Case Report - Biomedical Research (2017) Volume 28, Issue 7
Another case of new-born baby with Blueberry-Muffin rashes: congenital viral infection or immune related hematologic disorders?
Hua Wang1,2,#, Chao Chen1,2,#, Zhong Chen1,2, Sha Zhao3, Jun Tang1,2, Qin Yao4 and Dezhi Mu1,2*
1Department of Pediatrics Neonatology, West China Second University Hospital, Sichuan University, Chengdu, China
2 Key Laboratory of Birth Defects and Related Diseases of Women and Children, Ministry of Education, Chengdu, China
3Department of Pathology, West China School Of Medicine/West China Hospital, Sichuan University, Chengdu, China
4Division of Neonatology, Rainbow Babies and Children’s Hospital and Department of Pediatrics, Case Western Reserve University, Cleveland, Ohio
#The two authors contributed equally to this work and should be considered as co-first authors
- *Corresponding Author:
- Dezhi Mu
Department of Pediatrics Neonatology
West China Second University Hospital
Sichuan University, Renmin South Road, China
Accepted date: December 19, 2016
Abstract
A 2750 g female infant presented with multiple striking 1-6 mm purpuric, non-blanching macules Blueberry-Muffin rashes. After symptomatic treatment, the whole body skin rash were scabby, liver and spleen were also moderately enlargement. The diagnosis was confirmed by skin biopsy. The parents were reluctant to use hormone therapy, and discharged against medical advises 21 days after admission.
Keywords
Blueberry-Muffin rash, Infant, Langerhans cell histiocytosis.
Introduction
Langerhan cell tissue hyperplasia (langerhans cell histiocytosis, LCH) caused by the hyperplasia of langerhans cell is rare and the incidence is 1-2/1000, 000 [1]. The clinical manifestations are diversification involving skin, bone, lymph nodes, liver, spleen, lung, gastrointestinal tract, oral cavity and even the central nervous system. The classification of histiocytic disorders has been revised into three different classes by the Writing Group of the Histiocyte Society: Langerhans cell histiocytosis (LCH) (Class I), histiocytosis belonging to the mononuclear phagocyte system, or non-LCH (Class II), and malignant clonal proliferations (Class III) [2]. LCH can damage various tissues by the accumulation of a clonal population of cells with a Langerhans (LH) cell phenotype [3].
Case Report
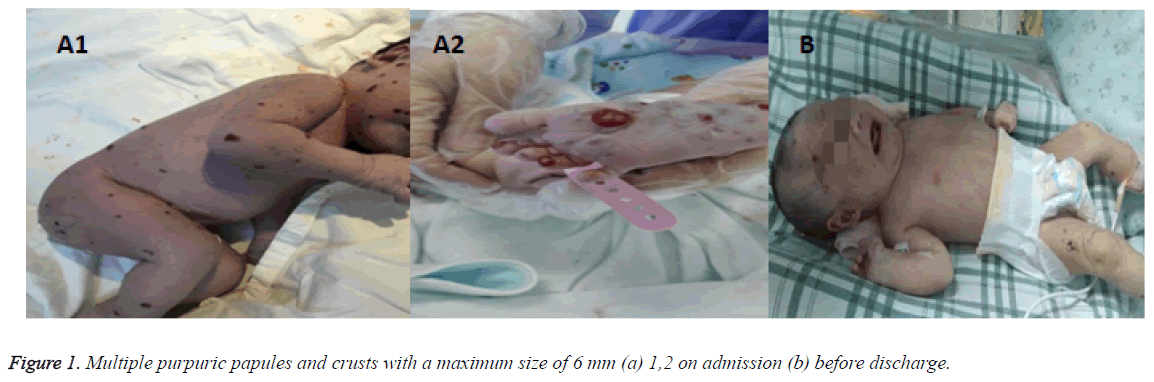
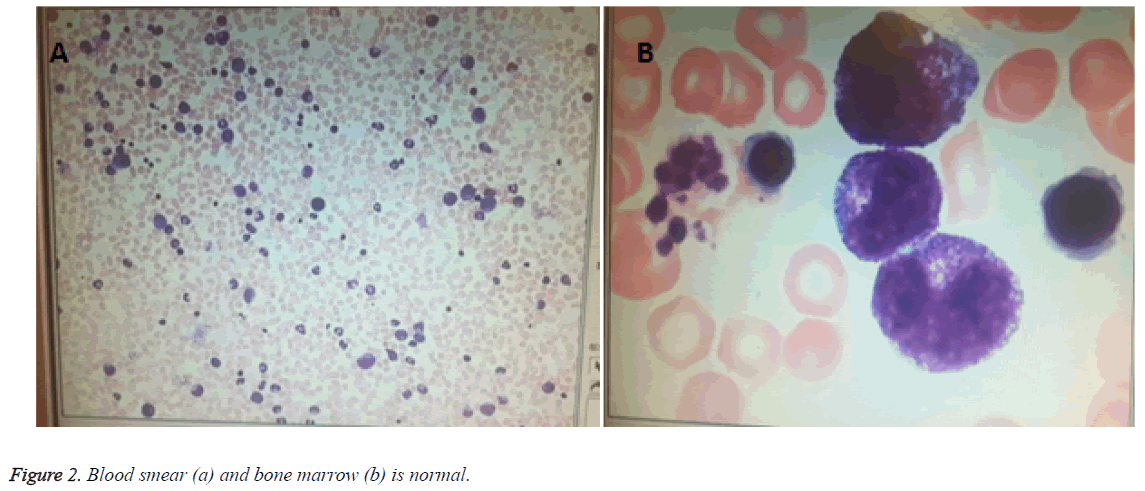
A 2750 g female infant, full term 40 1/7 week, appropriated for gestational age (AGA), was spontaneous vaginal delivery. Infant emerged vigorously with APGAR score of 9 at 1 min and 10 at 5 min. She presented with multiple striking 1-6 mm purpuric, non-blanching macules Blueberry-Muffin rashes scattered throughout whole body (face, lips, trunk, extremities, palms and soles) (Figure 1). Mom is healthy primigravida with an uncomplicated pregnancy. There was no history of autoimmune disease, HIV, Syphilis or animal or toxin exposure. Maternal serologies were negative for HIV, HCV, HBsAg, VDRL. The amniotic fluid, placental and umbilical cord appeared normal. On examination, the infant was healthyappearing and nondysmophic. Her vital signs were as follows: temperature, 36.5; RR, 45 times/min; blood pressure, 55/26 mmHg. The fontanelle measured 1.5 × 1.5 cm2, heart and lung exam were normal. She had mild hepatomegaly on palpation which was 2.5 cm below costal margin. Laboratory test showed WBC of 3.8 × 109/L, N of 78.1%, L of 16.8%, HGB of 165 g/L, PLT of 117 × 109/L, CRP of 21 mg/L, normal WBC differential on smear (Figure 2) and normal initial LFT. Differential diagnoses of an infant presented with Blueburrymuffin rashes, leukopenia, board line thrombocytopenia and mild hepatomegaly includes congenital infection, Hematologic disorders, malignancy/proliferative states and Neonatal lupus. Her subsequent laboratory tests revealed normal coagulation function and bone marrow (Figure 2). Ophthalmologic examination was also normal. Autoantibody presented positive resistance Ro-52 antibody (+ +), albumin of 23.5 g/L, Cr of 123 μmol/L, and troponin of 0.059 μg/L. Nucleated cell in cerebrospinal fluid was 2 × 106/L, while protein, sugar and chloride were within normal limits. TORCH, anti EBV IgM antibody, EB virus nucleic acid, against coxsackie virus IgM, adenovirus IgM, urinary and blood CMV DNA were all negative. Blood culture and sputum culture were also negative.
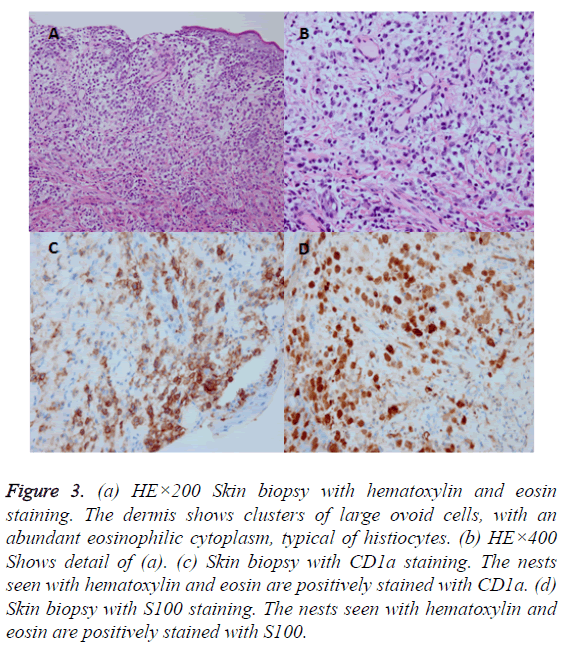
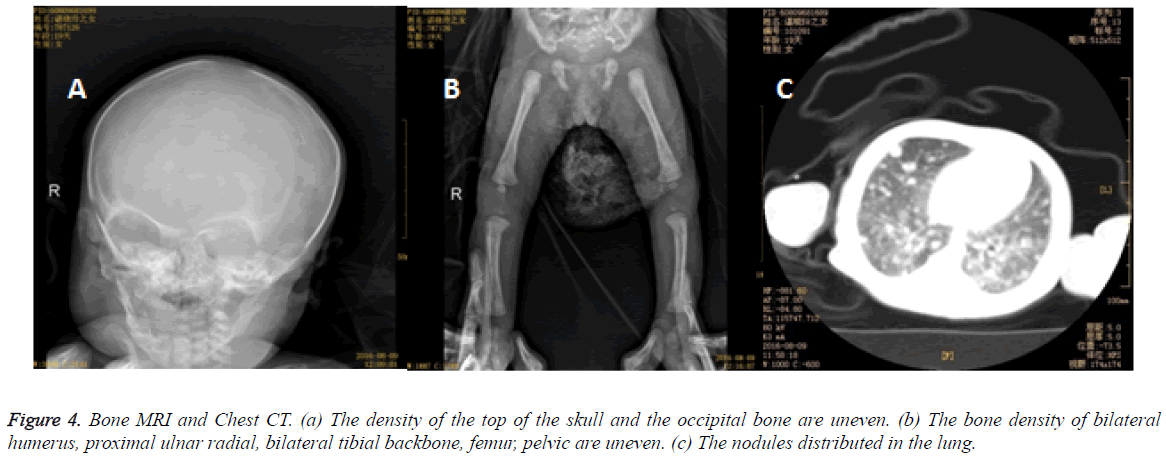
The skin biopsy disclosed that some mononuclear cell infiltrated the epidermis and the dermis courseware, and immunohistochemical study suggested CD1 and S100 positive (Figure 3). Gene mutated BRAF was found no 15 extra child point mutations (v600E) by gene mutation analysis. The patient brought computed tomography (CT) image of chest and abdomen which showed the nodules distributed in the lung, the density of the whole body bone is uneven with multiple small low density shadow. The left humerus exhibited low density shadow on MRI of bone.
Figure 3: (a) HE×200 Skin biopsy with hematoxylin and eosin staining. The dermis shows clusters of large ovoid cells, with an abundant eosinophilic cytoplasm, typical of histiocytes. (b) HE×400 Shows detail of (a). (c) Skin biopsy with CD1a staining. The nests seen with hematoxylin and eosin are positively stained with CD1a. (d) Skin biopsy with S100 staining. The nests seen with hematoxylin and eosin are positively stained with S100.
The bone density of bilateral humerus, proximal ulnar radial, bilateral tibial backbone, femur, pelvic were uneven as well as the top of the skull and the occipital bone (Figure 4). After symptomatic treatment, the whole body skin rash were scabby, liver and spleen were also moderately enlargement. The diagnosis was confirmed by skin biopsy. The parents were reluctant to use hormone therapy, and discharged against medical advises 21 days after admission. In the last telephone follow-up, the rash scabby was fade without new rash appearing, dyspnea, jaundice, bleeding and repeated infection. The child has been advised to be followed clinically with bone, liver and spleen surveys for at least two years. And pediatrician should be called if rash appears again. More attentions should be payed by parents to persistent swelling or bruising in the injured areas.
Discussion
Newborn skin rash, benign or malignant, is a very common problem. This female newborn baby was born with rash which may occur through infection and non-infection. Infection causes include herpes simplex virus, chicken pox, cytomegalovirus, B group hemolytic Streptococcus, Staphylococcus aureus, Listeria monocytogenes, Pseudomonas and Candida albicans. On the other hand, non-infection causes cover toxicity erythema, epidermolysis bullosa, congenital leukemia and relatively rare LCH. The major cause is usually infection, so we screened all types of infectious etiologies and perfected powerful anti-infection treatment before excluded it. Because of the resistance Ro-52 antibody was positive (++) in our case, we must rule out the Neonatal Lupus Erythematosus. Although other laboratory testing and the cardiac image were normal, it was finally ruled out by skin biopsy.
From 1985 to 2002, 894 children under 15 years with LCH were registered at German Childhood Cancer Registry. Sixteen of these patients (1.8%), 7 male and 9 female, were diagnosed within the first 28 days of life (15 of those were enrolled into LCH clinical trials and are therefore part of the neonatal cohort described below). The incidence of LCH in infants diagnosed under 1 year is calculated 25 per million infants (calculation based on 339 cases diagnosed in Germany between 1998 and 2002) [4]. 16 neonatal cases account for less than 5% of these cases. The diagnosis of LCH in all patients was established by histopathological examination of biopsy specimens according to the criteria of the Histiocyte Society [5,6]. Histopathologic examination should be performed to confirm the diagnosis, and typically reveals clusters of large ovoid cells with an abundant eosinophilic cytoplasm and a grooved indented nucleus in the upper dermis, sometimes with the sign of epidermotropism. Tumor cells contain Birbeck granules visible on electron microscopy, which can now also be identified by immunocytochemical and enzyme histochemical techniques, using CD1a and S100 as identifying markers [7,8].
Hashimoto et al. described a congenital self-healing reticulohistiocytosis (Hashimoto-Pritzker disease) with cutaneous lesions, mostly in the neonate stage [9]. In contrast to the isolated cutaneous LCH, only a couple of case reports concerning spontaneous resolution of MS-LCH have been reported [10,11]. In the review report, complete resolution of MS-LCH without systemic therapy was observed in 3/36 (8%) patients (2 untreated and 1 treated with topical steroids), all without risk organs at diagnosis. It is widely accepted that MSLCH presented in the younger is associated with poor prognosis [12,13]. Patient’s age has been implemented as a prognostic factor for stratifying LCH [12]. It seems, however, a close association between young age and disseminated disease [14-21].
Many different treatment regimens have been attempted for MS-LCH. Vinblastine and etoposide, both with activity against diverse disorders of the monocyte-macrophage system, appear to be the most effective. The results of this trial showed that both drugs, given with an initial high dose of methylprednisolone, are equally effective, but patients who show no response within 6 weeks may require different therapy. Recurrent disease affects more than 50% of patients who achieve remission, and LCH thus remains to be an orphan disease that warrants better treatment options. Conceivably, modern biologicals, such as (humanized) antibodies directly against CD1a, may be an interesting option to pursue.
Overall, in evaluating these patients, it is important to consider a differential diagnosis of a newborn presenting with skin lesions or mimicking extramedullary hematopoiesis. The differential diagnosis may include intrauterine toxoplasmosis, other infections (syphilis, varicella-zoster, parvovirus B19, rubella, cytomegalovirus, and herpes), leukemia cutis, neuroblastoma, LCH, various forms of hemolytic anemia, infantile hemangiomatosis, and other vascular anomalies. Diagnosis of LCH can be made according to histopathologic examination of a skin biopsy specimen. Hematoxylin and eosin stain typically demonstrates a dermal proliferation of mononuclear cells with reniform nuclei often associated with epidermotropism. Positive staining of these cells with CD1a and S100 confirms the presence of Langerhans cells.
Our case emphasizes the importance of recognizing the cutaneous manifestations for LCH, as well as the prevalence of multiorgan disease in this population. In such cases, close monitoring for disease progression is mandatory. At diagnosis, neonates with MS-LCH have less favorable prognosis compared with patients who are not in the neonatal period. Adapted treatment is mandatory for all neonates with MSLCH. In our case, although the patient may show poor prognosis, she has not developed serious complications by now, and she will continue to be followed.
Acknowledgment
The present study was supported by funds from the Natural Science Foundation of China (grant nos. 81401239 and 81401233). And thanks to Dr. Qin Yao for guidance and help in the diagnosis, treatment and follow-up process of our case.
Conflicts of Interest
The authors have no conflicts of interest relevant to this article.
References
- Kanitakis J, Zambruno G, Schmitt D, Cambazard F, Jacquemier D, Thivolet J. Congenital self-healing histiocytosis (Hashimoto-Pritzker). An ultrastructural and immunohistochemical study. Cancer 1988; 61: 508-516.
- SavaÅŸan S. An enigmatic disease: childhood Langerhans cell histiocytosis in 2005. Int J Dermatol 2006; 45: 182-188.
- Chu AC. Histiocytoses. In: Burns T, Breathnach S, Cox N (Eds). Rook’s Textbook of Dermatology. Malden: Blackwell Publishing, 2004; 521-523.
- Kaatsch P, Spix J. German childhood cancer registry-Annual report 2003 (1980–2002). Institute for Biostatistics, Epidemiology and Informatics, University of Mainz, 2004.
- Favara BE, Feller AC, Pauli M, Jaffe ES, Weiss LM, Arico M, Bucsky P, Egeler RM, Elinder G, Gadner H, Gresik M, Henter JI, Imashuku S, Janka-Schaub G, Jaffe R, Ladisch S, Nezelof C, Pritchard J. Contemporary classification of histiocytic disorders. The WHO committee on histiocytic/reticulum cell proliferations. Reclassification Working Group of the Histiocyte Society. Med Pediatr Oncol 1997; 29: 157-166.
- Avram MM, Gobel V, Sepehr A. Case records of the Massachusetts General Hospital. Case 30-2007. A newborn girl with skin lesions. N Engl J Med 2007; 357: 1327-1335.
- McKee P. Cutaneous lymphoproliferative diseases and allied disorders: Langerhans’ cell histocytosis. In: Whitehouse F, Russell D (Eds). Pathology of the Skin , Mosby, London, 1996, 1: 1251-1255.
- Hashimoto K, Bale GF, Hawkins HK, Langston C, Pritzker MS. Congenital self-healing reticulohistiocytosis (Hashimoto-Pritzker type). Int J Dermatol 1986; 25: 516-523.
- Broadbent V, Pritchard J, Davies EG, Levinsky RJ, Heaf D. Spontaneous remission of multi-system histiocytosis X. Lancet 1984; 1: 253-254.
- Corbeel L, Eggermont E, Desmyter J, Surmont I, De Vos R. Spontaneous healing of Langerhans cell histiocytosis (histiocytosis X). Eur J Pediatr 1988; 148: 32-33.
- Komp DM, Herson J, Starling KA, Vietti TJ, Hvizdala E. A staging system for histiocytosis X: a Southwest Oncology Group Study. Cancer 1981; 47: 798-800.
- Greenberger JS, Crocker AC, Vawter G, Jaffe N, Cassady JR. Results of treatment of 127 patients with systemic histiocytosis. Medicine (Baltimore) 1981; 60: 311-338.
- Braier J, Chantada G, Rosso D, Bernaldez P, Amaral D, Latella A, Balancini B, Masautis A, Goldberg J. Langerhans cell histiocytosis: Retrospective evaluation of 123 patients at a single institution. Pediatr Hematol Oncol 1999; 16: 377-385.
- Gadner H, Grois N, Arico M, Broadbent V, Ceci A, Jakobson A, Komp D, Michaelis J, Nicholson S, Pötschger U, Pritchard J, Ladisch S. Histiocyte Society. A randomized trial of treatment for multisystem Langerhans’ cell histiocytosis. J Pediatr 2001; 138: 728-334.
- Howarth DM, Gilchrist GS, Mullan BP, Wiseman GA, Edmonson JH. Langerhans cell histiocytosis: diagnosis, natural history, management, and outcome. Cancer 1999; 85: 2278-2290.
- Lucaya J. Histiocytosis X. Am J Dis Child 1971; 121: 289-295.
- Matus-Ridley M, Raney RB Jr, Thawerani H, Meadows AT. Histiocytosis X in children: patterns of disease and results of treatment. Med Pediatr Oncol 1983; 11: 99-105.
- Minkov M, Grois N, Heitger A, Pötschger U, Westermeier T, Gadner H. Response to initial treatment of multisystem Langerhans cell histiocytosis: An important prognostic indicator. Med Pediatr Oncol 2002; 39: 581-585.
- Nezelof C, Frileux-Herbet F, Cronier-Sachot J. Disseminated histiocytosis X: Analysis of prognostic factors based on a retrospective study of 50 cases. Cancer 1979; 44: 1824-1838.
- Rivera-Luna R, Alter-Molchadsky N, Cardenas-Cardos R, Martínez-Guerra G. Langerhans cell histiocytosis in children under 2 years of age. Med Pediatr Oncol 1996; 26: 334-343.
- Grana N. Langerhans cell histiocytosis. Cancer Control 2014; 21: 328-334.