- Biomedical Research (2015) Volume 26, Issue 2
Effects of polysaccharides of Radix ranunculi ternati on Type I Skin Hypersensitivity in rats.
Zhongxia Zhang1, Meiying Wang2 and Tianying Yu3*1Department of Rehabilitation Medicine, Affiliated Hospital of Weifang Medical University, Weifang 261031, Shandong province, China
2Department of Pediatrics, Affiliated Hospital of Weifang Medical University, Weifang 261031, Shandong province, China
3Department of Anesthesiology, Affiliated Hospital of Weifang Medical University, Weifang 261031, Shandong province, China
- *Corresponding Author:
- Tianying Yu
Department of Anesthesiology
Affiliated Hospital of Weifang Medical University
Weifang 261031, Shandong province
China
Accepted date: January 11 2015
Abstract
Radix Ranunculi Ternati the dried root tuber of Ranunculus ternatus Thunb. is neutral, sweet, pungent and warm, and enters the liver and lung meridians. It has the effects of removing toxicity, resolving phlegm and dissipating nodulation. We undertook this work to study the effect of extracted polysaccharides from Radix Ranunculi Ternati on Type I skin hypersensitivity. Water extraction and ethanol precipitation was used in extraction. Phenol-sulfuric acid method was used in determination of polysaccharides and type I hypersensitivity was established in rats. The rats were randomized into the blank control group, chlorphenamine group, cortisone group and Radix Ranunculi Ternati polysaccharide low-, medium- and high-dose groups (0.50, 1.00 and 1.50 g/kg). Polysaccharide content in Radix Ranunculi Ternati extract was determined to be 6.7%; the three doses could significantly inhibit the diameter of antiserum-induced blue reaction spots on dorsal skin of rats (P<0.01), of which the high- and medium-doses had efficacies equivalent to chlorpheniramine. Three Radix Ranunculi Ternati polysaccharide dose groups could inhibit histamine- and serotonin-induced increase in capillary permeability in rats, and inhibit histamine- induced paw edema in rats (P<0.01), of which the efficacies of high- and medium-dose groups were not significantly different from the chlorpheniramine group. Hence, polysaccharides from Radix Ranunculi Ternati may suppress type I skin hypersensitivity by the pathway of anti-allergic inflammatory mediators.
Keywords
Radix Ranunculi Ternati; polysaccharide; type I hypersensitivity; anti-allergy
Introduction
Radix Ranunculi Ternati is the dried root tuber of Ranunculus ternatus Thunb. in the family Ranunculaceae. Radix Ranunculi Ternati is neutral, sweet, pungent, warm, and enters the liver and lung meridians. It has the effects of removing toxicity, resolving phlegm and dissipating nodulation, and is used in the treatment of scrofula, lymph node tuberculosis, pharyngitis, etc. [1]. Chemical constituents of Radix Ranunculi Ternati are mainly polysaccharides, as well as some fatty substances and a small amount of alkaloids, etc. [2-5]. There have been studies indicating that saponins and polysaccharides from Radix Ranunculi Ternati have varying degrees of effects on growth and colony formation of three tumor cell lines, namely sarcoma S180, Ehrlich ascites tumor EAC and human breast cancer MCF-7 cell lines [6-9]. The author further studied the anti-type I hypersensitivity effect of polysaccharides from Radix Ranunculi Ternati.
Materials
Instruments
Model 721 spectrophotometer (Chengdu Fifth Factory of Optical Instruments); HY-250C electronic thermostatic water bath (Chengdu Hengyu Instrument Factory); Soxhlet extractor. ED-120 centrifuge (Beijing Hengtong Medical Machinery Factory).
Drugs and reagents
Radix Ranunculi Ternati medicinal material was purchased from crude drug market in Anyang, which was identified by Prof. Wang Zhen-xin of our college as the dried root tuber of Ranunculus ternatus Thunb. Glucose reference substance (AR); phenol reagent (redistilled). All other reagents were of analytical grade. Histamine phosphate (Chongqing Biological Reagent Factory, batch number 1302658); Evans blue (Chongqing Biological Reagent Factory, batch number 1304265); 0.9% sodium chloride (Chongqing Biological Reagent Factory, batch number 13012153644). Cortisone tablets (Xianju Pharmaceutical Co., Ltd., batch number 130256), chlorpheniramine (Zhengzhou Hasen Pharmaceutical Co., Ltd., batch number 2010334781).
Animals
SD rats, half male and half female, provided by the SPF Laboratory Animal Center of Chengdu Medical University, inspection certificate numbers were CDYK–C 2013- 0236 and CDYK-E 2013-0428, respectively
Preparation of polysaccharides from Radix Ranunculi Ternati
Extraction of polysaccharides from Radix Ranunculi Ternati
100 g of dried Radix Ranunculi Ternati powder was accurately weighed, placed into a Soxhlet extractor, and extracted under reflux successively with petroleum ether (30℃~60℃), ethyl ether and ethanol for 4 h, after evaporation of solvent, the residue was again extracted under reflux with water for 4 h. The extract was then concentrated under reduced pressure to half volume, decolorized with 0.1% active carbon, and filtered; the filtrate was added with 95% ethanol to make the solution contain 80% ethanol, and allowed to stand overnight. The filtration residue was washed repeatedly with ethyl ether and anhydrous ethanol to obtain polysaccharides of Radix Ranunculi Ternati, which was freeze-dried for later use.
Determination of polysaccharide content in Radix Ranunculi Ternati
25.2 mg of dried glucose standard substance was accurately weighed, dissolved in an appropriate amount of water, then transferred into a 250 mL volumetric flask, dilute to the mark with water, and shaken well to prepare a 100.8 μg/mL standard glucose solution and set aside.
100 g of phenol was taken, and distilled with 0.1 g of aluminum and 0.05 g of NaHCO3, fractions at 182℃ were collected, 7.5 g of the fraction was weighed, dissolved in 150 mL of water, placed in an amber bottle, sealed, and stored at 4℃.
0.0, 0.1, 0.2, 0.3, 0.4, 0.5, 0.6 and 0.7 mL of glucose standard solutions were accurately weighed, placed in dry test tubes, respectively, the volumes were made to 1.0 mL with water, then 1.6 mL of 5% phenol solution was added, respectively, after shaking well, 7.0 mL of concentrated H2SO4 was added, fully shaken, and allowed to stand at room temperature for 25 min, maximum absorbance was shown at a wavelength of 490 nm, the above experimental data were regressed to obtain regression equation: A=0.06873C+6.8581×10-3, r=0.998920.00 mg of polysaccharides from Radix Ranunculi Ternati were accurately weighed, placed in a 100 mL volumetric flask, dissolved and diluted to the mark with water, and shaken well to be used as the stock solution.
2.00 mL of the stock solution was accurately drawn, and absorbance was measured, followed by calculation of conversion factor according to the following formula: f=W/CD. Where, f is the conversion factor; W is the weight of polysaccharides (μg); C is the concentration of glucose in polysaccharides (μg/mL); and D the dilution factor of Radix Ranunculi Ternati polysaccharides. The conversion factor is measured to be f=1.70.
0.20 g of dried Radix Ranunculi Ternati powder was accurately weighed, soaked overnight with 80% ethanol, and then extracted under reflux with 80% ethanol in a Soxhlet extractor for 2 h, followed by filtration. The residue was then extracted under reflux with water for 2 h, the entire extract was transferred into a 250 mL volumetric flask, diluted to the mark, and shaken well to be used as the sample solution. 1 mL of the sample solution was accurately pipetted, and its absorbance was measured, followed by calculation of polysaccharide content, which was 6.70%, according to the following formula.
Polysaccharide content % = CDf/w × 100. Where: C is the concentration of glucose in the sample solution (μg/mL), D is the dilution factor of the sample solution, f is the conversion factor, and w the weight of sample (μg).
0.15 g of Radix Ranunculi Ternati powder was accurately weighed, added with 7 mg of polysaccharides, and refluxed in a Soxhlet extractor by addition of 80% ethanol for 2 h, after removal of solvent, the residue was continuously extracted under reflux with water for 2 h, washed repeatedly, then placed into a 250 mL volumetric flask, and diluted to the mark, after shaken well, 1 mL of the solution was accurately pipetted for measurement of absorbance, polysaccharide content was calculated, and the average recovery rate was converted to be 101.98% (n = 5).
Anti-type I hypersensitivity test
Grouping and administration methods
The rats were randomly divided into six groups: normal group (saline group), chlorpheniramine 4 mg/kg group, cortisone 5 mg/kg group; and Radix Ranunculi Ternati polysaccharide 0.50, 1.00, 1.50 g/kg groups. Each group was ig administered qd for 7 consecutive days.
Establishment of mouse model of histamine-induced increased capillary permeability
1.5 h after last administration, rats were injected in the tail vein with 0.5% 5 mL/kg Evans blue solution in saline, then immediately injected sc with 0.1% histamine phosphate in the middle of the abdomen at 0.1 mL/mouse to form a small papule. 1 h later, the rats were sacrificed by cervical dislocation, abdomens were dissected, and the blue stained abdominal skins were removed, cut into pieces with surgical scissors, placed into the test tube, added with 4 mL of acetone-saline (7:3) mixture, and shaken. After soaking for 24 h, the skin pieces were centrifuged at 2000 r/min for 20 min, then the supernatant was taken and its absorbance (A) was measured at 610 nm.
Effect of polysaccharides from Radix Ranunculi Ternati on serotonin-induced increased capillary permeability in rats
Grouping, administration and test methods were identical with the "Grouping and administration methods" with the exception that 0.2% serotonin creatinine sulfate was used instead of 0.1% histamine phosphate.
Effect on histamine-induced paw edema in rats
Grouping and administration methods were identical with the "Grouping and administration methods", administration was continued for 4 consecutive days. On the 4th day, 1 h after the last administration, right hind paw of each mouse was injected sc with 10 μL of 0.05% histamine phosphate solution in saline. Thickness of right hind paw was measured before and 15, 30, 60, 120 min after injection, respectively, and the degree of edema was expressed by the difference in thickness before and after injection.
Effect on passive cutaneous anaphylaxis in rats
Preparation of anti-ovalbumin serum
5 SD rats were taken, and given sc injection of 5% fresh egg white solution in saline in the inner hind leg at 0.5 mL/rat, as well as ip injection of 0.1 mL of 5% Al(OH)3 gel, on the 14th day, blood sample was collected from carotid artery, and serum was separated at 2000 r/min for 10 min, after mixing well, the serum was stored in a refrigerator (-4℃) for later use.
Grouping and administration methods
Grouping and administration methods were the same as above, on the 7th day, 1 h after the last administration, the rats were lightly anesthetized with ethyl ether, dorsal hair was shaved on both sides of dorsal midline 1.5 cm from the spine, three sites were shaved on each side, and each site was spaced 1.5 cm apart. The above IgE anti-serum was taken, and diluted with saline at three different dilution ratios of 1:30, 1:40 and 1:50, the different concentrations of anti-serum were successively injected ic into the shaved sites at 0.1 mL per site. On the 8th day, antigen challenge was performed: 1 ml of 0.5% Evans blue solution containing 1 mg ovalbumin was injected into the tail vein. 20 min later, the rats were sacrificed by cervical dislocation, dorsal skins were removed, and the diameter of blue reaction spot on each injection site was examined.
Statistical methods
Data were processed using SPSS 15.0 statistical software, measurement data were expressed as x — ±s, comparison between groups was performed by t test, P<0.05 was considered statistically significant.
Results
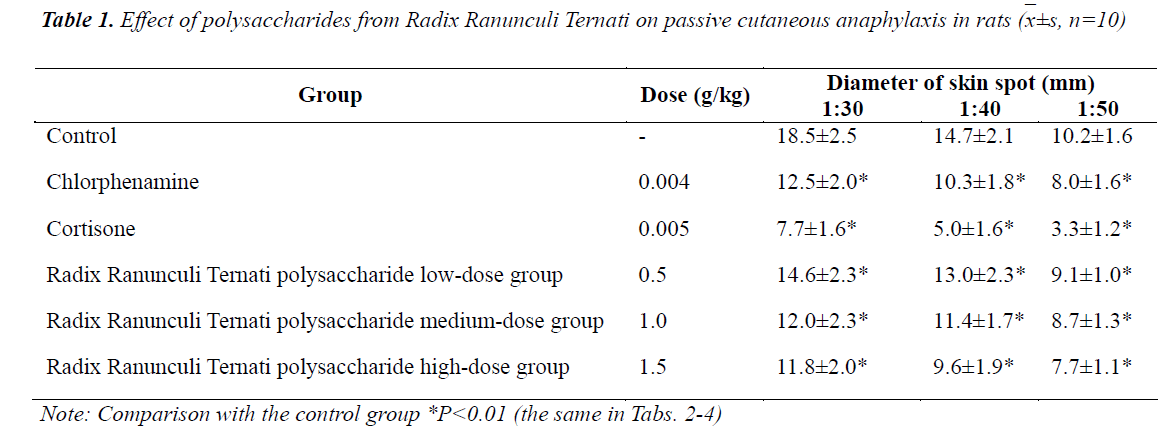
Effect of polysaccharides from Radix Ranunculi Ternati on passive cutaneous anaphylaxis in rats
Compared with the control group, the three Radix Ranunculi Ternati polysaccharide doses could significantly inhibit the diameter of blue reaction spots induced by each dilution ratio of antiserum on dorsal skin of rats (P<0.01), of which the high- and medium-doses had efficacies equivalent to chlorpheniramine. See Table 1.
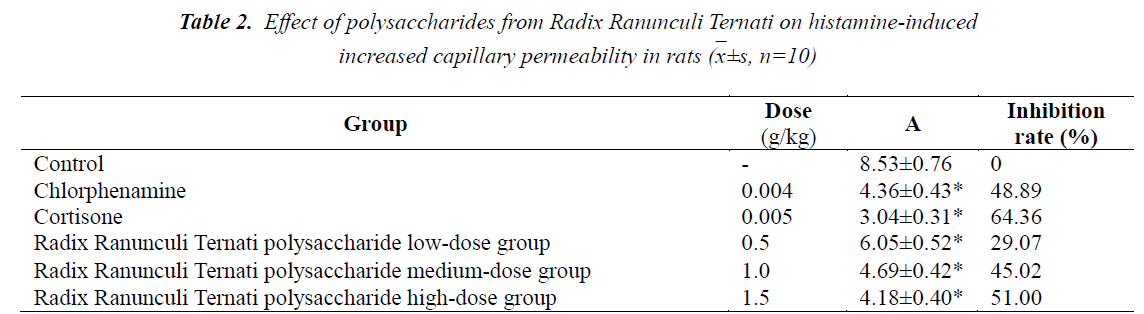
Effect of polysaccharides from Radix Ranunculi Ternati on histamine-induced increased capillary permeability in rats
Compared with the control group, the three Radix Ranunculi Ternati polysaccharide dose groups could all reduce the A value, and the differences were statistically significant (P<0.01); the A value reducing effects of Radix Ranunculi Ternati polysaccharide high- and medium- dose groups were equivalent to that of the chlorpheniramine group. See Table 2.
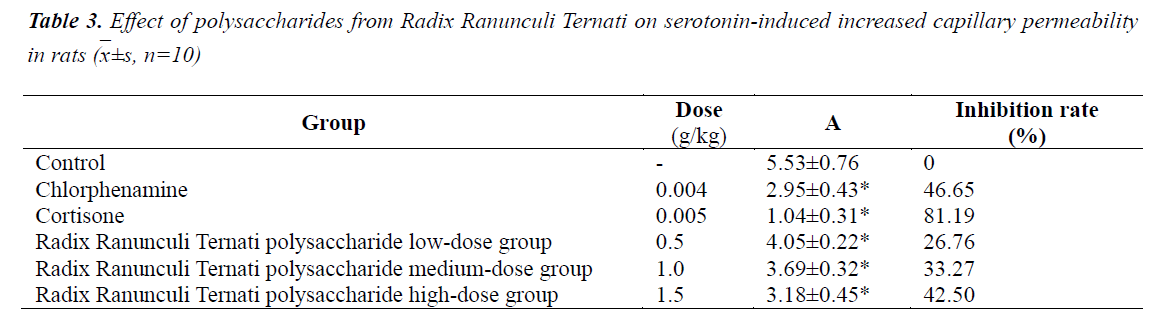
Effect on serotonin-induced increased capillary permeability in rats
The results are shown in Table 3. Compared with the control group, the three Radix Ranunculi Ternati polysaccharide dose groups could all reduce the A value, and the differences were statistically significant (P<0.01); the A value reducing effects of Radix Ranunculi Ternati polysaccharide high- and medium-dose groups were equivalent to that of the chlorpheniramine group.
Effect on histamine-induced paw edema in rats
The results are shown in Table 4. Compared with the control group, the three Radix Ranunculi Ternati polysaccharide dose groups could all reduce the degree of histamineinduced paw edema in rats at each observation time point, and the differences were statistically significant (P<0.01); the histamine-induced paw edema degree reducing effects of Radix Ranunculi Ternati polysaccharide high- and medium- dose groups were equivalent to that of the chlorpheniramine group at each time point.
Discussion
Type I hypersensitivity is mediated by specific IgE, and is accompanied by release of large amounts of bioactive media by effector cells such as mast cells, basophils and eosinophils. Histamine and serotonin are released by cell granules, which can cause vasodilation of venules and capillaries, increase vascular permeability, and promote mucosal and glandular secretion, etc. Histamine acts on H1 and H2 receptors on the surface of target cells, thereby producing inflammatory reactions, which are mainly manifested as the vasodilation of small blood vessels, increased capillary permeability, and induction of cutaneous pain or itching skin [10-12].
The present study showed that the three doses of Radix Ranunculi Ternati polysaccharides could significantly inhibit the diameter of blue reaction spots on dorsal skin of rats, of which the high- and medium-doses had efficacies equivalent to chlorpheniramine, indicating that the Radix Ranunculi Ternati polysaccharides has a good antiallergic effect on type I hypersensitivity model; Radix Ranunculi Ternati polysaccharides could inhibit histamine- and serotonin-induced increase in capillary permeability in rats, and histamine-induced paw edema in rats, of which the efficacies of high- and medium-doses were equivalent to chlorpheniramine, suggesting that Radix Ranunculi Ternati polysaccharides may exert anti-type I skin hypersensitivity effect by the pathway of anti-allergic inflammatory mediators. Ranunculus ternatus Thunb. is grown and widely cultivated in provinces such as Henan and Anhui, the present study has demonstrated that Radix Ranunculi Ternati contains polysaccharides, and has anti-hypersensitivity actions, providing a research basis for clinical application and development.
References
- Chinese Pharmacopoeia Commission. Chinese Phar- macopoeia, Vol. I. Beijing: Chemical Industry Press 2005: 223-224.
- Zhan Z, Feng Z, Yang Y, Li L, Jiang J, Zhang P. Ternatusine A, a New Pyrrole Derivative with an Ep- oxyoxepino Ring from Ranunculus ternatus. Org Lett 2013; 15: 1970-1973.
- Tian JK, Sun F, Cheng YY. Chemical constituents from the roots of Ranunculus ternatus. Journal of Asian Natural Products Research 2006; 8: 35-39.
- Chen J, Yao C, Xia LM, Ouyang PK. Determination of fatty acids and organic acids in Ranuncu- lus ternatus Thunb using GC-MS. SPECTROSCOPY AND SPECTRAL ANALYSIS 2006; 26: 1550-1552.
- Li JY, Liu DJ, Liu JK. Analysis of 180 cases of tuber- culous cervical lymphadenitis treated with Traditional Chinese medicine Radix Ranunculi Ternati. Tianjin Medical Journal 1964; 6: 958-962.
- Wang AW, Wang M, Yuan JR, Tian JK, Wu LM, Geng H. The study on antitumor effects in vitro of different extracts in Radix Ranunculi Ternati. Natural Product Research and Development 2004; 16: 529-532.
- Oyaizu M. Studies on product of browning reaction prepared from glucose amine. Jpn J Nutr 1986; 44: 307-309.
- Jiang YH, Jiang XL. The antitumorand antioxidative activities of polysaecharides isolated from Isaria fari- nosa B05. Microbiol Res 2006; 3: 112-114.
- Marx JL. Oxygen free radicals linked to many dis- eases. Science 1987; 235: 529-529.
- Li J, Yang WP, Xiao HB. Experimental study on treatment of anaphylactoid purpura by Xijiaodihuang Decoction. Acta Chinese Medicine and Pharmacology 2004; 3: 9-12.
- Chen YZ, Fan SL, Ou Q. Kangmin Tang Combined with Auto-blood Therapy for the Treatment of Chronic Urticaria. Chinese Journal of Experimental Traditional Medical Formulae 2011; 17: 254-255.
- Zhang YB, Qu X, Ren Y, Li YQ. Experimental study on the effect of Xijiao (Shuiniujiao) Dihuang Decoc- tion on animal model of acute dermatitis and allergic dermatitis. Chinese Journal of Experimental Traditional Medical Formulae 2008; 14: 61-63.
 ±s, n=10)
±s, n=10)