeruginosa were used.
Disinfectant
For this assay, the diverse concentration of thymol-eugenol solution was used for the determination of optimal disinfectant concentration. The solutions of thymol-eugenol (1:1) were freshly prepared as 1.0% v/v, 5% v/v, 7.5% v/v and 10% v/v for 5 min, and 10 min. Moreover, the sterile hard water was used as a diluent and as a control. The 2% Sodium thiosulfate was used as a neutralizer to verify whether it was suitable to inactivate every concentration of chemicals. Moreover, the bovine serum albumin (Cohn Fraction V, Sigma) was used to determine the effect of dirty conditions.
Detection of bactericidal activity
The bacterial suspension test was used for the determination of disinfectant activity. Briefly, the tryptic soy broth was used for the inoculation of bacterial strains for 24 hours at 37°C. After incubation, the bacterial suspension was centrifuged for 15 minutes at 2000 rpm and the resulting cell pellets were washed with sterile distilled water. The turbidity of bacterial suspension was adjusted with distilled water to 0.5 McFarland standard.
In the next instance, the bacterial suspension was added to the prepared solution of disinfecting agent (1:10) at the room temperature and then 1 ml was removed for neutralizing solution after the defined contact times in order to terminate the activity. The inoculation was performed on tryptic soy agar Petri plates by the spread-plate technique in triplicate. The plates were then incubated for 24 hours at 37°C. The expressed bacterial colonies were observed in colony-forming units per milliliter (c.f.u./mm). Thus, the plates that have colony count between 30 and 300 were considered significant for the assay. The Reduction Factor (RF) was calculated as the expression of the disinfectant efficacy, on the basis of following relationship
RF=log Nc - log Nd
Nc: Bacterial colony number corresponds to control plates
Nd: Bacterial colony number after contact with disinfectant
The reduction in Log10 with 5 or more suggests satisfactory microbicidal activity. In the case of dirty condition, each of the concentration of the solution was also tested with albumin. For this, bovine serum albumin was added to the disinfectant solution before adding bacterial inoculum.
Docking calculations
Docking calculations were carried out using DockingServer [16,17]. Gasteiger partial charges were added to the ligand atoms. Non-polar hydrogen atoms were merged, and rotatable bonds were defined. Docking calculations were carried out on enoyl ACP reductase (FabI) of E. coli protein model. Essential hydrogen atoms, Kollman united atom type charges, and solvation parameters were added with the aid of AutoDock tools [18]. Affinity (grid) maps of 60 × 60 × 60 Å grid points and 0.375 Å spacing were generated using the Autogrid program [19]. AutoDock parameter set- and distancedependent dielectric functions were used in the calculation of the van der Waals and the electrostatic terms, respectively.
Docking simulations were performed using the Lamarckian Genetic Algorithm (LGA) and the Solis & Wets local search method (Solis and Wets, 1981). Initial position, orientation, and torsions of the ligand molecules were set randomly. All rotatable torsions were released during docking. Each docking experiment was derived from 10 different runs that were set to terminate after a maximum of 250000 energy evaluations. The population size was set to 150. During the search, a translational step of 0.2 Å, and quaternion and torsion steps of 5 were applied.
Results and Discussion
The disinfection of the various concentrations of thymoleugenol was assayed for the potential to attenuate the growth of the bacterial organism responsible for HAI. The results of the investigation have been tabulated in Table 1 in terms of logarithmic reduction values corresponding to contact times of tested disinfectants. Numerous studies showed that, contaminated endoscopes are the major source of nosocomial infections. Whereas, after thorough disinfection of used endoscopes, there is a greater possibility that, it remains became contaminated and facilitates the transmission of the multidrug-resistant organisms across the individuals. Thus, the processing of reusable medical instruments with suitable disinfect is a major concern for medical institutions worldwide.
| Thymol:Eugenol Concentration (in % v/v) | E. coli | S. aureus | P. aeruginosa | |||
|---|---|---|---|---|---|---|
| 5 min | 10 min | 5 min | 10 min | 5 min | 10 min | |
| 1 | 0.22 | 0.22 | 2.31 | 2.31 | 1.03 | 1.03 |
| 5 | 1.34 | 1.35 | 5.52 | 5.52 | 2.50 | 2.50 |
| 7.5 | 6.45 | 6.50 | 6.32 | 6.32 | 4.72 | 4.89 |
| 10 | 9.21 | 9.26 | 8.05 | 8.05 | 5.56 | 5.63 |
Table 1. Disinfectant activity of Thymol-Eugenol against various microorganisms.
In this regard, endoscopes are more vulnerable and need a specialized disinfectant which can able to clean the last remains of the bacteria in short effective time. Glutaraldehyde is a most preferred chemical for the efficient disinfection of the heat-labile endoscopes. As it’s a chemical agent, it possesses certain disadvantage in handling, like, irritation to eyes, nose and throat. Therefore, it is much imperative to develop a green agent that is devoid of any of these characteristics and efficient in cleaning. In the present study, we have used a solution of thymol-eugenol (TE), which belongs to the natural origin and does not have any handling caution. As shown in Table 1, in the case of E. coli, the 1 % concentration of TE showed the same level of reduction at 5 and 10 min duration. While, on increasing the concentration to five-fold (5%), the level of attenuation has been become more than doubled, with slight improvement in the case of 10 min. More pronounced activity has been recorded on further increasing the concentration to 7.5% and 10% with caused logarithmic reduction factor above 5, i.e., 6.45 and 9.21 at 5 min, respectively.
The activity becomes more enhanced on further increasing the residence time. The past two decades have witnessed the increase in incidence of S. aureus infection with simultaneous rise in antibiotic-resistant strains also-in particular, methicillinresistant S aureus (MRSA) and, more recently, vancomycinresistant strains. Thus, it becomes more crucial to determine the effect of TE against S. aureus. The results as presented in Table 1 showed that, TE at a concentration above 5%, showed a logarithmic reduction of bacterial viability above 5. The results also confirmed that, the reduction efficiency is found unaltered on increasing the concentration. The TE solution was found to exhibit moderate activity against P. aeruginosa. It has been found that at all tested concentration except 10%, TE does not show any prominent activity. While at the highest concentration, it showed improved activity, i.e., a logarithmic reduction of 5.56 and 5.63 at 5 min and 10 min, respectively.
Moreover, to assess the effect of dirty condition on the bactericidal activity of TE, the bovine serum albumin was added to TE solutions before adding the bacterial inoculum. The results of the investigation have revealed that, the bactericidal activity of the TE has not been altered in the presence of a simulated organic environment. The results of the study have been found in agreement with the earlier studies which showed that, both thymol and eugenol have potent antibacterial activity. As a result, in combination, the antibacterial activity of both the chemicals might be summed up together to exert potent antibacterial action.
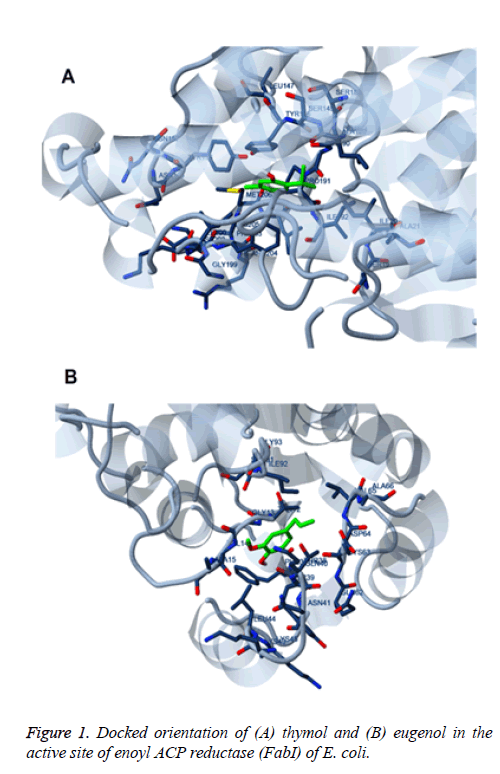
The present study enumerate that, TE mixture showed excellent bactericidal activity against E. coli. Thus, to determine the mechanism of action of these compounds as a disinfectant, we have performed docking with enoyl ACP reductase (FabI) of E. coli. The 3-D protein crystal structure was obtained from Protein Data Bank (http:// www.rcsb.pdb.org; PDB entry: 1C14). This enzyme was responsible for reduction of enoyl-acyl-carrier-protein (ACP) substrates, a critical step necessary for fatty acid elongation of E. coli. Thus, inhibition of Fabl serves as important step in controlling the pathogenesis of E. coli. The thymol and eugenol were docked individually to determine their binding affinity and their docked orientations has been depicted in Figure 1. The Figure 1 elucidates that, both thymol and eugenol bind to the active site in an efficient manner via formation of efficient hydrogen bonds and pi-pi stacking interactions. Particularly, thymol was identified to create more interactions, for example, it has been shown to create hydrophobic interaction with Ile20, Thr146, Ala189 and pi-pi and cation-pi with Tyr146 and Tyr156. Moreover, the eugenol interacts with Val65 and Ile92 via hydrophobic interaction. It also showed to interact with Thr38, Gln40 and Leu44. In terms of scoring parameters, as shown in Table 2, both thymol and eugenol found as efficient ligand to engage the active site of protein with estimated free energy of binding of -5.19 kcal/mol and -4.73 kcal/mol, respectively. The estimated inhibition constant of thymol and eugenol was also found in agreement with the excellent bioactivity, 156.07 uM and 343.24 uM, respectively.
| Compound | Hydrophobic Interaction | Hydrogen Bond | Pi-Pi | Cation-Pi | Other |
|---|---|---|---|---|---|
| Thymol | Ile20, Tyr146, Ala189, Pro191, Ile200, Met206 |
Tyr156 | Tyr146, Tyr156, Phe203 |
Tyr146, Tyr156 |
Tyr146, Ty156 |
| Eugenol | Val65, Ile92 | Nil | Nil | Nil | Thr38, Gln40, Leu44, Asp64 |
Table 2: Interaction of Thymol-eugenol with enoyl ACP reductase (FabI) of E. Coli
| Compound | Est. Free Energy of Binding | Est. Inhibition Constant, Ki | vdW + Hbond + desolv Energy | Electrostatic Energy | Total Intermolec. Energy |
|---|---|---|---|---|---|
| Thymol | -5.19 kcal/mol | 156.07 uM | -5.81 kcal/mol | -0.09 kcal/mol | -5.89 kcal/mol |
| Eugenol | -4.73 kcal/mol | 343.24 uM | -5.86 kcal/mol | -0.07 kcal/mol | -5.93 kcal/mol |
Table 3. Scoring of Thymol-Eugenol with enoyl ACP reductase (FabI) of E. Coli
Conclusion
The present study corroborated that, thymol-eugenol mixture efficiently inhibit the growth of bacterial organism which easily survive on the medical instruments. The effectiveness of the disinfectant activity was easily achieved in optimum contact period. Thus, thymol-eugenol could be used as a potential disinfecting agent. However, more studies are needed to explicate the detailed mechanism of action.
References
- Chan GJ, Lee AC, Baqui AH, Tan J, Black RE. Prevalence of early-onset neonatal infection among newborns of mothers with bacterial infection or colonization: a systematic review and meta-analysis. BMC Infect Dis 2015; 15: 118.
- Brown KA, Daneman N, Stevens VW, Zhang Y, Greene TH, Samore MH, Arora P. Integrating Time-Varying and Ecological Exposures into Multivariate Analyses of Hospital-Acquired Infection Risk Factors: A Review and Demonstration. Infect Control Hosp Epidemiol 2016; 16: 1-9.
- Phu VD, Wertheim HF, Larsson M, Nadjm B, Dinh QD, Nilsson LE, Rydell U, Le TT, Trinh SH, Pham HM, Tran CT, Doan HT, Tran NT, Le ND, Huynh NV, Tran TP, Tran BD, Nguyen ST, Pham TT, Dang TQ, Nguyen CV, Lam YM, Thwaites G, Van Nguyen K, Hanberger H. Burden of Hospital Acquired Infections and Antimicrobial Use in Vietnamese Adult Intensive Care Units. PLoS One 2016; 11: e0147544.
- Lalwani S, Punia P, Mathur P, Trikha V, Satyarthee G, Misra MC. Hospital acquired infections: preventable cause of mortality in spinal cord injury patients. J Lab Physicians 2014; 6: 36-39.
- Reichman DE, Greenberg JA. Reducing Surgical Site Infections: A Review. Rev Obstetrics Gynecol 2009; 2: 212-221.
- Anderson DJ, Podgorny K, Berríos-Torres SI. Strategies to Prevent Surgical Site Infections in Acute Care Hospitals: 2014 Update. Infect Control Hosp Epidemiol 2014; 35: 605-627.
- Sydnor ERM, Perl TM. Hospital Epidemiology and Infection Control in Acute-Care Settings. Clinical Microbiol Rev 2011; 24: 141-173.
- Rutala WA, Weber DJ. Disinfection and Sterilization in Health Care Facilities: What Clinicians Need to Know. Clin Infect Dis 2004; 39: 702-709.
- Maillard JY. Antimicrobial biocides in the healthcare environment: efficacy, usage, policies, and perceived problems. Ther Clin Risk Manag 2005; 1: 307-320.
- Cowan MM. Plant Products as Antimicrobial Agents. Clin Microbiol Rev 1999; 12: 564-582.
- Cragg GM, Newman DJ. Natural Products: A Continuing Source Of Novel Drug Leads. Biochimica et biophysica acta 2013; 1830: 3670-3695.
- Veras HNH, Rodrigues FFG, Botelho MA, Menezes IRA, Coutinho HDM, da Costa JGM. Antimicrobial Effect of Lippia sidoides and Thymol onEnterococcus faecalis Biofilm of the Bacterium Isolated from Root Canals. Sci World J 2014; 2014: 471580.
- Wechsler JB, Hsu C-L, Bryce PJ. IgE-mediated mast cell responses are inhibited by thymol-mediated, activation-induced cell death in skin inflammation. J Allergy Clin Immunol 2014; 133: 1735-1743.
- Ali SM, Khan AA, Ahmed I. Antimicrobial activities of Eugenol and Cinnamaldehyde against the human gastric pathogen Helicobacter pylori. Ann Clin Microbiol Antimicrob 2005; 4: 20.
- Yadav MK, Chae S-W, Im GJ, Chung J-W, Song JJ. Eugenol: A Phyto-Compound Effective against Methicillin-Resistant and Methicillin-SensitiveStaphylococcus aureus Clinical Strain Biofilms. Beloin C PLoS ONE 2015; 10: e0119564.
- Bikadi Z, Hazai E. Application of the PM6 semi-empirical method to modeling proteins enhances docking accuracy of AutoDock. J Cheminf 2009; 1: 15.
- Halgren, TA. Merck molecular force field. I. Basis, form, scope, parametrization, and performance of MMFF94. J Comput Chem 1998; 17: 490-519.
- Morris GM, Goodsell DS. Automated docking using a Lamarckian genetic algorithm and an empirical binding free energy function. J Comput Chem 1998; 19: 1639-1662.
- Solis FJ, Wets RJB. Minimization by Random Search Techniques. Math Operation Res 1981; 6: 19-30.