Research Article - Biomedical Research (2017) Volume 28, Issue 5
Cost and effectiveness comparison of thoracic epidural anaesthesia combined with general anaesthesia in esophagectomy
Li-Jun Wang1, Jing Cang2* and Zhang-Gang Xue21Department of Anesthesiology, Ear Eye Nose and Throat Hospital, Fudan University, Shanghai, PR China
2Department of Anesthesiology, Zhongshan Hospital, Fudan University, Shanghai, PR China
- *Corresponding Author:
- Jing Cang
Department of Anesthesiology
Zhongshan Hospital
Fudan University Shanghai, PR China
Accepted on October 06, 2016
Abstract
Thoracic epidural anaesthesia (TEA) combined with general anaesthesia (GA) is widely used in clinics, but its cost-effectiveness has rarely been studied. We hypothesised that TEA combined with GA is beneficial for patients undergoing esophagectomy without increasing their costs. Eighty patients scheduled for thoracic esophagectomy were randomised to receive TEA combined with GA (TEA group) or general anaesthesia alone (control group). The following indicators were recorded: hospital stay, patient satisfaction, time to extubation, intraoperative anaesthetic dosages and fluid transfusion, postanaesthesia care unit stay, visual analogue scale score and anaesthesia-related complications. Statistical analysis utilised the Kruskal-Wallis rank sum test and the χ2 test. The TEA group exhibited significantly improved indicators on time to extubation, postanaesthesia care unit stay, visual analogue score, anaesthesia-related complications, hospital stay (P<0.05 for all measurements), and patient satisfaction (P<0.01) compared with the control group. However, the direct medical expenses did not differ significantly between the two groups. TEA combined with GA resulted in better prognosis and higher satisfaction without increasing the medical expenses. This combination is also a cost-effective anaesthesia regimen for esophagectomy.
Keywords
Anesthesia, Epidural, Cost effectiveness, Sevoflurane, Esophagectomy.
Introduction
Medical expenses have recently been steadily increasing in China. The Ministry of Health of China stated that the total medical expenses of the Chinese government from 2009 to 2011 were 732.6 billion RMB, with an annual growth rate of 30.17%. Therefore, the appropriate use of limited medical resources is paramount. Thoracotomy surgery can cause severe patient morbidity and significantly affects the respiratory and circulatory systems [1]. Thoracic epidural anaesthesia (TEA) combined with general anaesthesia (GA) has also been widely used in clinical practice [2], especially for thoracic or upper abdominal surgeries and high risk patients [3,4]. Thoracic epidural anaesthesia and analgesia (TEAA) remains a preferred technique for the challenging and painful perioperative period of thoracic surgery [5]. TEAA with local anaesthetics has been reported to benefit stable haemodynamics intraoperatively and lessen pulmonary complications postoperatively [6,7]. Efficient analgesia with TEAA also results in early mobilisation, enabling painless ventilation and decreased morbidity and mortality [8]. Epidural block has been argued to increase the cost and risk of thoracic surgery anaesthesia [9]. Costs and benefits are both important for patients. Thus, the cost and effectiveness of TEA combined with GA should be further studied. In this prospective, randomised and double-blind study, the authors aimed to compare the clinical features and costs of TEA combined with GA and GA alone for thoracic esophagectomy. We tested the hospital stay and patient satisfaction as the primary endpoints and time to extubation, intraoperative anaesthetic dosages and fluid transfusion, postanaesthesia care unit stay, visual analogue scale score and anaesthesia-related complications as the secondary endpoints.
Subjects and Methods
Subjects
This study was an open-label, randomised and controlled clinical trial. Following IRB approval (Zhongshan Hospital, Fudan University), a total of 80 patients [18 years old to 65 years old; body mass index of 18-30; American Society of Anaesthesiologists (ASA) physical status I and II] undergoing selective thoracic esophagectomy were enrolled in this study. The exclusion criteria included dysfunction of the heart, liver or kidney, electrolyte disturbance, neuromuscular disorder, immune disease, endocrinological disorder, drug allergy and contraindications for TEA. The patients were removed from the study if: 1) postoperative mechanical ventilation or sedatives were required or 2) a second surgery was required during the study. The patients were randomised using a random number table to receive TEA combined with GA (TEA group) or GA alone (control group). This study was conducted in accordance with the declaration of Helsinki. This study was conducted with approval from the Ethics Committee of Zhongshan Hospital, Fudan University. Written informed consent was obtained from all participants.
Anaesthesia regimens
An epidural catheter was placed at the T7-8 interspace in the TEA group, and the analgesic effect was confirmed. Anaesthesia was induced intravenously in both groups with fentanyl (2 μg/kg to 3 μg/kg), propofol (1.5 mg/kg to 2 mg/kg) and succinylcholine (1 mg/kg to 2 mg/kg). A left-sided, double-lumen thoracostomy tube (Batch number: USA0604-2005; male, #37; female, #35) was inserted and confirmed by bronchofiberoscopy. Anaesthesia was maintained with sevoflurane (Baxter, Deerfield, USA; batch number, 00784 S046F712) at 0.7 to 1.1 minimal alveolar concentration (MAC), and muscle relaxation was provided by vecuronium bromide. In the TEA group, additional 5 mL epidural doses of bupivacaine 0.25% and adrenalin (1:2,000,000) were administered at 1 h intervals. Heart rate, arterial blood pressure, central venous pressure, pulse oximetry (SpO2) and bispectral index (BIS) were monitored intraoperatively (Aspect Medical Systems, Mansfield, USA). BIS was maintained between 40 and 60, and the heart rate and arterial blood pressure were maintained within 20% of the preoperative values. Additional fentanyl and vecuronium were used as needed. Patient-controlled analgesia was used after extubation with IV doses of morphine in the control group (continuous 0.6 mg/h to 1 mg/h, bolus 2 mg to 3 mg, 300 mL total) and epidural analgesia in the TEA group (bupivacaine 0.125% + morphine 20 μg/mL, continuous 3 mL/h to 4 mL/h, bolus 3 mL to 4 mL, 300 mL total). The patients were monitored postoperatively in the postanaesthesia care unit (PACU) and returned to the ward when the Aldrete score was ≥ 9.
Study endpoints
The primary endpoint of this study investigated whether TEA combined with GA would improve the hospital stay and patient satisfaction indicators. The secondary endpoints focused on whether this kind of anaesthesia would affect time to extubation, intraoperative anaesthetic dosages and fluid transfusion, PACU stay, visual analogue scale (VAS) scores at 4 h and 1 d to 5 d postoperative and anaesthesia-related complications. The patients were asked to answer a questionnaire with 10 questions to assess their satisfaction scores before being discharged from the hospital (0=most unsatisfactory, 10=most satisfactory).
Expenses
The documented expenses did not cover the operation room routine costs, medical staff salaries or depreciation expenses of the monitoring equipment. We only calculated the direct medical expenses, including bed, treatment, medication, laboratory test, surgery and anaesthesia expenses. As both groups had similar types of surgical procedures with assumptions of similar costs, surgery costs were excluded. The bed costs were calculated for each patient based on their length of hospital stay. The treatment costs referred to all costs of treatment given in the ward. The anaesthesia expenses included intraoperative anaesthetic expenses (costs of anaesthetic procedure, materials, monitoring and drugs) and postoperative anaesthetic expenses (costs of analgesic pump, analgesic drugs in the pump, additional analgesic drugs, antiemetics in the pump and additional antiemetics). The costs of sevoflurane were estimated by calculating the product of cost per MAC hour and surgery time. All estimated costs per MAC hour were based on a duration of 60 min and the following formula: Cost per MAC hour (RMB)=[(Concentration) (FGF) (duration) (MW) (cost/mL)]/[(2412) (D)], where FGF is the fresh gas flow rate in L/min, MW is the molecular weight in g, cost per mL is in dollars based on the average wholesale price and D is the density in g/mL. Sevoflurane calculations were based on a concentration of 2.05%, MW of 201 g and density of 1.51 g/mL [10].
Statistical analysis
Sample size determination was based on data from our preliminary experiment. We calculated that a sample of at least 68 patients would need to be enrolled to achieve 80% power (α=0.05, β=0.2). We chose 80 patients for this study to avoid off cases. The continuous data were represented as mean ± SD and compared using the student’s t-test. Non-parametric data were analysed using the Kruskal-Wallis rank sum test, and categorical data were analysed using the Pearson’s Chi-square test. A P value>0.05 was considered to indicate statistical significance. Statistical analyses were conducted using SPSS 11.5 (SPSS, Chicago, IL).
Results
Clinical and demographic characteristics
Eighty patients scheduled for thoracic esophagectomy were enrolled in this study. Table 1 summarises the demographic and clinical characteristics of the study population. No significant differences were found between the two groups in terms of age, sex, height, weight and ASA physical status (P>0.05).
| Subject Characteristics | Control Group (n=40) | TEA group (n=40) | P-value |
|---|---|---|---|
| Age (years) | 58.9 ± 3.9a | 55.9 ± 6.6a | 0.075 |
| Female n (%) | 14 (35) | 16 (40) | 0.624 |
| Height (cm) | 165.9 ± 7.3a | 164.6 ± 5.3a | 0.514 |
| Weight (kg) | 64.1 ± 6.5a | 63.4 ± 6.7a | 0.745 |
| ASA I n (%) | 14 (35) | 12 (30) | 0.423 |
| ASA II n (%) | 26 (65) | 28 (70) | 0.546 |
| aMean ± SD. ASA: American Society of Anesthesiologists. Age, height and weight were compared by using the student’s t-test. Female and ASA were compared by using the Pearson’s Chi-square test. | |||
Table 1. Clinical characteristics.
Intraoperative anaesthetic dosages and fluid volume
Table 2 summarises the anaesthetic dosages, crystalloid and colloid fluid transfusion, blood loss and urine volume during the operation. The doses of fentanyl, propofol, vecuronium and sevoflurane in the TEA group were significantly decreased compared with those in the control group (P<0.05).
| Anesthetic dosages and fluid volume | Control group (n=40) | TEA group (n=40) | P-value |
|---|---|---|---|
| Fentanyl (mg) | 0.6 ± 0.08 | 0.3 ± 0.1# | 0.001 |
| Propofol (mg) | 164.3 ± 41.9 | 126.2 ± 27.2# | 0.003 |
| Succinylcholine (mg) | 95.8 ± 10.2 | 97.5 ± 9.6 | 0.828 |
| Vecuronium (mg) | 6.8 ± 0.9 | 5.8 ± 1.6# | 0.009 |
| Bupivacaine (mg) | 0 | 80.3 ± 18.1# | <0.01 |
| Sevoflurane (mL) | 27.5 ± 2.9 | 19.7 ± 5.2# | 0.001 |
| Crystalloid fluid (mL) | 1809.5 ± 392.7 | 1595.2 ± 374.8 | 0.082 |
| Colloid fluid (mL) | 833.3 ± 235.7 | 809.5 ± 248.8 | 0.879 |
| Blood loss (mL) | 420.5 ± 130.9 | 409.5 ± 222.8 | 0.655 |
| Urine volume (mL) | 553.5 ± 157.1 | 573.8 ± 440.9 | 0.288 |
| Data were compared by using the Kruskal-Wallis rank sum test. #compared with the control group, P<0.01. | |||
Table 2. Intraoperative anesthetic dosages and fluid volume.
Surgery time, time to extubation and PACU stay
The surgeries were uneventful in both groups. No significant difference was found in surgery time between the two groups (mean: 202.9 ± 18.9 versus 182.6 ± 32.1 min, P>0.05). Both the time to extubation (mean: 12.1 ± 6.3 versus 21.4 ± 7.3 min, P=0.03) and PACU stay (mean: 36.7 ± 18.9 versus 56.5 ± 14.1 min, P=0.001) were significantly shorter in the TEA group compared with the control group.
Postoperative condition
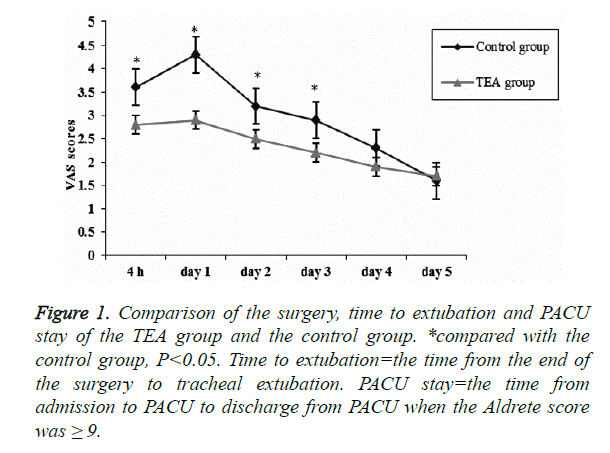
VAS score increased significantly within 24 h and started to decrease from the second day postoperatively. The VAS scores in the TEA group were significantly lower than those for the control group at 4 h, and on days 1, 2 and 3 postoperatively (P=0.001, 0.049, 0.026 and 0.015, respectively), as shown in (Figure 1). Compared with the control group, fewer patients exhibited hypersomnia (4 versus 16, P=0.028) and pain requiring intervention (6 versus 18, P=0.038), whereas more patients exhibited pruritus (24 versus 10, P=0.001) in the TEA group. No significant differences were found in patients in terms of vertigo, nausea and vomiting between the two groups (P>0.05). The postoperative hospital stay (mean: 10.1 ± 2.2 versus 12.1 ± 2.4, P=0.008) and patient satisfaction (mean: 8.7 ± 1.2 versus 7.2 ± 1.3, P=0.001) were significantly better in the TEA group than in the control group (Table 3).
Figure 1. Comparison of the surgery, time to extubation and PACU stay of the TEA group and the control group. *compared with the control group, P<0.05. Time to extubation=the time from the end of the surgery to tracheal extubation. PACU stay=the time from admission to PACU to discharge from PACU when the Aldrete score was ≥ 9.
| Anesthesia-related complications and primary endpoints | Control group (n=40) | TEA group (n=40) | P-value |
|---|---|---|---|
| Hypersomnia (n) | 16 | 4* | 0.028 |
| Vertigo (n) | 10 | 4 | 0.212 |
| Nausea (n) | 8 | 9 | 0.826 |
| Vomiting (n) | 6 | 4 | 0.718 |
| Pruritus (n) | 10 | 24# | 0.001 |
| Pain (n) | 18 | 6* | 0.038 |
| Postoperative hospital stay (days) | 12.1 ± 2.4 | 10.1 ± 2.2* | 0.008 |
| Satisfaction with the anesthesia | 7.2 ± 1.3 | 8.7 ± 1.2# | 0.001 |
| The persons of hypersomnia, vertigo, nausea, vomiting, pruritus and pain were compared by using the Pearson’s Chi-square test. Postoperative hospital stay and satisfaction with the anesthesia were compared by using the Kruskal-Wallis rank sum test. *compared with the control group, P<0.05. #compared with the control group, P<0.01. | |||
Table 3. Anesthesia-related complications and primary endpoints of two groups.
Comparison of expenses between the two groups
The intraoperative anaesthetic expenses covered the anaesthetic procedure, materials, monitoring and drugs. Compared with the control group, the TEA group had significantly increased anaesthetic procedure costs (780 versus 1,180 RMB), but significantly lower drug and monitoring costs (P=0.001 and 0.0009, respectively). The material expense did not differ significantly between the two groups (P>0.05). The TEA group had significantly higher intraoperative anaesthetic expenses (P=0.003), but significantly lower postoperative expenses for such items as analgesic pumps, analgesic drugs and antiemetics (P=0.001, 0.016 and 0.001, respectively), as shown in (Table 4). The direct medical expenses in the TEA group were 28,997.5 ± 4,687. 2 RMB, including the bed cost of 1,743.7 ± 842.1 RMB and the treatment cost of 2,821.8 ± 960.9 RMB, which were significantly lower than those in the control group (P=0.036 and 0.012, respectively). All other expenses did not differ significantly between the two groups (P>0.05). The direct medical expenses in the TEA group were lower than those in the control group, but the difference was not statistically significant (mean: 37,452.9 ± 4,687.2 versus 31,890.87 ± 3,424.9 RMB, P>0.05).
| Anesthesia-related expenses (RMB) | Control group (n=40) | TEA group (n=40) | P-value |
|---|---|---|---|
| Intraoperative expenses | |||
| Anesthetic procedure | 780 | 1180 | <0.01 |
| Materials | 2231.3 ± 0.7 | 2230.3 ± 3.1 | 0.937 |
| Monitoring | 384.8 ± 12.7 | 366.7 ± 20.4# | 0.001 |
| Drugs | 1153.1 ± 72.9 | 1020.3 ± 118.2# | 0.001 |
| Total | 4549.2 ± 76.3 | 4797.3 ± 134.2# | 0.003 |
| Postoperative expenses | |||
| Analgesic pump | 633.3 ± 270.8 | 325.7 ± 242.8# | 0.001 |
| Analgesic drugs in pump | 53.3 ± 22.8 | 38.8 ± 28.9 | 0.096 |
| Additional analgesic drugs | 4.6 ± 3.5 | 1.3 ± 0.1* | 0.02 |
| Antiemetics in pump | 392.1 ± 104.5 | 273.2 ± 93.9# | 0.001 |
| Additional antiemetics | 51.4 ± 21.2 | 43.1 ± 14.5 | 0.638 |
| Data were compared by using the Kruskal-Wallis rank sum test. *compared with the control group, P<0.05. #compared with the control group, P<0.01. | |||
Table 4. Perioperative anesthesia-related expenses in two groups.
Discussion
The major objective of this study was to compare the cost and effectiveness of TEA combined with GA and GA alone in esophagectomy. Previous studies usually focused on the clinical effects of anaesthesia, but few studies have investigated the cost. The principal finding of the present study is that TEA combined with GA could reduce the postoperative hospital stay and increase patient satisfaction without increasing the direct costs. Patient satisfaction is significantly correlated with postoperative pain control and the occurrence of complications. In our study, the VAS scores in both groups increased significantly within 24 h postoperatively but started to decrease from the second day postoperatively. TEA showed a significantly better analgesic effect than IV morphine from 24 h postoperatively until postoperative day 3. Postoperative analgesic efficacy is a major influencing factor of patient satisfaction with anaesthesia [11]. The trauma and postoperative pain resulting from thoracotomy surgery can lead to restricted breathing, decreased tidal volume and decreased frequencies of coughing and sputum, all of which compromise lung function and postoperative recovery. Effective postoperative analgesia can relieve patients’ pain, thereby enabling patients to cough, drink, breathe deeply and ambulate early [12]. Previous works reported that TEA could provide better postoperative analgesia and recovery [13,14], which were validated in our research.
In our study, the incidence of pruritus was 60% (24/40) with epidural dose of morphine and 25% (10/40) with IV doses of morphine, which was consistent with the findings of previous studies [15]. However, the symptoms in both groups were mild, and no intervention was needed. Intravenous use of morphine can lead to dizziness and hypersomnia, which are closely related to the blood concentration of morphine. Epidural use of morphine can significantly decrease the frequency of these side effects. We also found that the TEA group had fewer patients with dizziness and hypersomnia, and thus had significantly higher patient satisfaction. Postoperative nausea and vomiting (PONV) served another important function in patient satisfaction and cost. PONV was reportedly as unbearable as postoperative pain, and 78% of the patients were willing to pay $61 to $113 in the prophylactic treatment of vomiting to improve the postoperative recovery [16,17]. PONV did not differ significantly between the TEA group and the control group in this study. The reasons might be that: 1) male patients predominated in both groups, 2) thoracic surgery was not a risk factor for PONV and the surgical time did not differ significantly between the two groups and 3) sevoflurane and opioids were used in both groups.
Our observations suggest that the TEA group had significantly shorter time to extubation compared with the control group, which might have resulted from the lower dose of sevoflurane, muscle relaxants and analgesics used during the operation. TEA was reported to have potential sedative effects and could decrease the dose of sevoflurane required in general anaesthesia [18,19]. The local anaesthetics used in TEA can diffuse through the dura and spread cranially, directly affecting the central nervous system and resulting in decreased MAC [20-22]. Meanwhile, decreased doses of muscle relaxants and analgesics can minimise the postoperative muscle relaxation and respiratory inhibition, resulting in prompt and smooth emergence. This scenario might also be the reason why the TEA group had significantly shorter PACU stay compared with the control group. High turnover rates in the PACU can improve the utilisation efficiency of the operation room and the medical staff schedules, especially when high workloads are expected [23].
According to our results, the TEA group had significantly increased intraoperative anaesthetic procedure costs because of epidural block. The use of epidural anaesthesia reduced the drug doses and thus decreased the drug costs. Earlier extubation in the TEA group caused lower monitoring costs. Patients in the control group had higher VAS than those in the TEA group until day 3 after surgery. These patients had to pay more for analgesia pumps, antiemetics and additional analgesic drugs, such that their postoperative expenses were significantly higher than those of patients in the TEA group. Bed costs were closely related to the length of hospital stay. Mean hospital stay can determine the medical costs and is also a direct indicator of the efficient use of medical resources [24]. The efficient use of medical resources is an important aspect of any treatment regimen [25]. Shorter hospital stays can improve the bed utilisation and turnover rates, is effectively minimising the costs of hospital resources. In agreement with our findings, previous work has demonstrated that TEA and postoperative epidural analgesia can significantly decrease the frequency of perioperative adverse cardiac events and lung infections, leading to shorter hospital stays and better prognoses [26]. Our study consistently found that the TEA group had two days less hospital stay compared with the control group, which significantly lowered the medical costs.
Our study has a number of limitations. First, we did not calculate the indirect medical expenses such as the operation room routine costs and medical staff salaries. Second, the observation time was relatively short, and we did not perform a post-discharge follow-up. In the future, we may conduct further research with a larger sample size and a longer follow-up time.
Conclusion
The anaesthetic regimen serves an important function in the recovery and prognosis after esophagectomy. The choice of anaesthesia must not only meet the requirements of the surgery, but also promote postoperative recovery. TEA has higher intraoperative anaesthetic expenses and possible complications owing to the epidural block. However, our results show that TEA does not increase the total medical expenses and results in shorter hospital stay, better postoperative analgesia, less anaesthesia-related complications and improved patient satisfaction compared with GA alone. No severe intraspinal anaesthesia-related complications occurred in this study as well. Hence, TEA combined with GA is a cost-effective anaesthesia regimen for thoracic esophagectomy.
References
- Fernandes O, Teixeira C, Silva LC. Thoracic surgery: risk factors for postoperative complications of lung resection. Rev Assoc Med Bras 2011; 57: 292-298.
- Onan IS, Onan B, Korkmaz AA, Oklu L, Kilickan L, Gonca S, Dalcik H, Sanisoglu I. Effects of Thoracic Epidural Anesthesia on Flow and Endothelium of Internal Thoracic Artery in Coronary Artery Bypass Graft Surgery. J Cardiothorac Vasc Anesth 2011; 25: 1063-1070.
- Cok OY, Eker HE, Turkoz A, Findikcioglu A, Akin S, Aribogan A, Arslan G. Thoracic Epidural Anesthesia and Analgesia During the Perioperative Period of Thoracic Surgery: Levobupivacaine Versus Bupivacaine. J Cardiothorac Vasc Anesth 2011; 25: 449-454.
- Bakhtiary F, Therapidis P, Dzemali O, Ak K, Ackermann H, Meininger D, Kessler P, Kleine P, Moritz A, Aybek T, Dogan S. Impact of high thoracic epidural anesthesia on incidence of perioperative atrial fibrillation in off-pump coronary bypass grafting: A prospective randomized study. J Thorac Cardiovasc Surg 2007; 134: 460-464.
- Joshi GP, Bonnet F, Shah R, Wilkinson RC, Camu F, Fischer B, Neugebauer EA, Rawal N, Schug SA, Simanski C, Kehlet H. A systematic review of randomized trials evaluating regional techniques for postthoracotomy analgesia. Anesth Analg 2008; 107: 1026-1040.
- De Cosmo G, Congedo E, Lai C, Sgreccia M, Amato A, Beccia G, Aceto P. Ropivacaine vs. levobupivacaine combined with sufentanil for epidural analgesia after lung surgery. Eur J Anaesthesiol 2008; 25: 1020-1025.
- Clemente A, Carli F. The physiological effects of thoracic epidural anesthesia and analgesia on the cardiovascular, respiratory and gastrointestinal systems. Minerva Anestesiol 2008; 74: 549-563.
- Amini A, Patanwala AE, Maegawa FB, Skrepnek GH, Jie T, Gruessner RW, Ong ES. Effect of epidural analgesia on postoperative complications following pancreaticoduodenectomy. Am J Surg 2012; 204: 1000-1004.
- Bartha E, Rudin A, Flisberg P, Lundberg CJ, Carlsson P, Kalman S. Could benefits of epidural analgesia following oesophagectomy be measured by perceived perioperative patient workload? Acta Anaesthesiol Scand 2008; 52: 1313-1318.
- Golembiewski J. Economic considerations in the use of inhaled anesthetic agents. Am J Health Syst Pharm 2010; 67: S9-S12.
- Bujedo BM, Santos SG, Azpiazu AU. A review of epidural and intrathecal opioids used in the management of postoperative pain. J Opioid Manag 2012; 8: 177-192.
- Muller S, Zalunardo MP, Hubner M, Clavien PA, Demartines N. A Fast-Track Program Reduces Complications and Length of Hospital Stay after Open Colonic Surgery. Gastroenterology 2009; 136: 842-847.
- Monaco F, Biselli C, Landoni G, De Luca M, Lembo R, Covello RD, Zangrillo A. Thoracic epidural anesthesia improves early outcome in patients undergoing cardiac surgery for mitral regurgitation: a propensity-matched study. J Cardiothorac Vasc Anesth 2013; 27: 445-450.
- Guay J. The benefits of adding epidural analgesia to general anesthesia: a meta analysis. J Anesth 2006; 20: 335-340.
- Reich A, Szepietowski JC. Opioid-induced pruritus: an update. Clin Exp Dermatol 2010; 35: 2-6.
- Habib AS, White WD, Eubanks S, Pappas TN, Gan TJ. A randomized comparison of a multi- modal management strategy versus combination antiemetics for the prevention of postoperative nausea and vomiting. Anesth Analg 2004; 99: 77-81.
- Gan T, Sloan F, Dear Gde L, El-Moalem HE, Lubarsky DA. How much are patients willing to pay to avoid Postoperative nausea and vomiting. Anesth Analg 2001; 92: 393-400.
- Kim SH, Chun DH, Chang CH, Kim TW, Kim YM, Shin YS. Effect of caudal block on sevoflurane requirement for lower limb surgery in children with cerebral palsy. Paediatr Anaesth 2011; 21: 394-398.
- Ashgan RA, Sameh AS, Shawky A, Rahman MA. Bilateral sphenopalatine ganglion block as adjuvant to general anaesthesia during endoscopic trans-nasal resection of pituitary adenoma. Egyp J Anaesth 2010; 26: 273-280.
- Zhang J, Zhang W, Li B. The effect of epidural anesthesia with different concentrations of ropivacaine on sevoflurane requirements. Anesth Analg 2007; 104: 984-986.
- Shin S, Bai SJ, Rha KH, So Y, Oh YJ. The effects of combined epidural and general anesthesia on the autonomic nervous system and bioavailability of nitric oxide in patients undergoing laparoscopic pelvic surgery. Surg Endosc 2013; 27: 918-926.
- Zhou X, Song FH, He W, Yang XY, Zhou ZB, Feng X, Zhou LH. Neonatal exposure to sevoflurane causes apoptosis and reduces nNOS protein expression in rat hippocampus. Mol Med Rep 2012; 6: 543-546.
- Gonano C, Kettner SC, Ernstbrunner M, Schebesta K, Chiari A, Marhofer P. Comparison of economical aspects of interscalene brachial plexus blockade and general anaesthesia for arthroscopic shoulder surgery. Br J Anaesth 2009; 103: 428-433.
- Fontaine P, Jacques J, Gillain D, Sermeus W, Kolh P, Gillet P. Assessing the causes inducing lengthening of hospital stays by means of the Appropriateness Evaluation Protocol. Health Policy 2011; 99: 66-71.
- Oddoye JP, Jones DF. Combining simulation and goal programming for healthcare planning in a medical assessment unit. Eur J Oper Res 2009; 193: 250-261.
- Ellison AM, Bauchner H. Socioeconomic status and length of hospital stay in children with vaso-occlusive crises of sickle cell disease. J Natl Med Assoc 2007; 99: 192-196.