Research Article - Biomedical Research (2017) Volume 28, Issue 11
Diagnostic value of the pachymetry parameters measured by RTVueOCT for keratoconus
Xiaobo Zhu1, Yuehua Zhou2* and Jing Zhang21Department of Ophthalmology, China Meitan General Hospital, Beijing, PR China
2Department of Ophthalmology, Beijing Tongren Hospital, Capital Medical University, Beijing, PR China
- *Corresponding Author:
- Yuehua Zhou
Department of Ophthalmology
Beijing Tongren Hospital
Capital Medical University
PR China
Accepted on April 10, 2017
Abstract
Background: This study aimed to investigate the diagnostic value of pachymetry parameters measured by RTVueOCT for keratoconus.
Methods: 74 patients with 118 eyes diagnosed with keratoconus were enrolled in keratoconus group and 60 patients with normal 120 eyes as health controls. Pachymetry parameters were measured. The major indicators were tested: 1) S-I value: difference between the mean superior and the mean inferior corneal thickness; 2) SN- IT value: difference between the mean superonasal and the mean inferotemporal corneal thickness; 3) MIN: values of minimum corneal thickness; 4) Min-Med: difference between minimum and median corneal thickness; 5) Min-Max: difference between minimum and maximum corneal thickness; 6) the thinnest X-location; 7) the thinnest Y-location. The seven parameters were measured within 5 mm of optical center.
Results: The thinnest corneal thickness in keratoconus group and control group was 423.5 ± 58.72 um and 539.15 ± 25.64 um respectively. There were significance differences of SN-IT, S-I, Min, Min-Med and Min-Max between keratoconus group and control group. AROC of IT-SN, I-S, Min, Min-Med and Min-Max were 0.869, 0.814, 0.991, 0.996 and 0.996 respectively. Cut-off values of SN-IT, S-I, Min, Min- Median and Min-Max in distinguishing control group and keratoconus group were -28.5 μm, -33.5 μm, 497.0 μm, -22.5 μm and -53.0 μm respectively; sensitivities were 0.839, 0.627, 0.946, 0.966 and 0.975; and specificities were 0.842, 0.917, 0.967, 1.000 and 0.958 respectively.
Conclusions: The pachymetry parameters measured by RTVueOCT can be used in diagnosis of keratoconus effectively.
Keywords
RTVueOCT, Pachymetry parameters, Corneal topography, Keratoconus.
Introduction
Keratoconus is a noninflamed, chronic, progressive dilatated disease occurring in bilateral local cornea. The major characteristics of keratoconus are that corneal stroma in central and paracentral zone become thinner and conical macula. Moreover, the arc of corneal changes and leads to the formation of irregular astigmatism, myopia and scar. Generally, keratoconus may occur with monocular, and progress to binocular involvement. Clinical keratoconus is often accompanied with significant clinical manifestations and impaired vision [1], while the sub-clinical keratoconus has normal vision with slight or without any clinical manifestation, making it hard to be identified. Moreover, the morbidity of patients who undergo refractive surgery is about 5.7% [2,3]. For those who are not diagnosed with abnormal corneal dilatation before laser-assisted in situ keratomileusis (LASIK), their corneal dilatation may be promoted faster after operation, which has great impact on prognosis. Therefore, it is crucial to know clinical characteristics of sub-clinical keratoconus and general diagnostic criteria. Besides, accurately screening keratoconus before operation also plays an important role in completing corneal refractive surgery and preventing keratoconus effectively. Corneal topography based on placido plate principle is a standard method for identifying abnormal dilatation [4-10]. Quantitative index of corneal topography helps to diagnose sub-clinical keratoconus [11-14]. However, corneal topography examination cannot identify all sub-clinical keratoconus. Patients having normal morphology without any other risk factor may still progress to corneal dilatation after LASIK [15]. Furthermore, it is difficult to identify corneal deformation, non-pathological irregular astigmatism, epithelial deposition or scar, and uneven tear film caused by keratoconus and contact lens only by using morphology [16-18]. Progressive thinning of cornea is a typical pathological change and corneal thickness is also a clinical indicator of keratoconus. In clinical practice, it was found that corneal thickness might become thinner in the unaffected eye of keratoconus patient, even if corneal topography did not show any change. Therefore, precise pachymetry may be helpful in improving the diagnosis of sub-clinical keratoconus.
Corneal pachymetric features have been described since the optical pachymetry was available. Horizontal pachymetry profile in keratoconus has been described by Mandell and Polse [19] and corneal thickness spatial profile by using Scheimpflug technology [20]. Zeiss company in German first introduced the Visante OCT, designed for the anterior segment of the eye. The eidelberg company in German launched the anterior segment SL-OCT, combined with slit lamp examination technology [21]. Optical Coherence Tomography (OCT) is a novel optical diagnostic technique and can be used as non-contact and non-invasive coherence tomography in microscopic living tissues [22]. Fourier optical coherence tomography (RTVueOCT) (RTVuel00®, Optovue, America) is a new generation OCT based on Fourier technique with a minimum resolution of 5 μm. In addition, it has a rapid scanning speed and 10214 times of accurate scan can be completed within 0.04 s. Accurate pachymetry map and parameters for thickness can be obtained by using RTVueOCT for anterior segment check with a short scanning time and good comfort, thus the pain can be relieved during examination. Meanwhile, OCT resolution is higher than other imaging modes, which is able to draw normal and post-operational pachymetry map and presents image of non-transparent cornea [23]. OCT pachymetry has increasingly become the optimal choice to measure the corneal thickness.
At present, computer-assisted corneal topography detection system is the major technology used in diagnosis of subclinical keratoconus, which can not only reveal corneal surface shape through levels but also offer a plenty of measured parameters, such as corneal mean diopter, average corneal diopter with different diameter-circumference, surface regularity index and potential visual acuity. Moreover, TMS system is equipped with keratoconus diagnostics. The fact that corneal topography can be used for sub-clinical keratoconus has gotten clinical proven [24]. Smolek/Klyce index is indicated as keratoconus severity index (KSI). KSI is obtained by combinatorial analysis with backbone network model and tree analysis. KSI represents the severity of keratoconus which is linearly correlated with keratoconus course. It may be keratoconus if KSI>0.15; however, the keratoconus can be identified if KSI>0.3.
The patients were divided into keratoconus and control group in this study. In addition, pachymetry parameters were measured by RTVueOCT to investigate the change pattern of pachymetry parameters in patients with keratoconus. Then reference values of RTVueOCT parameters for diagnosis of keratoconus would be calculated in order to improve keratoconus screening. In this way, the rate of refractive surgery for the keratoconus patients without any clinical manifestation can be reduced effectively.
Materials and Methods
Ethics approval and consent to participate
This study was approved by the Ethic Committee of Beijing Tongren Hospital (2012-05-1J), and complying with the Helsinki Declaration. All subjects gave their consent to participate in this study. Consent to publish has been obtained from the participant to report individual patient data.
Patients
One hundred and thirty-four patients in Tongren Hospital from October 2012 to October 2015 were enrolled, and then they were divided to keratoconus group (74 patients with 118 eyes) and health control group (60 patients with 120 eyes). Objectives in keratoconus group and control group were all come from the refractive outpatient clinic, who wanted to take off their glasses. Patients with keratoconus had refractive error. Therefore, they came to the refractive outpatient clinic. The objectives in control group were all patients with refractive errors.
Inclusion criteria for keratoconus group were as follows: 1) the patients who had a history of myopic and astigmatism; 2) Corrected Visual Acuity (CVA)<1.0; 3) abnormal alter of corneal topography and KSI>30%; 4) there was at least one positive reaction for the following manifestations in slit-lamp examination: a) corneal stroma is progressive thinning; b) corneal shows as cone shaped with forward exhibition like Munson sign; c) Fleischer annulus; d) Vogt line; e) conic node epithelio-scar or scar under the epithelium. Inclusion criteria for control group were as follows: 1) patients with myopia who came to our hospital for LASIK; 2) young adults aging from 18 to 40 years; 3) the patients who had normal corneal topography; 4) no other eye diseases.
Exclusion criteria were as follows: 1) the patients who had the history of eye surgery, trauma and strabismus amblyopia diseases; 2) the patients who have histories of other eye disease: such as keratoconus, glaucoma and retinal disease, etc.; 3) the patients who have eye diseases such as serious dry eye, eyelid calculi, conjunctivitis and pterygium which may influence imaging; 4) corneal lens wearers have deficient time for getting rid of glasses (hard lens>four weeks or soft lens>one week); 5) the patients who have poor fixed views and cannot cooperate during examination; 6) the patients who had secondary keratectasia after operation, or had secondary keratectasia caused by other reasons.
Pachymetry parameters measured by RTVueOCT (all parameters were within 5 mm of optical center)
Pachymetry parameters: 1) S-I: differential value between the mean superior corneal thickness and the mean inferior corneal thickness; 2) SN-IT: differential value between the mean superonasal and the mean inferotemporal corneal thickness; 3) MIN: values of minimum corneal thickness; 4) MIN-MED: differential value between minimum and median corneal thickness; 5) MIN-MAX: differential value between minimum and maximum corneal thickness; 6) the thinnest Xlocation; 7) the thinnest Y-location.
Inspection equipment and methods
RTVueOCT (software version: #4, 0, 5, 100) and Tomey corneal topography (software version: 4.2D) were used in this study. All patients experienced successive examinations: far and near vision check, history taking, examination with slit lamp microscope, non-contact intraocular pressure measurement, manifest refraction, keratometer measurement, Tomey topographic screening, cycloplegic retinoscopy, binocular indirect ophthalmoscope for fundus examination, OCT pachymetry map and ultrasonic A pachymeter. Corneal curvature parameters obtained by Tomey topographic screening include Ks, Kf, Avek, Mink, Cyl, SRI, SAI, KCI, KSI and so on. Parameters obtained by RTVueOCT pachymetry map include SN-IT, S-I, MIN, MIN-MED, MINMAX, X-location and Y-location. All operations were executed by the same well-trained doctors. Corneal topography and OCT were checked three times for each eye and the results with good repeatability were retained. The working principle of Fourier optical coherence tomography (RTVueOCT) was measuring values after operation by taking corneal vertex as adjust center.
Statistical analysis
Data were analysed by using SPSS 13.0 statistical software. The mean ± SD of each pachymetric parameter was calculated for both the keratoconus and normal groups. Continuous variables with normal distributions were compared by t test, whereas those with non-normal distributions were compared by the Mann-Whitney U rank sum test. P<0.05 was considered statistically significant. After that, pachymetry parameters with significant difference were drawn into ROC curve to determine the sensitivity and specificity in distinguishing patients in control group and in keratoconus group.
Results
This study included 57 male and 17 female in the keratoconus group. The mean age was 32.13 years old (range: 14-40 years old). There were 26 male and 34 female in the control group. The mean age was 29.4 years old (range: 16-43 years old).
Mean corneal thickness values of the thinnest location in keratoconus group and control group were 423.5 ± 58.72 μm and 539.15 ± 25.64 μm respectively, which indicated that the thickness in keratoconus group is thinner than that in control group. In addition, the average SN-IT, S-I, MIN-MED and MIN- MAX of both keratoconus group and control group were 51.98 ± 31.07 μm and 20.47 ± 9.71 μm, 45.41 ± 31.34 μm and 19.42 ± 10.89 μm, -57.65 ± 30.45 μm and -12.68 ± 3.09 μm, -117.03 ± 51.56 μm and -36.93 ± 8.77 μm, respectively. After comparing absolute value of five parameters (SN-IT, S-I, MIN, MIN-MED and MIN-MAX) in both groups, it could be observed that all values in keratoconus group was higher than those in control group. Besides, the differences of the abovementioned five parameters in both groups were significant (P<0.05). When comparing X-location and Ylocation value in both groups, the differences were not significant (P>0.05). When comparing values of SN-IT, S-I, MIN, MIN-MED and MIN-MAX between the two groups, the differences were significant; however, the differences of XLocation and Y-Location values were not significant (Table 1).
| Keratoconus group | Control group | P value | |||
|---|---|---|---|---|---|
| Mean | Standard deviation | Mean | Standard deviation | ||
| SN-IT | 51.9831 | 31.06706 | 20.4667 | 9.71262 | 0 |
| S-I | 45.4068 | 31.34103 | 19.4167 | 10.88564 | 0 |
| MIN | 423.5 | 65.58437 | 539.15 | 25.63501 | 0 |
| Min-Med | -57.6525 | 30.44608 | -12.675 | 3.08469 | 0.000a |
| Min-Max | -117.025 | 51.55753 | -36.925 | 8.76793 | 0.000a |
| X-Location | -0.0467 | 0.600152 | -0.0751 | 0.600108 | 0.618 |
| Y-Location | -0.6401 | 0.394347 | -0.58005 | 0.459309 | 0.184a |
Note: a: this value is result by t testing; all p values without marking a were results of Mann-Whitney U test; all the pachymetry parameters were within 5 mm of optical center.
Table 1. Comparison of pachymetry parameters in clinical keratoconus group and control group.
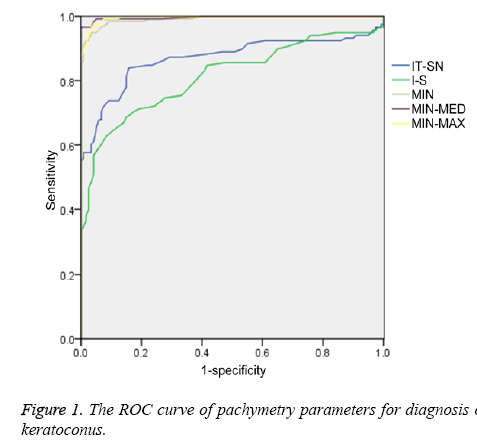
ROC curve analysis was made for those pachymetry parameters with significant difference. When distinguishing keratoconus group and control group, area under ROC curve (AROC) of IT-SN, I-S, MIN, MIN-MED and MIN-MAX were 0.869, 0.814, 0.991, 0.996 and 0.996 respectively (Figure 1). Moreover, AROC of MIN-MED, MIN-MAX and MIN declined in turn and all of them above 0.9 with higher accuracy. The cut-off values of IT-SN, I-S, MIN, MIN-MED and MIN-MAX in discriminating keratoconus group and control group were -28.5 μm, -33.5 μm, 497.0 μm, -22.5 μm and -53.0 μm. Sensitivities of IT-SN, I-S, MIN, MIN-MED and MIN-MAX in differentiating keratoconus group and control group were 0.839, 0.627, 0.946, 0.966 and 0.975 respectively. Specificities of IT-SN, I-S, MIN, MIN-MED and MIN-MAX in differentiating keratoconus group and control group were 0.842, 0.917, 0.967, 1.000 and 0.958 respectively (Tables 2 and 3). These results showed that IT-SN, I-S, MIN, MIN-MED and MIN-MAX were all valid data in distinguishing keratoconus group and control group. MIN-MED and MIN-MAX had the maximum diagnostic capability of 0.996. Besides, MIN also had a high accuracy.
| Parameters of corneal thickness | AROC | Standard deviation | 95% CI |
|---|---|---|---|
| IT-SN | 0.869 | 0.025 | 0.820-0.919 |
| I-S | 0.814 | 0.027 | 0.760-0.868 |
| Min | 0.991 | 0.004 | 0.983-0.999 |
| Min-Med | 0.996 | 0.003 | 0.000-1.000 |
| Min-Max | 0.996 | 0.002 | 0.992-0.999 |
Note: all the pachymetry parameters were within 5 mm of optical center.
Table 2. Area under ROC curve of pachymetry parameters in keratoconus group and control group.
| Parameters of corneal thickness | Keratoconus group and control group | ||
|---|---|---|---|
| Cut-off value | Sensitivity | Specificity | |
| IT-SN | -28.5 | 0.839 | 0.842 |
| I-S | -33.5 | 0.627 | 0.917 |
| Min | 497 | 0.946 | 0.967 |
| Min-Med | -22.5 | 0.966 | 1 |
| Min-Max | -53 | 0.975 | 0.958 |
Note: all the pachymetry parameters were within 5 mm of optical center.
Table 3. SN-IT, S-I, MIN, Min-Med and Min-Max were used for distinguished keratoconus group and control group.
Discussion
Currently, corneal topography is still a conventional method for screening keratoconus before corneal refractive surgery. Corneal curvature measured by conventional placido based computer-assisted corneal analysis system is converted according to specular reflection angle of front surface which is not the real corneal height. Besides, conventional corneal topographer is operated by assuming that measured optical axis, corneal fixed point and reference surface are in the common axis. Corneal vertex is often discrepancy with optical axis and the result may have some errors accordingly. As a new technique, the operation of OCT is based on different optical scatterings of ocular tissues; moreover, it takes on twodimensional image and makes quantitative analysis by adopting interference measurement. In addition, OCT is a noncontact technique with a high resolution in contrast with other testing methods. Except biological measurement and disease study for preocular structures such as cornea, anterior chamber angle and crystalline lens, OCT can also be used for the imaging and detection of retina and optic nerve. Moreover, it can be used for dynamic observation and real-time imaging during operation. The measured values in this study were obtained after operation by taking corneal vertex as the adjust center. The OCT pachymetric measurements were accurate and highly repeatable. The Fourier-domain OCT system used in this study was capable of a speed of 26,000 axial scans per second, which is 10 more times faster than the time-domain OCT systems based on original OCT technology. The 840 nm Fourier-domain OCT system also has an axial resolution of 5 μm, which is three more times better than a conventional 1310 nm time-domain OCT system. Faster scan speed can reduce data acquisition time, minimize eye movement during the scan, and improve repeatability of pachymetric measurements [25]. High-resolution Fourier-domain OCT was capable of mapping corneal, epithelial, and stromal thicknesses. Characteristic corneal and its sub layer thickness changes in subclinical keratoconus could be detected with a very high accuracy by using PSD variables. These new diagnostic variables might be useful in the detection of early keratoconus [26].
In the research done by Yan et al. [27], keratoconus was tested by OCT pachymetry map. Diagnostic criteria were confirmed by setting the parameter range from 1% to 99% of healthy adults. The diagnostic criteria included following contents: 1) irregular pachymetry parameters: SN-IT or S-I>45 μm; 2) corneal thickness of the thinnest location<470 μm; 3) local thinning parameters Min-Max value<-100 μm. Keratoconus should be considered in-patient with a single abnormal parameter. Keratoconus or other expansion lesions are likely to be considered when two or more parameters are abnormal.
In this study, the pachymetry parameters of Chinese population measured by RTVueOCT were obtained for comparison between keratoconus patients and healthy controls. The results of corneal thickness in keratoconus group and control group measured by RTVueOCT showed that differences of IT-SN, IS, MIN, MIN-MED and MIN-MAX were significant between keratoconus patients and health controls. Furthermore, variation trend of these parameters were associated with progress of keratoconus. In other words, IT-SN, I-S, MIN, MIN-MED and MIN-MAX may change along with the progress of keratoconus. Corneal thickness of the thinnest location for healthy controls was 539.15 ± 25.64 μm, which was similar to the result (546 ± 23.7 μm) of Li et al. [27]. However, Keratoconus thickness of the thinnest location 423.5 ± 58.72 μm was lower than the result (452.6 ± 60.9 μm) of Li et al. [27].
In terms of distinguishing keratoconus group and control group by IT-SN, I-S, MIN, MIN-MED and MIN-MAX, it was found that AROC of MIN-MED and MIN-MAX is 0.966 by ROC curve, indicating that MIN-MED and MIN-MAX had highest accuracy in distinguishing between keratoconus patients and health controls. In addition, cut-off value of MIN-MED in this study was -22.5 μm, which was much lower than the result (-62.6 μm) of Li et al. [27]. Secondly, AROC of MIN was 0.991, which also showed a high accuracy, and their diagnostic capacity was similar to MIN-MED. The cut-off value of MIN was 497.0 μm which was similar to the results (491.6 μm) of Li et al. [27]. However cut-off value of MIN-MAX was -53.0 μm, which was significant different from the result (-100.0 μm) of Li et al. [27]. AROC of IT-SN was 0.869 with certain accuracy and its cut-off value was -33.5 μm, which was different from the result (-48.2 μm) of Li et al. [27]. AROC of I-S was 0.814 with certain accuracy and its cut-off value was -28.5 μm, which was similar to the result (-31.3 μm) of Li et al. [27]. All results showed that pachymetry parameters measured by RTVueOCT such as IT-SN, I-S, MIN, MIN-MED and MINMAX had high sensitivity and specificity in distinguishing keratoconus patients from health controls.
This research reveals that some pachymetry parameters measured by RTVueOCT can be used to improve diagnosis of keratoconus effectively. MIN, MIN-MED and MIN-MAX were the important parameters. MIN-MED and MIN-MAX of which AROC was 0.996 had maximal diagnostic efficiency. Thus, keratoconus could be identified by using MIN-MED, MIN-MAX and MIN values. Meanwhile, IT-SN and I-S can also be used to screen keratoconus. Besides, cut-off values of keratoconus group and control group can be calculated through these parameters, and keratoconus can be identified by using these cut-off values.
References
- Rabinowitz YS. Keratoconus. Surv Ophthalmol 1998; 42: 297-319.
- Wilson SE, Klyce SD. Screening for corneal topographic abnormalities before refractive surgery. Ophthalmology 1994; 101: 147-152.
- [No authors listed]. Keratoconus, videokeratography, and refractive surgery. Refract Corneal Surg 1992; 8: 403-407.
- Avitabile T, Franco L, Ortisi E, Castiglione F, Pulvirenti M, Torrisi B, Castiglione F, Reibaldi A. Keratoconus staging: a computer-assisted ultrabiomicroscopic method compared with videokeratographic analysis. Cornea 2004; 23: 655-660.
- Li X, Rabinowitz YS, Rasheed K, Yang H. Longitudinal study of the normal eyes in unilateral keratoconus patients. Ophthalmology 2004; 111: 440-446.
- Maguire LJ, Bourne WM. Corneal topography of early keratoconus. Am J Ophthalmol 1989; 108: 107-112.
- Rabinowitz YS, Garbus J, McDonnell PJ. Computer-assisted corneal topography in family members of patients with keratoconus. Arch Ophthalmol 1990; 108: 365-371.
- Wilson SE, Lin DT, Klyce SD. Corneal topography of keratoconus. Cornea 1991; 10: 2-8.
- Maeda N, Klyce SD, Tano Y. Detection and classification of mild irregular astigmatism in patients with good visual acuity. Surv Ophthalmol 1998; 43: 53-58.
- Ambrosio JR, Klyce SD, Smolek MK, Wilson SE. Pellucid marginal corneal degeneration. J Refract Surg 2002; 18: 86-88.
- Maeda N, Klyce SD, Smolek MK, Thompson HW. Automated keratoconus screening with corneal topography analysis. Invest Ophthalmol Vis Sci 1994; 35: 2749-2757.
- Rabinowitz YS. Videokeratographic indices to aid in screening for keratoconus. J Refract Surg 1995; 11: 371-379.
- Rabinowitz YS, Rasheed K. KISA% index: a quantitative videokeratography algorithm embodying minimal topographic criteria for diagnosing keratoconus. J Cataract Refract Surg 1999; 25: 1327-1335.
- Schwiegerling J, Greivenkamp JE. Keratoconus detection based on videokeratoscopic height data. Optom Vis Sci 1996; 73: 721-728.
- Randleman JB, Woodward M, Lynn MJ, Stulting RD. Risk assessment for ectasia after corneal refractive surgery. Ophthalmology 2008; 115: 37-50.
- Cheng HC, Lin KK, Chen YF, Hsiao CH. Pseudokeratoconus in a patient with soft contact lens-induced keratopathy: assessment with Orbscan I. J Cataract Refract Surg 2004; 30: 925-928.
- Wilson SE, Lin DT, Klyce SD, Reidy JJ, Insler MS. Topographic changes in contact lens-induced corneal warpage. Ophthalmology 1990; 97: 734-744.
- Maeda N, Klyce SD, Smolek MK. Comparison of methods for detecting keratoconus using videokeratography. Arch Ophthalmol 1995; 113: 870-874.
- Mandell RB, Polse KA. Keratoconus: spatial variation of corneal thickenss as a diagnostic test. Arch Ophthalmol 1969; 82: 182-188.
- Amano S, Honda N, Amano Y, Yamagami S, Miyai T, Samejima T, Ogata M, Miyata K. Comparison of central corneal thickness measurements by rotating Scheimpflug camera, ultrasonic pachymetry, and scanning-slit corneal topography. Ophthalmology 2006; 113: 937-941.
- Müller M, Hoerauf H, Geerling G, Pape S, Winter C. Filtering bleb evaluation with slit-lamp-adapted 1310-nm optical coherence tomography. Curr Eye Res 2006; 31: 909-915.
- Chen J, Lee L. Clinical applications and new developments of optical coherence tomography: an evidence-based review. Clin Exp Optom 2007; 90: 317-335.
- Khurana RN, Li Y, Tang M, Lai MM, Huang D. High-speed optical coherence tomography of corneal opacities. Ophthalmology 2007; 114: 1278-1285.
- Ertan A, Muftuoglu O. Keratoconus clinical findings according to different age and gender groups. Cornea 2008; 27: 1109-1113.
- Qin B, Chen S, Brass R, Li Y, Tang M, Zhang X, Wang X, Wang Q, Huang D. Keratoconus diagnosis with optical coherence tomography-based pachymetric scoring system. J Cataract Refract Surg 2013; 39: 1864-1871.
- Li Y, Chamberlain W, Tan O, Brass R, Weiss JL, Huang D. Subclinical keratoconus detection by pattern analysis of corneal and epithelial thickness maps with optical coherence tomography. J Cataract Refract Surg 2016; 42: 284-295.
- Li Y, Meisler DM, Tang M, Lu AT, Thakrar V, Reiser BJ, Huang D. Keratoconus diagnosis with optical coherence tomography pachymetry mapping. Ophthalmology 2008; 115: 2159-2166.