- Biomedical Research (2012) Volume 23, Issue 2
Comparative Study of Intrathecal Midazolam and Ketamine with Bupivacaine for Post-Operative Analgesia in lower limb and perianal surgery
Abdul Muthalib Hussain*, Badurudeen Mahmood Buhary; Zikrullah Tamanna
Main Hospital, King Fahad Medical City, Riyadh PO Box 59046, Saudi Arabia.
- *Corresponding Author:
- Abdul Muthalib Hussain, MD.,
King Fahad Medical City
Main Hospital, PO Box 59046
Riyadh 11525
Saudi Arabia.
Accepted date: February 22 2012
Abstract
Spinal anesthesia with bupivacaine is routinely administered for lower limb and perianal surgery. The ensuing nerve blockage is sufficient to provide adequate motor blockage, which facilitates the surgeon’s work and also provides effective pain relief during the initial postoperative period. In order to maximize post-operative analgesia, a number of adjutants have been added to local spinal anesthetics. Intrathecal supplements for post-operative pain relief are intriguing as they eliminate the need for intravenous and intramuscular analgesics and their associated complications. The objective of this study is to compare the intensity and duration of post-operative pain relief using intrathecal ketamine and midazolam with bupivacaine in patients undergoing lower limb and perianal surgery. This prospective, open label, parallel assignment, randomized, single-center trial, included eighty patients, who admitted for lower limb and perianal surgeries to the M.S. Ramaiah Medical College and Hospital, University- affiliated tertiary care center in Bangalore, India, were studied for 6 months. ASA (American Society of Anesthesiology) grade I and II patients between the ages of 20 and 60 years were included in this study. The onsets of action, intra-operative vital signs, postoperative vital signs, pain assessment by visual analogue scale, and post- operative analgesia time were recorded. A significantly higher VAS score were seen in group I (Ketamine). Postoperative analgesia was supplemented in all patients in group I at a mean duration of 482 ± 68.22 minutes and in group II at a mean duration of 645 61.28 minutes. The difference in mean post-operative supplemental analgesic time between the 2 groups was very highly significant (p < 0.001). Intrathecal midazolam with bupivacaine provides very good and prolonged post-operative analgesia compare to intrathecal ketamine with bupivacaine.
Keywords
Analgesia, intrathecal analgesia, Ketamine, Midazolam, Post-operative analgesia
Abbreviations
CSF – Cerebro Spinal Fluid
HS – Highly Significant
NS - Not Significant
SD – Standard Deviation
VAS – Visual Analogue Scale
VHS – Very Highly Significant
Introduction
Spinal anesthesia with bupivacaine is routinely administered for lower limb and perianal surgery with sufficient motor blockage to facilitate the surgeon’s work. Bupivacaine is a long acting, amide group, local anesthetic agent that is 4 times more potent than lignocaine. The minimal toxic blood concentration of bupivacaine is 2 to 4 micrograms/ cc, which makes it 15 times more toxic compared to lignocaine [5]. The Cardiovascular Collapse/Central Nervous System (CC/CNS) ratio of bupivacaine is 2.7 and a higher incidence of irreversible cardio vascular collapse is seen with bupivacaine because of the narrow CC/CNS ratio. Bupivacaine also provides effective pain relief in the initial post-operative period.
In order to maximize post-operative analgesia, a number of adjutants have been added to local spinal anesthetics. The discovery of encephalin by Hughes and endorphins by Pert and Snyder in 1975 initiated the opioid receptor theory and studies on pain mechanisms. In 1976, Yaks and Rudy reported the presence of opioid receptors in the spinal cord and they demonstrated that intrathecal administration of morphine produced dose-dependent pain relief in rats. Benzodiazepine receptors are present throughout the nervous system, including the spinal cord. Midazolam is a water-soluble benzodiazepine with sedative, amnesic, anxiolytic, muscle relaxant, and anticonvulsant properties [30, 31]. Midazolam given by intrathecal or epidural injection can also produce an antinociceptive effect. This may be Gamma-Aminobutyric Acid (GABA) mediated. The Gamma- Aminobutyric Acid has been shown to have analgesic properties. There are many uses for midazolam during the pre-operative period including premedication, anesthesia induction, and maintenance of sedation for diagnostic and therapeutic procedures [32]. Ketamine is a potent analgesic that was released in 1968 and is still employed in a variety of clinical settings. Ketamine modulates pain perception at the dorsal horn of spinal cord. NMethyl- D-Aspartate (NMDA) receptor interaction may mediate general anaesthetic effects as well as some analgesic actions of ketamine [18].
Ketamine is also the only hypnotic agent with analgesic properties. Analgesia induced by ketamine is mediated by the opiate receptors [3]. The advantages of ketamine include a good analgesic effect, cardio vascular stability in a hypotensive state, bronchodilatation in asthmatics, and the absence of awareness [9,15]. Disadvantages include increased heart rate and blood pressure, emergence phenomenon, laryngospasm and apnea, increases in intracranial and intraocular pressure, and the lack of visceral anesthesia. In 1968, ketamine was used in 1508 patients for various surgical and diagnostic procedures [2]. In 1982, further studies on the mechanisms of ketamine induced analgesia were performed [3]. Ketamine appears to be relatively free of any serious side effects and possesses advantages over local anaesthetics because it stimulates both the cardiovascular and respiratory systems [15, 17,19].
Post-operative pain relief is an unresolved issue. One of the methods of providing post-operative analgesia is by prolonging the duration of intrathecal bupivacaine using additives such as opioid [37], ketamine [19,20], or other drugs. The discovery of benzodiazepine receptors in the spinal cord has triggered the use of intrathecal midazolam for analgesia [23,24]. Intrathecal supplements for postoperative pain relief are intriguing prospects as they eliminate the need for intravenous and intramuscular analgesics and their associated complications. There are only a handful of studies that have assessed the efficacy of the combination of intrathecally administered ketamine and midazolam with bupivacaine.
We performed this study in order to compare the intensity and duration of post-operative pain relief using intrathecal ketamine and midazolam with bupivacaine in patients undergoing lower limb and perianal surgeries.
Material and Methods
Source of Data:
The study protocol was approved by the Institutional Research Ethical Committee. All patients gave written informed consent. This prospective, open label, parallel assignment, randomized, single- center trial study included eighty patients, who admitted for lower limb and perianal surgeries to the M.S. Ramaiah Medical College and Hospital, University-Affiliated tertiary care center in Bangalore, India, were studied for 6 months. American Society of Anesthesiology (ASA) grade I and II patients between the ages of 20 and 60 years were included in this study. Patients with a history of cardiac, renal, hepatic, respiratory, neurological, endocrine, coagulation disorders, and known sensitivities to study drugs or emergency surgeries were excluded from the study.
Pre-operative Preparation:
Pre-operative evaluation done by the on call anesthetists on the day prior to the surgery. Preoperative assessment was done according to ASA guideline. The patients were explained about the spinal anesthesia technique and educated regarding the Visual Analogue Scale (VAS) [Figure 1]. Advocated by Revill and Robinson in 1976. The VAS consists of a 10 cm line anchored at one end by the label “no pain” and at the other end with “the worst pain imaginable”. The main disadvantage of the VAS is the time required to measure the scale [11]. The preanaesthetic preparation of the patients included overnight fasting and preanesthesia medication consisting of oral diazepam 0.2 mgkg-1 the night before surgery. Boyles Anaesthesia Machine was checked and a standard intubation kit was prepared. In the operating theatre, the Kits were preloaded with 15 mlkg-1 intravenous Ringer’s lactate solution before administering the subarachnoid block.
Procedure
Patients were randomly allocated into 1 of 2 groups. Group I (Ketamine) received 25 mgs of preservative free ketamine with 10 mgs of 0.5% bupivacaine containing 22.5% dextrose made up to a volume of 3ml with a specific gravity of 1.036. Group II (Midazolam) received 2.5 mgs of preservative free midazolam with 10 mgs of 0.5% bupivacaine containing 16% dextrose made up to a volume of 3ml with a specific gravity of 1.035. The specific gravity of spinal anesthetic medication was maintained in both groups. Subarachnoid block was performed with the patients in the right lateral position with the table in horizontal level. With all aseptic precautions suing a 23 G spinal needle block was performed at L3-L4 level. Respective drugs were administered over a period of 15 seconds after free flow of CSF was obtained. Patients were immediately returned to the supine position and the table was maintained in the horizontal level. Standard monitoring was carried out in the from of ECG, pulse oximetry, and respiratory rate, and non invasive arterial blood pressure was recorded every 5 minutes, intra-operatively.
Hypotension, defined as a 20% decrease in systolic blood pressure from baseline values [37], was treated with intravenous fluids and 6 mg mephenteramine intravenous boluses. Bradycardia, defined as a pulse rate < 60 min-1 was treated with intravenous atropine sulphate. The sensory blockage was assessed by the loss of sensation in response to pinprick. The time to onset of the sensory block, maximum level of sensory block achieved, and time to achieve maximum sensory block were noted. A dermatomal sensory loss from T8 to S4 was considered satisfactory. Intensity of the motor blockade was assessed by the Bromage scale [33] [Table 1]. The duration of surgery for each case was noted.
No other sedative or analgesics were given to the patients during surgery. Post-operatively, patients were examined every 30 minutes for 7 hours to evaluate the duration and quality of post-operative pain relief. Pain assessment was determined using the Visual Analogue Scale (VAS) (Figure 1). Supplemental analgesia was given when the result of the VAS was greater than 4. The time of supplemental analgesia administration was noted. Following recovery, the ensuing parameters were observed: Time to regression to level L5-S1, motor power assessed by Bromage scale [Table 1], and the time of voiding urine, in minutes.
Statistical Analysis
The data are presented as mean ± SD. The results were statistically analyzed by using independent t' test described by Bonferonni. The independent t test was done to determine the statistical significance between the two groups.
In this study, we analyzed the statistical significance of the differences between Group I (Ketamine) and Group II (Midazolam). A p value > 0.05 was considered statistically not significant (NS), a p < 0.05 was considered statistically significant, a p < 0.01 was considered highly significant (HS), and a p < 0.001 was considered very highly significant (VHS).
Results
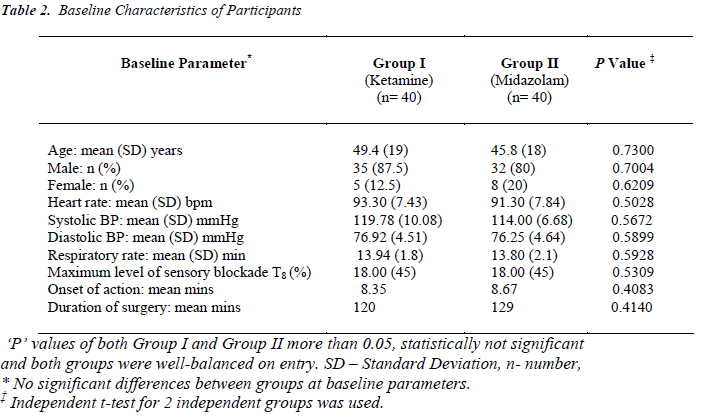
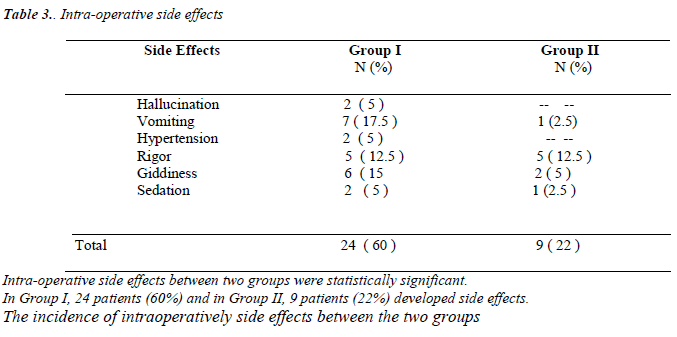
Eighty patients of both sexes participated in this study. Both groups had predominantly male patients, as shown in Table 2. The 2 treatment groups were well-balanced on entry [Table 2]. There was no significant difference in the mean onset of action between both groups; group I was 8.35 mins and Group II was 8.67 mins. The maximum level of sensory block as well as the time to onset of action was not statistically significant between groups [Table 2]. The average duration of surgery in both groups were nearly equal. In Group I, the average duration of surgery was about 120 minutes and in Group II, about 129 minutes. In Group I, 24 patients (60%) developed side effects intraoperatively. Incidence of adverse effects like vomiting, giddiness and rigor were noticed in 17.5 percent, 15 percent and 12.5 percent, respectively.
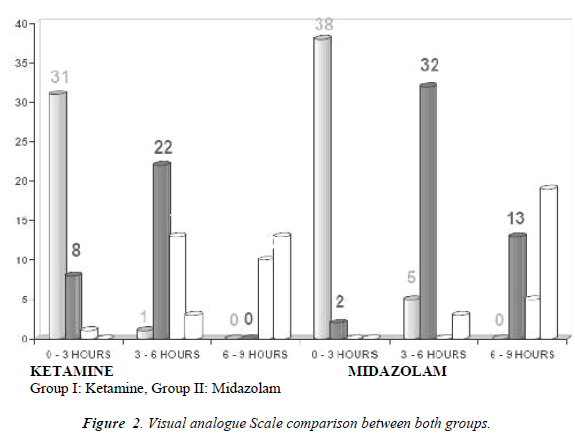
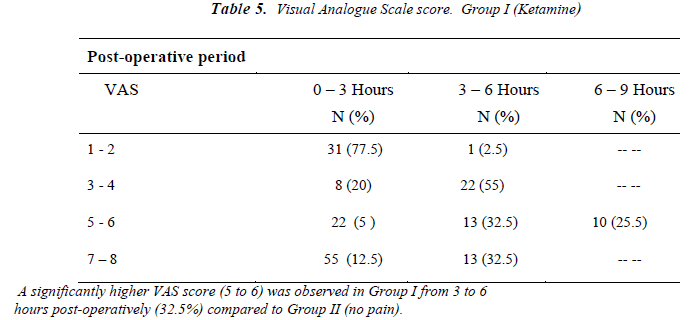
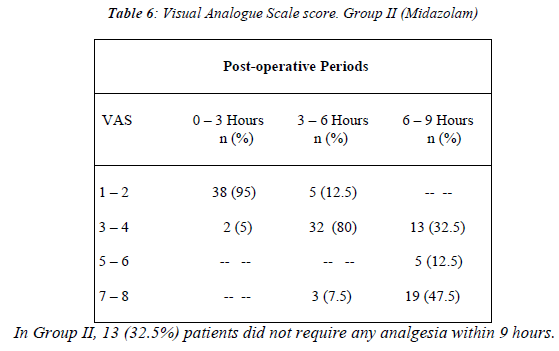
In the Group II, only 9 (22%) patients developed side effects and 37 patients did not develop any side effects. The incidence of intra-operative side effects between the two groups was statistically very highly significant (P < 0.001). [Table. 3] The VAS scores were comparable between both groups during the first 3 hours of immediate post-operative period. After 3 hours of post-operative period, the VAS score was statistically significant between two groups [Figure 2; Tables 5]. In Group II, 32.5% of patients did not require any analgesia within 9 hours [Table 6]. A significantly higher VAS score (5 to 6) was observed in Group I from 3 to 6 hours post-operatively as compared to the VAS (3 to 4) of Group II during this period [Tables 5].
VAS is a simple assessment tool consisting of a 10 cm line with 0 on one end, representing no pain, and 10 on the other, representing the worst pain ever experienced, which a patient indicates so the clinician knows the severity of the patient.
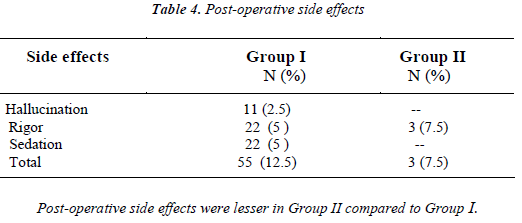
In Group I, 5 percent of patients developed sedation and rigor, and 2.5 percent patients developed hallucination during the post-operative period. In Group II, 7.5 percent of patients developed rigor during post-operative period. Post-operative side effects were lesser in Group II compared to Group I [Table 7].
The VAS scores were comparable between both groups during the first 3 hours of immediate post-operative period. After 3 hours of post-operative period, the VAS score was statistically significant between two groups.
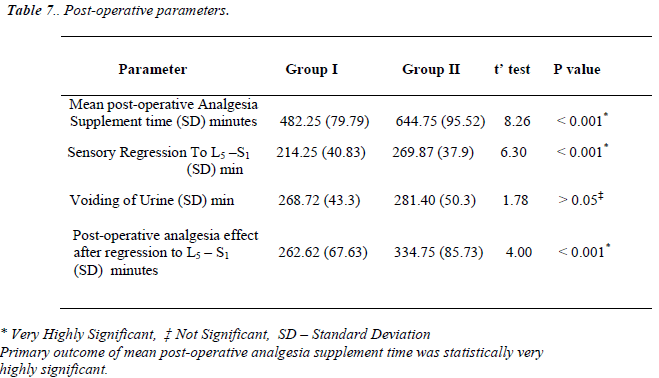
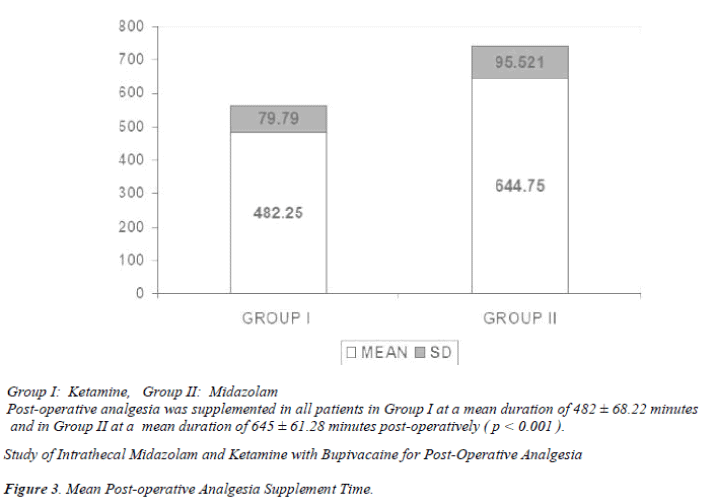
Post-operative analgesia was supplemented in all patients in Group I at a mean duration of 482 ± 68.22 minutes post-operatively (p < 0.01; VHS) [Table 7]. Only 2 patients in Group II demanded post-operative analgesia within this period. Post-operative analgesia was supplemented in all patients in Group II at a mean duration of 645 ± 61.28 minutes post-operatively. The difference in mean post-operative supplemental analgesic time between the 2 groups was very highly significant (p < 0.001) [Table 7], [Figure 3]. The time required for the sensory level to reduce to L5 – S1 was longer in Group II compared to Group I (p < 0.001). Table 7 shows that the difference in post-operative analgesia effect after regression to L5 – S1 level was statistically longer in Group II (p < 0.001).
Discussion
Every surgical procedure produces pain. Intra-operative pain, which continues into the post-operative period, is a matter of major concern as far as anesthesiologists are concerned. The importance of spinal anesthesia with the addition of local anesthesia is well established, as it reduces the severity of post-operative pain and prolongs analgesia even after recovery from sensory and motor blockades. In this study, we compared 2 additives, ketamine and midazolam, for their analgesic effect in the postoperative period following spinal anesthesia. Bansal [12], Ohri [17], and Upadhyay [19] concluded that the hemodynamic stability was remarkable with intrathecal ketamine in patients who underwent lower limb and lower abdominal surgeries. In our study, the cardiovascular profile of our patients was found to be stable throughout the intra-operative period in both groups. There was no significant variation in pulse rate or respiratory rate between both groups [Table 2]. Bansal [12] noticed a mild increase in respiratory rate with intrathecal ketamine (mean 20.8 ± 0.3 to 30.8 ± 0.4), Bion [6] did not observe any significant change in respiratory rate, both correlates with our study [Table 2].
Our study shows that the addition of midazolam to intrathecal bupivacaine significantly prolongs the duration of post-operative analgesia. The time to first rescue analgesic was 645 ± 61.28 minutes in Group II compared to 482.25 ± 79.79 minutes in Group I. Kim MH, reported that the time to rescue analgesic was prolonged by only 2 hours and 4.5 hours when midazolam 1mg and 2mg, respectively, were added to bupivacaine intrathecally [34]. The administration of the benzodiazepine antagonist flumazenil and the GABA-A antagonist bicuculline has been reported to reverse the analgesic effect of intrathecal midazolam, suggesting that the antinociceptive actions are mediated via the benzodiazepine, Gamma-Aminobutyric Acid-A receptor complexes, which are abundantly present in lamina II of the dorsal horn ganglia of the spinal cord [25]. Intrathecal midazolam probably also causes the release of an endogenous opioid acting on the spinal delta receptor as naltrindole, a delta selective opioid antagonist, suppresses the analgesic effect of intrathecal midazolam [26].. In our study, 38 of 40 patients in Group II did not require any rescue analgesia for more than 645 ± 61.28 minutes [Table 7]. The time of regression to the sensory level of L5-S1 was longer in Group II (269 ± 37.98) compared to Group I (214 ± 40.88). Y K Batra [23] observed that the mean duration of time to recede to the L5-S1 sensory level was 267 ± 67.38 minutes, which correlates with our study. The mean post-operative analgesia period after regression to L5-S1 was statistically very highly significant (P < 0.001) [Table 7]. In Group I, 60% had intraoperative side effects compared to only 22% in Group II. [Table 3]. The incidence of side effects was more in Group I. In Group I, 42.5% had pain in the first 6 hours compared to only 7.5% in Group II (p < 0.01) [Tables 3]. All patients experienced pain (VAS > 4) in Group I within 9 hours, whereas in Group II, 67.5 percent developed pain (VAS > 4) within 9 hours and 32.5 percent did not require any supplemental analgesia within 9 hours (p < 0.001) [Tables 5 and 6]. We observed superior and prolonged post-operative analgesia in Group II, which was comparable to that observed by Y K Batra [23].
Conclusion
This study was undertaken to compare the analgesic efficacy of intrathecally administered ketamine and midazolam with bupivacaine for lower limb and perianal surgeries. The quality of analgesia was assessed by VAS. The VAS score was statistically significant between both groups after 3 hours of the post-operative period. A significantly higher VAS score was observed in Group I. The incidences of side effect are less in Group II in compare with Group I. In Group I, 42.5% of patient experienced pain in the first 6 hours compared to only 7.5% in Group II (p< 0.01) [Tables 5 and 6]. In our study, 38 of 40 patients in Group II did not require any rescue analgesia for more than 645 ± 61.28 minutes (p < 0.001)[Table 7].
Thus, we conclude that intrathecal midazolam provide very good and prolonged post-operative analgesia without significant intra-operative and post- operative side effects compared to intrathecal ketamine.
Competing interests
None of the authors has a financial relationship with a commercial entity that has an interest in the subject of this manuscript.
References
- Corrsen G, Domini EF. Dissociative Anaesthesia: Further pharmacologic studies and first clinical experience with the phencyclidine derivative C1-581. Anaesth. Analgesia 1996; 45: 29-40.
- Crawford ME, Jensen FM, Toftdahl DB, Madsen JB. Direct spinal effect of intrathecal and epidural midazolam on visceral noxious stimulation in rabbits. Br J Anaesth 1993; 70: 642-646.
- Fink AD, Nagai SH. Opiate receptor mediation of Ketamine analgesia. Anaesthesiology 1982, 56:291-297
- Ahuja BR: Analgesia effect of intrathecal Ketamine in rats. Br J Anaesthesia 1983; 55: 991.
- Burm AG, Vankleefal JW. Plasma concentration lidocaine and Bupivacaine after subarachnoid administration. Anaesthesiology 1983; 59:191-195.
- Bion JF. Intrathecal Ketamine for war surgery. A preliminary study under field conditions. Anaesthesia 1984; 39: 1023-1028.
- Green NM. Distribution of local anaesthetic solution within the subarachnoid space. Anaesth Analgesia 1985; 64: 715-730.
- Mohler H, Okada T. Benzodiazepine receptor: demonstration in central nervous system Science 1977; 198: 849-851.
- Zeisser M, Robilart A. Our experience of spinal anaesthesia for transurethral resection of the bladder and prostate. Retrospective study of 334 patients operated in 1986. J.Urology 1990; 96: 267-270.
- Godchild CS, Noble J. The effects of intrathecal midazolam on sympathetic nervous System reflexes in man - A pilot study. Br J Clin Pharmacol 1987; 23:279-285.
- Bonica’s Management of Pain. Practical Management of Pain. JAMA 2001; 286 (15): 1906-1907.
- Bansal SK, Bhatia VK: Evaluation of intrathecal Ketamine in emergency surgery. Ind. J.Anaesth 1994; 42: 32-36.
- Gebhardt B. Pharmacology and clinical results with epidural and intrathecal administration of Ketamine. Anaesthesia 1994; 43 (Suppl. 2): 534-540.
- Hampl KF, Scheider MC. Transient neurological symptoms after spinal anaesthesia. Anaesth Analg 1995, 81: 1148-1153.
- Shekaran NM, Neelakandan B: Spinal Ketamine anaesthesia for hemithyroidectomy. Can J Anaesth 1996; 37: 537.
- Chung Yue Yang, Chih-Shung Wong. Intrathecal Ketamine reduces morphine requirements in patients with terminal cancer pain. Can J Anaesth 1996, 43:379-383
- Ohri AR. Intrathecal Ketamine and spinal anaesthesia. Ind J Anaesth 1997; 40: 41-43.
- Bullingham, Res, McQuay HJ. Extradural and intrathecal narcotics. In Atkinson R.S., Longthon C, Eds. Recent advances in anaesthesia and analgesia 1982; 41-54
- Upadhyay MR: Intrathecal Ketamine for short lower limb surgeries. Ind J. Anaesth 1998; 42: 70-73.
- Collins VJ: spinal; anaesthesia technique, chapter 56 in Principles of Anaesthesiology, Collins VJ, Philadelphia: Lea and Febiger 1993; 1511-1570.
- Reves, Gerald T, Glass, Peter SA, Lubaesky, Smith A: Non barbiturate intravenous anaesthetics, chapter 11 in Anaesthesia, Miller Ronald 1994; 259-264.
- Batra YK, Jain K, Chari P. Addition of intrathecal Midazolam to Bupivacaine produces better postoperative analgesia without prolonging recovery. Int J Clin Pharmacology Therapeutics 1999; 37 (10).
- Valetine MJ, Lyons G, Bellamy MC. The effect of intrathecal Midazolam on postoperative pain. European journal of Anaesthesiology 1996; 13: 589-593.
- Edwards M, Serrao JM, Gent JP. On the mechanism by which Midazolam causes spinally mediated analgesia. Anaesthesiology 1990; 23: 273-277.
- Goodchild CS, Guo Z, Gent JP. Antinociception by intrathecal Midazolam involves endogenous neurotransmitters acting at spinal cord delta opioid receptors. British Journal of Anaesthesia 1996; 77: 758-763.
- Nicol ME, Holdcroft A. Density of intrathecal agents. British Journal of Anaesthesia 1992; 68: 60-63.
- Mohamed Naguib, Mohamed EI Gammal, Yasser, Mahammed Seraj. Midazolam for caudal analgesia in children: comparison with caudal Bupivacaine. Can J Anaesth 1995; 42 (9): 758-764.
- Nishiyama, Hirasaki T, Odaka A. Epidural Midazolam with Bupivacaine – optimal dose for postoperative pain relief. Mausi Japanese Journal of Anaesth 1992; 41 (7): 1113-1118.
- Aaltonen L, Kanto J. The usefulness of radio receptor assay and gas liquid chromatography in pharmacokinetic studies on Midazolam. International Journal of Clinical Pharmacology, therapy and Toxicology 1985; 23 (5): 247-252.
- Kanto J, Aaltonen L, Aatimaa L. Pharmacokinetics and selective effects of Midazolam in connection with caesarean section performance under epidural analgesia. Acta Anaesthesiologica Scandinavia 1984; 28 (1): 116- 118.
- Audrey Shafer. Complication of selection with Midazolam in the intensive care unit and a comparison with the other selective regimens. Critical care Medicine 1998;. 26 (5): 947-952.
- Bromage PR. Practice of spinal narcotic analgesia – Basic consideration. Anaesthesia Analgesia 1981; 60: 461.
- Kim MH, Lee YM. Intrathecal midazolam increases the analgesic effects of spinal blockade with bupivacaine in patients undergoing haemorrhoidectomy. Br J Anaesth 2001; 86(1):77-79
- Kwok RF, Lim J, Chan MT, Gin T, Chiu WK. Preoperative Ketamine improves postoperative analgesia after gynecological laparoscopic surgery. Anesth analg 2001; 93(5): 1174-1177.
- Karthivel S, Saxena TR, Ganjoo P. Effects of intrathecal ketamine added to bupivacaine for spinal anaesthesia. Anaesthesia 2001; 55(9): 899-904
- 36. Tan PH, Chia YY, Lo Y. Intrathecal bupivacaine with morphine or neostigmine for postoperative analgesia after total knee replacement. Can J Anaesth 2001; 48(6): 551-556.