Research Article - Biomedical Research (2017) Volume 28, Issue 7
Effect on various organs in TNBS-induced ulcerative colitis
Chen-yang Zhi1, Zhe Lv2, Ning Kong2, Yan Chen2, Zhe-hui Wang3* and Hai-cheng Gao1*
1Department of Clinical Pharmacy, School of Pharmaceutical Sciences, Jilin University, Changchun, PR China
2Changchun University of Traditional Chinese Medicine, Changchun, Jilin, PR China
3Department of Surgery, China-Japan Union Hospital of Jilin University, Changchun, PR China
- *Corresponding Authors:
- Zhe-hui Wang
Department of Surgery
China-Japan Union Hospital of Jilin University, PR China
- Hai-cheng Gao
Department of Clinical Pharmacy, School of Pharmaceutical Sciences
Jilin University, PR China
Accepted date: December 21, 2016
Abstract
Purpose: In this study, the effect on heart, kidneys and liver organs in TNBS-induced Ulcerative colitis was investigated.
Method: Twenty mice were randomly divided into two groups, which included normal group and model group. The model of chronic colitis was induced by 50% TNBS. The model of chronic colitis was verified by body weight, H&E staining. Heart, kidneys and liver organs were tested by H&E staining and Relative weight of heart and spleen.
Results: Colon of mice appeared obviously pathological changes by injection of 50% TNBS. In addition, mortality of chronic colitis was 50%. Mortality with 70% and 55% TNBS-induced colitis was 100%. Meanwhile, heart, kidneys and liver organs also occurred pathological changes.
Conclusion: 50% TNBS could induce successfully the model of Ulcerative colitis. At the same time, 50% TNBS can also induce heart, kidneys and liver damage. In addition, these results indicate that the occurrence of chronic colitis may induce heart, liver, kidney damage.
Keywords
Chronic colitis, TNBS, Various organs, Mice.
Introduction
Ulcerative colitis (UC) is chronic inflammatory bowel diseases (IBD), which affects the colon and is morphologically characterized by inflammation, epithelial damage and crypt erosions/ulcerations [1]. UC have evil change tendency which poses a serious threat to the patient [2]. It is one of the most serious diseases in the gastrointestinal tract crime [3]. Its incidence is the highest in Europe and North America [4]. In addition, UC has a rising trend in Asia and other regions [5]. At present, to create a model of UC by exogenous chemical stimulation-induced intestinal mucosal immune response is more common used methods. According to reports, 2,4,6- trinitrobenzenesulfonicacid (TNBS) was used to induce-UC model [6]. In that way, when TNBS cause damage to colon organ, which could cause heart, kidneys and liver organs damage. In this article, we investigated the effect on various organs in TNBS-induced Ulcerative colitis. The colitis was causing damage to other organs, which can provide a useful clinical basis.
Materials and Methods
Materials
Weigh in mice was 17 ± 2.0 g, which was purchased from the military academy of medical sciences animal centre. TNBS was purchased from Sigma Company. Before using, the TNBS was formulated as 50%, 55% and 70%.
Methods
Twenty mice were randomly divided into two groups, normal group (10) and model group (10). After the experimental animals were fasted for 24 h, TNBS module slow injection 21% TNBS, dose of 100 mg/kg body weight.
HE staining
Thin slices of colon, heart, spleen and liver tissue for all cases received in our histopathology unit were fixed in 4% formaldehyde solution (pH 7.0) for periods not exceeding 24 h. The tissues were processed routinely for paraffin embedding, and 5 μm-thick sections were cut and placed on glass slides. Tissue samples were stained with hematoxylin and eosin. A total of 20 sections in ablation region, each section randomly selected five fields at x400 magnification and photographed counting the number of cells.
Results
Mortality in mice
Normal mice responded flexibly and had normal food intake. Model in BALB/c mice induced by 50% TNBS, which appeared bloody stools, loose stools, less moving, dull hair, body curled up and reduced food intake. The above symptoms were aggravated after 3 days, and then gradually stabilized. Mouse mortality was 50% in injection 50% TNBS. However, Mouse mortality was 100% injection 55% and 70% TNBS (Table 1).
| Group | N | Death in mice | mortality |
|---|---|---|---|
| Control | 10 | 0 | 0% |
| 50% Model | 10 | 5 | 50% |
| 55% Model | 5 | 5 | 100% |
| 70% Model | 5 | 5 | 100% |
Table 1: The mortality of mice on injection of TNBS.
Changes in body weight
In this experiment, there was significant difference between the first weeks and the second weeks in body weight (Table 2). Body weight in control mice was increased in the second weeks. However, Body weight in model mice was reduced in the second weeks.
| Group | N | The first week (weight) |
The second week (weight) |
|---|---|---|---|
| Control | 10 | 15.2 ± 0.4 | 17.07 ± 0.6 |
| Model | 10 | 16.9 ± 0.3 | 15.8 ± 0.2 |
Table 2: The change of weight.
Relative weight of heart, kidney and spleen
In this experiment, there was significant difference in weight of heart, spleen and kidney (Table 3). Weight of heart, spleen and kidney in control mice reduced by using 50% TNBS.
| Group | N | Heart (weight) | Spleen (weight) | Kidney (weight) |
|---|---|---|---|---|
| Control | 4 | 0.090 ± 0.007 | 0.080 ± 0.005 | 0.184 ± 0.002 |
| Model | 4 | 0.087 ± 0.005 | 0.070 ± 0.010 | 0.178 ± 0.003 |
Table 3: The changes of weight on heart, kidney and spleen.
Animal figure
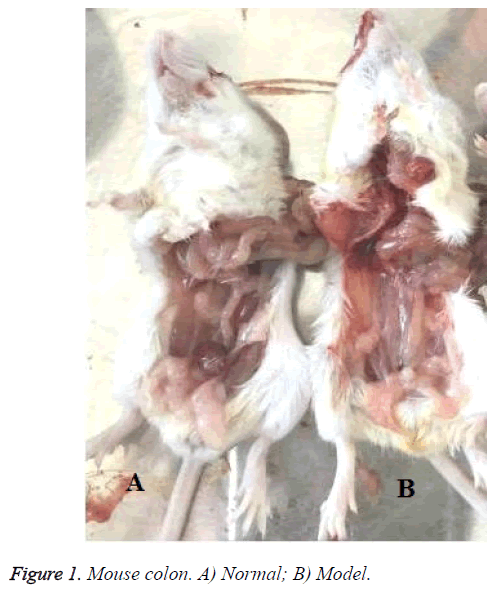
There was significant difference between Figures 1A and 1B in the colon. It was red in the colon Figure 1B. However, body weight in model mice was reduced in the second weeks.
Colon
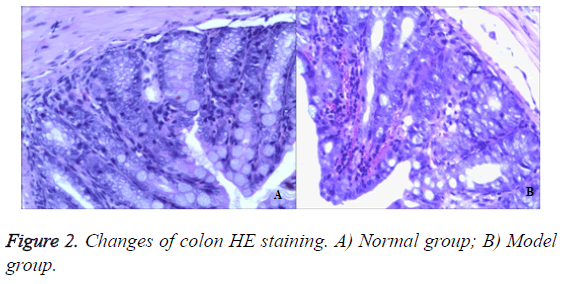
In the control group, there was no abnormality seen in the mucosa and ulceration or hyperplasia in the surface. Intestinal peristalsis was normal and moderate engorged degree. In the model group, there was interstitial lymphocyte in injection 50% TNBS. Colonic mucosa could see chronic inflammation. Glandular epithelial cells saw neutrophils and lymphocytes, plasma cells (Epithelial cells saw neutrophils, which was characterized by acute inflammation and injury. Plasma cells and lymphocytes were chronic inflammatory features). In addition, the blood vessel congestion could be seen in figure by injection 50% TNBS (Figure 2).
Heart
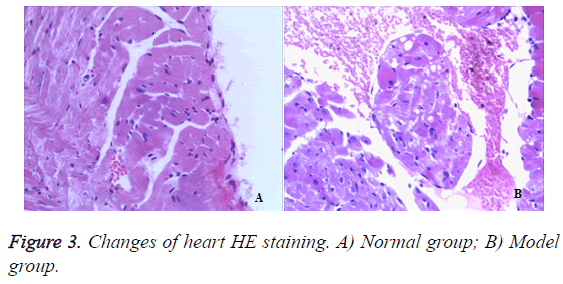
In the normal mice, myocardial cells were neatly arranged, sarcomeres were arranged regularly, subcellular structure, such as mitochondrias, were rich, clustered and in normal morphology (Figures 3A and 3B). However, 50% TNBS induced ulcerative colitis which induced cardiomyocytes degeneration and focal inflammatory cell infiltration as well as interstitial connective tissues hyperplasia.
Kidney
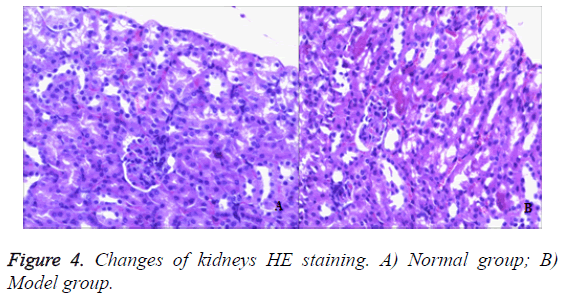
In the control group, no obvious pathological changes were observed in the kidneys of mice. In the model group, mouse tubular epithelial cells had shedding and accompanied with edema. There was focal inflammatory cell infiltration, glomerular mesangial cell proliferation. Most of the glomerular basement membrane appeared thickening. It showed that the model group had significant renal lesion and the model was established successfully by injection 50% TNBS induced ulcerative colitis (Figure 4).
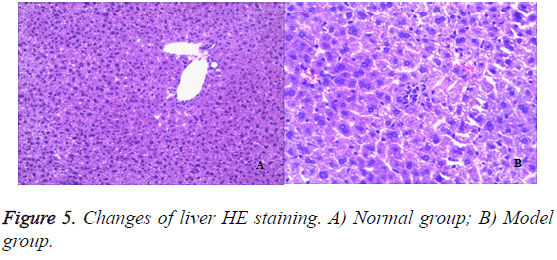
Liver
To assess the effect of the different liver architecture, paraffin section prepared from the hepatic tissues of the different groups were stained with hematoxylin/eosin and examined. From the histological point of view, liver from rats in the healthy control group showed a normal liver lobular architecture and hepatocyte structure (Figures 5A and 5B). In contrast, 50% TNBS-induced ulcerative colitis resulted in histopathological lesions and extensive hepatocellular damage, as represented by the presence of portal inflammation, fatty change and venous congestion.
Discussion
Inflammatory bowel disease is quite common in Western countries [7]. Although the incidence in Asian countries is lower than in the West [8]. But it also showed an upward trend in recent 10 years. The domestic has been that the disease is rare and not enough attention in the past [9]. But according to reports have more than 2 million people in recent years, and new cases are constantly being discovered. IBD etiology and exact pathogenesis is unclear, the treatment is quite difficult. The ideal animal model is an indispensable tool for the study of pathology, etiology and related therapeutic drugs [10]. At present, TNBS-induced rat intestinal injury model has been widely used in the pathogenesis of various intestinal diseases [11]. TNBS is a weak organic acid, cannot induce immune response in the body alone [12]. In combination with the organization of protein and other high molecular substances can induce immune response. So when modelling the need for ethanol as intestinal mucosal barrier damage agent, making the TNBS and intestinal mucosal keratin combined into the whole antigen, thus stimulating the local immune response. TNBSinduced intestinal injury, which is typical of the occurrence of transmural necrosis on the colon and extensive inflammatory cell infiltration, and accompanied with a sustained decline in body weight [13]. In this study, mice were enemaed with 50% TNBS. From the experimental results showed that body weight of mice was continued to decline in 50% TNBS-induced model. Histopathological observation after modelling showed that the intestinal wall appeared necrosis and deep muscle layer, infiltration of inflammatory cells. These were typical inflammatory bowel disease characteristics, indicated that the method of preparation on inflammatory bowel disease model was successful. In addition, the heart, liver and kidney were also pathological changes. These results suggested that 50% TNBS-induced chronic colitis, which might lead to other organ damage. This model was of great significance for the future development of drugs.
References
- Randall K, Henderson N, Reens J, Eckersley S, Nyström AC, South MC, Balen- dran CA, Böttcher G, Hughes G, Price SA. Claudin-2 Expression Levels in Ulcerative Colitis: Development and Validation of an In-Situ Hybridisation Assay forrapeutic Studies. PLoSOne 2016; 11: e0162076.
- Anaraki F, Vafaie M, Behboo R, Maghsoodi N, Esmaeilpour S, Safaee A. Clinicalprofile and post-operative lifestyle changes in cancer and non-cancer patients withostomy. Gastroenterol Hepatol Bed Bench 2012; 5: S26-30.
- Kedia S, Ahuja V, Makharia GK. Golimumab for moderately to severely active ulcerative colitis. Expert Rev Clin Pharmacol 2016; 9: 1273-1282.
- Juyal G, Amre D, Midha V, Sood A, Seidman E, Thelma BK. Evidence of allelic heterogeneity for associationsbetween the NOD2/CARD15 gene and ulcerative colitis among North Indians. Aliment Pharmacol Ther 2007; 26: 1325-1332.
- Tan YM, Goh KL. Ulcerative colitis in a multiracial Asian country: racial differences and clinical presentation among Malaysian patients. World J Gastroenterol 2005; 11: 5859-5862.
- Liu R, Li Y, Zhang B. The effects of konjac oligosaccharide on TNBS-induced colitis in rats. Int Immunopharmacol 2016; 40: 385-391.
- Gonciarz M, Szkudlapski D, Mularczyk A, Smagacz J. Hygienic hypothesis in immun-opathogenesis of inflammatory bowel disease. Pol Merkur Lekarski 2016; 40: 384-387.
- Coolsen M, Leedham SJ, Guy RJ. Non-steroidal anti-inflammatory drug-induced diaphragm disease: an uncommon cause of small bowel obstruction. Ann R Coll Surg Engl 2016; 98: e189-e191.
- Ng SC, Leung WK, Shi HY, Li MK, Leung CM, Ng CK, Lo FH, Hui YT, Tsang SW, Chan YK, Loo CK, Chan KH, Hui AJ, Chow WH, Harbord M, Ching JY, Lee M, Chan V, Tang W, Hung IF, Ho J, Lao WC, Wong MT, Sze SF, Shan EH, Lam BC, Tong RW, Mak LY, Wong SH, Wu JC, Chan FK, Sung JJ. Epidemiology of Inflammatory Bowel Disease from 1981 to 2014: Results from a Territory-Wide Population-Based Registry in Hong Kong. Inflamm Bowel Dis 2016; 22: 1954-1960.
- Bito LZ, Kaufman PL, DeRousseau CJ, Koretz J. Presbyopia: an animal Modeland experimental approaches for the study of the mechanism of accomm- odation and ocular ageing. Eye (Lond) 1987; 1: 222-230.
- Szalai Z, Szász A, Nagy I, Puskás LG, Kupai K, Király A, Berkó AM, Pósa A, Strifler G, Baráth Z, Nagy LI, Szabó R, Pávó I, Murlasits Z, Gyöngyösi M, Varga C. Anti-inflammatory effect of recreational exercise in TNBS-induced colitis in rats: role of NOS/HO/MPO system. Oxid Med Cell Longev 2014; 2014: 925-981.
- Lipinski T, Luu T, Kitov PI, Szpacenko A, Bundle DR. A structurally diversified linker enhances the immune response to a small carbohydrate hapten. Glycoconj J 2011; 28: 149-164.
- Zeeh JM, Zorlu, Riley NE, Hoffmann P, Ruwe M, Goebell H, Gerken G, Dignass AU. Mycophenolate mofetil reduces tissue damageand inflammation in an experimental model of colitis in rats. Scand J Gastroen-terol 2001; 36: 66-70.