Research Article - Biomedical Research (2016) Volume 27, Issue 4
Component analysis of prostatic calculi and the causes of calculus formation
Wei Guo1#*, Meini Chen2#, Xiaolong He1, Ansheng Bai1, Jixue Gao1, Ye Zhang1, Yi Li1 and Jing Wen11Department of Urinary Surgery, Yanan University Affiliated Hospital, Yanan 71600, PR China
2Experimental Centers of Medical College, Yanan University, Yanan 716000, PR China
#These authors contributed equally to this work.
- *Corresponding Author:
- Wei Guo
Department of Urinary Surgery
Yanan University Affiliated Hospital
No. 250 Center Street
Yanan 716000
China
Accepted date: April 14, 2016
Abstract
Objective: To analyze the chemical components of prostatic calculi.
Method: An infrared automatic analyzer was used to determine the chemical components of prostatic calculi from 86 patients, and the causes of formation and related clinical data were examined.
Results: Magnesium ammonium phosphate and carbapatite were found in 84.9% of calculi, and the difference in the comparison with urinary infection was statistically significant. The components of prostatic calculi differed significantly from those of the urinary tract, and their formation was related to many factors.
Conclusions: Prevention of prostate infection can effectively reduce the occurrence of prostatic calculi; healthy lifestyles and habits are of great significance in calculus formation.
Keywords
Prostatic calculi, Component, Analysis, Urinary calculi.
Introduction
Prostatic calculi have a high occurrence rate, which is related to the improvement in living standards and changes in dietary structure and lifestyle. Prostatic calculi are common, and are related to prostatitis, which can lead to prostatic cancer [1,2], but there is no link between calculi and prostatic cancer [3]. Calculi are formed in prostatic tissue and acini, and are caused by the deposition of calcified substances on the corpora amylacea [4]. Some studies have reported that prostatic calculi are seen in patients with recurrent bacterial urinary infection [5], which may be a cause of calculi. Other causes include radiotherapy, transurethral prostatectomy, prostate stent therapy, and ochronosis due to alkaptonuria. Patients with prostatic calculi usually have no symptoms, but large calculi may result in uroschesis, prostatitis, and chronic pelvic pain syndrome [6,7]. Bladder stones in men with benign prostatic hyperplasia are thought to be secondary to bladder outlet obstruction induced by urinary stasis [8]. It is still not clear whether formation of prostatic calculi is related to benign prostatic hyperplasia. Moreover, there are no uniform standards for treatment and prevention of prostatic calculi. It has been reported that prostate cancer is related to chronic prostatitis [9], but not to urinary calculi [10].
Most patients with prostatic calculi present with symptoms of prostate hyperplasia, urethral stenosis, or chronic prostatitis. Calculi are usually detected when patients are evaluated for other conditions. Prostatic calculi generally cause no obvious symptoms and need no treatment. For some patients with clinical symptoms, there is no optimal treatment, and the calculi are often removed by transurethral resection. When younger men undergo prostate surgery, the postoperative incidence of impotence and retrograde ejaculation is increased. Therefore, prevention of prostatic calculi has gained increasing attention. Whether prostatic calculi are involved in the occurrence and development of prostate cancer, and whether calculi are associated with dietary structure and lifestyle remain unclear. This study analyzed the chemical components of prostatic calculi from 86 patients, and explored the causes of calculus formation. The objective is to provide guidelines for prevention and treatment of prostatic calculi.
Materials and Methods
General data
A total of 86 specimens were collected from 86 patients with prostatic calculi combined with prostate hyperplasia, which underwent surgery in our hospital from October 2009 to October 2013. Very small calculi were excluded. This study was conducted in accordance with the declaration of Helsinki. This study was conducted with approval from the Ethics Committee of Yanan University. Written informed consent was obtained from all participants.
Methods
All agents were milled to powder in a mortar, and completely desiccated in an HW-3A temperature-controlled drying cabinet for about 48 h (temperature range: 110-150°C). Calculi were collected and washed, and then dried at room temperature. The calculi were also milled to powder. Then, 200-300 mg of dried potassium bromide and 1-2 mg of powdered calculi were combined in a mortar, resulting in an optimal dispersive concentration range of calculi in potassium bromide of 5%-10%. The mixture was then dried in the drying cabinet (100°C) for 5 min, and then milled for about 30 s. Next, the powder was compressed with a performing machine (Qingdao Huaqing Group) at 20 mPa for 1 min; the tablet thickness was about 0.3-0.5 mm. The major components of the calculi were determined using an LIIR20 infrared spectroscopy automatic analyzer. The types of calculi were confirmed according to characteristic band peaks.
Statistical analysis
Statistical analysis was performed using SPSS 17.0 software and the major indicator was a component ratio. The chi-square test was used for comparisons of multiple groups, and P<0.05 indicated a significant statistical difference.
Results
Distribution of prostatic calculus composition
Of 86 samples, 69 were mixed calculi (80.2%) and 17 were simple calculi (19.8%). Calcium oxalate and carbapatite were detected in 70.9% (61/86) and 68/6% (59/86) of samples, respectively, followed by 53.4% (46/86) with ammonium magnesium phosphate hexahydrate and 7.0% (6/86) with uric acid. Infectious calculi mostly contained ammonium magnesium phosphate and carbapatite.
In this study, 73 of 86 samples were infectious calculi (84.9%) and the others were non-infectious calculi (15.1%). The compositions of prostatic calculi are shown in Table 1.
| Calculi composition | N | Percentage% | |
|---|---|---|---|
| Simplex (n=17) |
Calcium oxalate | 7 | 41.2 |
| Carbapatite | 4 | 23.5 | |
| Ammonium magnesium phosphate hexahydrate | 4 | 23.5 | |
| Uricite | 2 | 11.8 | |
| Mixture (n=69) |
Carbapatite+calcium oxalate monohydrate+calcium oxalate dehydrate | 23 | 33.3 |
| Ammonium magnesium phosphate hexahydrate+carbapatite+calcium oxalate monohydrate+calcium oxalate dehydrate | 16 | 23.2 | |
| Ammonium magnesium phosphate hexahydrate+carbapatite | 15 | 21.8 | |
| Ammonium magnesium phosphate hexahydrate+calcium oxalate monohydrate+calcium oxalate dehydrate | 10 | 14.5 | |
| Calcium oxalate monohydrate+calcium oxalate dehydrate+uricite | 4 | 5.8 | |
| Ammonium magnesium phosphate hexahydrate+carbapatite+calcium oxalate monohydrate | 1 | 1.4 |
Table 1: Distribution of the prostate calculi compositions.
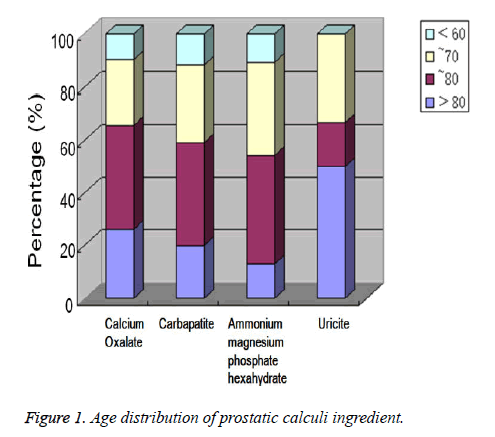
Distribution of prostatic calculus components by age
Figure 1 shows that of 86 patients with an age range from 56 to 87 years (mean: 68.21 years), 7 (8.1%) patients were aged less than 60, 26 (30.2%) were 61 to 70, 31 (36%) were 71 to 80, and 22 (25.6%) were over 80 years old. The highest detection rate for prostatic calculi in all age groups was for calcium oxalate and carbapatite. A few cases had uricite and were not analyzed. The numbers with calcium oxalate, carbapatite, and ammonium magnesium phosphate hexahydrate were compared for all ages, and showed no significant difference (P>0.05).
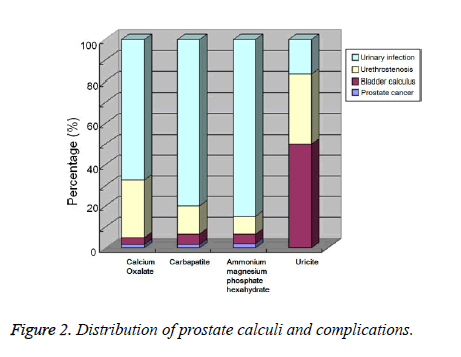
Distribution and complications of prostatic calculi
Of 86 cases, 61 (73.3%) were associated with urinary infection, 21 with urethral stenosis (24.4%), 3 with bladder calculi (3.5%), and only 1 with prostate cancer (1.2%). In the urinary infection group, the detection rates of calcium oxalate, carbapatite, and ammonium magnesium phosphate hexahydrate were obviously higher than those for uricite. In the urethral stenosis group, the detection rates of calcium oxalate, carbapatite, and ammonium magnesium phosphate hexahydrate were obviously lower than those for uricite. Because there were few cases with calcium oxalate, carbapatite, and ammonium magnesium phosphate hexahydrate, the results were subjected to a statistical analysis. There was a statistically significant difference between the compositions of prostatic calculi and urinary complications (P<0.05, Figure 2).
Discussion
Prostatic calculi show a high incidence rate in prostatic disease and the rate increases with age. About 5% of men have prostatic calculi, especially those more than 50 years old. Prostatic calculi account for 5.3% of urinary calculi. Benign prostatic hyperplasia is also a common disease of older men, and the incidence rate of those more than 50 years is 48.91%. Furthermore, the incidence rate of prostatic calculi combined with prostatic hyperplasia is as high as 68.52% [10], which reflects the fact that prostatic calculi are part of the aging process. It was reported that the number of patients found to have prostatic calculi increased with age, but the association was not statistically significant [7,11]. This study collected prostatic calculi from elderly patients with bladder outlet obstruction or lower urinary tract symptoms, including 85 cases with prostatic hyperplasia and 1 with prostate cancer; the ages ranged from 56 to 87, including 7 cases more than 60 years old (8.1%), 26 from 61 to 70 (30.2%), and 31 from 71 to 80 (36.0%). This study mainly included elderly patients and the incidence rate increased with age. Calcium oxalate calculi and carbonated apatite were commonly seen in clinical practice in all age groups, and age was considered an important factor in the formation of prostatic calculi.
Presently, the mechanism of prostatic calculus formation remains unclear; some reports showed that prostatic calculi are made up of the deposition of sloughed epithelium and prostatic secretions, and the major components include calcium phosphate and calcium carbonate [12]. Possible contributors to the formation of prostatic calculi include infections, which promoted the shedding of epithelial cells and the blockage of prostatic crypts. For example, when chronic inflammation develops in the prostate, the alveoli dilate and the ducts become narrowed; this might result in a backflow of urine into the prostate, and crystals in the urine could precipitate in the glandular tissue and form calculi. In addition, when inflammation occurs, the components of prostatic fluid change, and foreign matter including leukocytes and bacteria increase; inorganic salts or Ca2+ adhere more easily and then form calculi. Other authors suggested that microcalcification inside the prostate was a benign process of intra-prostate dystrophy and prostate dysfunction [13,14]; similarly, there was also a correlation between inflammation, location of bacterial infection, and their duration [15]. Recent studies confirmed that nanobacteria may be a critical etiological factor in prostatitis [16]. It is still unclear whether infections play primary roles or are just a minor complication during the formation of prostatic calculi. This study analyzed the components of 86 calculi using infrared spectroscopy, and the results showed that mixed calculi were the predominant type, with a rate of 80.2%; 26.7% were a mixture of carbapatite, calcium oxalate monohydrate, and calcium oxalate dehydrate; 18.6% were a mixture of ammonium magnesium phosphate hexahydrate, carbapatite, calcium oxalate monohydrate, and calcium oxalate dihydrate. Calcium oxalate calculi and carbapatite had high detection rates in all components of prostatic calculi, at 70.9% and 68.6%, respectively. The above results were in line with previous studies.
Studies have demonstrated that the occurrence of prostatic calculi was related to prostate hyperplasia, prostatitis, and prostatic cancer [17]. Calculi are classified into four types, according to the primary components: calcium, infectious, uric acid, and cystine. Infectious calculi contain ammonium magnesium phosphate and carbapatite [18]; 84.9% of calculi in this study were infectious. Furthermore, an analysis of the relationship between prostatic calculi and hyperplasia revealed that 70.73% of patients had an associated urinary infection, and that infectious calculi comprised of an average 68.6% carbapatite and 53.4% ammonium magnesium phosphate hexahydrate, indicating that prostatic calculi were correlated with urinary infection. It is still unclear whether these symptoms are caused by the calculi or the associated glandular inflammation. Some studies also reported that the major types of lower urinary tract calculi in adult men were simple uric acid or mixed uric acid calculi [19]. Uric acid could promote the formation of calcium oxalate calculi. With prostate inflammation or hyperplasia, expanding acini cause tube narrowing and force urine reflux, inducing formation of calculi [20]. In this study, 6 calculi, including 3 bladder calculi, contained uric acid. This implied that prostatic uric acid calculi might be related to urine reflux. Therefore, in patients with prostatic calculi plus urinary infection, improvement in living conditions, enhanced medical care, and timely treatment of urinary infections would be very important in prevention of prostatic calculi.
In this study, the incidence of infectious prostatic calculi was 84.9%, which was significantly higher than that for urinary calculi reported in a previous study [21]. This indicated that the formation of prostatic calculi is related to prostate infection. Some studies suggested that the recurrence rate of infectious calculi was higher than for other types of calculi, which might be related to recurrent infection of the prostate with bacteria that are difficult to eradicate. One study found that patients with chronic prostatitis/recalcitrant chronic pelvic pain syndrome who received anti-nanobacterial therapy had a decrease in size of some prostatic calculi after therapy [22]. The formation of calcium oxalate calculi was associated with congenital urinary tract deformities, primary hyperparathyroidism, and renal tubular acidosis; moreover, patients whose diet includes foods with high oxalate, protein, salt, tea, and alcohol develop calculi more easily. Uric acid is the final product of purine metabolism, and uric acid calculi are caused by hyperuricemia and hyperuricuria. Acquired uric acid calculi are mostly found in patients with gout, malignant tumors, and gastrointestinal diseases; of course, patients who consume acidic foods, meat, seafood products, and alcohol have a greater risk of developing calculi.
The subjects in this study were patients with prostatic calculi who were treated in our hospital; thus, there were regional limitations, and the occurrence of prostatic calculi might be related to geographical environment, lifestyle, dietary habits, medical care, etc. In conclusion, this study analyzed the components of prostatic calculi and found that their formation is related to many factors, especially infection. Prevention of prostate infection can effectively reduce the occurrence of prostatic calculi; healthy lifestyles and habits are of great significance in prevention of prostatic calculi. This study can provide evidence to guide the prevention and treatment of prostatic calculi.
Acknowledgements
Supported by Scientific research of Yanan University affiliated Hospital (2012YDFY-Y316) and the Key Experimental Programs of Tumor Prevention of Yanan City (No.2014-78).
Conflicts of interest
All of the authors declare that they have no conflicts of interest regarding this paper.
References
- Nelson WG, De Marzo AM, De Weese TL, Isaacs WB. The role of inflammation in the pathogenesis of prostate cancer. J Urol 2004; 172:S6-S11.
- Palapattu GS, Sutcliffe S, Bastian PJ, Platz EA, De Marzo AM, Isaacs WB, Nelson WG. Prostate carcinogenesis and inflammation: emerging insights. Carcinogenesis 2005; 26: 1170-1181.
- Hwang EC, Choi HS, Im CM, Jung SI, Kim SO, Kang TW, Kwon DD, Park KS, Ryu SB. Prostate calculi in cancer and BPH in a cohort of Korean men: presence of calculi did not correlate with cancer risk. Asian J Androl 2010; 12: 215-220.
- Kodaka T, Hirayama A, Sano T, Debari K, Mayahara M, Nakamura M. Fine structure and mineral components of primary calculi in some human prostates. J Electron Micros 2008; 57: 133-141.
- Zhao WP, Li YT, Chen J, Mayahara M, Nakamura M. Prostatic calculi influence the antimicrobial efficacy in men with chronic bacterial prostatitis. Asian J Androl 2012; 14: 715-719.
- Bedir S, Kilciler M, Akay O, Erdemir F, Avci A, Ozgök Y. Endoscopic treatment of multiple prostatic calculi causing urinary retention. Int J Urol 2005; 12: 693-695.
- Geramoutsos I, Gyftopoulos K, Perimenis P, Thanou V, Liagka D, Siamblis D, Barbalias G. Clinical correlation of prostatic lithiasis with chronic pelvic pain syndromes in young adults. Eur Urol 2004; 45: 333-337.
- Childs MA, Mynderse LA, Rangel LJ, Wilson TM, Lingeman JE, Krambeck AE. Pathogenesis of bladder calculi in the presence of urinary stasis. J Urol 2013; 189: 1347-1351.
- Vral A, Magri V, Montanari E, Gazzano G, Gourvas V, Marras E, Perletti G. Topographic and quantitative relationship between prostate inflammation, proliferative inflammatory atrophy and low-grade prostate intraepithelial neoplasia: a biopsy study in chronic prostatitis patients. Int J Oncol 2012; 41: 1950-1958.
- Chung SD, Liu SP, Lin HC. Association between prostate cancer and urinary calculi: a population-based study. PLoS One 2013; 8: e57743.
- Mori R, Kodaka T, Sano T. Preliminary report on the correlations among pineal concretions, prostatic calculi and age in human adult males. Anatsci int 2003; 78:181-184.
- Demir O, Kefi A, Cahangirov A, Cihan A, Obuz F, Esen AA, Celebi I. The giant calculus within the prostatic urethra. Urol Res 2011; 39: 319-321.
- Sfanos KS, Wilson BA, De Marzo AM, Isaacs WB. Acute inflammatory proteins constitute the organic matrix of prostatic corpora amylacea and calculi in men with prostate cancer. Proc Natl Acad Sci 2009; 106: 3443-3448.
- Mi H, Gao Y, Yan Y, Wu Y, Tan A, Yang X, Zhang H, Zhang Y, Lv W, Mo Z. Research of correlation between the amount of leukocyte in EPS and NIH-CPSI: result from 1242 men in Fangchenggang Area in Guangxi Province. Urology 2012; 79: 403-408.
- Shoskes DA, Lee CT, Murphy D, Kefer J, Wood HM. Incidence and significance of prostatic stones in men with chronic prostatitis/chronic pelvic pain syndrome. Urology 2007; 70: 235-238.
- Shen X, Ming A, Li X, Zhou Z, Song B. Nanobacteria: a possible etiology for type III prostatitis. J Urol 2010; 184: 364-369.
- Usta MF, Baykara M, Erdogru T. Idiopathic prostatic giant calculi in a young male patient. IntUrolNephrol 2005; 37: 295-297.
- Köseoğlu H, Aslan G, Sen BH, Tuna B, Yörükoğlu K. Prostatic calculi: silent stones. ActasUrolEsp 2010; 34: 555-559.
- Bellanato J, Cifuentes JL, Salvador E, Medina JA. Urates in uric acid renal calculi. Int J Urol 2009; 16: 318-321.
- Marchini GS, Sarkissian C, Tian DV, Gebreselassie S, Monga M. Gout, stone composition and urinary stone risk: a matched case comparative study. J Urol 2013; 189: 1334-1339.
- Sun X, Shen L, Cong X, Zhu H, He L, Lu J. Infrared spectroscopic analysis of 5,248 urinary stones from Chinese patients presenting with the first stone episode. Urol Res 2011; 39: 339-343.
- Shoskes DA, Thomas KD, Gomez E. Anti-nanobacterial therapy for men with chronic prostatitis/chronic pelvic pain syndrome and prostatic stones: preliminary experience. J Urol 2005; 173: 474-477.