Research Article - Biomedical Research (2017) Volume 28, Issue 12
EHRCT is superior to the pulmonary function test at showing small airway disease in patients with isolated mitral valve stenosis
Ayşegül Karadeniz1 and Mevlüt Karataş2*
1Department of Radiology, Trabzon Ahi Evren Cardiovascular and Thoracic Surgery Research and Application Center, Saglik Bilimleri University, Trabzon, Turkey
2Atatürk Chest Diseases and Thoracic Surgery Training and Research Hospital, Ankara, Turkey
- *Corresponding Author:
- Mevlüt Karataş
Atatürk Chest Diseases and Thoracic Surgery Training and Research Hospital
Ankara, Turkey
Accepted date: April 27, 2017
Abstract
Introduction: We investigated whether expiratory high resolution computed tomography (EHRCT) is superior to the pulmonary function test (PFT) in showing small airway disease (SAD) in patients with isolated mitral valve stenosis (MVS).
Methods: Sixty patients contributed to the study prospectively. Cases were divided into three groups (mild, moderate, severe) on the basis of MVS and PAP.
Results: A highly significant relation was determined between severity of MVS and both FEV1 (p<0.002) and FVC (p<0.007). Mean FEV1 in patients with severe MVS were significantly lower than those of cases of mild and moderate MVS (p=0.015, p=0.004). Highly significant variation was also determined between FEF25-75% based on severity of MVS (p<0.003). Mean FEF25-75% of cases of severe MVS were significantly lower than those of the mild and moderate cases (p=0.008, p=0.009). Significant variation was determined between mean FEV1 depending on PAP classifications (p<0.038). Significant variation was also observed among mean FEF25-75% in terms of PAP (p<0.005). FEF25-75% of cases with air trapping in cases of moderate and severe MVS were significantly lower than those of cases without air trapping (p=0.043, p=0.016). Mean FEF25-75% of all cases with air trapping were highly significantly lower than those of cases without air trapping (p<0.001). A significant difference was determined when presence of air trapping was assessed according to EHRCT and FEF25-75% in cases with mild and moderate MVS (p=0.021, p=0.016).
Conclusions: According to our knowledge, this is the first study to show the superiority of EHRCT over PFT (FEF25-75%) in showing SAD in patients with isolated MVS (mild and moderate stages but not in severe MVS) without additional respiratory disease.
Keywords
EHRCT, Mitral valve stenosis, Small airway disease, Pulmonary function, FEV1, FEF25-75%.
Introduction
Small airways are structures with internal diameters of less than 2 mm containing no cartilaginous tissue. In healthy individuals, 80% of total airway resistance derives from the trachea and the large airways, while less than 20% derives from small airways. Small airway diseases (SAD) are defined as pulmonary dysfunction together with inflammatory and/or obliterate changes in small airways. Since the small airways represent a very small part of total resistance, there is no significant decrease in flow speeds in small airway obstruction, and it is therefore difficult to determine obstruction with routine spirometric tests. Forced expiratory flow at 25-75% of FVC (FEF25-75%) value is quite sensitive in determining SAD. FEF25-75% being below the expected value (FEF25-75% <80%) while FEV1 and FVC values are normal shows obstruction in the small airways [1].
Hypertrophic changes in the pulmonary arterioles associated with high pulmonary vascular pressure occur in mitral valve stenosis (MVS), particularly small airways disease (SAD), are seen in this condition [2,3]. As a result, the pulmonary parenchyma loses elasticity and becomes less compliant. Air trapping is added to the picture as this process advances [4,5]. Air trapping refers to retention of excess gas “air” in all or part of the lung, especially during expiration, either as a result of complete or partial airway obstruction or as a result of local abnormalities in pulmonary compliance. In pulmonary vascular diseases associated with local hypoxic reflex vasoconstriction in the small airways, a decreased dimension and number of vessels is observed in areas of pulmonary lucency in association with underlying hypoperfusion. These areas also emerge as air trapping in the small airways on expiratory high resolution computed tomography (EHRCT) scans. Differentiation is assisted if these areas do not remain hyperlucent at the end of expiration [6]. EHRCT is a special imaging method that facilitates the diagnosis of SAD showing the areas of air trapping in small airways. The presence of air trapping in EHRCT is the one of the indirect radiological finding of SAD. In additional to this, EHRCT plays a complementary role to PFTs in the small airways by showing SAD and air trapping in cases with normal PFT values [7].
Materials and Methods
The study was planned prospectively. EHRCT was compared with PFT in patients with isolated MVS. Patients’ demographic characteristics, PFT values, ECHO reports, symptoms and findings were collected from medical records and recorded.
Study population
The study commenced following receipt of local ethical committee approval. Sixty patients were included in the study. All patients had shortness of breath with isolated MVS determined by the same cardiologist at echocardiography (ECHO). PFT and inspiratory and EHRCT were performed for differential diagnosis of shortness of breath. Patients with leftsided heart failure, coronary artery disease, moderate or severe mitral insufficiency, myocardial disease, pulmonary or aortic valve disease, chronic obstructive pulmonary disease, allergy/ atopy or a history of smoking were excluded.
Patients were divided into three groups using ECHO, based on mitral valve orifice area (MVOA) and pulmonary artery pressure (PAP). MVS was classified as mild (1.5-2 cm2), moderate (1-1.5 cm2) or severe (<1 cm2) depending on MVOA. Based on mean PAP values, patients were classified as Group-1 (<40 mmHg), Group-2 (40-50 mmHg) and Group-3 (>50 mmHg).
Pulmonary function test
This was performed with the PFT (with a Sipro Analyzer ST-300-Fukuda-Sangyo device). The PFT was performed three times for each patient, and the highest value was included in the study. The observed or measured values were expressed as a percentage of predictive values which were computed according to age, sex, weight, and height and were considered normal if they were >80% of predictive values. FVC, FEV1 and FEF25-75% were measured at PFT.
FEF25-75% (L/sn)
This represents mid-expiratory flow, that between the first and last quarters of forced expiration (in other words, the flow of the 50% volume once the first 25% of air has been expelled). It constitutes the effort independent part of forced expiration. It shows obstruction in the airways in the early period. FEF25-75% being below the expected value (FEF25-75% <80%) while FEV1 and FVC values are normal shows obstruction in the small airways [1].
HRCT technique
HRCT was performed using a Siemens Somatom Sensation 16 (Siemens, Germany) device. Patients were placed in the supine position and asked to hold their breath following deep inspirium and scanned without contrast from the apexes of the lung to the lowest point of the hemidiaphragm. Following deep inspirium, patients performed forced expirium and were scanned again. Images for 16-detector CT were obtained with parameters of 120 kV, 125 active mA, 16 × 1.5 mm collimation, 3 mm section thickness and a 512 × 512 matrix. Axial images were obtained on standard mediastinal (50 HU, 400 HU) and parenchymal (-600 HU, 1500 HU) window settings. High geometric resolution images (bone algorithm) re-construction was performed with parameters of 120 kV, 170 active mA, 10 mm intervals and a 0.75 mm section thickness and a 512 × 512 matrix for HRCT. Images were examined at the parenchymal window setting (-600 HU, 2000 HU). Images were evaluated by a radiologist. Lucent areas identified at inspiratory and EHRCT were interpreted and evaluated as radiological appearance associated with SAD.
Statistical analysis
The findings obtained were analyzed using NCSS (Number Cruncher Statistical System) Statistical Software (Utah, USA). In addition to descriptive statistical methods (mean and standard deviation), one-way ANOVA was used to compare normally distributed parameters between groups at comparison of quantitative data and the Turkey HDS test to identify the group-responsible for variation. Student’s t test was used to compare normally distributed parameters between two groups and the Mann Whitney U test to compare non-normally distributed parameters between two groups. The chi square test and Fisher’s exact chi square test were used in the comparison of qualitative data. Significance was set at p<0.05.
Results
Sixty patients aged between 26 and 72, 50 (86.7%) female and 8 (13.3%) male, were included in the study. Mean age of the subjects was 49.96 ± 11.16 years. No statistically significant difference was observed between degree of severity of MVS and cases’ mean age or gender distribution (p=0.484, p=0.866).A statistically highly significant difference was determined between PAP levels and severity of MVS (p<0.001).
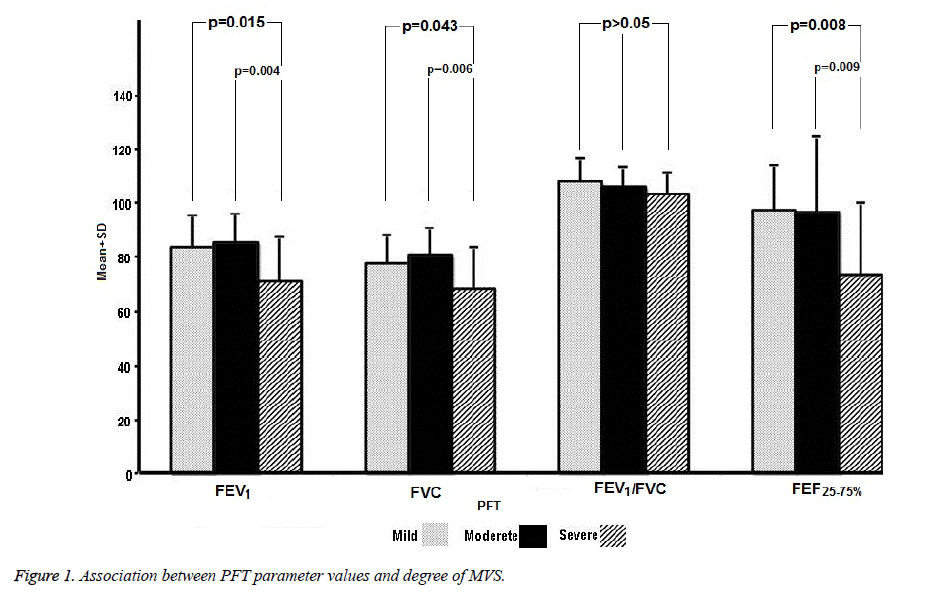
A significant relation was determined between severity of MVS and cases’ mean FEV1 values (p=0.002). Mean FEV1 values of patients with severe MVS were significantly lower than those of patients with mild or moderate MVS (p=0.015, p=0.004). No significant difference was observed between the mean FEV1 values of patients with mild or moderate MVS (p=0.872) (Figure 1).
There was also a statistically highly significant relation between mean FVC values and severity of MVS (p=0.007). Mean FVC values of cases with severe MVS were significantly lower than those of patients with mild or moderate MVS (p=0.043, p=0.006). No significant difference was observed in terms of mean FVC values between mild and moderate cases of MVS (p=0.656). No significant difference was determined in terms of FEV1/FVC% values depending on severity of MVS (p>0.05) (Figure 1).
A statistically significant difference was observed between mean FEF25-75% values and severity of MVS (p ≤ 0.003). Mean FEF25-75% values of cases with severe MVS were significantly lower than in cases of mild or moderate MVS (p=0.008, p=0.009). No significant difference was determined in terms of mean FEF25-75% values between mild and moderate cases of MVS (p=1.000) (Figure 1).
Statistically significant difference was determined in mean FEV1 values depending on cases’ PAP classifications (p=0.001). Mean FEV1 values of group-3 cases based on PAP values were significantly lower than those of group-1 cases (p=0.032). No significant difference was observed between group-2 cases’ FEV1 levels and those of groups-1 and groups-3 (p=0.299, p=0.494). No statistically significant variation was observed in mean FVC and FEV1/FVC% values between the groups (p>0.05).
A highly significant variation was determined between mean FEF25-75% values in the PAP groups (p=0.005). Group-3 cases’ mean FEF 25-75% values were significantly lower than those of group-1 cases (p=0.004). No statistically significant difference was observed between group-2 cases’ FEF25-75% levels and those of groups-1 and groups-3 (p=0.482, p=0.081).
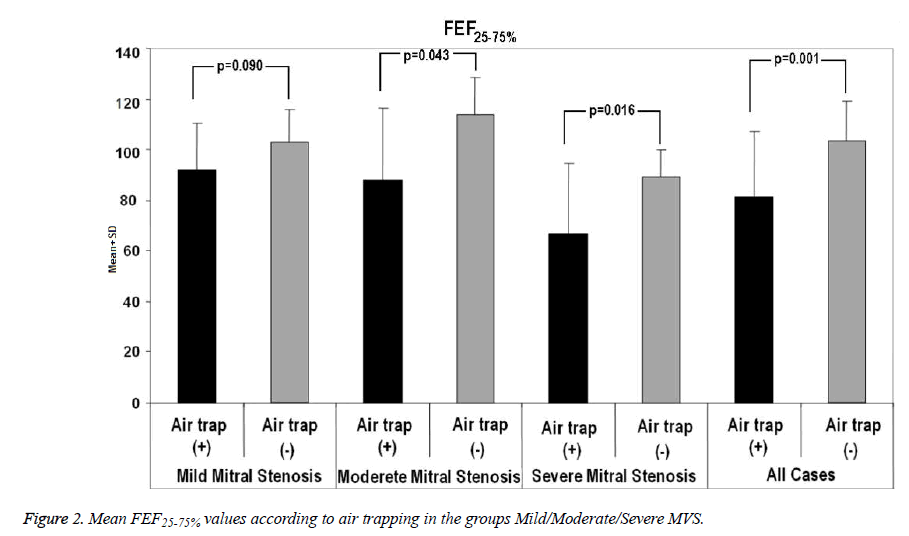
There was no statistically significant variation between presence of air trapping and mean FEF25-75% values in cases of mild MVS (p=0.090). However, in cases of moderate and severe MVS, FEF25-75% values of cases with air trapping were significantly lower than those of patients with no air trapping (p=0.043, p=0.016). In all cases, FEF25-75% values of cases with air trapping were highly significantly lower than those of cases with no air trapping (p=0.001) (Figure 2).
Examination of presence of air trapping at HRCT and based on FEF25-75% values revealed a difference between the two methods. In cases of mild MVS, air trapping was seen in 50% of cases at EHRCT, and in 10% of cases on the basis of FEF25-75% (p<0.021). In cases of moderate MVS, air trapping was seen in 65% of cases at EHRCT but in 30% of cases based on FEF25-75%. There was a significant difference between the two methods (p<0.016). In cases of severe MVS, air trapping was observed in 75% of cases at EHRCT and in 70% based on FEF25-75%. There was no significant difference between the two methods (p=1.000) (Table 1).
| Air Trap | |||||
|---|---|---|---|---|---|
| FEF25-75% | Air trap (+) n% | Air trap (-) n% | Total n% |
P value | |
| Mild Stenosis | |||||
| <80 | 1 (5%) | 1 (5%) | 2 (10%) | ||
| >80 | 9 (45%) | 9 (45%) | 18 (90%) | 0.021 | |
| Total | 10 (50%) | 10 (50%) | 20 (100%) | ||
| Moderate Stenosis | |||||
| <80 | 6 (30%) | 0 (0%) | 6 (30%) | ||
| >80 | 7 (35%) | 7 (35%) | 14 (70%) | 0.016 | |
| Total | 13 (65%) | 7 (35%) | 20 (100%) | ||
| Severe Stenosis | |||||
| <80 | 13 (65%) | 1 (5%) | 14 (70%) | ||
| >80 | 2 (10%) | 4 (10%) | 6 (30%) | 1.000 | |
| Total | 15 (75%) | 5 (25%) | 20 (100%) | ||
Table 1. Mean FEF25-75% values according to air trapping in the groups Mild/Moderate/Severe MVS.
Discussion
The most independent PFT value showing SAD is FEF25-75%. This value being below 80% is significant in terms of SAD. We observed a significant decrease in FEF25-75% in all patients with MVS such as to indicate SAD. The FEV1/FVC% values of groups were within normal limits and we determined no statistically significant variation in mean FEV1/FVC% values (Table 2). Expiratory HRCT is more sensitive than the PFT in determining the air trapping seen in SAD in most cases [7,8]. Patients with clinical respiratory symptoms with isolated MVS should be investigated by EHRCT, even though FEF25-75% values are normal. In present study, we observed the presence of air trapping at EHRCT and based on FEF25-75% values revealed a significant difference between the two methods in the cases of mild and moderate MVS (p=0.021, p=0.016). However, in cases of severe MVS, there was no significant difference between the two methods (p=1.000) (Table 1). The results revealed that EHRCT is superior to the PFT at showing SAD in patients with mild and moderate isolated MVS.
| Mitral stenosis | ||||
|---|---|---|---|---|
| Mild (n=20) Mean ± SD, n (%) | Moderate (n=20) Mean ± SD, n (%) | Severe (n=20) Mean ± SD, n (%) | P value | |
| Age | 51.45 ± 9.77 | 50.95 ± 13.11 | 47.50 ± 10.48 | 0.484 |
| Female | 17 (85.0%) | 17 (85.0%) | 18 (90.0%) | 0.866 |
| Male | 3 (15.0%) | 3 (15.0%) | 2 (10.0%) | |
| FEV1 | 83.35 ± 12.28 | 85.45 ± 10.64 | 71.14 ± 16.45 | 0.002 |
| FVC | 77.52 ± 10.92 | 80.92 ± 10.09 | 68.59 ± 15.09 | 0.007 |
| FEV1/FVC % | 107.83 ± 9.27 | 105.88 ± 7.45 | 103.52 ± 7.65 | 0.205 |
| FEF 25-75% | 97.36 ± 16.84 | 97.15 ± 27.49 | 73.85 ± 26.28 | 0.003 |
| PAP, mmHg | ||||
| <40 | 13 (65.0%) | 9 (45.0%) | 2 (10.0%) | 0.001 |
| 40-50 | 7 (35.0%) | 8 (45.0%) | 5 (25.0%) | |
| ≥ 50 | 0 (0%) | 3 (15.0%) | 13 (65.0%) | |
| PAP: Pulmonary arterial Pressure | ||||
Table 2. Mean values for SFT parameters and PAP according to degrees of MSV and demographic characteristics of patients.
When we compared the presence of air trapping identified at EHRCT with FEF25-75% values in cases of moderate and severe MVS, FEF25-75% values were significantly lower in subjects with air trapping than in those without air trapping (p=0.043, p=0.016). Similarly, FEF25-75% values among all cases were significantly lower in cases with air trapping than in those without air trapping (p=0.001). However, we observed no such significant difference in the mild MVS group (Figure 2).
We also determined a statistically significant difference between FEV1 and FEF25-75% values depending on PAP groups. FEV1 and FEF25-75% values differed significantly between patients in group-1(<40 mmHg ) and group-3 (>50 mmHg ), but no significant difference was observed in terms of mean FEV1 and FEF25-75% values between group-2 patients and the other groups (groups-1 and 3). This finding shows that PFT may be useful in showing SAD in subjects with PAP values <40 mmHg and >50 mmHg but that it is not useful between group-2 and the other groups. In addition, there was no statistically significant difference between mean FVC and FEV1/FVC% values in terms of PAP groups (p>0.05).
Chatterji et al. reported findings showing that FVC values decreased in direct proportion to PAP, and that PFT values were correlated with degree of severity of MVS and PAP, and also showed the presence of SAO [9]. Dogliotti et al. also observed similar spirometric findings [10]. Similar findings also emerged from our study. The lowest FEV1 and FEF25-75% values were observed in patients with severe MVS and PAP values >50 mmHg. FEV1 and FVC values decreased significantly as severity of MVS and PAP values increased, in such a way as to show restrictive type disorder (Table 3).
| PAP | ||||
|---|---|---|---|---|
| Mean ± SD | Mean ± SD | Mean ± SD | P value | |
| PAP, mmHg | <40 (n=24) | 40-50 (n=20) | ≥ 50 (n=15) | |
| FEV1 | 85.23 ± 13.16 | 78.86 ± 12.72 | 73.50 ± 16.61 | 0.038 |
| FVC | 79.80 ± 11.09 | 74.27 ± 12.38 | 71.25 ± 15.58 | 0.108 |
| FEV1/FVC% | 106.91 ± 9.44 | 106.48 ± 6.78 | 103.06 ± 4.95 | 0.260 |
| FEF25-75% | 99.31 ± 24.78 | 90.80 ± 27.97 | 72.98 ± 17.25 | 0.005 |
Table 3. Mean PFT parameter values according to PAP classification.
Several studies have shown a correlation between degree of severity of mitral valve disease and PFT values [9,11-13]. Some of these have reported a decrease in PFT values associated with mitral valve disease [11,12,14]. Some studies in the literature have reported a correlation between severity of MVS and a decrease in PFT values [9,15-19]. Other studies have reported no such correlation [11,12,20-22]. In a study of a group with mitral valve disease (MVD), all smokers and with additional respiratory disease, Carmo et al. observed decreased PFT values and a restrictive type respiratory pattern. However, they reported no correlation between the decrease in PFT values and patients’ respiratory symptoms and ECHO parameters [12]. Rhodes et al. reported a direct correlation between a decrease in PFT values and severity of MVD [17]. In addition to that association, Nour et al. also reported a correlation between MVS and respiratory symptoms [14]. Doo et al. showed a correlation between degree of severity of MVS and impaired PFT values [23]. Ohno et al. showed a significant improvement in PFT values with valve surgery in patients without pulmonary arterial hypertension (PAH) in addition to MVS and with low preoperative PFT values [24]. In a study of 30 patients with isolated MVS and pulmonary hypertension, Gölbaşı et al. reported significant improvement in PFT values after mitral valve replacement surgery and suggested that the changes which cause pulmonary dysfunction in cases with MVS, are mostly reversible (21). Our study differs from previous studies in that our patients had isolated MVS, none were smokers and none had additional respiratory diseases [13,15,21]. The presence of other respiratory diseases in additional to MVD in other studies may have affected PFT values in a restrictive or obstructive direction, but there was no possibility of this in our study.
In conclusion, to the best of our knowledge, this is the first study to show the superiority of EHRCT over PFT value (FEF25-75%) in showing SAD in patients with isolated MVS without additional respiratory disease. EHRCT is more useful than FEF25-75% in showing air trapping and SAD in patients with mild and moderate stages of isolated MVS but not in severe MVS. A decrease in FEF25-75% values, statistically significant and exhibiting reverse correlation with degree of severity of MVS and PAP elevation, occurs in patients with isolated MVS without additional respiratory disease. We think that these results will contribute to early diagnosis and treatment of diseases of the SAD with isolated MVS. We also think that early bronchodilator therapy can be clinically useful in cases with symptoms among patient with MVS.
There is some limitation of our study. The study was conducted in single centre and the number of patients participating in the study is relatively low. If the number of patients with mitral valve stenosis involved in the study was higher, the literature contribution of the results obtained at the end of this study might be higher. We nevertheless believe that this study may also open the door for similar studies to be made in the future.
References
- Grippi MA, Tino G. Pulmonary function testing. In: Fishman’s Pulmonary Diseases and Disorders (4thedn) Fishman AP, Elias JA, Fishman JA (eds.) The McGraw-Hill 2008; pp. 567-609.
- Honig EG. Tecniques of evaluating pulmonary function in patients with cardiac disease in Hurst’s the heart. In: Schlant RC, Alexander RW (8thedn) Mc Graw-Hill, New York, 1994; pp. 441-445.
- McFadden E Jr, Ingram RJ Jr. Relationship between disease of the heart and lung In Ed. Braunwald E. Heart disease, a textbook of cardiovascular medicine (2ndedn) WB Saunders, Philadelphia, 1988; 3: 1870.
- Depeursinge FB, Depeursinge CD, Boutaleb AK, Feihl F, Perret CH. Respiratory system impedance in patients with acute left ventricular failure: pathophysiology and clinical interest. Circulation 1986; 73: 386-395.
- Collins JV, Clark TJ, Brown DJ. Airway function in healthy subjects and patients with left heart disease. Clin Sci Mol Med 1975; 49: 217-228.
- Stern EJ, Frank MS. Small-airway diseases of the lungs: findings at expiratory CT. AJR Am J Roentgenol 1994; 163: 37-41.
- Lucidarme O, Coche E, Cluzel P, Mourey-Gerosa I, Howarth N. Expiratory CT scans for chronic airway disease: correlation with pulmonary function test results. AJR Am J Roentgenol 1998; 170: 301-307.
- Webb WR, Stern EJ, Kanth N, Gamsu G. Dynamic pulmonary CT: findings in healthy adult men. Radiology 1993; 186: 117-124.
- Chatterji RS, Panda BN, Tewari SC, Rao KS. Lung function in mitral stenosis. J Assoc Physicians India 2000; 48: 976-980.
- Dogliotti GC, Angelino PF, Brusca A, Garbagni R, gavosto F. Pulmonary function in mitral valve disease; hemodynamic and ventilatory studies. Am J Cardiol 1959; 3: 28-39.
- Palmer WH, Gee JB, Bates DV. Disturbances of pulmonary function in mitral valve disease. Can Med Assoc J 1963; 89: 744-750.
- Carmo MM, Ferreira T, Lousada N, Bárbara C, Neves PR, Correia JM, Rendas AB. The repercussions of pulmonary congestion on ventilatory volumes, capacities and flows. Rev Port Cardiol 1994; 13: 763-768.
- Bitner M, Nowak D. The lung function in operated acquired mitral and aortic valve diseases without left ventricular failure-preliminary observations before operation. Arch Med Sci. 2005; 1: 254-257.
- Nour MM, Shuhaiber H, Yousof AM. Lung function and severity of mitral stenosis. Med Principles Pract 1999; 8: 32-39.
- Vaidya R, Husain T, Ghosh PK. Spirometric changes after open mitral surgery. J Cardiovasc Surg (Torino) 1996; 37: 295-300.
- Seboldt H, Stunkat R, Keppeler F, Hoffmeister HE, Hilpert T. Reversibility or irreversibility of pulmonary function changes after cardiac surgery on insufficient or stenotic cardiac valves (author's transl)) Thoraxchir Vask Chir 1975; 23: 431-436.
- Rhodes KM, Evemy K, Nariman S, Gibson GJ. Relation between severity of mitral valve disease and results of routine lung function tests in non-smokers. Thorax 1982; 37: 751-755.
- Wood TE, McLeod P, Anthonisen NR, Macklem PT. Mechanics of breathing in mitral stenosis. Am Rev Respir Dis 1971; 104: 52-60.
- Wilhelmsen L. Lung mechanics in rheumatic valvular disease. Acta Med Scand 1968; 489: 3-114.
- Strzyzakowska M, Hryniewiecki T, Gutkowski P, Rawczynska-Englert I. Pulmonary function in patients with rheumatic mitral valve disease. Preoperative evaluation. Pol Arch Med Wewn 1995; 93: 63-68.
- Saxena P, Luthra S, Dhaliwal RS, Rana SS, Behera D. Early changes in pulmonary functions after mitral valve replacement. Ann Thorac Med 2007; 2: 111-117.
- Parvathy UT, Rajan R, Faybushevich AG. Pulmonary function derangements in isolated or predominant mitral stenosis - Preoperative evaluation with clinico-hemodynamic correlation. Interv Med Appl Sci 2014; 6: 75-84.
- Doo YC, Koh YS, Kim WS, Kim JJ, Park SW, Park SJ. Pulmonary function tests results in relation to degree of mitral stenosis in non-smoking mitral stenosis patients. The Korean Journal of Int Med 1991; 41: 67-72.
- Ohno K, Nakahara K, Hirose H, Nahano S, Kawashima Y. Effects of valvular surgery on overall and regional lung function in patients with mitral stenosis. Chest 1987; 92: 224-228.