- Biomedical Research (2012) Volume 23, Issue 2
Carbohydrate diet links to higher risk of significant coronary artery disease in young Indonesian patients: Cardiometabolic Investigation study
Sugiri1, Sefri Noventi1, Ichiro Hisatome2,3 and Udin Bahrudin1,4*
1Department of Cardiology and Vascular Medicine, Faculty of Medicine Diponegoro University – Dr. Kariadi Hospital, Semarang, Indonesia
2Division of Cardiovascular Medicine, Tottori University Hospital, Yonago, Japan
3Division of Regenerative Medicine, Tottori University Graduate School of Medical Science, Yonago, Japan
4Division of Molecular Cardiology, Center of Biomedical Research, Faculty of Medicine Diponegoro University, Semarang, Indonesia
Accepted date: January 15, 2012
Abstract
Coronary artery disease (CAD) remains the leading cause of cardiovascular death. The CAD risk factors dyslipidemia, hypertriglyceridemia, and diabetes are related to dietary intake and their metabolism. However, the association between carbohydrate, lipid, and protein components of dietary intake and the significant CAD remains elusive; and it never been reported among Asian population. The purpose of this study was to identify the risk factor profile of significant CAD of young Indonesian patients and to compare those components of dietary intake in those patients with and without significant CAD. Within the framework of this Cardiometabolic Investigation study, a case / control group was established comprising 22 patients with significant CAD (case group) and 14 patients without or non-significant CAD (control group) aged ≤50 years all of whom were admitted to the Dr. Kariadi Hospital. The multivariate analysis of the risk factors for significant CAD were energy intake >1750 kcal/day (OR=14.4; p=0.005) and smoking habit (OR=7.7; p=0.036). The energy intake of the patients with significant CAD was significantly higher than that of the control (2005.25±85.91 vs. 1695.56±128.19 kcal/day; p=0.003). While the lipid and protein component of the energy intake did not vary significantly between those two groups, the carbohydrate intake in the significant CAD group was significantly higher than that of the control group (1142.99±38.11 vs. 950.00±75.88 kcal/day; p=0.018). Conclusion: In addition to the smoking habit, a high carbohydrate intake could pose a risk factor for the significant CAD in young Indonesian patients.
Keywords
carbohydrate, coronary artery disease, young Indonesian patients
Introduction
Coronary artery disease (CAD) remains the leading cause of cardiovascular death and is most commonly caused by the narrowing of the coronary artery lumen by atheromatous plaque [1]. Although the Framingham Risk Score, which represents a significant advance in the primary prevention of cardiovascular disease, classifies virtually all younger adults as low risk, regardless of risk factor burden [2–4], necropsy studies demonstrated that atherosclerotic changes in the vessel wall begin early in life [5]. A study using intravascular ultrasonography showed that coronary atherosclerosis begins at a young age and the lesions are present in one of six teenagers [6]. Individuals with low short-term but high lifetime risk have a greater burden and progression of subclinical atherosclerosis, as can be measured by coronary artery calcium and carotid intima-media thickness, compared with those with low short-term and low lifetime risk, even at <50 years of age [7].
Identification of risk factors for the significant CAD in young individuals is important in the prevention of CAD in the productive population. A study in the Japanese population showed that male sex, body mass index (BMI) >25 kg/m2, current smoker, family history of CAD, dyslipidemia, and metabolic syndrome-like factor accumulation are coronary risk factors which are more prevalent in younger patients than in the older [8]. The Korean CAD registry found that compared with the older patients, young patients with CAD were predominantly male and active smokers, but showed a lower prevalence of hypertension, diabetesor any prior history of stroke and myocardial infarction [9]. CAD risk factors dyslipidemia, hypertriglyceridemia and diabetes are related to their dietary intake and their metabolism. Compared with those with a 40%-carbohydrate diet, subjects receiving a 60%- carbohydrate diet had a lower high-density lipoprotein (HDL) cholesterol, a higher fasting plasma triacylglycerol and very-low density-lipoprotein (VLDL) triacylglycerol and VLDL-cholesterol concentrations [10]. However, the association between carbohydrate, lipid, and protein components of dietary intake and the significant CAD remains elusive, and it never been reported among Asian population.
In this study, we identified the risk factor profile of significant CAD in those patients, and then compared the carbohydrate, lipid, and protein components of dietary intake between young Indonesian patients with and without significant CAD. We found that the risk factors for significant CAD in young Indonesian patients were energy intake >1750 kcal/day and smoking habit; and a high carbohydrate intake was the dominant contributor in the high energy intake.
Methods
Subject and design of study
As a part of the Cardiometabolic Investigation (Carmetin) study, the design of this study was a case control in one center (Dr. Kariadi General Hospital, Semarang, Indonesia). Informed consent for participation in this study was obtained and the investigation was approved by the institutional ethics committee of human research in the Faculty of Medicine Diponegoro University and conformed with the principles outlined in the Declaration of Helsinki [11].
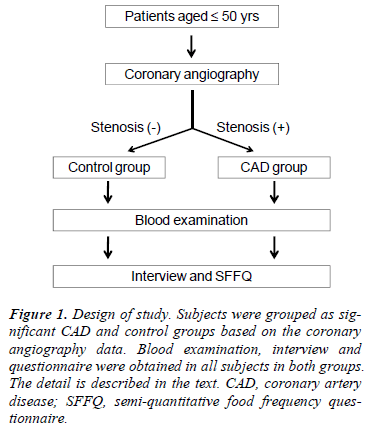
The design of study is shown in Figure 1. Subjects were eligible for inclusion in the study if their age was 50 years or less, the diagnosis of their condition for hospital admission was either stable angina pectoris or acute coronary syndrome, and they underwent percutaneus coronary angiography. We chose to study subjects who were aged (≤50 years), on the basis of previous work.7 Subjects were grouped as significant CAD if they had significant stenosis of their coronary artery (≥50%), and as a control if they did not have either stenosis of the coronary artery or their stenosis was not significant [12]. After an eight hours fasting, their blood samples were drawn from an antecubital vein. Total cholesterol, HDL cholesterol, LDL cholesterol, VLDL cholesterol, triglycerides and plasma glucose levels were determined in the clinical laboratory of Dr. Kariadi Hospital which has an acceptable standardization and quality control system. A personal interview was conducted with each patient to ask about smoking habits (never, past, or current smoker); past or current smokers were asked about the number of cigarettes smoked per day and the duration of smoking in years. To investigate dietary intake, we used a standardized semiquantitative food frequency questionnaire (SFFQ) which has been widely used to quantify food intake [13,14]. Food models were used to help patients fill in the questionnaire and to minimize recall bias. Dietary intake data collection has been described in detail elsewhere [15,16]. In brief, each subject completed two FFQs and 7-day weighed food intake record (DRs). The first FFQ was administered to provide data to compare with the second FFQ as a means of evaluating reproducibility, and the second FFQ was administered to obtain data to compare with the DRs to evaluate its validity. Only data from the second FFQ for validity has been used in this paper. Subjects with cancer, Kawasaki’s disease, or inherited metabolic disorder were excluded from this study. The diagnosis of the inherited metabolic disorder was done clinically as described in detail elsewhere [17].
Figure 1: Design of study. Subjects were grouped as significant CAD and control groups based on the coronary angiography data. Blood examination, interview and questionnaire were obtained in all subjects in both groups. The detail is described in the text. CAD, coronary artery disease; SFFQ, semi-quantitative food frequency questionnaire.
Statistical analysis
SPSS software version 17.0 (Polar Engineering and Consulting, USA) was used for the statistical analysis. Differences in basic clinical characteristics and dietary intakes between significant CAD and control groups were analyzed with chi-square test or t-test depending on the type of variables. The data were expressed in mean±SD, except where they were already mentioned. To assess the correlation between significant CAD and its risk factors, bivariate and multivariate analyses were used. Differences with a p value of <0.05 were considered statistically significant.
Results
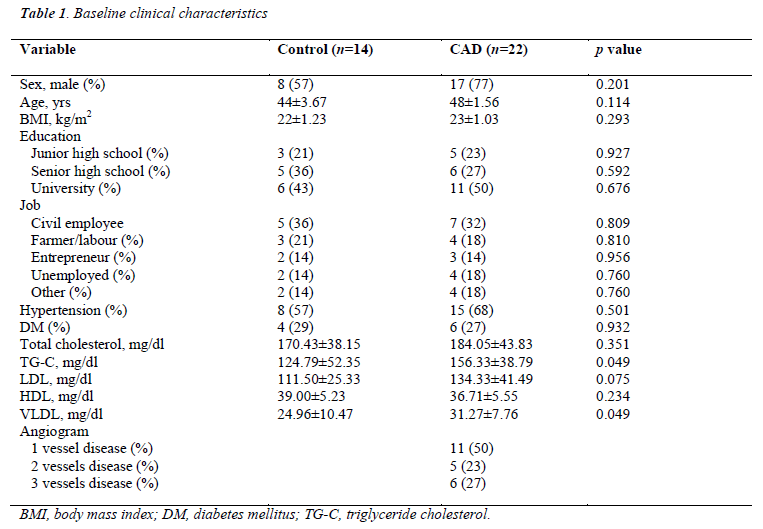
Baseline clinical characteristics between young patients (≤50 years old) with coronary artery disease and the control groups were not significantly different with the exception of the total triglyceride cholesterol and VLDL (Table 1). The number of cases of significant coronary stenosis within the subjects of CAD group varied. The number of subjects with one, two or three vessel diseases was 11 (50%), 5 (23%), and 6 (27%), respectively.
A high energy intake and smoking habit were risk factors for the significant CAD in young Indonesian patients
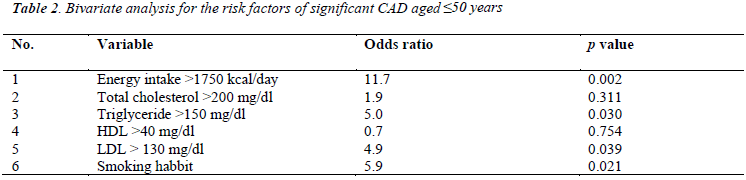
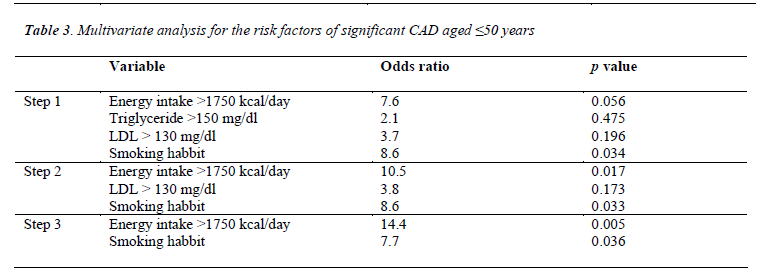
Bivariate analysis of the risk factors associated with significant CAD showed that energy intake >1750 kcal/day, triglyceride >150 mg/dl, LDL >130 mg/dl, and smoking habit had both a high odd ratio and a significant p value (Table 2), but the result of multivariate analysis showed that only energy intake >1750 kcal/day and smoking habit had both a high odd ratio and a significant p value (Table 3). This data suggests that energy intake >1750 kcal/day and smoking habit are risk factors associated with a significant CAD in young Indonesian patients.
A high carbohydrate intake contributed dominantly in the high energy intake of young CAD patients
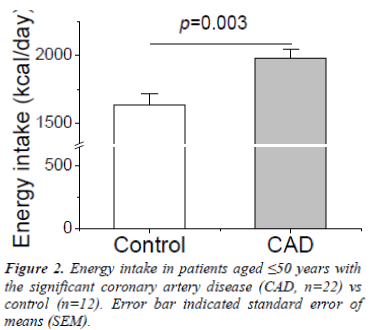
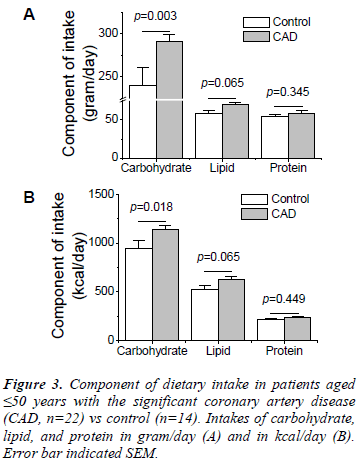
Figure 2 shows the comparison of intake of energy in young patients with a significant CAD and the control. The energy intake was significantly higher in the significant CAD group than in the control (2005.25±85.91 vs. 1695.56±128.19 kcal/day; p=0.003). Intake consisted of carbohydrate, lipid and protein. The components of each dietary intake are shown in Figure 3. The intake of lipid and protein were not significantly different between the significant CAD and control groups (lipid 69.65±3.85 vs. 58.73±3.97 gram/day; p=0.065 or 626.84±34.66 vs. 528.56±39.49 kcal/day; p=0.065; protein 58.86±3.28 vs. 54.25±3.21 gram/day; p=0.345 or 235.42±13.14 vs. 217±12.82 kcal/day; p=0.449), but carbohydrate intake was significantly higher in the significant CAD group than the control (290.69±8.56 vs. 240.28±20.27 gram/day; p=0.003 or 1142.99±38.11 vs. 950.00±75.88 kcal/day; p=0.018). Taken together, this data indicates that a high carbohydrate intake was a dominant contributor to the higher energy intake of the significant CAD group as opposed to the control.
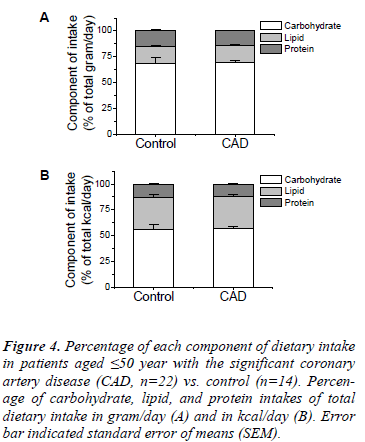
Figure 4 shows the percentage of each component of dietary intake in both significant CAD and control groups. Percentages of carbohydrate in significant CAD and control groups were 69.34±2.04% and 68.02±5.74% of the total daily intake in gram/day or 57.00±1.90% and 56.03±4.48% of the total daily intake in kcal/day, respectively. Percentages of lipid in significant CAD and the control groups were 16.61±0.92% and 16.63±1.12% of the total daily intake in gram/day or 31.26±1.73% and 31.17±2.33% of the total daily intake in kcal/day, respectively. The percentage of protein in significant CAD and control groups was 14.04±0.78% and 15.36±0.91% of the total daily intake in gram/day or 11.74±0.66% and 12.80±0.76% of the total daily intake in kcal/day, respectively. Thus, this data shows that the percentages of each component of dietary intake are not significantly different between both groups and the percentage of carbohydrate intake is high, equal to, almost 60% of the total daily energy intake in kcal/day.
Figure 4: Percentage of each component of dietary intake in patients aged ≤50 year with the significant coronary artery disease (CAD, n=22) vs. control (n=14). Percenage of carbohydrate, lipid, and protein intakes of total dietary intake in gram/day (A) and in kcal/day (B). Error bar indicated standard error of means (SEM).
Taken together, these data indicate that although the percentage of the carbohydrate intake as a percentage of the total intake in the significant CAD group is no different from that of the control, a high carbohydrate intake is a more dominant contributor to the higher energy intake of the significant CAD group than the control.
Discussion
In the present study, incidences of high energy intake and smoking habit were significantly higher in the patients with a significant coronary artery disease than that of the control, and the carbohydrate intake was a dominant contributor to the higher energy intake of the significant CAD group than the control. The finding that a high carbohydrate intake is significantly higher in the significant CAD patients is supported by other studies. The level of carbohydrate intake correlates with blood triglyceride levels in the Indonesian population [17]. Abassi et al. showed that a high carbohydrate diet decreases HDL-cholesterol and increases fasting triglycerides [18]. Reduced concentration of HDL and increased triglyceride as well as increased LDL are responsible for the genesis of atherosclerotic lesions [19–23]. A prospective study in US women by Liu et al. suggested that a high intake of rapidly digested and absorbed carbohydrates increases the risk of CAD independent of the conventional coronary risk factors [24].
A low-fat, high-carbohydrate diet can increase plasma triglyceride levels, known as carbohydrate-induced hypertriglyceridemia [25]. Triglycerides are composed of three fatty acids attached to a glycerol molecule. A dietary carbohydrate contributes to the formation of triglyceride molecules by forming either fatty acids through the Embden- Mayerhof pathway and Kreb’s cycle or glycerol moiety of the triglyceride molecule via u-glycerolphospate [26,27]. An increased triglyceride level in the plasma may occur by the decreased clearance of triglyceride-rich lipoprotein, i.e. VLDL and/or chylomicrons, and/or the increased triglyceride synthesis from either preformed or newly synthesized fatty acid and secretion from the liver [28-30]. Evidence of the enhancement of primary saturated fatty acid palmitate synthesis in a high-carbohydrate, low-fat diet was reported by Hudgkin et al. [31].
Our findings indicate that young Indonesian patients with a significant CAD have a significantly higher carbohydrate intake than those without CAD. Whether this finding represents the real population remains speculative at present. The limitations of this study may be as a result of the small number of subjects and because the results are based upon one center. Further study with a larger number of subjects and/or a multicenter study are warranted to re-affirm the findings in this present study. Lifestyle and biochemical data might be modified before and after onset of coronary artery disease, and the association between dietary intake profile and CAD could be be occured reversed. Nevertheless, several findings [9,10,-17-23] corroborate the suggestion that in addition to the smoking habit, a high carbohydrate intake could be an important risk factor for a significant coronary artery disease in young Indonesian patients.
Acknowledgements
This study was supported by the National Strategic Research Grant, Diponegoro University, 2009 (to Sugiri). We thank Prof. Endang Purwaningsih from the Department of Clinical Nutrition and Dr. Arie B Dwitaryo from the Department of Cardiology, Medical Faculty Diponegoro University for their collaboration.
References
- Morrow DA, Gersh BJ. Cronic coronary artery disease. In: Libby P, Bonow RO, Mann DL, Zipes DP, editors. Braunwald’s heart disease, a textbook of cardiovascular medicine, 8th edn. Philadelphia: Saunders Elsevier, 2008: 1353-1427.
- Grundy SM, Pasternak R, Greenland P, Smith S Jr, Fuster V. Assessment of cardiovascular risk by use of multiple-risk-factor assessment equations: a statement for healthcare professionals from the American Heart Association and the American College of Cardiology. Circulation 1999; 100: 1481-1492.
- Wilson PWF, D’Agostino RB, Levy D, Belanger AM, Silbershatz H, Kannel WB. Prediction of coronary heart disease using risk factor categories. Circulation1998; 97: 1837-1847.
- Berry JD, Lloyd-Jones DM, Garside DB, Greenland P. Framingham risk score and prediction of coronary heart disease death in young adults. Am Heart J 2007; 154: 80-86.
- Tuzcu EM, Kapadia SR, Tutar E, Ziada KM, Hobbs RE, McCarthy PM, et al. High prevalence of coronary atherosclerosis in asymptomatic teenagers and young adults: evidence from intravascular ultrasound. Circu-lation 2001; 103 (22): 2705-2710.
- Cole JH, Miller JI 3rd, Sperling LS, Weintraub WS. Long-term follow-up of coronary artery disease presenting in young adults. J Am Coll Cardiol 2003; 41 (4): 521-528.
- Berry JD, Liu K, Folsom AR, Lewis CE, Carr JJ, Polak JF, et al. Prevalence and progression of subclinical atherosclerosis in younger adults with low short-term but high lifetime estimated risk for cardiovascular disease: the coronary artery risk development in young adults study and multi-ethnic study of atherosclerosis. Circulation 2009; 119 (3): 382-389.
- Furukawa Y, Ehara N, Taniguchi R, Haruna Y, Ozasa N, Saito N, et al. Coronary risk factor profile and prognostic factors for young Japanese patients undergo- ing coronary revascularization. Circ J 2009; 73 (8): 1459-1465.
- Park JS, Lee HJ, Kim YJ, Seong IW, Lee JW, Kim JJ, et al. The epidemiological and clinical characteristics of patients admitted for coronary angiography to evaluate ischemic heart disease. Korean J Intern Med 2007; 22 (2): 87-92.
- Jeppesen J, Schaaf P, Jones C, Zhou MY, Chen YD, Reaven GM. Effects of low-fat, high-carbohydrate diets on risk factors for ischemic heart disease in postmeno- pausal women. Am J Clin Nutr 1997; 65 (4): 1027- 1033.
- World Medical Association Declaration of Helsinki. Recommendations guiding physicians in biomedical research involving human subjects. Cardiovasc Res1997; 35 (1): 2-3.
- Smith SC Jr, Feldman TE, Hirshfeld JW Jr, Jacobs AK, Kern MJ, King SB 3rd, et al. ACC/AHA/SCAI 2005 Guideline Update for Percutaneous Coronary Interven- tion-summary article: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (ACC/AHA/SCAI Writing Committee to Update the 2001 Guidelines for Percuta- neous Coronary Intervention). Circulation 2006; 113 (1):156-175.
- Fisberg RM, Colucci AC, Morimoto JM, Marchioni DM. Food frequency questionnaire for adults from a population-based study. Rev Saude Publica 2008; 42 (3): 550-554.
- Marks GC, Hughes MC, van der Pols JC. Relative validity of food intake estimates using a food frequency questionnaire is associated with sex, age, and other personal characteristics. J Nutr 2006; 136 (2): 459-465.
- Tsugane S, Sasaki S, Kobayashi M, Tsubono Y, Akabane M. Validity and reproducibility of a self- administered food frequency questionnaire in the JPHC Study Cohort I: study design, conduct and participant profiles. J Epidemiol 2003; 13 (1 Suppl): S2-S12.
- Ishihara J, Sobue T, Yamamoto S, Yoshimi I, Sasaki S, Kobayashi M, et al. Validity and reproducibility of a self-administered food frequency questionnaire used in the 5-year follow-up survey of the JPHC Study Cohort II: study design, participant profile and results in comparison with Cohort I. J Epidemiol 2003; 13 (1 Suppl): S 134-S147.
- Champion MP. An approach to the diagnosis of inher- ited metabolic disease. Arch Dis Child Educ Pract Ed2010; 95 (2): 40-46.
- Irawati ID. Correlation between recommended dietary allowances level of carbohydrate, fat, food fiber and blood triglyceride level. Study of Coronary Heart Dis- ease at the Sragen District General Hospital. Thesis, Faculty of Public Health, Diponegoro University. 2004.
- Abbasi F, McLaughlin T, Lamendola C, Kim HS, Tanaka A, Wang T, et al. High carbohydrate diets, triglyceride-rich lipoproteins, and coronary heart disease risk. Am J Cardiol 2000; 85 (1): 45-48.
- Singh RB, Mengi SA, Xu YJ, Arneja AS, Dhalla NS. Pathogenesis of atherosclerosis: A multifactorial proc- ess. Exp Clin Cardiol 2002;7 (1): 40-53.
- Gotto AM Jr. Triglyceride as a risk factor for coronary artery disease. Am J Cardiol 1998;82(9A):22Q–25Q.
- Chanu B. Hypertriglyceridemia: danger for the arteries. Presse Med 1999; 28 (36): 2011-2017.
- Patsch JR, Miesenböck G, Hopferwieser T, Mühlberger V, Knapp E, Dunn JK, et al. Relation of triglyceride metabolism and coronary artery disease. Studies in the postprandial state. Arterioscler Thromb 1992; 12 (11): 1336-1345.
- Geurian K, Pinson JB, Weart CW. The triglyceride connection in atherosclerosis. Ann Pharmacother 1992; 26 (9): 1109-1117.
- Liu S, Willett WC, Stampfer MJ, Hu FB, Franz M, Sampson L, et al. A prospective study of dietary glycemic load, carbohydrate intake, and risk of coronary heart disease in US women. Am J Clin Nutr2000; 71 (6): 1455-1461.
- Parks EJ. Effect of dietary carbohydrate on triglyceride metabolism in humans. J Nutr 2001; 131 (10): 2772S– 2774S.
- Macdonald I. Effects of dietary carbohydrate on lipid metabolism in primates. Proc Nutr Soc 1971; 30 (3): 277-282.
- Mohanlal N, Holman RR. A standardized triglyceride and carbohydrate challenge: the oral triglyceride tolerance test. Diabetes Care 2004; 27 (1): 89-94.
- Parks EJ, Hellerstein MK. Carbohydrate-induced hypertriacylglycerolemia: Historical perspective and review of biological mechanisms. Am J Clin Nutr 2000; 71 (2): 412-433.
- Nestel PJ, Carroll KF, Havenstein N. Plasma triglyceride response to carbohydrates, fats and caloric intake. Metabolism 1970; 19 (1): 1-18.
- Parks EJ, Krauss RM, Christiansen MP, Neese RA, Hellerstein MK. Effects of a low-fat, high-carbohydrate diet on VLDL-triglyceride assembly, production, and clearance. J Clin Invest 1999; 104 (8): 1087-1096.
- Hudgins LC. Effect of high-carbohydrate feeding on triglyceride and saturated fatty acid synthesis. Proc SocExp Biol Med 2000; 225(3): 178-183.