- Biomedical Research (2016) Volume 27, Issue 1
Antibacterial activity of crude extracts and fractions from Iranian wild-grown and cultivated Agaricus spp.
| Hadi Soltanian1, Sharareh Rezaeian1, Abolfazl Shakeri2, Javad Janpoor1, Hamid R Pourianfar1* 1Industrial Fungi Biotechnology Research Department, Research Institute for Industrial Biotechnology, Iranian Academic Centre for Education, Culture and Research (ACECR), Mashhad-Iran 2Department of Pharmacognosy, Faculty of Pharmacy, Mashhad University of Medical Sciences, Mashhad-Iran |
| Corresponding Author: Hamid R Pourianfar, Industrial Fungi Biotechnology Research Department Research Institute for Industrial Biotechnology Iranian Academic Centre for Education Culture and Research (ACECR) Mashhad-Iran |
Abstract
Agaricus species are among the most important commercial mushrooms in the world. Thus far, antibacterial activities of several Agaricus spp. have been reported. However, no study was undertaken to evaluate antibacterial activities of Iranian wild Agaricus spp. In addition, the information about compounds conferring antibacterial activity in these species is scarce. The aim of this study was to determine the antibacterial activity of methanol-dichloromethane (1:1) extracts of several wild and cultivated species of Agaricus through a colorimetric micro-dilution method. In addition, several active fractions obtained from the crude extract of cultivated A. bisporus was assayed for antibacterial activity. The findings revealed that the mushroom crude extracts showed quantifiable inhibition only towards the Gram-positive bacteria. The cultivated A. bisporus significantly exhibited higher antibacterial activity than did the wild strain, even though no complete inhibition was observed. In addition, A. devoniensis and A. gennadii exhibited slight inhibitory effects against only Staphylococcus aureus. Six different fractions were then eluted from the crude extract of cultivated A. bisporus through a step wise gradient elution, of which two fractions exerted quantifiable antibacterial activities. As opposed to the crude extract, these two fractions significantly inhibited both the Gram-positive and the Gram-negative bacteria particularly E. coli at 8 mg/ml. These fractions might promisingly possess bioactive compounds and warrant further investigations to develop new antimicrobials obtained from A. bisporus.
Keywords |
| Agaricus spp., Antiviral activity, Gradient elution, Active fraction. |
Introduction |
| Continuous acquisition of antibiotic resistance remains an imperative driving force for the search for natural antimicrobial agents [1]. In the context of food safety, there is also an increasing concern on chemical preservatives, warranting more attention towards the antimicrobial potential of natural products. Various natural sources may be investigated for antimicrobial compound discovery, including plants, herbs, prokaryotes, fungi, and animals [2]. In recent years, edible mushrooms have also been seen as one of the great sources of antibacterial agents. Edible mushrooms harbor a broad spectrum of pharmacological activity, including anticancer, antimicrobial, antifungal, antiviral, anti-inflammatory, and anti-cholesterol activities [3]. |
| Among edible mushrooms, species belonging to the genus Agaricus are the most important commercial mushrooms in many countries. Thus far, antibacterial activities of extracts of several Agaricus spp. have been reported, including A. bisporus, A. brasiliensis, A. bitorquis, A. essettei, A. silvicola, A. silvaticus, and A. cf. nigrecentulus [4]. However, the information about fractions or compounds conferring antibacterial activity in these species is scarce [4]. In addition, to the best of our knowledge, there is no report in the literature regarding antibacterial activity of two species of Agaricus: A. devoniensis, and A. gennadii. Moreover, there is limited data on antibacterial potency of wild-grown species of Agaricus as compared to the cultivated strains [5-7]. |
| During the year 2012, we have collected and identified several wild-grown species of Agaricus mushrooms from Iran, including A. devoniensis, A. gennadii, and brown strains of A. bisporus [8, 9]. Thus, it was of interest to investigate antibacterial activity of these wild mushrooms against several Gram-positive and Gram-negative bacteria. In addition, efforts were made to isolate several active fractions of the crude extract of cultivated A. bisporus. These fractions were also analyzed and assayed to determine their antibacterial activity. |
Materials and Methods |
| Chemicals and reagents |
| Dimethyl sulfoxide (DMSO), 2,3,5-Triphenyltetrazolium chloride (TTC), thin layer chromatography (TLC) plates, silica gel (Mesh 70-230), and solvents (analytical grade) including methanol (MeOH), dichloromethane (DCM), petroleum ether (PE), chloroform (CHCl3), and ethyl acetate (EtOAc ) were all acquired from Merck (Darmstadt, Germany). |
| Mushroom extracts and bacterial strains |
| Dried fruiting bodies of cultivated Agaricus bisporus (A15), a wild brown strain of A. bisporus (As003), and wild strains of A. devoniensis, and A. gennadii were prepared and macerated in DCM- MeOH (1:1, v/v) as described elsewhere [8]. The following freeze-dried bacterial strains were purchased from the Iranian Biological Resource Center (IBRC): Bacillus cereus (IBRC-M 10796), Staphylococcus aureus (IBRC-M 10690), Escherichia coli (IBRC-M 10698), Psudomonas aeroginosa (IBRC-M 10205), and Enterococcus faecalis (IBRC-M 10740). Freeze-dried Salmonella Typhi was obtained from the Persian Type Culture Collection (PTCC 1609). Brain- Heart infusion broth (BHIB) was used to revive the lyophilized bacteria and maintain bacterial broth cultures. |
| Silica gel column chromatography |
| The extract of cultivated A. bisporus (20 g) was reconstituted in 300 ml of DCM- MeOH (1:1) followed by centrifugation at 5000 rpm for five minutes. The aqueous phase was mixed with the silica gel powder until well adsorbed. A step-wise gradient elution was performed on a silica gel column (4 × 80 cm) using the following solvents: PE, PE/DCM (1:1), PE/DCM (1:4), DCM, DCM/CHCl3 (1:1), DCM/CHCl3 (1:4), CHCl3, CHCl3/EtOAc (1:1), CHCl3/EtOAc (1:4), EtOAc, EtOAc/MeOH (1:1), EtOAc- MeOH (1:4), and MeOH. The eluted fractions were pooled together according to the TLC profiles developed in the same solvents as those used in the column chromatography, and viewed under UV lamp (254 nm). The fractions were then evaporated until dryness and subjected to assay of antibacterial activity [10]. |
| Assay of antibacterial activity |
| The stocks were prepared in DMSO followed by making serial dilutions in the BHIB media to reach the range of 1.25-0.015 mg/ml and 8-0.015 mg/ml for the crude extracts and fractions, respectively. The difference in the range of concentrations was due to different solubility of stocks of the crude extract and that of the fractions in DMSO, ensuring the maximum concentrations of the crude extracts or fractions did not contain more than 10% (v/v) DMSO. To each well, 70 µl of the test mushrooms and 70 µl of the bacterial inoculum with density of 1.5 × 108 cfu/ml was added. Four controls comprising medium with 10% DMSO, medium with bacterial inoculum (negative control), medium with the test mushroom sample (to measure the turbidity of the extracts or fractions), and medium only were included in each experiment. All experiments were performed in triplicate and the plates were incubated at 37°C for 24 hours, after which 50 µl of TTC (0.25% w/v in water) was added into each well and the plates were incubated for further 30 minutes. The color change (from clear to red) was observed, photographed and then recorded using a microplate reader (Epoch- Biotek, USA) at 625 nm [11]. The dose-response curves for bacterial inhibition were drawn using the following equation: (1-t/c) × 100, where t represents the absorbance of the test mushroom sample from which the absorbance of the extract or fraction was subtracted, and c represents the absorbance of the negative control. The 50% minimum inhibitory concentration (MIC50) was determined based on the dose-response curves [11]. |
Results |
| Antibacterial activity of the mushroom crude extracts |
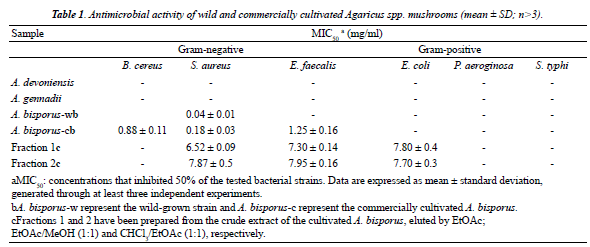
| The crude extracts of the Agaricus spp. could not quantifiably hinder the growth of the Gram-negative bacteria, while it prevented the growth of the Grampositive bacteria in a dose-dependent manner. Among the Gram-positive bacteria, Staphylococcus aureus was considerably inhibited by the extracts of the cultivated and wild strains of A. bisporus with MIC50s of 0.18 and 0.039 mg/ml, respectively, whereas it was less than 30% inhibited by A. devoniensis and A. gennadii at 1.25 mg/ ml. Enterococcus faecalis and Bacillus cereus were mostly susceptible to the cultivated A. bisporus. Table 1 summarizes MIC50s determined with the Agaricus spp., while Figure 1a depicts the dose-dependent antiviral activity of the crude extract of cultivated A. bisporus. Based on these findings, the crude extract of the cultivated A. bisporus proceed to further fractionation and isolation. |
| Antibacterial activity of fractions isolated from the A. bisporus extract |
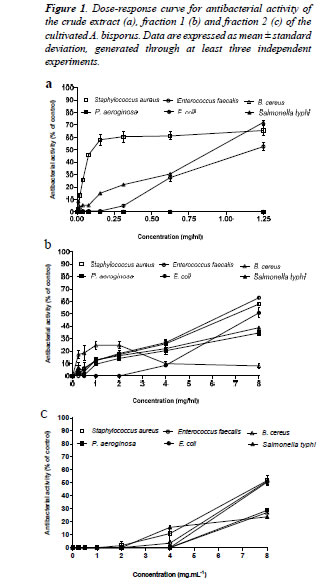
| Totally, more than 200 initial eluates were eluted through silica gel chromatography, of which six different fractions were prepared based on their TLC band patterns. The results revealed that fractions 1 (eluted by two eluent systems: EtOAc and EtOAc/MeOH 1:1) and 2 (eluted by CHCl3/ EtOAc 1:1) exerted quantifiable and dose-dependent antibacterial activities, against both gram-negative and gram-positive bacteria. No significant difference was observed between fractions 1 and 2 in the MIC50s (Table 1) or in the antibacterial activity towards the Gram-negative bacteria (p>0.05). On the contrary, significant differences were observed between two fractions against the Grampositive bacteria. While, fraction 1 exhibited significantly greater dose-dependent antiviral activity against the Gram-positive bacteria including Staphylococcus aureus and Enterococcus faecalis as compared to fraction 2 (p<0.05), fraction 2 inhibited Bacillus cereus higher than did fraction 1 (p<0.05) (Figures 1b and c). |
 |
 |
Discussion |
| This is the first report on potential antibacterial activities of the Iranian wild grown Agaricus spp, namely A. devoniensis, A. gennadii, that have been recently cultivated by us. Despite their distinguished antioxidant activity [8], they did not show considerable antibacterial activities in the current study. This is in agreement with the findings of Ozen et al. (2011) that there was not necessarily a correlation between the amount of total phenols and antimicrobial activity of wild Agaricus spp. [6]. |
| In this study, a dose-dependent inhibition of Grampositive bacteria by A. bisporus was observed, through a colorimetric dilution method. However, no MIC value could be drawn and no Gram-negative bacteria were inhibited. Micro-dilution methods coupled with a chromogenic reagent generate quantitative data and improve reliability and reproducibility, as only living microorganisms react with the chromogen reagent [12]. However, there have been very few studies employing colorimetric methods to determine antibacterial activity of A. bisporus [13]. Thus, studies that have used other methods should be also taken into consideration here. Accordingly, our findings are compatible with several studies [5, 7, 14], while they might contradict other studies [6, 14, 15] where low MIC values and susceptibility of Gram-negative bacteria was reported. These variations might be due to differences in methodology, extraction, bacterial strain susceptibility, or mushroom strain/type [6]. |
| Two fractions eluted by EtOAc/MeOH and CHCl3 obtained from the crude extract of cultivated A. bisporus exhibited quantifiable antibacterial activities towards both the Gram-negative and Gram-positive bacteria. The fractions had an improved solubility in DMSO so that we were able to test higher non-toxic concentrations as compared to crude extract. The results demonstrated that the crude extract was more efficient than the fractions, as it generated lower MIC50 values (Table 1) and quantifiable bacterial inhibition in lower concentrations (Figure 1). These findings might suggest that mycochemical constituents in combination may be having synergy in their efficacy. This is in agreement with many other reports that have shown higher antibacterial potency for crude extracts as compared to a fraction [16]. However, antibacterial potency of the fractions demonstrated in this research will be an important step towards purification and structural elucidation of components responsible in antibacterial activity in A. bisporus. At present, information on specific components responsible for antibacterial activity in A. bisporus is scarce. Alves et al. (2013) [17] showed potent antibacterial activity for 2,4-dihydroxybenzoic and protocatechuic acids, which have been previously isolated from several mushroom species including A. bisporus. However, direct association of these phenolic compounds to A. bisporus has not been elucidated. |
| In conclusion, these findings demonstrate that these fractions possess bioactive compounds that can be developed into new antimicrobials. Thus, further investigations will be required to separate and characterize components conferring antibacterial activity in cultivated A. bisporus. The effect of these components on a broad range of pathogens particularly resistant bacterial strains could also be further investigated. |
Acknowledgments |
| This research project was funded by a grant (code: 2173- 20) to HR Pourianfar from ACECR. |
Declaration of interest |
| There is no conflict of interest. The authors alone are responsible for the content and writing of the paper. |
References |
|