- Biomedical Research (2012) Volume 23, Issue 1
Effects of climate changes on dissolved heavy metal concentrations among recreational park tributaries in Pahang, Malaysia
Shantakumari Rajan1*, Nurul Nadiah Mohd Firdaus2, Mahenderan Appukutty3, Kalavathy Ramasamy4
1Faculty of Health Sciences, Universiti Teknologi MARA, Malaysia
2Faculty of Applied Science, Universiti Teknologi MARA, Malaysia
3Faculty of Sports Science and Recreation, Universiti Teknologi MARA, Malaysia
4Faculty of Pharmacy, Universiti Teknologi MARA, Malaysia
- Corresponding Author:
- Shantakumari Rajan Faculty of Health Sciences Universiti Teknologi MARA 42300 Puncak Alam Selangor Malaysia
Accepted date: October 14 2011
Abstract
River water quality is important for ecological health. The Tembeling River is the main river flowing through the Pahang National Park and is used as a resource for navigation as well as for recreation purposes. Recreational use of water may offer health benefits to human. Waters contaminated with chemicals that are either toxic or irritating to the skin or mucous membranes are unsuitable for recreational purposes. The objective of the study was to evaluate the heavy metal chemicals changes before and after monsoon. Water samples from five tributaries (Sg Tahan, Sg Keniam, Sg Trenggan, Sg Perkai and Sg Tenor) were monitored for temperature, pH, dissolved oxygen, and concentrations of eight dissolved metals (Ag, Cd, Cu, Cr, Fe, Ni, Pb and Zn) over six different sampling periods; 3 before the onset of the seasonal monsoon and three after the monsoon period. All sampling stations were located within the recreational zones, where conditions are homogenous. Cr, Cu, Fe, Pb, Ni and Zn concentrations were significantly higher before the monsoon compared to the period after. However, concentrations of Ag and Cd showed an opposite pattern compared to the other metals. Seasonal patterns of Ag and Cd in the forested area were characterized by decreased concentrations of them following monsoon but gradual increased levels of Ag and Cd were recorded as the water flow reduced. Ag, Fe, Pb, and Cd were found to be present at higher concentrations than the recommended standards. The sources contributing to high concentrations of metals were found to be of natural origin. The results suggest that the water supply in the National Park is of acceptable quality for recreational purposes. However, continuous monitoring of environmental pollutants is essential in order to prevent any recreational water illness.
Keywords
Heavy metals; Recreational water, Forest ecosystem; Temporal distribution
Introduction
Toxic metals, including "heavy metals," are individual metals and metal compounds that negatively affect people's health. In very small amounts, many of these metals are necessary to support life. However, in larger amounts, the heavy metals become toxic and may build up in biological systems and create a significant health hazard. Once liberated into the environment through the air, drinking water, food, or countless human-made chemicals and products, heavy metals are taken into the body via inhalation, ingestion, and skin absorption [1]. If heavy metals enter and accumulate in body tissues faster than the body's detoxification pathways can dispose them off, a gradual buildup of these toxins will occur [2].
Human exposure to heavy metals has risen dramatically over the years, as a result of an exponential increase in the use of heavy metals in industrial processes and products often resulting in the release of toxic heavy metals [3]. At present, the chronic exposure comes in many ways namely from mercury-amalgam dental fillings, lead in paint and tap water, chemical residues in processed foods, and “personal care” products such as cosmetics and toiletries [4].
In today's industrial society, there is no escaping exposure to toxic chemicals and metals. In addition to the hazards at home, even outdoor recreational water may be exposed to pollution. The heavy metals can directly influence behavior by impairing mental and neurological function, influencing neurotransmitter production and utilization, and altering numerous metabolic body processes [5]. The systems in which toxic metal elements can induce impairment and dysfunction include the blood and cardiovascular [6], energy production pathways [7], gastrointestinal [8], nervous (central and peripheral) [9], reproductive, and urinary [10].
Pollutant metals are usually non-degradable and there is no known homeostasis mechanism for them. Thus, any high levels of heavy metals will threaten biological life [11,12]. Nutritional status, metabolic rate, the integrity of detoxification pathways (ability to detoxify toxic substances), and the mode and degree of heavy metal exposure all affect how an individual responds. Children and the elderly, whose immune systems are either underdeveloped or age-compromised, are more vulnerable to toxicity [13].
Recreational water parks become a common leisure place for many individuals but many are not aware of the quality of water, which may harm their health if polluted. Till to date there is not much local data available on heavy metal content in recreational water. Results of our investigation will be important at the different levels of recreational water park management authorities to promote future conservation actions for this aquatic resource to have a clean environment.
Therefore, this study was carried out to investigate the monsoon-related changes in dissolved metal concentrations in channel waters of the Tembeling River tributaries in the Pahang National Park.
Materials and methods
Site description: The main tributaries of Sungai Tembeling are Sungai Tahan arising from Mount Tahan (elevation 2187 m) and Sungai Keniam from the east. The climate is moderate and the annual average temperature is 22 - 26°C. The average yearly rainfall is approximately 2200 mm (lowland) and 3800 mm (highland). The water quantity of Sungai Tembeling is plentiful, also an important source of water downstream.
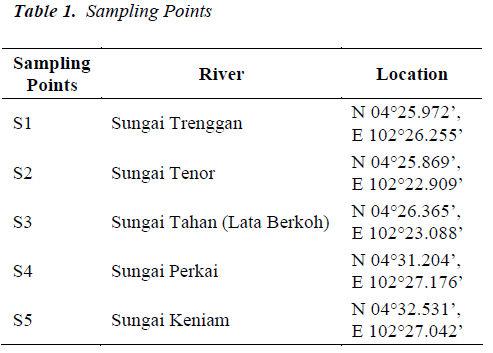
Water Sampling: Surface water samples were collected before and after the monsoon/ rainy season in a year calendar (Table 1). The sampling times were planned in advanced without capturing temporary events. The sampling strategy was designed to cover a wide range of determinants at key sites and account for tributary inputs that can have important impacts upon downstream water quality. The number of the sampling sites was optimized in such a way that only representative sites from each river would be used. Since the variability of trace-element concentration may be important even within a 24 hour period [14], sampling was performed in the same day.
The physicochemical characteristics of the water recorded directly in the field at each sampling station were temperature, pH and dissolved oxygen. Acid-washed highdensity polyethylene bottles were filled upstream of the sampler to avoid any material disturbed from the streambed, and below the water surface to avoid floating material and the surface film. A total of two replicates were taken for every sample. Samples were transported in sealed polyethylene bags to prevent contamination and all bottles were kept in a cool box. All sampling, preservation and analyses were performed as per standard methods [15].
Chemical analysis: The “dissolved” fraction was operationally defined as that which passes through a 0.45-mm membrane filter. Filtrates were acidified with 1% (v/v) nitric acid as a preservative and stored in acid washed polyethylene bottles.
Metal concentrations in surface waters were determined by Inductively Coupled Plasma Mass Spectrometry (ICPMS) [16]. Analytical precision was in good agreement and the recoveries ranged from 91% to 104%.
Data Analysis: Descriptive data were generated for all variables and presented as means ± standard deviation (SD). Statistical analyses were performed with the Statistical for Applied Science (SAS) statistical package for Windows. Statistical analysis of variance (ANOVA) was used to determine the significance differences at P<0.05 for comparison between samples before and after monsoon.
Results and Discussion
Taman Negara (National Park) straddles the state borders of Kelantan, Terengganu and Pahang in Peninsular Malaysia. A totally protected area of 4,343 sq km, Taman Negara is one of the world's oldest tropical rain forests. Sungai Tembeling is the main river flowing through the park and converges with Sungai Jelai to form the Pahang River. The river acts as the chief artery for river borne traffic wishing to go upstream. The water itself is being utilized for various recreational activities, in addition to being the main source of water. In recent years, with an increase in population and tourism, settling around Kuala Tembeling has shown an increase. The entry point of the Pahang National Park at Kuala Tahan and Sungai Relau has the highest record of 81,974 visitors in the year 2007 [17].
Seasonal variations in precipitation, surface runoff, interflow, groundwater flow and pumped in and outflows have a strong effect on river discharge and subsequently on the concentration of pollutants in river water [18]. During periods of low-to-mean discharges, heavy metal associated sediments accumulate in the river channels. During floods, they are resuspended and transported downstream as well as dispersed onto the flood plains in the form of vertically accreted overbank alluvium [19, 20]. Although the periods of low-to-mean discharge occur for as much as 99% of any given year, 50% of the annual suspended load may be transported during a short flood event [21].
Long-term and seasonal changes of heavy metal concentrations in channel water and sediments can be attributed to changes in heavy metal load discharged to a river channel and the river discharges are related to the amount and intensity of precipitation [22]. Also, higher heavy metal concentrations are exhibited in suspended sediments and water during a wet season than in a dry season [23]. The National Park can be considered as a pristine environment with natural concentrations of elements in rivers because of the absence of any industrial or agricultural activity and limited accessibility. As such, we expect only the presence of long-range worldwide atmospheric pollution without any local source of contaminants, with the exception of sources from the natural rock.
In view of the spatial and temporal variations in the hydrochemistry of rivers, regular monitoring programs are required for reliable estimates of water quality [24]. Thus, it has become a timely requirement to build up records and understanding of the pollution processes and trends in order to keep this water body free from contamination. We were, therefore interested to investigate the effects of monsoon-related changes in dissolved metal concentrations in channel waters of the Tembeling River tributaries in the Pahang National Park. The seasonal variability of the studied elements and statistical correlations between them were determined.
Six sets of data were obtained from repeated sampling and analysis, whereby three sets were taken preceding the monsoon season of 2007 and three sets taken following the monsoon season. A total of 10 metals were analyzed for each set of data.
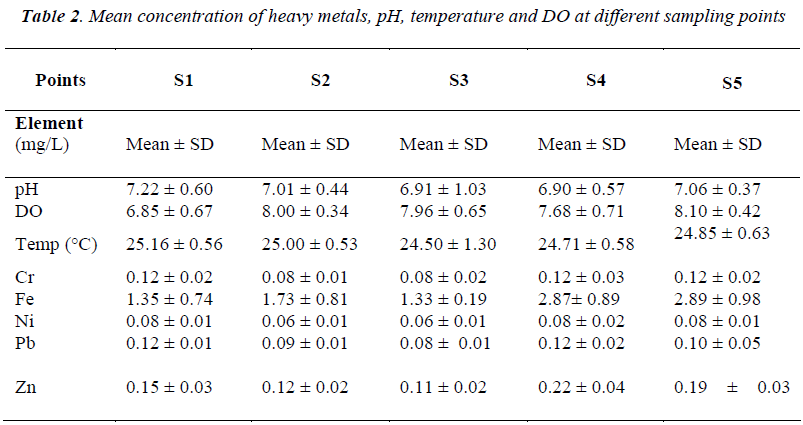
Average pH for all streams ranged from 6.9 to 7.2 (Table 2). Highest pH was observed in a small headwater site (S1) and lowest pH was observed in Sungai Tahan (S3), the largest stream in the study. This sampling site was downstream from Mount Tahan and the water chemistry influenced by processes at the higher elevation. Measured mean heavy metal concentrations in filtrates are shown in Table 2.
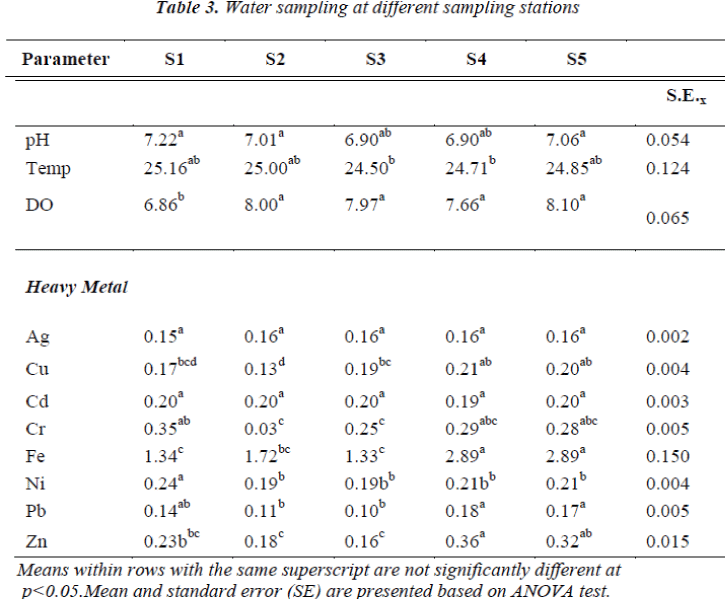
In the forested catchment we observed no difference in pH for the different headwater streams. A similar pattern was observed for the surface water temperature. However, for dissolved oxygen (DO) content, S1 had the lowest reading compared to the rest. Temporal patterns showed increasing pH and temperature after the monsoon season. Nevertheless, temperatures were the highest just prior to the monsoon season in October 2008. The DO did not present any pattern, though the highest DO was recorded after the monsoon season in February 2008 (Tables 3 and 4). There was no significant correlation between pH, DO and temperature. This could be due to the diminutive seasonal variation in these parameters. All stations had temperatures representative of Class I of the Interim National Water Quality Standard (INWQS) [25]. The results for pH showed the same pattern as temperature with all stations within the range of 6.5 – 8.5 and falling under Class I of the INWQS.
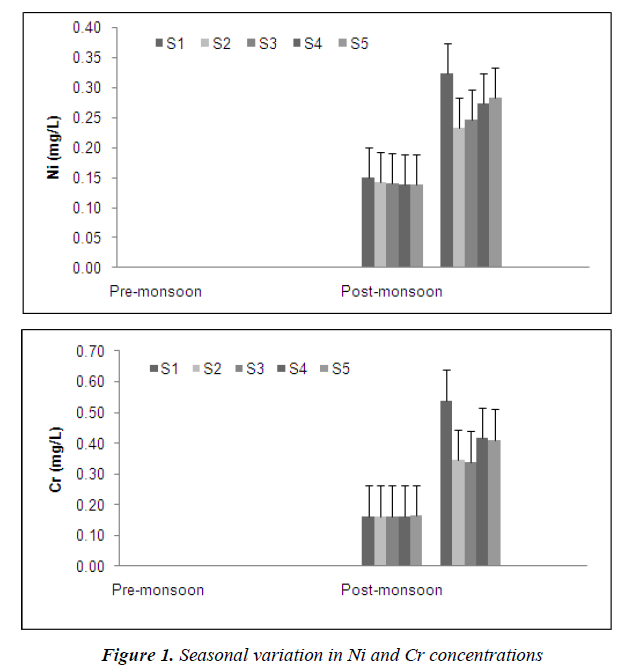
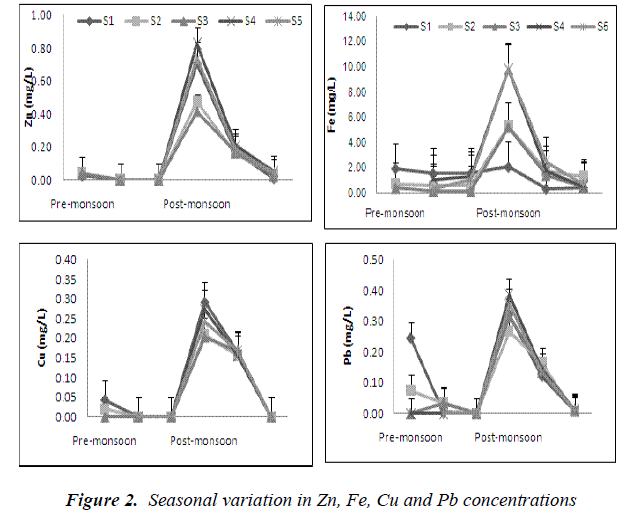
The temporal patterns of Ni and Cr showed similarity (Figure 1), both increased just after the monsoon season and reduced to being undetected by June the following year. Similar temporal patterns were also observed for Cu, Fe, Pb and Zn at the various forested headwater streams (Figure 2). The presence of ocher-colored water in Sungai Tahan (S3) and further upstream indicates that Fe is in the dissolved forms in this stream. Throughout the study, concentrations of iron were the highest in all streams. It can be concluded that higher heavy metals concentrations are exhibited in the water subsequent to the monsoon season. Similar findings were reported by He et al. [23]. Since this area is free from anthropogenic inputs, the only likely source of metals in the water is from the natural geology of the area. This pattern is typical for this type of forested area [26] and other studies have also reported increased concentrations of metals during periods of higher discharge [27, 28]. During periods of low volume, dissolved metals tend to become bound to sediments. With increase in water turbulence, sediments are resuspended and the metals become dissolved in the water again.
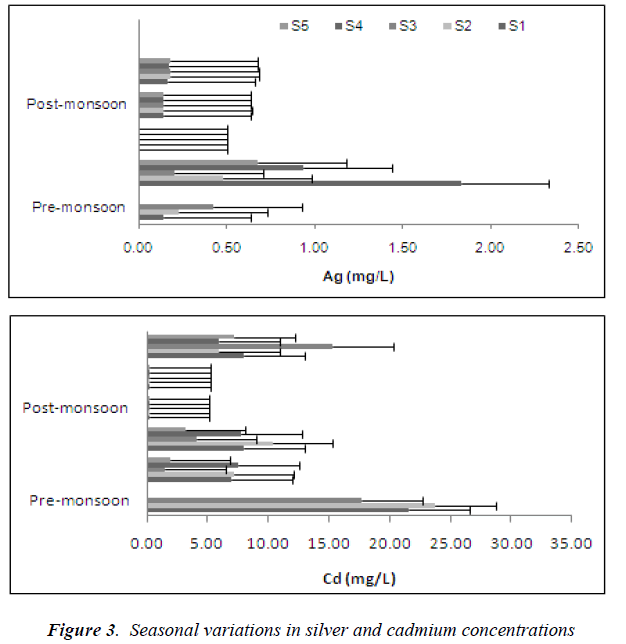
However, concentrations of silver and cadmium showed an opposite pattern compared to all the other metals. In this study, the seasonal pattern of Ag and Cd in the forested area were characterized by a decrease after the monsoon season and a gradual increase as the water flow reduced (Figure 3). Cadmium is very soluble and can be leached especially from sandy soils [29]. During the high volume/flood period concentrations of Cd and Ag are diluted, hence the reduced concentrations.
In the study area, formations of Triassic rocks dominate the western section of the Park, including the valley of the Tembeling up to Kuala Tahan and the entire Tahan valley. Cretaceous-Jurassic sedimentary rocks exist between the Kuala Keniam and Kuala Tahan. Both these formations consist of mainly thick cross-bedded sandstone deposits with subordinate shale and conglomerates and scattered limestone outcrops and caves [30]. Because sampling was not done during the wet spell when the streams were overflowing, the variability between the pre-monsoon and post-monsoon flood period could not be assessed. Water quality upstream was generally good except for those tributaries having heavy erosion and siltation problems.
Metal concentrations compared against the Interim National Water Quality Standards (INWQS) [25] showed copper, chromium and zinc falling under Class I and were at natural concentrations, with nickel at Class III and lead at Class IV. Exceedances of the INWQS occurred for silver, cadmium and iron; with iron being the most common and widespread contaminant. The water bodies in Taman Negara sampling stations that contained high mineral and metal content are natural and sourced from mineral- rich strata. According to our results, the water volume is of importance for trace metal concentrations in stream water.
Maintaining and protecting recreational water quality are important public health and resource management issue. Further studies on how variations in landscape properties affect surface water chemistry are needed to evaluate the temporal and spatial variation of trace metals. In conclusion, our study highlights the importance of including a landscape perspective when studying stream water chemistry. The chemical contaminants in any recreational water environment should be assessed on a local basis. The relevant authorities which include the local councils, health authorities, environmental agencies, policy makers and recreational water managers, have a responsibility to educate the public about hazards related to the recreational water quality.
Acknowledgments
This study is funded by the Fundamental Research Grant Scheme (FRGS) from the Ministry of Higher Education, Malaysia. The authors are grateful to the Director General, Department of Wildlife and National Parks, Peninsular Malaysia and the Superintendent, Pahang National Park for permission to carry out this project in the National Park.
References
- WHO. Aluminum. In Guidelines for Drinking-Water Quality, Second Edition,. Geneva: World Health Organization 1998; pp. 3-13.
- Klaassen CD, Watkins JB. Casarett, Doull’s essentials of toxicology. New-York: McGraw-Hill 2003.
- Basel Action Network. Exporting Harm: the High Tech Thrashing of Asia. 2002.
- Al-Saleh I, Al-Enazi S, Shinwari N. Assessment of lead in cosmetic products. Regulatory Toxicol Pharmacol 2009; 54: 105-113.
- Bush AI, Curtain CC. Twenty years of metalloneurobiology: where to now? Eur Biophys J Biophys Lett 2008; 37: 241-245.
- Lippmann M, Ito K, Hwang JS, et al. Cardiovascular effects of nickel in ambient air. Environ. Health Perspect. 2006; 114: 1662-1669.
- Kapoor SC, Van Rossum GDV. Effects of Pb2+ added in vitro on Ca2+ movements in isolated mitochondria and slices of rat kidney cortex. Biochem Pharmacol 1984; 33:1771-1778.
- Naughton DP, Nepusz T, Petroczi A. Metal ions affecting the gastrointestinal system including the liver. Met Ions Life Sci. 2011; 8: 107-132.
- Chuang HY, Tsai SY, Chao KY et al. The Influence of Milk Intake on The Lead Toxicity to The Sensory Nervous System in Lead Workers. NeuroToxicol 2004; 25: 941-949.
- Mahaffey KR, Capar SG, Gladen BC et al. Concurrent exposure to lead, cadmium, and arsenic. Effects on toxicity and tissue metal concentrations in the rat. J Lab Clin Med. 1981; 98: 463-481.
- Tong STY, Lam KC. Home sweet home? A case study of household dust contamination in Hong Kong. Sci Total Environ 2000; 256: 115-123.
- Ellenhorn MJ, Barceloux DG. Medical Toxicology. Diagnosis and Treatment of human poisoning. New York: Elsevier 1998.
- Acosta JA, Cano AF, Arocena JM, et al. Distribution of metals in soil particle size fractions and its implication to risk assessment of playground in Murcia City (Spain). Geoderma 2009; 149: 101-109.
- Brick CM, Moore JN. Diel variation of trace metals in the Upper Clark Fork River, Montana. Environ. Sci. Technol 1996; 30: 1953-1960.
- APHA. Standard Methods For The Examination Of Water And Wastewater, 20th Ed. American Public Health Association, Washington 1998.
- ICP-MS for the Analysis of Metals on Membrane Filters, Am Indust Hygine Assoc J 1987; 48: 977- 979.
- DWNP. Annual Report 2007. Department of Wildlife and National Parks, Putrajaya, Malaysia.
- Vega M, Pardo R, Barrado E, et al. Assessment of seasonal and polluting effects on the quality of river water by exploratory data analysis. Water Res 1998; 32: 3581-3592.
- Macklin MG. In: Floodplain Processes. Wiley, Chichester 1996; pp 441-460.
- Klimek K. In: Fluvial Processes and Environmental Change. Chichester 1999; pp 329-343.
- Webb BW, Walling DE. The magnitude and frequency characteristics of fluvial transport in a Devon drainage basin and some geomorphological implications. Catena 1982; 9: 9-23.
- Heininger P, Pelzer J, Claus E, et al. Contamination and toxicity trends for sediments-case of the Elbe River. Water Sci. Technol. 1998; 37: 95-102.
- He M, Wang Z, Tang H. Spatial and temporal patterns of acidity and heavy metals in predicting the potential for ecological impact on the Le An River polluted by acid mine drainage. Sci Total Environ. 1997; 206: 67- 77.
- Singh KP, Malik A, Sinha S. Water quality assessment and apportionment of pollution sources of Gomti river (India) using multivariate statistical techniques - a case study. Analytica Chimica Acta. 2006; 538: 355 - 374.
- DOE. Water Quality Criteria and Standard for Malaysia. Department of Environment, Ministry of Science and Technology, Malaysia. 1995.
- Cory N, Buffam I, Laudon H, et al. Landscape control of stream water aluminum in a boreal catchment during spring flood. Environ. Sci. Technol. 2006; 40: 349-350.
- Shafer MM, Overdier JT, Hurley JP, et al. The influence of dissolved orgaAnic carbon, suspended particulates, and hydrology on the concentration, partitioning and variability of trace metals in two contrasting Wisconsin watersheds (USA). Chem Geol 1997; 136: 71- 97.
- Rember RD, Trefry JH. Increased concentrations of dissolved trace metals and organic carbon during snowmelt in rivers of the Alaskan arctic. Geochim Cosmochim Acta 2004; 68: 477-489.
- Beyer NW, Cromartie EJ. A survey of Pb, Cu, Zn, Cd, Cr, As and Se in earthworms and soil from diverse sites. Environ Monitor Asses 1987; 8: 27-36.
- DWNP. Taman Negara Master Plan. Department of Wildlife and National Parks, Ministry of Science Technology and the Environment, Malaysia 1987.