- Biomedical Research (2010) Volume 21, Issue 2
Daytime hormonal rhythms in practitioners of the transcendental meditation-Sidhi program
Jose R. Infantea*; Fernando Peranb; Juan I. Rayoa; Justo Serranoa; Maria L. Domíngueza; Lucia Garciaa; Carmen Durana; Roman Sancheza; Ana Roldanc.
aNuclear Medicine Service, Infanta Cristina Hospital, Avda. Elvas, s/n. E-06080, Badajoz, Spain.
bClinical Analysis. Virgen de las Nieves Hospital, Avd. Fuerzas Armadas nº2. E-18014, Granada, Spain.
cArea Health Service, Mérida, Badajoz, Spain.
- *Corresponding Author:
- Jose R. Infante
Nuclear Medicine Service
Infanta Cristina Hospital
Carr. Portugal s/n. E-06080
Badajoz, Spain.
Telephone: +34-924-218095
Fax: +34-924-218038
E-mail: infantetorre@yahoo.com
Accepted date: November 27 2009
Abstract
The aim of the study was to evaluate the daytime rhythm of hormones in subjects who are practitioners of the advanced meditation technique TM-Sidhi. The study group consisted of five subjects, two women and three men, with at least two years of experience in the practice of the technique. Blood samples were taken at hourly intervals from 09:00h to 21:00h. The subjects practiced meditation for one hour between 12:00h and 13:00h. Cortisol and ACTH levels were measured using radioimmunoassay methods, whereas catecholamines concentrations were determined by high performance liquid chromatography. Cortisol showed a normal rhythm, with values significantly higher in the morning than in the afternoon (p=0.003, Kruskal-Wallis test), with a non significant decline in the levels during the period of meditation. For ACTH, there were no diurnal variations (p= 0.4), but with elevated levels of the hormone after meditation. Catecholamines showed no significant differences between the morning and afternoon concentrations (p= 0.09 for NE, p= 0.08 for E). After the TM-Sidhi program their values declined, reaching the minimum level at 14:00h. The present study found patterns of the daytime secretion of ACTH and catecholamines in practitioners of the TM-Sidhi technique that were different from those normally described. After the period of meditation, there was an increase in the levels of the hypophyseal hormone to reach its maximum daytime concentration. This was associated with decreased plasma catecholamines levels. The technique of meditation seems to have a significant effect on the sympathetic-adrenal medullary system and the neuroendocrine axis
Keywords
ACTH, Cortisol, Catecholamines, Circadian rhythms, Neuroendocrine axis, Transcendental Meditation, Yoga.
Introduction
Transcendental Meditation (TM) is a simple and effortless stress management technique. This procedure, the main technology of Maharishi Consciousness-Based Health Care, has been the subject of numerous studies of its physiological and psychological effects [1,2]. These include hormonal changes, with decreased blood levels of stress-associated hormones being observed both during the practice of TM and outside the periods of meditation [3, 4]. Specific electroencephalographic and metabolic patterns have also be en demonstrated in these subjects [5].
A few studies have focused on the circadian rhythms of practitioners of the technique, demonstrating, amongst other findings, variations in the excretory level of urinary 5-hydroxyindole related to the periods of meditation, and the acrophase of the metabolite occurring during or immediately following the period of meditation [6]. Similarly, elevated plasma levels of melatonin have been described in the period following meditation, leading to the postulation of a possible effect of TM on the diurnal rhythm of hormone release via an effect on melatonin [7].
In previous studies on TM practitioners, our group has observed lowered blood levels of catecholamines and modifications in the diurnal secretion patterns of both β-endorphins and adrenocorticotropic hormone (ACTH) [8, 9]. On the basis of these findings, we speculated that there might be a modification of hypothalamic and hypophyseal activity due to the regular practice of meditation.
In order to confirm these findings, and to attempt to establish the exact diurnal evolution of these hormones, in the present study we quantified the daytime rhythm of various hormones in subjects who are practicing the more advanced meditation technique TM-Sidhi.
Methods
Subjects
The study group consisted of five Caucasian subjects, two women and three men, between the age of 32 and 45 years, and leading family lives with normal daily routines (meal times, working hours, hours of sleep) except for two periods of meditation during the day. They participated in our previous research [8, 9] where all of them showed different daytime secretion pattern of catecholamines and pituitary hormones compared with a control non-meditator group.
They volunteered to participate in the trial, which was approved by the Hospital Research Ethics Committee, and underwent a prior medical examination that included a physical check-up and a blood analysis of twenty biochemical parameters determined using a Hitachi 747 autoanalyzer. None of the participants presented physical or mental disease, obesity or under medical treatment, or regularly performed any sport. They were asked not to smoke or ingest caffeine for one week before the trials. The women took part in the trial during the follicular phase of their menstrual cycle.
All participants were experienced with the TM technique and had at least two years of experience in the practice of TM-Sidhi, including "yogic flying", having been taught the two techniques by trained professionals according to standard procedures. During TM, the thought process quietens down, reaching a state of alert hypometabolism [10]. TM-Sidhi is an advanced meditation technique, practised for an additional 20 to 30 minutes after TM, using aphorisms known as sutras instead of mantras [11].
Procedure
The study was carried out in spring at a private building adjacent to the Hospital Virgen de las Nieves in Granada, Spain. The subjects were previously familiarized with this place, and were asked to abstain from exercise for two days prior to the study. Each subject had a catheter inserted into the vein of the elbow flexure one hour before drawing the first blood sample in order to avoid effects of the stress of the cannulation. Successive blood samples were taken at hourly intervals from 09:00h to 21:00h. For each sample, the first 5 mL of blood were discarded, and, after the extraction was complete, the extraction tube was rinsed with heparinized saline solution to prevent obstruction.
The volunteers fasted for 12 hours before the first morning sample. During the trial, the subjects practiced the meditation techniques for one hour between 12:00h and 13:00h. They were allowed oral hydration with water ad libitum and a single meal at 15:00h. No high-level physical activity was permitted.
Hormone Analysis
Plasma concentrations of cortisol, ACTH, norepinephrine (NE), and epinephrine (E) were determined. Blood samples were collected in vacutainer tubes with anticoagulant EDTA(K3). After centrifugation in a cold centrifuge, the separated plasma was decanted into plastic tubes and frozen at –40° C for subsequent hormone determination.
The plasma levels of catecholamines were determined by high pressure liquid chromatography. As a mobile phase a mix was used with a determined proportion of methanol, phosphate tampon and an ionic pair former, supplied by ESA, Inc. (Bedford, MA, USA). As an external standard a mix of norepinefrina and epinefrina was used at determined concentrations, supplied by the same company, and dihydroxybenzylamine was used as the internal standard. Using a Beckman 342 chromatograph, the ESA Plasma Catecholamine Analysis System marketed by ESA, Inc. (Bedford, MA, USA) was applied. A Gilson 307 dosing pump with a flow rate of 1 ml/min was connected to a reverse-phase Catecholamine HR-80 Column. A model 5100A Coulochem Electrochemical Detector was used, with its signal registered and integrated by the Beckman Instruments System Gold 500-APP registry and calculation terminal.
Cortisol and ACTH levels were measured using radioimmunoassay methods (Cortisol-ICN Biochemical Inc. Costa Mesa, CA. ACTH - Nichols Institute Diagnostics. San Juan Capistrano, CA).
Data Analysis
Through the application of the statistical program Statistical Graphics System (STATGRAF, Statistical Graphics Corporations), data were analysed using the Kruskal-Wallis test. P values less than .05 were considered statistically significant.
Results
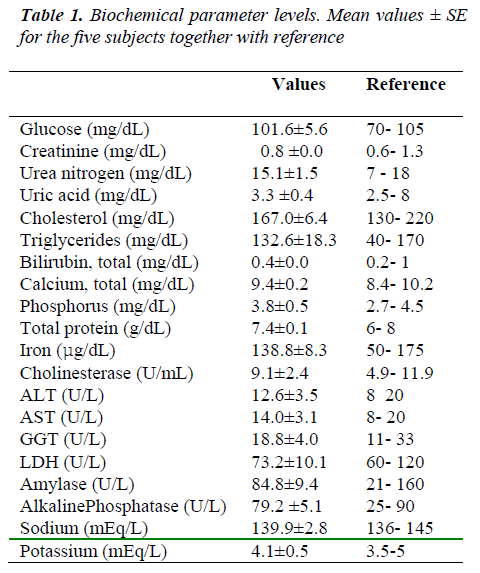
Table 1 shows mean values ± standard errors (SE) of 20 biochemical parameters for the five subjects. These concentrations were within the normal values of our laboratory coinciding with the general literature [12].
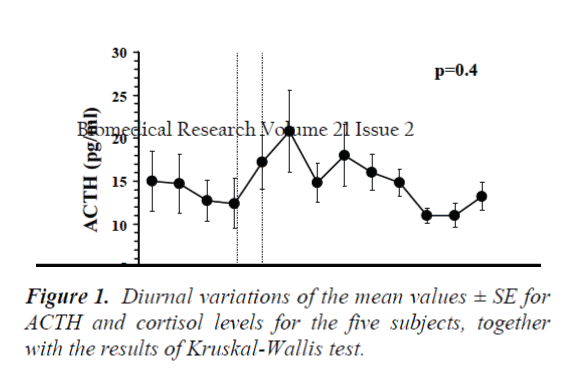
Figure 1 presents the diurnal variations of the mean values ± SE of ACTH and cortisol for the five subjects. For cortisol, one appreciates a normal rhythm, with values significantly higher in the morning than in the afternoon (p=0.003, Kruskal-Wallis test). There was a decline in the levels during the period of meditation, although this was not significant, and a clear peak in secretion after the mid-day meal (16:00 hours). For ACTH, no diurnal variation (p=0.4, Kruskal-Wallis test) was detected, the morning and afternoon levels being similar. There were elevated levels of the hormone after the meditation period, reaching the diurnal peak at 14:00h (20.8 ± 4.8 pg/ml). As was the case with cortisol, there was a peak secretion coinciding with the mid-day meal.
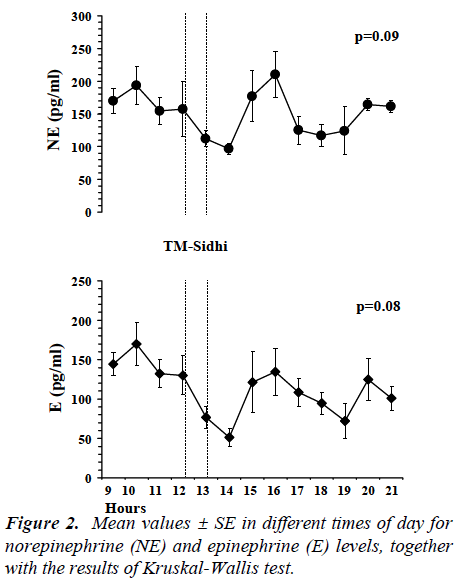
Figure 2 presents the diurnal variations of the mean catecholamine values. Neither in this case were there any significant differences between the morning and afternoon concentrations of the two hormones (p=0.09 for NE, p = 0.08 for E, Kruskal-Wallis test). After the TM-Sidhi program, the values declined, reaching in both cases their minimum level at 14:00h (NE 96.7 ± 8.7 pg/ml; E 51.2 ± 11.7 pg/ml). There was also a peak in secretion after the midday meal.
Discussion
The present study found patterns of the daytime secretion of ACTH and catecholamines in practitioners of the TM-Sidhi technique that were different from those normally described. After the period of meditation, there was an increase in the levels of the hypophyseal hormone to reach its maximum daytime concentration. This was associated with decreased plasma catecholamine levels.
These results are consistent with our previous studies in which TM practitioners presented similar morning and afternoon blood concentrations of ACTH and catecholamines [8, 9].
Various studies have reported the effect of TM on the neuroendocrine system, including changes in hormone levels during practice of the technique and with its regular use. Altered responses to acute stress for cortisol, GH, TSH and testosterone after four months of TM practice have been demonstrated [13]. Low cortisol and elevated serum dehydroepiandrosterone sulfate levels have been reported in TM practitioners compared to non-practising subjects [14, 15].
However, there have been just a few studies of circadian rhythms in meditators, including the aforementioned two carried out by our group. Walton et al. [6] studied the diurnal fluctuations of the urinary metabolite of serotonin, 5-hydroxyindole. They described a pattern of excretion in which the acrophase tended to occur during or immediately after the period of meditation, and suggested that the practice of TM might cause alterations in neural regulation, behaving as a zeitgeber to synchronize hormone release. This hypothesis has also been considered by other workers on the basis of their estimated plasma concentrations of different hormones [16,17].
The present results seem to be coherent with the above, because the concentrations of ACTH and cortisol that we measured were in synchrony with each other, the peaks of secretion occurring at 14:00h following the practice of the technique.
The TM-Sidhi technique involves the use of certain mental formulas called sutras. This use is accompanied by a marked increase in electroencephalographic coherence, particularly in the beta frequency, in the frontal and central areas of the brain [18]. Furthermore, TM itself is associated with increased cerebral blood flow, which implies increased brain activity [19]. Current theories on the effects of TM-Sidhi on the CNS consider the hypothalamus, thalamus, and hippocampus to be the possible sites of action [20]. Meditation would alter the activity of these neural structures which, via their connections with other structures (cortex, autonomic nervous system, medullary nuclei) would be responsible for the electroencephalographic, hormonal, and respiratory effects described in the literature.
The existence of rhythms of secretion of both hormones of the adrenal axis and catecholamines is well known, with high levels in the morning followed by a progressive decline over the course of the day [21].
Circadian rhythms are under the control of the hypothalamic suprachiasmatic nucleus (SCN), and are synchronized by zeitgebers with γ-aminobutyric acid probably being the neurotransmitter involved. Serotonin is extensively distributed throughout the hypothalamus, with its greatest concentration in the SCN, and at the hypothalamic level exerts a regulatory action controlling the circadian rhythm of ACTH [22].
Melatonin, which is the principal hormone of the pineal gland, is strongly linked to biological rhythms, and is postulated as being an internal zeitgeber in mammals. Recently, Tooley et al. [7] have described increased plasma melatonin levels following the practice of TM-Sidhi. They suggested that the mechanism by which this meditation induced increase could be either due to a decrease in the rate of melatonin metabolism via reduced hepatic blood flow, or an increased production and secretion by the pineal gland.
Also, increased levels of prolactin have been described following TM, considering that these changes in the pituitary hormone' secretion are due to modulations of the hypothalamic GABA-ergic tone [23]. In this regard, it is interesting to note that this neurotransmitter is involved in regulating the secretion of ACTH.
Together with the aforementioned studies showing changes in the concentrations of melatonin and the metabolite of serotonin, our own results support the hypothesis of a modulatory effect of the TM-Sidhi relaxation technique on hormonal circadian rhythms.
Clinical situations can alter these rhythms. In Cushing's disease patients show high concentrations of cortisol and a loss of its rhythm [24]. In obese individuals, the β-endorphin circadian rhythm is altered, although those of cortisol and ACTH are unchanged [25]. Finally, in major depressive illness hypercortisolism with the persistence of the circadian rhythm of both cortisol and ACTH does exist [26]. None of these patterns fits that described in the present study.
It was noteworthy that concurrent with the peaks in cortisol and ACTH secretions after the performance of the TM-Sidhi technique were the minimum measured NE and E concentrations. This finding is indicative that the peak of the hormones of the hypothalamic-hypophyseal axis is unrelated to situations of stress. The low blood catecholamine levels can be interpreted as an effect of meditation on the sympathetic tone, and could explain in part the results obtained with the use of TM in hypertensive subjects [27, 28].
Although no significant differences were demonstrated, it is interesting to note that the subjects with longer experience in the practice of the technique had lower mean values of ACTH and cortisol relative to those with less experience. In this sense, there has been described a steady decline in blood concentrations of anterior pituitary hormones after subjects learn the TM-Sidhi program, suggesting that this technique could influence the secretion of these hormones through regulating the release of the hypothalamic peptides [17].
Various considerations involving possible confounders need to be taken into account. It has been reported that prolonged decubitus could interfere with catecholamine rhythms [29]. In our case, this possible interference can be discarded, since all the study subjects were allowed to remain standing throughout the study. Furthermore, the protocol used for the sample collection included a delay between the introduction of the catheter and the extraction of the first blood sample, and the first 5 mL of blood of each extraction were discarded, eliminating any possible effect of the stress caused by cannulation.
The ACTH, cortisol, and catecholamine peaks observed at 16:00h immediately after food intake are coherent with results described in the literature [30].
Finally, although the number of subjects was limited, one must appreciate the difficulty of finding volunteers for this type of study. Moreover, this limitation is a common norm in the literature on human circadian rhythms.
In conclusion, the results are coherent with those previously described by our group, showing the patterns of diurnal hormone secretion in TM-Sidhi practitioners to be different from those normally described. The technique of meditation seems to have a significant effect on the sympathetic-adrenal medullary system and the neuroendocrine axis.
Acknowledgement
We are grateful to Mister Humberto Fariñas, Miss Rosa Arenas and Miss Concha Ruiz for their excellent technical assistance.
References
- Wallace R.K. The physiology of consciousness. MIU Press. Fairfield, Iowa 1993.
- Jevning R, Wallace RK, Beidebach M. The physiology of meditation: a review. A wakeful hypometabolic integrated response. Neurosci Biobehav Rev 1992; 16: 415-424.
- Walton KG, Fields JZ, Levitsky DK, Harris DA, Pugh ND, Schneider RH. Lowering cortisol and CVD risk in postmenopausal women: a pilot study using the transcendental meditation program. Ann N Y Acad Sc. 2004; 1032: 211-215.
- Jevning R, Wells I, Wilson AF, Guich S. Plasma thyroid hormones, thyroid stimulating hormone, and insulin during acute hypometabolic states in man. Physiol Behav 1987; 40: 603-606.
- Dillbeck MC, Bronson EC. Short-term longitudinal effect of the Transcendental Meditation technique on EEG power and coherence. Int J Neurosci 1981; 14: 147-151.
- Walton KG, Lerom M, Salerno J, Wallace RK. Practice of the Transcendental Meditation (TM) and TM-Sidhi program may affect the circadian rhythm of urinary 5-hydroxyindole excretion. Soc Neur Abstr 1981; 7: 48.
- Tooley GA, Armstrong SM, Norman TR, Sali A. Acute increases in night-time plasma melatonin levels following a period of meditation. Biol Psychol 2000; 53: 69-78.
- Infante JR, Peran F, Martinez M, Roldan A, Poyatos R, Ruiz C, Samaniego F, Garrido F. ACTH and ├ā┬¤-endorphin in Transcendental Meditation. Physiol Behav 1998; 64: 311-315.
- Infante JR, Torres-Avisbal M, Pinel P, Vallejo JA, Peran F, Gonzalez F, Contreras P, Pacheco C, Roldan A, Latre JM. Catecholamine levels in practitioners of the transcendental meditation technique. Physiol Behav 2001; 72: 141?146.
- Nader T. Human physiology ? Expression of Veda and the Vedic Literature. Maharishi University Press. Vlodrop, Holland 1995.
- Patanjali. Yoga sutras (R. Prasada, Trans.). Oriental Books Reprint Corporation. New Delhi 1978.
- Tietz NW. Clinical guide to laboratory tests. W.B. Saunders Co. Philadelphia 1990.
- MacLean CR, Walton KG, Wenneberg SR, Levitsky DK, Mandarino JP, Waziri R, Hillis SL, Schneider RH. Effects of the Transcendental Meditation Program on adaptive mechanisms: changes in hormone levels and responses to stress after 4 months of practice. Psychoneuroendocrinology 1997; 22: 277-295.
- Bevan AJ. Endocrine changes in Transcendental Meditation. Clin Exp Pharmacol Physiol 1980; 7: 75-76.
- Glaser JL, Brind J, Vogelman JH, Eisner MU, Dillbeck MC, Wallace RK, Chopra D, Orentreich N. Elevated serum dehydroepiandrosterone sulfate levels in practitioners of the Transcendental Meditation (TM) and TM-Sidhi programs. J Behav Med 1992; 15: 327-341.
- Jevning R, Wilson AF, Davidson JM. Adrenocortical activity during meditation. Horm Behav 1978; 10: 54-60.
- Werner OR, Wallace RK, Charles B, Janssen G, Stryker T, Chalmers RA. Long-term endocrinologic changes in subjects practicising the transcendental meditation and TM-Sidhi program. Psychosom Med 1986; 48: 59-66.
- Travis F, Orme-Johnson DW. EEG coherence and power during yogic flying. Int J Neurosci; 1990, 54: 1-12.
- Jevning R, Anand R, Biedebach M, Fernando G. Effects on regional cerebral blood flow of transcendental meditation. Physiol Behav 1996; 59: 399-402.
- Travis F, Wallace RK. Autonomic and EEG patterns during eyes-closed rest and transcendental meditation (TM) practice: the basis for a neural model of TM practice. Conscious Cogn 1999; 8:302-318.
- Linsell CR, Lightman SL, Mullen PE, Brown MJ, Causon RC. Circadian rhythms of epinephrine and norepinephrine in man. J Clin Endocrinol Metab 1985; 60: 1210-1215.
- Patel YC. Neurotransmitters and hypothalamic control of anterior pituitary function. In: DeGroot LJ, Jameson JL eds. Endocrinology. 4th ed. W.B. Saunders Company. Philadelphia 2001; pp183-200.
- Elias AN, Wilson AF. Serum hormonal concentrations following transcendental meditation - potential role of gamma aminobutyric acid. Med-Hypotheses 1995; 44: 287-291.
- Boyer RM, Witkin M, Carruth A, Ramsey J. Circadian cortisol rhythms in Cushing's disease. J Clin Endocrinol Metab 1979; 48: 760-765.
- Facchinetti F, Giovannini G, Barletta C, Petraglia F, Buzzetti R, Burla F, Lazzari R, Genazzani AR, Scavo D. Hyperendorphinemia in obesity and relationships to affective state. Physiol Behav 1986; 36: 937- 940.
- Claustrat B, Chazot G, Brun J, Jordan D, Sassolas G. A chronobiological study of melatonin and cortisol secretion in depressed subjects: plasma melatonin, a biochemical marker in major depression. Biol Psychiatry 1984; 19: 1215-1228.
- Schneider RH, Alexander CN, Staggers F, Orme-Johnson D, Rainforth M, Salerno J, Sheppard W, Castillo-Richmond A, Barnes V, Nidich S. A randomized controlled trial of stress reduction in the treatment of hypertension in African Americans over one year. Am J Hypertens 2005; 18: 88-98.
- Alexander CN, Schneider RH, Staggers F, Sheppard W, Clayborne BM, Rainforth M, Salerno J, Kondwani K, Smith S, Walton KG, Egan B. Trial of stress reduction for hypertension in older African Americans. II. Sex and risk subgroup analysis. Hypertension 1996; 28: 228-237.
- Young JB, Landsberg L. Catecholamines and the adrenal medulla. In: Wilson JD, Foster DW, Kronenberg M, Larsen PR, eds. Williams textbook of Endocrinology. 9th ed. W.B. Saunders Company. Philadelphia 1998; pp 665-728.
- Follenius M, Brandenberger G, Hietter B. Diurnal cortisol peaks and their relationships to meals. J Clin Endocrinol Metab 1982; 55: 757-761.