Research Article - Biomedical Research (2017) Volume 28, Issue 11
Correlation between hyperhomocysteine and serum cystatin C in diabetic nephropathy
Jian-Jin Guo1*, Wei Ren2, Xing Li1, Guang-Xia Xi2 and Jie Liu3
1Department of Endocrinology and Metabolism, the Second Clinical Medical College of Shanxi Medical University, Taiyuan, PR China
2Department of Endocrinology and Metabolism, Shanxi Academy of Medical Sciences, Shanxi Da Yi Hospital, Taiyuan, PR China
3Shanxi Province People’s Hospital, Taiyuan, PR China
- *Corresponding Author:
- Jian-Jin Guo
Department of Endocrinology and Metabolism
The Second Clinical Medical College of Shanxi Medical University, PR China
Accepted date: April 10, 2017
Abstract
The aim of this study was to investigate the changes and correlations of plasma Homocysteine (Hcy) and serum Cystatin C (Cys C) under different urinary albumin excretion rates in diabetic nephropathy. According to the 24 h urinary albumin excretion rate, 90 type 2 diabetes patients were divided into three groups: normal albuminuria group (n=27), microalbuminuria group (n=37), and macroalbuminuria group (n=17); 30 healthy subjects were included as controls. Fasting plasma glucose, glycosylated hemoglobin, triglyceride, cholesterol, serum creatinine, Hcy, and Cys C levels were determined, and the bivariate correlation analysis was used for linear correlation analysis. The Hcy values of the three groups were 8.46 ± 2.72, 9.43 ± 2.13, and 13.52 ± 5.24, respectively; these values were significantly different (P<0.01). The Cys C values of the three groups were also significantly different (P<0.01). As urine albumin excretion and aggravated glomerular filtration function increased, Hcy and Cys C levels also increased, showing a positive correlation (r=0.641, r=0.892, and r=0.765, P<0.01 in three groups, respectively). Combined detection of Hcy and Cys C can be used to predict the impacts of Hcy on glomerular filtration function and degree of renal damage of diabetic nephropathy. Blood Hcy levels should be evaluated at an early clinical stage.
Keywords
Blood homocysteine, Cystatin C, Diabetic nephropathy
Introduction
Diabetic Nephropathy (DN) is a major microvascular complication of diabetes and the main cause of end-stage renal disease. With the rapidly increasing incidence of diabetes worldwide and as survival time of diabetic patients is prolonged, the prevalence ratio of diabetes-caused chronic kidney diseases is also increasing each year [1]. In recent years, numerous domestic and international epidemiological surveys have shown that Homocysteine (Hcy) is an independent risk factor of cardiovascular diseases [2]. Hcy has also been linked to neurological disorders [3], aging [4], and all-cause mortality [5]. Most researchers also predict that Hcy is related to the microvascular complications of diabetes [6,7]. Elevated Hcy levels in the blood caused by deficiencies in sulfur amino acid metabolism were reported to be associated with vascular disease [8,9]. Protein homocysteinylation and oxidative stress are directly triggered by Hcy [10]. Hcy is formed during methionine metabolism by S-adenosyl-Lhomocysteine hydrolase, which catalyses the reversible hydrolysis of AdoHcy to Hcy and adenosine. Hcy has emerged as a more sensitive indicator of cardiovascular disease and of microvascular complications of diabetes. Cystatin C (Cys C) is a secreted protein found in cerebrospinal fluid, saliva, urine, and semen. The highest and lowest concentrations are in the cerebrospinal fluid and urine, respectively [11]. Cys C can also freely permeate the glomerulus. After degradation of renal tubular reabsorption, nearly no effect is detected under physiological conditions Cys C stability factor. Cys C is constantly produced via glomerular filtration excretion in the renal tubules that is reabsorbed completely degraded [12]. In many diabetic patients with renal failure requiring renal dialysis, Glomerular Filtration Rate (GFR) in renal clearance can be estimated and the status of renal function can be further evaluated in diabetic patients [13]. Numerous reports confirmed that serum Cys C is an ideal endogenous indicator of glomerular function and can be used as a specific and accurate assessment marker for evaluating glomerular filtration function. Therefore, Hcy and Cys C in type 2 diabetes patients were detected to determine the changes in these indicators at different DN stages and to analyze the relevance of these changes.
Methods
Subjects
Ninety type 2 diabetes patients diagnosed in our hospital from August 2007 to December 2007 were enrolled and met the diagnostic criteria of ADA (2014 edition); 32 cases were male and 28 cases were female, subjects were aged 30-69 years, and diabetes duration was 0.5-18 years. This study was conducted in accordance with the declaration of Helsinki. This study was conducted with approval from the Ethics Committee of Shanxi Medical University. Written informed consent was obtained from all participants. According to the 24 h urinary albumin excretion rate, the patients were divided into three groups: normal albuminuria group (UAER<30 mg/24 h, n=27), microalbuminuria group (UAER 30-300 mg/24 h, n=37), and macroalbuminuria group (UAER>300 mg/24 h, n=17). Three groups were matched for age, gender, and disease duration, and the serum creatinine levels of all selected subjects were<115 M. The exclusion criteria were as follows: had fever, urinary tract infections, or other kidney diseases within the last month, had ketoacidosis or acute complications such as hyperosmolar nonketotic coma, combined with other cardiovascular and peripheral major vascular complications and high blood disease, administrated folic acid, vitamin B12, and vitamin B6 within the past 3 months, administrated metformin, carbamazepine, phenytoin, and diuretics within the past month, others diseases, such as non-diabetic nephropathy-caused renal failure, pernicious anemia, leukemia, thyroid function loss, and psoriasis, which would disturb the metabolism of vitamin B12, vitamin B6, and folic acid, smoking or quit-smoking for less than 1 year, caffeine administration, long-term alcohol consumption and high methionine diet, or had performed regular alternative therapy. Thirty healthy subjects were included as controls, including 10 males and 10 females, aged 30-65 years, without hypertension, cardiovascular diseases, endocrine diseases, or other chronic diseases, and did not take folic acid, vitamin B12, or vitamin B6 for the past three months.
Sampling treatment
Twenty-four-hour urine specimens were collected from all subjects and preserved in 10 ml benzoic acid. The total amount of urine was recorded after mixing, and a 3 ml specimen was sent for UAER detection; after fasting for 10 h, 3 ml overnight fasting venous blood was sampled, placed in a heparin tube, centrifuged within 1 h of sampling at 4°C to separate the plasma for immediate Hcy level detection, and stored in the cold. Another 3 ml blood was centrifuged within 3 h of sampling and the serum was sent for Cys C determination.
Detection methods
UAER was detected by radioimmunoassay, plasma Hcy was detected by the cyclic enzyme method, and serum Cys C was evaluated by immunoturbidimetric assay (kit provided by Beijing Jiuqiang Co., Beijing, China). Fasting plasma glucose (FPG), triglyceride, and Serum creatinine (Scr) were detected with an automatic biochemical analyser. HbA1c was detected by high-performance liquid chromatography. The MDRD equation was used to determine the estimated GFR (eGFR)=186 (Scr/88.4)-1.154 × (Age)-0.203 × (0.742 female), and Scr values were used to calculate the Scr clearance rate (Ccr).
Statistical analysis
The data were expressed as ͞x ± SD; statistical analysis was conducted using SPSS 12.0 statistical software (SPSS Inc., Chicago, IL, USA). Multiple group comparison was conducted by multivariate analysis of variance, while bivariate correlation analysis was used for linear correlation analysis. A P<0.05 was considered to indicate a significant difference.
Results
Clinical data
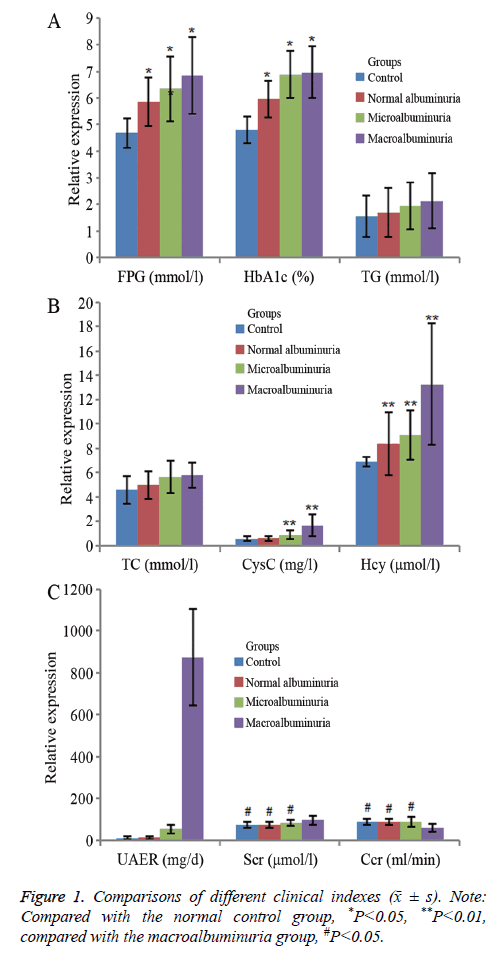
Comparison of clinical parameters among the DN groups and normal control group, which included subjects of similar ages and genders, revealed that FPG and HbA1c levels were higher in the DN groups (P<0.05), while FPG, HbA1c, and triglyceride levels among the DN subgroups showed no significant difference (P>0.05). The Scr and Ccr levels in the microalbuminuria group were not significantly different from the normal albuminuria group and control group (P>0.05). The Ccr level was significantly lower in the macroalbuminuria group than in the microalbuminuria group (P<0.05, Figure 1).
Comparison of Hcy and Cys C
Comparison of Hcy and Cys C among groups revealed that the Cys C levels in the normal control group showed a gradually increasing trend, but there was no significant difference in Cys C between the normal control group and normal albuminuria group (P>0.05). Among the DN subgroups, Cys C levels were significantly significant (P<0.01), and Cys C was significantly negatively correlated with Ccr (r=-0.842, P<0.01). Hcy levels in all DN groups were higher than in the normal control group (P<0.05); among the DN subgroups, Hcy increased as microalbuminuria level increased. Comparisons of the three groups revealed significant differences (P<0.01), which were also negatively correlated with Ccr (r=-0.623, P<0.01).
Correlation analysis
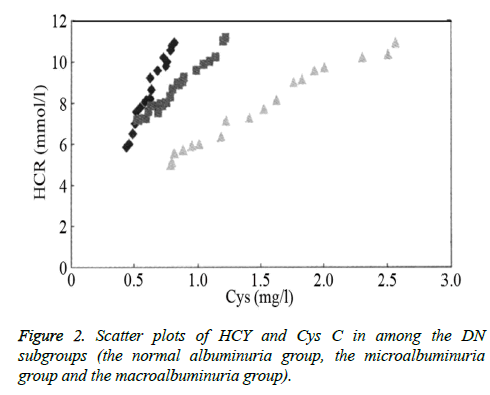
Correlation analysis of Hcy and Cys C among the DN subgroups, and the levels of Hcy and Cys C in each group were positively correlated (normal albuminuria group r=0.641, microalbuminuria group r=0.892, macroalbuminuria group r=0.765, P<0.01), among which the microalbuminuria group showed the most significant results; thus, there was a significant linear positive correlation between the two indices (Figure 2).
Discussion
DN has a complex pathogenesis and is a severe microvascular complication of diabetes and the main cause of diabetes-related mortality and mutilation. Epidemiological surveys have shown that the mortality rate of DM patients with DN is 30-fold higher than in those without kidney disease complications; therefore, early diagnosis and treatment of DN is important for improving the quality of life of DM patients [14].
DN is known to be closely related to high blood sugar, hypertension, and high cholesterol. Numerous domestic and foreign studies in recent years have shown that Hcy, as a vascular injury amino acid, is associated with DN. Additionally, Cys C was confirmed to be a sensitive and specific indicator of glomerular filtration function. Thus, we examined the changes and correlation of Hcy and Cys C in different DN periods. Hcy is a cytotoxic and genotoxic endogenous pathogenic factor and may cause blood vessel damage through various mechanisms, thus leading to atherosclerosis [15,16]. The kidney is not only the Hcy excretion organ, but also its metabolic organ. Hyperhomocysteinaemia (Hhcy) would mutually influence renal function impairment in DN patients [17]. Most studies showed that Hhcy was related with UAER and DN and considered Hcy as the UAER factor, independently from hypertension, diabetes, protein intake, and renal function [18]. This study detected Hcy levels during different DN phases and found that with decreased glomerular filtration function, blood Hcy concentrations increased, and Ccr and Hcy were significantly negatively correlated (r=-0. 623, P<0.01).
Cys C is one of the 9 subtypes of serum cystatin C and can freely pass through the glomerular filtration membrane and completely reabsorbs in the proximal tubule but does not return to the blood cycle. However, the renal tubular does not secrete Cys C [19]. Thus, Cys C can be used as an indicator of GFR and is considered a sensitive and specific index of renal functions. Recent studies suggested that the renal clearance rate of Cys C is the same as that of the radionuclide mTc- DTPA. Recently, Rule et al. also evaluated Cys C as a predictor of GFR and found partial improvement in the accuracy of GFR prediction values using this marker [19-21]. Numerous studies have confirmed that serum Cys C concentrations are significantly negatively correlated with GFR. When glomerular filtration function is subjected to minor injury, the blood Cys C concentration increases, gradually aggravating disease situations [22,23]. This study also showed that Cys C was significantly negatively correlated with UAER and Ccr. When Scr and Ccr were normal, Cys C was increased; thus, Cys C may be useful for earlier detection of glomerular filtration dysfunction.
Cys C may be useful for measuring GFR; as a factor involved in DN vascular injury, Hcy can be detected in combination with Cys C to determine the effect of Hcy on GFR, detect early DN damage, and more comprehensively evaluate early diabetic kidney damage from different perspectives. Additionally, detection of this marker may enable early clinical intervention for Hhcy in diabetic patients to reduce DN incidence, prevent and slow DN progression, and help to change the DN course. We found that the Cys C level in the microalbuminuria group was significantly higher than in the normal albuminuria group, indicating that during the early diabetic kidney damage stage, Cys C shows corresponding changes and thus can be used as an indicator for the early diagnosis of type 2 DN. Simultaneous detection of Cys C and Hcy may improve the accuracy of UAER in DN diagnosis. Hcy increases during early DN stages, and Hcy levels were positively correlated with UAER levels, further suggesting that Hhcy is related to the degree of DN microalbuminuria. Thus, with increasing urine albumin, glomerular filtration dysfunction, Hcy level, and Cys C level also increase; Hcy and Cys C showed a positive correlation and were much more significantly correlated during early DN stages.
Acknowledgements
This study was supported by National Natural Science Foundation of China Youth Found (81400836), The Science Foundation of Health and Family Planning Commission of Shanxi Province (20161004, 201601052).
Conflicts of Interest
All of the authors declare that they have no conflicts of interest regarding this paper.
References
- Afkarian M. Diabetic kidney disease in children and adolescents. Pediatr Nephrol 2015; 30: 65-74.
- Huang T, Chen Y, Yang B, Yang J, Wahlqvist ML, Li D. Meta-analysis of B vitamin supplementation on plasma homocysteine, cardiovascular and all-cause mortality. Clin Nutr 2012; 31: 448-454.
- Zou Z, Lu Y, Dong M, Yang H. Effect of homocysteine on voltage-gated sodium channel currents in primary cultured rat caudate nucleus neurons and its modulation by 2-arachidonylglycerol. J Mol Neurosci 2015; 57: 477-485.
- Tassino M, Campos TF, Guerra RO. Homocysteine (Hcy) and cognitive performance in a population sample of elderly Brazilians. Arch Gerontol Geriatr 2009; 48: 142-145.
- El-Khairy L, Vollset SE, Refsum H, Ueland PM. Plasma total cysteine, mortality, and cardiovascular disease hospitalizations: the Hordaland Homocysteine Study. Clin Chem 2003; 49: 895-900.
- Karabag T, Kaya A, Temizhan A, Koē F, Yavuz S, Cam S. The influence of homocysteine levels on endothelial function and their relation with microvascular complications in T2DM patients without macrovascular disease. Acta Diabetologica 2007; 44: 69-75.
- Upadhyay TR, Kothari N, Shah H. Association between serum b12 and serum homocysteine levels in diabetic patients on metformin. J Clin Diagn Res 2016; 10: 1-4.
- Akalin A, Alatas O, Colak O. Relation of plasma homocysteine levels to atherosclerotic vascular disease and inflammation markers in type 2 diabetic patients. Eur J Endocrinol 2008; 158: 47-52.
- Ozdemir O, Yakut A, Dinleyici EC, Aydogdu SD, Yarar C, Colak O. Serum asymmetric dimethylarginine (ADMA), homocysteine, vitamin B 12, folate levels, and lipid profiles in epileptic children treated with valproic acid. Eur J Pediatr 2011; 170: 873-877.
- Jakubowski H. Homocysteine thiolactone: metabolic origin and protein homocysteinylation in humans. J Nutr 2000; 130: 377-381.
- Pavkov ME, Knowler WC, Hanson RL, Williams DE, Lemley KV, Myers BD, Nelson RG. Comparison of serum cystatin C, serum creatinine, measured GFR, and estimated GFR to assess the risk of kidney failure in American Indians with diabetic nephropathy. Am J Kidney Dis 2013; 62: 33-41.
- Steffes MW. Estimating GFR from cystatin C in serum: seeking to enhance its clinical application. Am J Kidney Dis 2006; 48: 842-843.
- Biesenbach G, Pohanka E. Dialysis in diabetic patients: hemodialysis and peritoneal dialysis. Pros and cons. Minerva Urol Nefrol 2012; 64: 173-182.
- Zürbig P, Jerums G, Hovind P, Macisaac RJ, Mischak H, Nielsen SE, Panagiotopoulos S, Persson F, Rossing P. Urinary proteomics for early diagnosis in diabetic nephropathy. Diabetes 2012; 61: 3304-3313.
- Ezekwe KA, Adegboye ARA, Gamborg M, Heitmann BL. Associations between initial change in physical activity level and subsequent change in regional body fat distributions. Obesity Facts 2013; 6: 552-560.
- Rossing K, Mischak H, Dakna M, Zürbig P, Novak J. Urinary proteomics in diabetes and CKD. J Am Soc Nephrol 2008; 19: 1283-1290.
- Sen U, Pushpakumar SB, Amin MA, Tyagi SC. Homocysteine in renovascular complications: Hydrogen sulfide is a modulator and plausible anaerobic ATP generator. Nitric Oxide 2014; 41: 27-37.
- Ash S, Campbell KL, Bogard J, Millichamp A. Nutrition prescription to achieve positive outcomes in chronic kidney disease: a systematic review. Nutrients 2014; 6: 416-451.
- Oh MY, Lee H, Kim JS, Ryu WS, Lee SH. Cystatin C, a novel indicator of renal function, reflects severity of cerebral microbleeds. BMC Neurol 2014; 14: 127.
- Stevens LA, Coresh J, Schmid CH, Feldman HI, Froissart M, Kusek J, Rossert J, Van Lente F, Bruce RD 3rd, Zhang YL, Greene T, Levey AS. Estimating GFR using serum cystatin C alone and in combination with serum creatinine: a pooled analysis of 3,418 individuals with CKD. Am J Kidney Dis 2008; 51: 395-406.
- Rule AD, Bergstralh EJ, Slezak JM, Bergert J, Larson TS. Glomerular filtration rate estimated by cystatin C among different clinical presentations. Kidney Int 2006; 69: 399-405.
- Wagner D, Kniepeiss D, Stiegler P, Zitta S, Bradatsch A, Robatscher M, Müller H, Meinitzer A, Fahrleitner-Pammer A, Wirnsberger G, Iberer F, Tscheliessnigg K, Reibnegger G, Rosenkranz AR. The assessment of GFR after orthotopic liver transplantation using cystatin C and creatinine-based equations. Transpl Int 2012; 25: 527-536.
- Ensrud KE, Parimi N, Fink HA, Ishani A, Taylor BC, Steffes M, Cauley JA, Lewis CE, Orwoll ES, Osteoporotic Fractures in Men Study Group. Estimated GFR and risk of hip fracture in older men: comparison of associations using cystatin C and creatinine. Am J Kidney Dis 2014; 63: 31-39.