- Biomedical Research (2016) Volume 27, Issue 2
Assessment of thyroid function in critically ill patients.
| Kiran Bhat1, Sumita Sharma1, Kapil Sharma1, RK Singh2* 1Departments of Biochemistry and Internal Medicine, Shri Mahant Indiresh (SMI) Hospital , Shri Guru Ram Rai Institute of Medical and Health Sciences, Dehradun-248001, India 2Department of Biochemistry, TSM Medical College and Hospital, Lucknow-226008, India |
| Corresponding Author: Rajesh Kumar Singh, Departments of Biochemistry TSM Medical College and Hospital India |
| Accepted: January 30, 2016 |
Abstract
The present study included 340 inpatients of SMI Hospital admitted during the period January 2014 to December 2014 to study thyroid hormone profile in critically ill patients. The patients with history of thyroid disorders or any family history of thyroid disorders were excluded from the study. Serum free triiodothyronine (fT3), free tetraiodothyronine (fT4) and TSH (Thyroid Stimulating Hormone) levels were assessed in critically ill patients admitted in wards and Intensive Care Units (ICUs). 59% of the critically ill patients admitted in wards and ICUs showed abnormality in one or more than one parameters of the thyroid profile. Low fT3 level was the most common abnormality found in these patients. High TSH and low fT4 levels were the other common abnormalities. Assessment of thyroid function should be carried out in the critically ill patients, only if the clinical suspicion is very high. Otherwise the results may be misleading. The clinicians should consider that abnormal thyroid function test results in critically ill patients are more likely to be due to the alteration in the metabolism of thyroid hormones secondary to the disease process, rather than the thyroid disorder itself.
Keywords |
||||
| Hypermetabolism, Thyroid function, fT3, fT4, TSH, Critically ill patients. | ||||
Introduction |
||||
| The metabolic responses to sepsis involve every organ and tissue of the body and yet, surprisingly, little is known about the underlying mechanisms. During sepsis and other critical illnesses, the state of stress results in hypermetabolism, increased energy expenditure, hyperglycemia and muscle loss [1,2]. Critical illness is often associated with alterations in thyroid hormone concentrations in patients with no previous intrinsic thyroid disease [3-5]. Euthyroid Sick Syndrome (ESS) is the commonest endocrine change seen in critically ill patients [6]. | ||||
| It is described as abnormalities in circulating thyroid hormone levels without pre-existing hypothalamic pituitary or thyroid gland dysfunction in the setting of a Non-Thyroidal Illness (NTI) [7]. It reverts back to normal after recovery from the NTI. ESS is characterized by low triiodothyronine (T3), low or normal thyroxine (T4) and normal Thyroid Stimulating Hormone (TSH). Three patterns are described in ESS; Type1 or low T3 syndrome (seen in moderately sick patients), type 2 or low T4 syndrome (low T3 and T4, seen in very sick patients and associated with poor prognosis) and type 3 or low TSH syndrome. These probably reflect different stages on a continuum and severity of illness [8]. | ||||
| The most common thyroid hormonal change reported in critically ill patients is reduced serum T3 level. Under normal circumstances 100% of T4 and 10-20% of T3 are directly secreted by the thyroid gland. 5`deiodinase causes peripheral monodeiodination of T4 contributing to 80-90% of T3 and also increases the clearance of the inactive isomer reverse T3 (rT3) (which is derived by the action of 5` deiodinase on T4) [9]. | ||||
| Critical illness decreases 5`deiodinase activity, thereby, decreasing T4 to T3 conversion and rT3 clearance [8]. Increased metabolic clearance of T4 in critical illness further diverts T4 to form the inactive isomer rT3 [10,11].Thus, T3 decreases and rT3 increases [12]. Several mechanisms can contribute to the inhibition of 5`-monodeiodination and therefore to the low serum T3 concentration in critically ill patients with nonthyroidal illness: | ||||
| 1. Exogenous glucocorticoid therapy [13]. | ||||
| 2. Circulating inhibitors of deiodinase activity, such as free (non-esterified) fatty acids [14]. | ||||
| 3. Treatment with drugs that inhibit 5`-monodeiodinase activity, such as amiodarone and high doses of propranolol. | ||||
| 4. Cytokines (such as tumor necrosis factor, interferon- alpha, NF-kB and interleukin-6) [15-17]. | ||||
| Serum total T4 levels can be decreased (i.e., low T4 syndrome) typically in patients with more chronic and severe systemic illness [18-20]. Majority of patients have serum free-T4 either being normal or slightly decreased, but occasionally elevated [21]. This variability in free-T4 level reflects both the assay method used and the underlying illness. As the severity of illness, progresses, there is gradual development of a more complex syndrome associated with low T3 and low T4 levels that may correlate with the bad prognosis [22]. | ||||
| Serum TSH alterations in euthyroid patients with non-thyroidal illnesses include transiently reduced or elevated basal TSH values, blunted TSH response to TRH, diminished or absent diurnal rhythms of TSH, and altered TSH glycosylation and bioactivity [23,24]. | ||||
| Some hospitalized patients have transient elevations in serum TSH concentrations (up to 20 mU/L) during recovery from nonthyroidal illness. Few of these patients prove to have hypothyroidism when re-evaluated after recovery from their illness. Patients with serum TSH concentrations over 20 mU/L usually have permanent hypothyroidism [25]. The present study was conducted to evaluate the magnitude of alteration in thyroid hormone profile in non-thyroidal illnesses. | ||||
Material and Methods |
||||
| In this single centre, retrospective, observational study, 340 inpatients (91 Males; 249 Females) admitted to medical wards and ICUs, irrespective of diagnosis were included. Patients with previous thyroid disorders and those with family history of thyroid disorders were excluded. | ||||
| Fasting venous blood samples received from these patients were subjected for hormone analyses. Admission into wards or ICUs was based on the presentation of the patient and underlying clinical conditions unrelated to the study objectives. fT3, fT4 and TSH levels were assessed using chemilumniscence slide technique. | ||||
| The normal reference range for thyroid hormones in our laboratory is as follows: fT3 (4.26-8.10 pmol/l), fT4 (10.0-28.2 pmol/L) and TSH (0.465-4.68 mIU/l). Any deviation from the normal range is considered to be abnormal (low or elevated). We did not repeat the hormonal assays a second time or prior to discharge in survivors. | ||||
Results |
||||
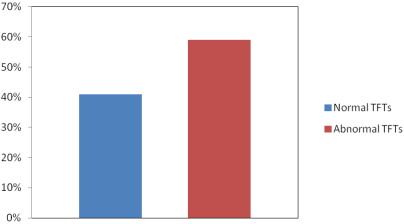
| Thyroid profile was assessed in 340 critically ill inpatients. Out of these 249 (73.2%) were females and 91 (26.8%) were males. 201 (59.1%) of the patients showed abnormality in one or more than one parameter of thyroid function tests (TFTs) (Figure 1 and Figure 2). Mean values of fT3, fT4 and TSH were as shown in Table 1. | ||||
Discussion |
||||
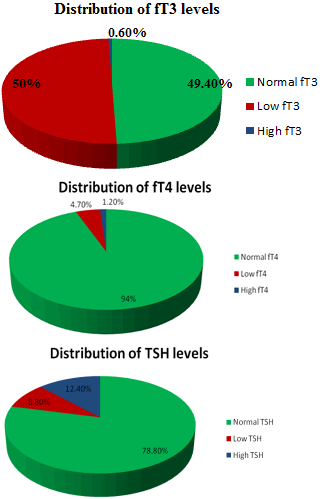
| Out of all the critically ill patients screened for thyroid disorders, 73.2% were found to be females. This preponderance of the female patients in the present study might be attributed to the fact that thyroid disorders being more common in females are more likely to raise physician’s suspicion. This may be the reason for high screening rate in the female patients. Moreover, hormonal differences between the two genders, working traditions, body weight, meal schedules, dietary factors or ingestion of drugs among other factors may be responsible for the observed gender differentiation in this study. Furthermore, it was found that in critically ill patients, low fT3 (50%) was the commonest abnormality followed by high TSH (12.4%), low TSH (8.8%) and low fT4 (4.7%) concentration. Occurrence of thyroid hormones abnormalities in sick patients confirms the previous observations that euthyroid sick syndrome is a common disorder in hospitalized sick patients. Half of the critically ill patients had low fT3 levels, which is mainly due to inhibition of deiodinase activity. Low fT4 levels were found in very few patients as serum concentrations of free T4 remain in the normal range in most patients unless the illness is severe and protracted. | ||||
| Multiple alterations in serum thyroid function test findings have been recognized in patients with a wide selection of Non- Thyroidal Illness Syndrome (NTIS) without evidence of preexisting thyroid or hypothalamic- pituitary disease. The most prominent alterations are low serum triiodothyronine (T3) and elevated reverse T3 (rT3), leading to the general term “low T3 syndrome.” TSH, T4, free T4 (fT4), and Free T4 Index (FTI) also are affected in variable degrees based on the severity and duration of the NTIS. As the severity of the NTIS increases, both serum T3 and T4 levels drop and gradually normalize as the patient recovers [26]. In the work of Sahana et al., NTIS was detected in 80% of the ICU patients [6] while Tognini et al., reported lower prevalence of 31.9% [27]. | ||||
| Changes in thyroid hormones (especially T3) and TSH may be seen as early as 24 h after the onset of non-thyroidal illness [28]. During recovery from intercurrent illness, thyroid hormones and TSH concentrations return to normal, although in some patients TSH may become overtly elevated for a short period of time. This rise in TSH typically precedes the increase in T4 and T3 concentrations, suggesting that it is required for the restoration of euthyroidism [29]. | ||||
| The goal of TFTs in the ICU should mainly be the identification of previously unrecognized thyroid dysfunction that would require therapeutic intervention. When hypothyroidism is suspected clinically in an ICU patient (e.g. hypothermia, bradycardia, respiratory acidosis, pleural effusions, failure to wean), and the evaluation suggests central hypothyroidism, one should consider, that the probability of euthyroid sick syndrome is much higher than the pituitary or hypothalamic disease. If hyperthyroidism is suspected (e.g. tachyarrhythmias, widened pulse pressure, respiratory alkalosis, high-output heart failure) and low TSH is detected, true hyperthyroidism, is unlikely unless the TSH is suppressed fully on a third-generation assay and the free T4 is elevated or at least in the upper limits of the normal range [30]. In conclusion, a common pitfall in the interpretation of thyroid function tests is to overlook the confounding effects of ‘Non- Thyroidal Illness’ (NTI). NTI (or sick euthyroid syndrome) is a relatively common finding following any acute or chronic illness which could be deliberated as a secondary adaptive change. It is important to be aware of this transient phenomenon in order to avoid inappropriate diagnosis and treatment. However, the knowledge of hypothalamic-pituitarythyroid axis, various factors governing thyroid hormone action at the cellular level and interpretation of different ranks of thyroid function tests may be considered to be central in establishing the correct diagnosis and further management. | ||||
Tables at a glance |
||||
|
||||
Figures at a glance |
||||
|
||||
References
|