Research Article - Biomedical Research (2017) Volume 28, Issue 6
Efficacy of 4-2-1 principle-guided laser treatment in diabetic retinopathy
Ding Xu1, Chun-Xia Li2, Chun Zhao1, Wan-Cheng Jia3, Yi Zhang4, Xiao-Qiang Liu1, Qing-Yu Liu1 and Fang Wang1*1Department of Ophthalmology, Shanghai Tenth People’s Hospital, Tongji University School of Medicine, Shanghai, PR China
2Department of Ophthalmology, Shanghai TCM-integrated Hospital, Shanghai, PR China
3Department of Ophthalmology, Shanghai Fengxian District Central Hospital, Shanghai, PR China
4Department of Ophthalmology, Shanghai Pudong Gongli Hospital, Shanghai, PR China
- *Corresponding Author:
- Fang Wang
Department of Ophthalmology, Shanghai Tenth People’s Hospital
Tongji University School of Medicine, PR China
Accepted date: November 4, 2016
Abstract
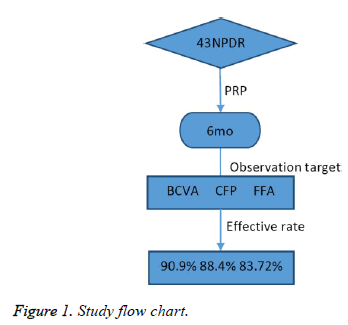
This study aimed to observe the efficacy of 4-2-1 principle-guided Pan-Retinal Photocoagulation (PRP) for treating severe Non-Proliferative Diabetic Retinopathy (NPDR). This multi-center, prospective, and non-comparative clinical study involved performance of PRP in 43 patients (43 eyes) with severe NPDR, who were diagnosed according to 4-2-1 principles, and were followed up for 6 months. Best corrected visual acuity (BCVA) tests, Colored Fundus Photography (CFP), and Fluorescence Fundus Angiography (FFA) were performed before and after PRP. Assessments were again performed in the 6-month followup to evaluate efficacy. The mean BCVAs before and after PRP were 59.91 ± 14.58 and 62.86 ± 15.41 letters, respectively (t=1.782, P>0.05). Thirty-nine eyes exhibited effective BCVA results (90.7%), 38 eyes exhibited effective CFP results (88.4%), and 36 eyes exhibited effective FFA results (83.72%); no statistically significant difference was found (χ2=0.19, P>0.05). Thus, 4-2-1 principle-guided PRP treatment was an effective approach in the absence of FFA. Using this approach, disease progression could be effectively controlled in the vast majority of patients with severe NPDR, preventing severe visual impairment.
Keywords
Diabetic retinopathy, Photo coagulation, 4-2-1 principle.
Introduction
Diabetic Retinopathy (DR) is a serious complication of diabetes, and is a major cause of blindness in people aged 20-70 years [1]. There are already 9.84 million adults with diabetes in China, which is the highest number in the world [2]. The latest survey of DR in Shanghai showed an incidence of 27.29% [3]; it is therefore the second most common disease that could result in blindness in the elderly in Shanghai [4]. Timely retinal photocoagulation could prevent severe visual impairment in DR patients, and it has been recognized as the “gold standard” treatment for retinal ischemia and neovascularization. However, in China, the number of individuals receiving regular fundus examination and laser treatment is far lower than that expected, which is the main reason why DR is the sixth-ranked blinding disease in China [3]. Typically, Fluorescence Fundus Angiography (FFA) would be performed during screening patients for DR laser treatment. However, due to a lack of FFA equipment in some hospitals, some patients would undergo retinal laser treatment directly after fundus examination, and some DR patients are restricted to FFA due to their general condition. According to the Fundus Manifestations and Treatment Recommendations of the International DR Staging Criteria issued in 2003 [5], non-FFAdependent photocoagulation could also be performed. It has been reported that fundus examination and FFA exhibited good consistency in selecting patients for retinal laser treatment [6,7], but few multi-center systematic studies have been performed in China to date. This study was jointly conducted by the Department of Ophthalmology in one third-level grade- A hospital and two second-level hospitals in Shanghai; patients with severe Non-Proliferative DR (NPDR) were selected in accordance with the 4-2-1 principle for PRP, and the efficacies of photocoagulation were then observed and compared.
Study Subjects and Methods
General information
Forty-three NPDR patients (43 eyes) were recruited from the Ophthalmological Departments of Shanghai Tenth People’s Hospital, Tongji University School of Medicine, Shanghai TCM-Integrated Hospital, Shanghai Fengxian District Central Hospital, and Shanghai Pudong Gongli Hospital, from April 2010 to April 2012. The patients’ details are shown in Table 1. This study was conducted in accordance with the Declaration of Helsinki. This study was conducted with approval from the Ethics Committee of Tongji University. Written informed consent was obtained from all participants.
| Eye (n) | Gender (n) | Mean age (years) | Blood pressure (mmHg) | Mean fasting blood sugar (mmol/L) | Mean HbA1c (%) | Disease duration (years) | BCVA (EDTRS) | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Right | Left | M | F | SBP | DBP | |||||
| 25 | 18 | 19 | 24 | 60.97 ± 7.69 | 134.53 ± 15.55 | 82.37 ± 11.09 | 8.70 ± 4.13 | 7.75 ± 1.75 | 10.93 ± 4.56 | 59.91 ± 14.58 |
Table 1. General info of the 43 patients with severe NPDR (43 eyes).
Inclusion criteria
Patients were included if they were diagnosed with severe NPDR (type 2 diabetes) by Color Fundus Photography (CFP), according to the staging criteria approved by the International Council of Ophthalmology (ICO) in 2003 [5]. All the patients were enrolled into a single registry, and all were investigated for the same factors. The main inspection items included disease duration, blood pressure, fasting blood glucose and glycosylated haemoglobin levels. Best Corrected Visual Acuity (BCVA) tests, Intraocular Pressure (IOP) measurements, fundus examination, and CFP were performed at baseline and at each follow-up; further, FFA was performed at the beginning and during the 6-month follow-up. The test results were only used for determining the efficacies on completion of the treatment.
Exclusion criteria
Patients who had severe cataract or opacity of the refracting media, which could affect inspection, were excluded. Combined with macular oedema, other non-DR retinal diseases or a poor general condition (active or uncontrolled heart or liver diseases, non-diabetic metabolic diseases, or neurological or blood diseases that had important clinical significance), were excluded.
Methods
This was a multicenter, prospective, and non-comparative clinical study. The follow-up lasted 6 months.
Treatment protocols
All patients underwent Pan-Retinal Photocoagulation (PRP) after using one argon ion laser instrument for full mydriasis (Lumenis, CA, USA). The treatment was divided into four sessions, with a 1 week interval between treatments. Standard PRP was performed. The photocoagulation range included 1 Papilla Disc (PD) superior and inferior to the Discus Opticus (DO) and nasal side, and 2 PD superior and inferior to the central fovea of the macula and temporal side. The posterior pole was retained among the papillomacular bundle of the DO and the superior and inferior temporal vascular arches, and peripheral spreading to the equator section or slightly over the equator of the eye was allowed. The spot size was 200-500 μm; exposure time was 0.15-0.2 s, spot intensity grade III photocoagulation, power of 200-500 mW, and light spots ranging 800-1000 points. The follow-up was performed monthly after PRP; this mainly included visual acuity tests (using the ETDRS eye chart) and IOP measurements, and the key items were the recorded. CFP and FFA (Heidelberg, Heidelberg, Germany) were conducted in the 6th month, and the efficacies were determined (Figure 1).
CFP
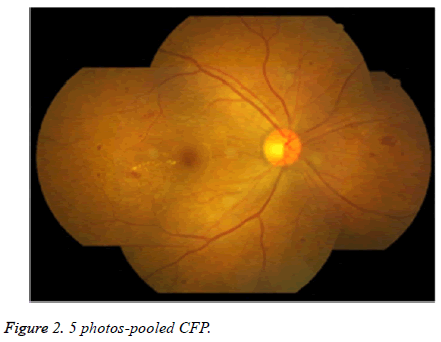
One fundus camera (Canon, Tokyo, Japan) was used to obtain color fundus photos: the first photo was taken, targeting the macula lutea-centralized posterior pole. The second photo was obtained with the macula lutea-DO line as the horizontal axis and the line perpendicular to the DO axis as the perpendicular axis, targeting the site that reached the center of macula lutea along the nasal edge on the horizontal extension from the temporal side. The three quadrants (superior, inferior, and nasal), centered on the node of the two perpendicular axes, were then imaged in one photograph. Thus, a total of five non-3D 50° CFP photos were obtained for each eye (Figure 2).
Criteria for determining clinical efficacies
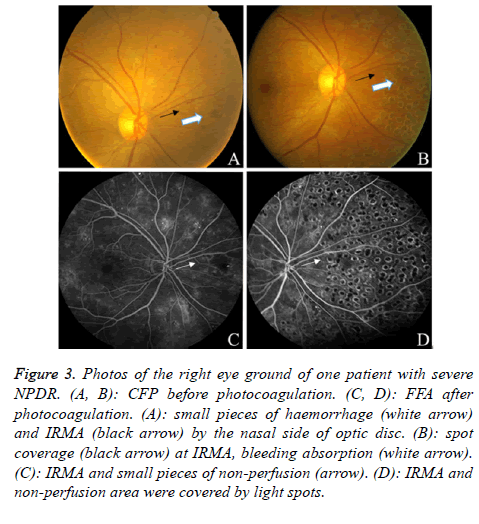
Efficacies could be divided into improved, stabilized, or declined visual acuity, according to whether BCVA increased by more than 10 letters, changed within 10 letters, or declined by more than 10 letters, respectively. The improved and stabilized visual acuity was defined as indicating efficacy, whereas declined visual acuity was defined as indicating ineffectiveness. Retinal signs were also used to categorize the approach as effective or ineffective. If total or partial retinal haemorrhage, venous beading, or Intraretinal Micro Vascular Abnormalities (IRMA) were absorbed or decreased after photocoagulation as compared to before, the treatment was defined as effective (Figure 3). If there were no changes, lesions had increased, or neovessels had appeared, the treatment was defined as ineffective. An increase in lesions or newly appeared neovessels that were found by comparing the FFA results before and after the treatment defined the treatment as ineffective. The appearance of vitreous haemorrhage during the treatment, which affected further photocoagulation treatments, was defined as ineffective.
Figure 3. Photos of the right eye ground of one patient with severe NPDR. (A, B): CFP before photocoagulation. (C, D): FFA after photocoagulation. (A): small pieces of haemorrhage (white arrow) and IRMA (black arrow) by the nasal side of optic disc. (B): spot coverage (black arrow) at IRMA, bleeding absorption (white arrow). (C): IRMA and small pieces of non-perfusion (arrow). (D): IRMA and non-perfusion area were covered by light spots.
Statistical analysis
SPSS 17.0 statistical software package was used for analysis. Normally distributed measurement data were used to calculate mean values, standard deviation, and range. The count data were expressed as percentages. Visual acuities before and after the treatment were evaluated using paired t-tests; the effectiveness rates based on CFP and FFA were determined using chi-square tests. The relationships of PRP results with pre-treatment visual acuity, disease duration, disease stage, blood pressure, and blood glucose and glycated haemoglobin levels were evaluated using the multivariate logistic regression analysis, with P ≤ 0.05 considered as statistically significant.
Informed consent
All the patients signed informed consent before treatment, and this study was approved by the Ethics Committee of Shanghai Tenth People's Hospital, Tongji University School of Medicine.
Results
From April 2010 to April 2012, this study enrolled 50 patients with severe NPDR (50 eyes), among whom five patients (five eyes) were excluded due to being diagnosed with PDR based on their final FFA, and two patients (two eyes) failed to complete the follow-up. Therefore, 43 patients (43 eyes) were finally included in the study.
Visual acuity
Before the treatment, the mean BCVA of these 43 eyes was 59.91 ± 14.58 (45.33-74.49) letters, and after treatment, it was 62.86 ± 15.41 (47.45-78.27) letters. The visual acuity of 12 patents was improved (27.9%), 27 patients exhibited stable results (62.79%), and four patients exhibited a decline in visual acuity (9.30%); these data are shown in Table 2. Comparison of visual acuity before and after the treatment revealed no statistically significant difference (t=1.782; P>0.05).
| Effective (eyes/%) | Ineffective (eyes/%) | |
|---|---|---|
| Visual acuity | 39 (90.70) | 4 (9.30) |
| Retinopathy (CFP) | 38 (88.37) | 5 (11.63) |
| Retinopathy (FFA) | 36 (83.72) | 7 (16.28) |
Table 2. Efficacies of 43 patients (43 eyes).
Retinal lesions
Efficacy as judged by CFP and FFA are shown in Table 2; there were no statistically significant differences (χ2=0.19, P>0.05).
Multivariate logistic regression analysis
The “laser effects” was set as the dependent variable, and the independent variables included the “pre-treatment visual acuity”, “disease duration”, “blood pressure”, “glycated haemoglobin”, and “fasting blood glucose”. Multivariate logistic regression analysis of these 43 eyes revealed no correlation between the post-laser visual acuity and the above inspection items (r2=0.31, P>0.05). This result excluded the confounding factor of age (OR=1.12).
Discussion
Timely photocoagulation treatments could reduce the risk of serious vision loss in DR patients by 50% [8]; however, the exact underlying mechanisms remain unclear. The underlying mechanisms may include facilitation of oxygen and nutrient transport from the choroid to the retina, facilitation of metabolite elimination from the retina, and reduction of the retinal metabolic burden, thereby reducing the production of Vascular Endothelial Growth Factor (VEGF) [9]. Clinically, the classical pathway of laser photocoagulation therapy for DR patients is as follows: first, performing FFA before photocoagulation, and then deciding whether to choose to use photocoagulation, or choosing the photocoagulation type according to the FFA results [10]. However, for various reasons, such as lack of imaging equipment, sources of contrast agents, or the patients’ own health problems, FFA may not be performed; therefore, partial DR patients could not enter the therapeutic approach of “first FFA, then laser treatment”. As an alternative, the laser treatment would have to be guided by the signs of retinopathy, typically using the DR staging criteria established by the Third Academic Committee of National Ophthalmology in 1985 [11]. As they are convenient to use and easy to grasp, these criteria are still widely used. However, this staging cannot adequately reflect the severity of the condition, and does not show a progressive relationship with stage II and III lesions.
In 2003, the American Academy of Ophthalmology (AAO) and ICO announced new DR staging approaches, relying on evidence-based medicine, and described the corresponding classifications in greater detail. In particular, the description of severe NPDR by the 4-2-1 principle also made it easy for nonophthalmologists to understand the severity of the diseased eye, thus providing reasonable treatment recommendations [5]. The 4-2-1 principle is a standard formulated by the Early Treatment DR Study group (ETDRS, Treatment Diabetic Retinopathy Study) in the USA in 1991, and emphasized the use of post-dilated ophthalmoscope or seven standard 35° CFP photographs. This principle allowed clinicians to obtain information from such photographs, on which judgment and analysis was based, in order to assess whether NPDR patients had transitioned to a state of high risk of proliferation [12]. Severe NPDR is the critical risk period, where NPDR may enter the proliferative stage; therefore, severe NPDR would be the best time for photocoagulation. The consensus for PRP treatment of severe NPDR patients in countries other than China was as follows: patients with 1) poor compliance; 2) requirement for cataract surgery; 3) preparing for pregnancy; 4) blindness of the contralateral eye; and 5) elderly and type 2 diabetes mellitus [13-16]. However, indication selection criteria, and the reliability of efficacy of treatment based on the 4-2-1 principle has been limited to assessing disease stages, both in China and abroad [7,17].
This project was a 2-year joint clinical ophthalmological research study co-performed by the Ophthalmological Departments of one third-level hospital and two second-level hospitals. Diabetic patients in China are often first diagnosed in second-level hospitals, and therefore, DR patients would normally be initially diagnosed in second-level hospitals. For this reason, it is necessary to popularize the international DR staging approach and to specify the use of laser photocoagulation to ophthalmologists.
This project used five 50° CFP photos for the clinical staging of DR cases [18]; this differed from the seven 35° standard photos used by the ETDRS; yet, the information obtained was essentially the same, and covered the major sites of DR.
The photocoagulation method used standard PRP. In addition to the unified treatment parameters, the distribution of light spots during photocoagulation should also receive more attention. The light spots were slightly smaller (approximately 200 μm) when they were close to the posterior pole, and were 500 μm at other sites; the light spots should not be too small or too large, in order to ensure an effective spot area.
In this study, severe NPDR patients were screened for treatment by PRP based on the 4-2-1 principle, and the visual acuity in almost 90% of the patients did not decrease, while the effective rate of retinopathy was 88.37% and the effective rate of FFA determination was 83.72%. Two cases that were determined as effective were re-determined as ineffective by FFA; this was mainly due to the interference of observation during angiogenesis by laser spots. In particular, angiogenesis in the surrounding area was more difficult to identify, so that when image analysis is conducted, every part of the image should be carefully compared with that part in an image taken before treatment, to avoid being missed. This also confirmed that FFA remains the gold standard for guiding the treatment of DR [19]. Lobestam et al. reported the effective rate of laser in severe NPDR in Type 1 DM patients as 65%, which was lower than our results, and this may be due to the relatively long follow-up time [20].
Our multi-center study demonstrated that, under conditions where FFA is not available, using 4-2-1 principle-based photocoagulation in DR is an effective approach. Using this approach, disease progression could be effectively controlled in the vast majority of severe NPDR patients, thus preventing severe visual impairment.
Acknowledgments
Projects of appropriate techniques co-developed and promoted among Shanghai municipal hospitals (SHDC1201027).
Conflicts of Interest
All of the authors declare that they have no conflicts of interest regarding this paper.
References
- Gardner TW, Antonetti DA, Barber AJ, LaNoue KF, Levison SW. Diabetic retinopathy: more than meets the eye. Surv Ophthalmol 2002; 47: 253-262.
- Xu Y, Wang L, He J, Bi Y, Li M. Prevalence and control of diabetes in Chinese adults. JAMA 2013; 310: 948-959.
- Zou HD, Xu X, He J, Xu X, Du X, Bai X, Zhang B, He X, Lu L, Zhu J, Zhao J. Prevalence and risk factors of mild vision loss in patients with type 2 diabetes in Beixinjing Community of Shanghai. Zhonghua Yi Xue Za Zhi 2016; 96: 210-215.
- Xu X, Zou H, Ning G. Active response to high momentum of diabetic retinopathy with the strategy of combined treatment and prevention. Zhonghua Yan Ke Za Zhi 2015; 51: 801-803.
- Wilkinson CP, Ferris FL 3rd, Klein RE, Lee PP, Agardh CD, Davis M, Dills D, Kampik A, Pararajasegaram R, Verdaguer JT. Global diabetic retinopathy project group. Proposed international clinical diabetic retinopathy and diabetic macular edema disease severity scales. Ophthalmol 2003; 110: 1677-1682.
- Klein R, Klein BE. Screening for diabetic retinopathy, revisited. Am J Ophthalmol 2002; 134: 261-263.
- Khalaf SS, Al-Bdour MD, Al-Till MI. Clinical bio-microscopy versus fluorescein angiography: effectiveness and sensitivity in detecting diabetic retinopathy. Eur J Ophthalmol 2007; 17: 84-88.
- Early treatment diabetic retinopathy study research group. Fundus photographic risk factors progression of diabetic retinopathy. ETDRS report number 12. Ophthalmol 1991; 98: 823-833.
- Blumenkranz MS. The evolution of laser therapy in ophthalmology: a perspective on the interactions between photons, patients, physicians, and physicists: the LXX Edward Jackson Memorial Lecture. Am J Ophthalmol 2014; 158: 12-25.
- Early treatment diabetic retinopathy study research group. Classification of diabetic retinopathy from fluorescein angiograms. ETDRS report number 11. Ophthalmol 1991; 98: 807-822.
- Retinal academy group. The grading criteria of diabetic retinopathy. Zhonghua Yan Ke Za Zhi 1985; 21: 113.
- Early treatment diabetic retinopathy study research group. Grading diabetic retinopathy from stereoscopic color fundus photographs-an extension of the modified Airlie House classification. Ophthalmol 1991; 98: 786-806.
- Ghanchi F. The Royal College of Ophthalmologists clinical guidelines for diabetic retinopathy: a summary. Eye 2013; 27: 285-287.
- Neubauer AS, Ulbig MW. Laser treatment in diabetic retinopathy. Ophthalmologica 2007; 221: 95-102.
- American Academy of Ophthalmology. Diabetic Retinopathy PPP-updated October 2012; 2013.
- Mitchell P, Foran S. Guidelines for the management of diabetic retinopathy, 2014.
- Gao LQ, Zhang F, Zhou HY, Yan W, Xiong Y, Wang GL. Comparison of fundus photography and fluorescein angiophy in grading diabetic retinopathy. Zhonghua Yan Ke Za Zhi 2008; 44: 12-16.
- Moss SE, Meuer SM, Klein R, Hubbard LD, Brothers RJ, Klein BE. Are seven standard photographic fields necessary for classification of diabetic retinopathy? Invest Ophthalmol Vis Sci 1989; 30: 823-828.
- Early photocoagulation for diabetic retinopathy. Early treatment diabetic retinopathy study research group. Ophthalmol 1991; 98: 766-785.
- Lovestam-Adrian M, Agardh CD, Torffvit O, Agardh E. Type 1 diabetes patients with severe non-proliferative retinopathy may benefit from panretinal photocoagulation. Acta Ophthalmol Scand 2003; 81: 221-225.